Abstract
Background:
Reward-processing deficits have been demonstrated in obsessive–compulsive disorder (OCD) and this has been linked to ventral striatal abnormalities. However, volumetric abnormalities of the nucleus accumbens (NAcc), a key structure in the reward pathway, have not been examined in OCD. We report on the volumetric abnormalities of NAcc and its correlation with illness severity in drug-naïve, adult patients with OCD.
Method:
In this cross-sectional study of case–control design, the magnetic resonance imaging (MRI) 1.5-T (1-mm) volume of NAcc was measured using 3D Slicer software in drug-naïve OCD patients (n = 44) and age, sex and handedness-matched healthy controls (HCs) (n = 36) using a valid and reliable method. OCD symptoms were assessed using the Yale–Brown Obsessive Compulsive Scale (Y-BOCS) Symptom checklist and severity and the Clinical Global Impression-Severity (CGI-S) scale.
Results:
There was no significant difference in NAcc volumes on either side between OCD patients and HCs (F = 3.45, p = 0.07). However, there was significant negative correlation between the right NAcc volume and Y-BOCS compulsion score (r = −0.48, p = 0.001).
Conclusions:
Study observations suggest involvement of the NAcc in the pathogenesis of OCD, indicating potential reward-processing deficits. Correlation between the right NAcc volume deficit and severity of compulsions offers further support for this region as a candidate for deep brain stimulation treatment in OCD.
Introduction
Considerable progress has been made in the last two decades in understanding the neuro-anatomical correlates of obsessive–compulsive disorder (OCD) largely due to advances in brain imaging (Atmaca, 2011). Abnormalities in the specific corticostriatal-thalamocortical loop involving orbitofrontal cortex, cingulum, striatum and thalamus have been implicated in the manifestation of OCD. The neuropsychological deficits in OCD also suggest abnormalities in these brain regions that are involved in important cognitive functions postulated to be abnormal in OCD including response inhibition, error monitoring and executive functioning (Kuelz et al., 2004; Rao et al., 2008, 2010; Viswanath et al., 2009). In addition, it has been speculated that some pathological behaviors in OCD patients are connected to a maladaptive perception of reward (Figee et al., 2011). Compulsive washers find it difficult to perceive a sense of satiety and continue to feel forced to wash themselves (Phillips et al., 2000). By performing a compulsion, which is a negative reinforcement, there is a temporary relief of anxiety, which acts as a reward. In relation to this, the search for an immediate reward (relief of anxiety from compulsions), lack of behavioral flexibility (continuous repetition of the same behavior) and blindness to negative future consequences, the ‘myopia for the future’ (resulting in compromised life-quality), have all been described as characteristic traits of OCD patients (Cavedini et al., 2006).
Animal models and functional imaging have allowed the neurocircuitry of reward processing to be delineated, and show that the ventral striatum plays a key role (Knutson et al., 2001). Among the particular neurotransmitters involved in mediating the processing of rewards, dopamine plays a very important role (Fiorillo et al., 2003). This understanding comes primarily from theories of drug addiction. Patients with substance use show abnormalities in the neurobiology of reward processing, and may respond to treatments acting on these pathways (Bechara et al., 2002). This theory has been extended to explain other psychiatric disorders also. Reward processes are theorized to be disrupted in a range of disorders (Naranjo et al., 2001). A number of conditions involve repetitive goal-seeking behaviors. Pathological gambling (Stein and Grant, 2005) and hypersexual disorder (Stein et al., 2001), for example, may be characterized by disturbances in the processing of rewards. Likewise, subjects with OCD were found to be impaired in adjusting their behavior following monetary incentives (Nielen et al., 2009). The aberrant response of the ventral striatum has been demonstrated in medication-free subjects with OCD with altered incentive processing, convincingly suggesting that reward abnormalities exist in OCD and these abnormalities have been linked to the ventral striatum (Jung et al., 2011).
Symptoms in OCD are not always anxiety-driven. They could be related to cognitive and behavioral inflexibility, reflected by impairments in response inhibition and attentional set shifting (Chamberlain et al., 2005) because of dysfunctional frontostriatal circuitry (Chamberlain et al., 2008). OCD could also be conceptualized as an illness similar to behavioral addiction, with obsessions reflective of loss of voluntary control and compulsions indicative of addictive behaviors that are performed repetitively because of their ability to reduce anxiety generated by obsessions (Denys et al., 2004a, 2004b; Holden, 2001). It is possible that OCD, like other addictive behaviors, is associated with defective processing of rewards, a function that is critically dependent on ventral striatal–orbitofrontal circuitry (Tobler et al., 2009). The reward circuitry comprises a circuit that links the ventral tegmental area, nucleus accumbens and ventral pallidum through the medial forebrain bundle (Gardner, 2011). Striatal volumetric abnormalities have been reported in OCD. For instance, caudate and globus pallidus volume reduction has been documented in OCD patients compared with healthy controls (Robinson et al., 1995; Szeszko et al., 2004). On the other hand, a meta-analytical study of gray matter volume in OCD has reported that striatal structures were increased in volume among patients compared with healthy individuals (Radua and Mataix-Cols, 2009). Even though structural imaging studies have so far shown abnormalities in the dorsal striatal areas in OCD (Rotge et al., 2010), ventral striatal structures are possibly overlooked and have only occasionally been demonstrated (Menzies et al., 2008; Pujol et al., 2004). Indeed, functional imaging (Figee et al., 2011) and positron emission tomography (PET) (Hesse et al., 2011) studies have demonstrated an important role for the ventral striatum in OCD.
The nucleus accumbens (NAcc) receives strong afferents from the baso-lateral amygdala through the ventral amygdalofugal pathway and from the central and medial amygdaloid nuclei through the sublenticular and supracapsular parts of the extended amygdala (de Olmos and Heimer, 1999). Its main efferents innervate the pallidum, striatum, mediodorsal thalamus, prefrontal – including cingulate – cortex and mesolimbic dopaminergic areas. The NAcc attains, thus, a central position between limbic as well as mesolimbic dopaminergic structures, basal ganglia, mediodorsal thalamus and prefrontal cortex. The excitatory input from baso-lateral amygdala to the NAcc and the dopaminergic signaling within this structure is demonstrated to have a major role in cue-triggered motivated behaviors (Stuber et al., 2011). It is thus evident that the NAcc plays a significant role in reward function.
In a recent functional MRI study, OCD patients showed attenuated reward anticipation activity in the NAcc compared with healthy control subjects (Figee et al., 2011). A subset of treatment-resistant OCD patients who showed greater dysfunction in reward processing subsequently were successfully treated with deep brain stimulation (DBS) of the NAcc. Other studies have also shown that the NAcc, as part of the ventral striatum, is a successful target for DBS in OCD treatment (Denys et al., 2011). These studies have shown that the severity of compulsions reduces on surgical manipulation of this brain structure. In addition, a recent animal study reports that compulsive behavior becomes significantly reduced with electrical manipulation of NAcc (Mundt et al., 2009).
Studying reward processing and its neuroanatomical correlates in OCD could thus be a useful method to elucidate the pathogenesis of the condition. Based on the evidence from DBS studies and from preliminary work suggesting dysfunctional reward circuitry in OCD, we hypothesized that there would be volumetric abnormalities of NAcc in OCD patients in comparison with healthy controls. Further, we expected to find a correlation between the volume of NAcc and severity of OCD. The aim of the present study was to examine the volumetric differences of NAcc between drug-naïve OCD patients and healthy controls. In addition, we sought to examine the relationship between OCD symptom severity and NAcc volume.
Methodology
Subjects
The study subjects consisted of 44 drug-naïve patients with OCD and 36 matched healthy controls (HCs). The patients were recruited from the National Institute of Mental Health and Neurosciences (NIMHANS), Bangalore, India. Written informed consent was taken from all subjects before assessment. Approval from the ethics committee of the Institute was obtained. The diagnosis of OCD was made according to Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV) criteria (APA, 1994). Patients had never had exposure to psychotropic medications in the past. In addition, none had received cognitive behavior therapy. All the patients were evaluated with the Yale–Brown Obsessive Compulsive Scale (Y-BOCS) that includes a symptom checklist, a severity rating scale and item 11 for insight (Goodman et al., 1989), the Mini International Neuropsychiatric Interview plus (MINI plus) (Sheehan et al., 1998) and the Clinical Global Impression (CGI) scale (Guy, 1976). The Montgomery-Åsberg Depression Rating Scale (MADRS) (Montgomery and Åsberg, 1979) was used to measure the depressive symptoms.
Healthy controls who volunteered for participation in the study were recruited by word of mouth. The absence of any psychiatric diagnosis was confirmed by applying the MINI plus (Sheehan et al., 1998). They had no history of any medical illness or of substance dependence. There was no family history of psychiatric illness, including alcohol-dependence syndrome, in their first-degree relatives. All subjects were right handed. No subject had any contraindications for MRI. None of the subjects had any of the following: a medical or neurological illness that may have significantly influenced CNS function or structure, a history suggestive of delayed developmental milestones (suggestive of mental retardation), a family history of a hereditary neurologic disorder that may complicate diagnosis, a co-morbid DSM-IV psychoactive substance dependence and lifetime history of significant head injury (loss of consciousness longer than 10 minutes, seizures, neurological deficit, depressed skull fracture, surgical intervention, or central nervous system infection). Female subjects were neither pregnant nor within the postpartum period.
MRI acquisition
MRI was done with a 1.5-T scanner (Magnetom ‘Vision’; Siemens, Erhlangen, Germany) in the Department of Neuroimaging and Interventional Radiology at the National Institute of Mental Health and Neurosciences, Bangalore, India. A T1-weighted three-dimensional magnetization-prepared rapid acquisition gradient echo (MP-RAGE) sequence was performed (TR = 9.7 ms, TE = 4 ms, nutation angle = 12 degrees, and slice thickness = 1 mm with no inter-slice gap), yielding 1-mm3 isotropic voxels.
Measurement of the region of interest
The NAcc was traced in the coronal plane (Ballmaier et al., 2004). The first slice containing the most rostral end of the ventral caudate–putamen junction marks the anterior boundary of the NAcc (internal capsule separate from white matter). On slices rostrally with respect to the anterior commissure, the superior border of the NAcc is demarcated by a line that connects the inferior-most tip of the lateral ventricle to the most ventral point of the internal capsule at the level of the ventral putamen. From this last point, a vertical line is drawn from the tip of the internal capsule down to the white matter, thus defining the lateral border with the putamen. The medial border is identified by a line which includes all gray matter next to the white matter tract (olfactory radiation, radiation of the corpus callosum, olfactory tubercle). Once the globus pallidus appears, the superior border is extended from the internal capsule to the lateral inferior-most tip of the external medullary lamina of the globus pallidus. A vertical line is then traced straight down to adjacent white matter. Once the anterior commissure starts to decussate, a line is started from the junction of the internal capsule and the anterior commissure and then arced to the end point of the anterior commissure. The last slice, clearly containing the midline decussation of the anterior commissure, demarcates the posterior boundary of the NAcc. Complete details of the anatomical protocol can be found at resource.loni.ucla.edu/resources/downloads/research-protocols/masking-regions/nucleus-accumbens/).
Image analysis
All measurements were automatically calculated by computer using the 3D Slicer software (Version 3.4; www.slicer.org/) (Pieper et al., 2004). The NAcc contours were outlined in all brain images by a single investigator (JCN) using the computer mouse controlled pointer. The rater was blind to the subjects’ clinical details at the time of the brain measurements on coded MRI sections. A high inter-rater reliability (two raters: JCN and DJ) was established (intra-class correlation coefficient, two-way mixed effect, absolute agreement, ICC > 0.90) by independent blinded measurements of 11 training scans. The intracranial volume was calculated automatically using the FreeSurfer (Version 4.5; surfer.nmr.mgh.harvard.edu/).
Statistical analysis
The Statistical Package for Social Sciences (Version 13; SPSS, Chicago, IL, USA) was used for analysis. Data were assessed using the Shapiro-Wilk test and were found to be normatively distributed. The independent t-test and chi-squared test were used for continuous and discrete variables for baseline comparisons. The differences between OCD patients and HCs were examined using repeated measures analysis of covariance (ANCOVA) with age, sex and intracranial volume as covariates and with hemisphere as the repeated measure. For the subjects with OCD, we employed partial correlation analysis with age and intracranial volume as covariates in order to examine the relationship between NAcc volume and illness severity scores on either side. In view of eight tests of correlation (four on each side: Y-BOCS obsessions score, Y-BOCS compulsion score, Y-BOCS insight score and CGI-severity), we employed Bonferroni correction for multiple comparisons and, hence, a p-value of < 0.006 was considered statistically significant.
Results
The subjects with OCD were comparable to control subjects at baseline with respect to age (26.64 ± 6.78 years and 25.56 ± 5.60 years, t = 0.76, p = 0.44) and sex (males: 56.8% vs 66.7%, χ2 = 0.81, p = 0.36). For the subjects with OCD, the mean age of illness onset was at 20.97 ± 7.62 years. Table 1 shows the demographic profile and differences in brain volumes of subjects with OCD compared to HCs. Table 2 shows the various clinical parameters for the subjects with OCD. As shown, the subjects with OCD had moderate to severe illness (mean Y-BOCS total score = 26.70 ± 8.00). The mean duration of illness in months was 68.48 ± 60.36 (median value of 60.28). The following were the comorbid psychiatric conditions for OCD patients: depression (15.9%), social phobia (5.12%) and generalized anxiety disorder (2.4%). The mean MADRS score (mean ± SD) for subjects with OCD was 13.08 ± 9.41.
Demographic and volumetric comparison of OCD patients and healthy controls.
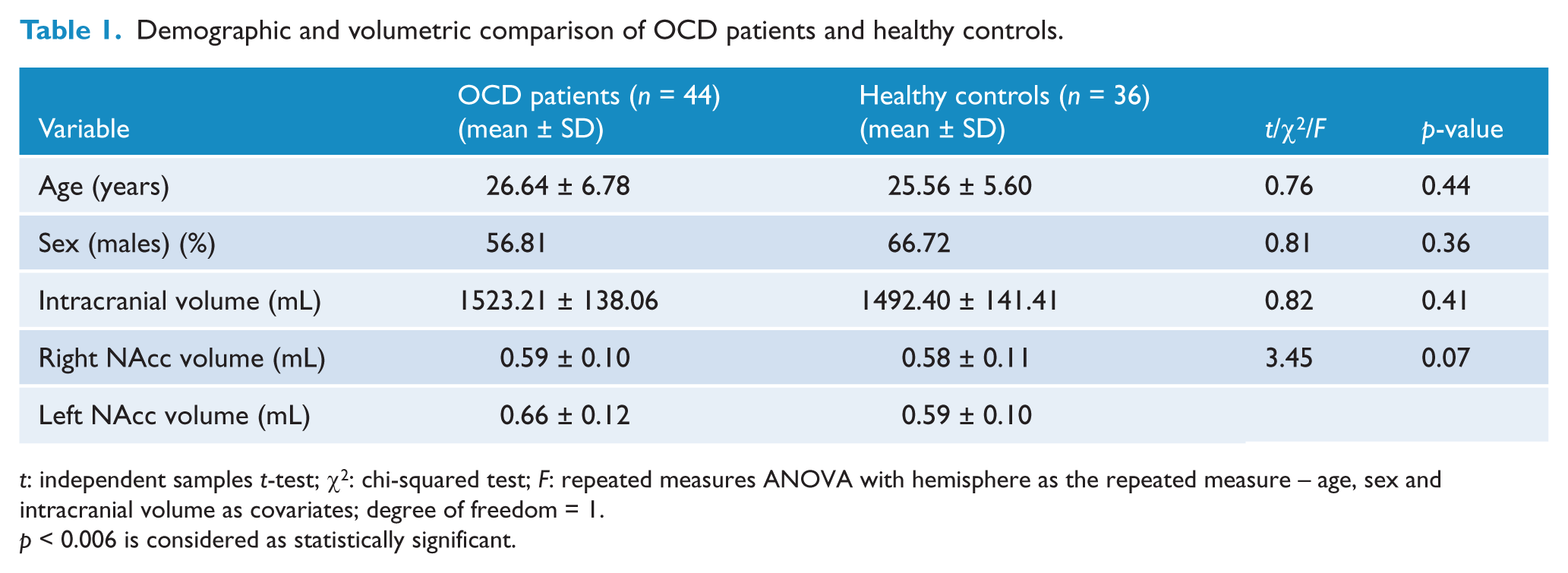
t: independent samples t-test; χ2: chi-squared test; F: repeated measures ANOVA with hemisphere as the repeated measure – age, sex and intracranial volume as covariates; degree of freedom = 1.
p < 0.006 is considered as statistically significant.
Clinical profile of subjects with OCD (n = 44).
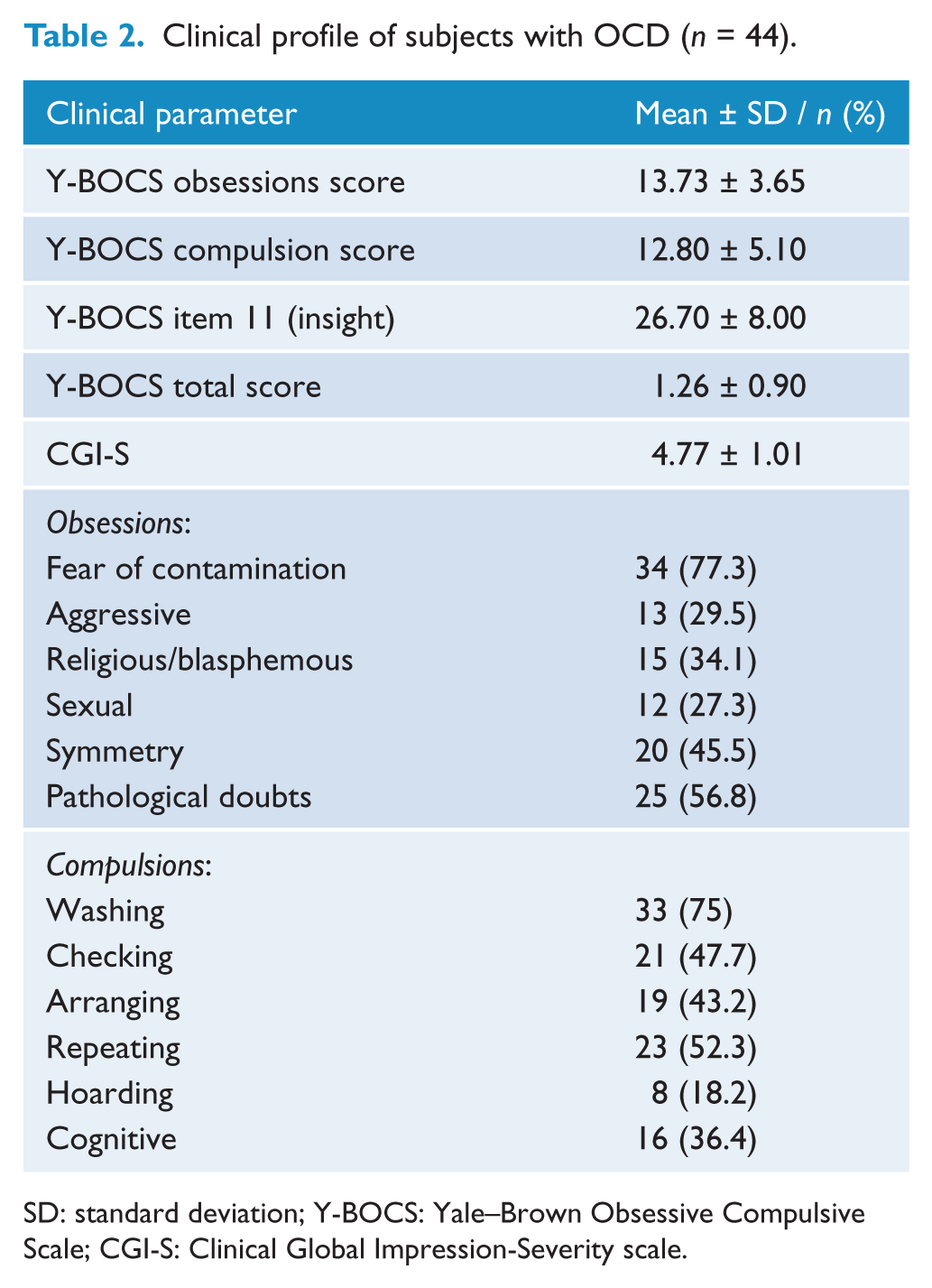
SD: standard deviation; Y-BOCS: Yale–Brown Obsessive Compulsive Scale; CGI-S: Clinical Global Impression-Severity scale.
There was no significant difference in NAcc volumes on both right (0.59 ± 0.10 vs 0.58 ± 0.11) and left (0.66 ± 0.12 vs 0.59 ± 0.10) sides between OCD patients and HCs on repeated measures ANCOVA with hemisphere as the repeated measure using age, sex and intracranial volume as the covariates (F = 3.45, df = 1, p = 0.07), as shown in Table 1.
Bivariate correlation revealed that age at assessment had a significant negative correlation with the right (r = −0.40, p < 0.001) and left (r = −0.24, p = 0.03) NAcc volumes. However, the depression severity score did not correlate with either right (r = 0.28, p = 0.07) or left (r = 0.19, p = 0.23) NAcc volumes. Additionally, illness duration also did not have significant correlation with right (r = 0.54, p = 0.12) or left (r = 0.11, p = 0.45) NAcc volumes. Partial correlation analysis with age and intracranial volume as covariates to assess the relationship between volumes measured and clinical severity indices of illness revealed that the compulsion severity and total Y-BOCS severity correlated with the right NAcc volume. Right NAcc volume had a significant negative correlation with the Y-BOCS compulsion score (r = −0.48, p = 0.001) (Table 3; Figure 1). However, there was no significant correlation between left NAcc volume and any of the clinical variables.
Correlation analysis of volumes of NAcc in OCD patients (n = 44) with illness severity scores (after controlling for age at assessment and intracranial volume).
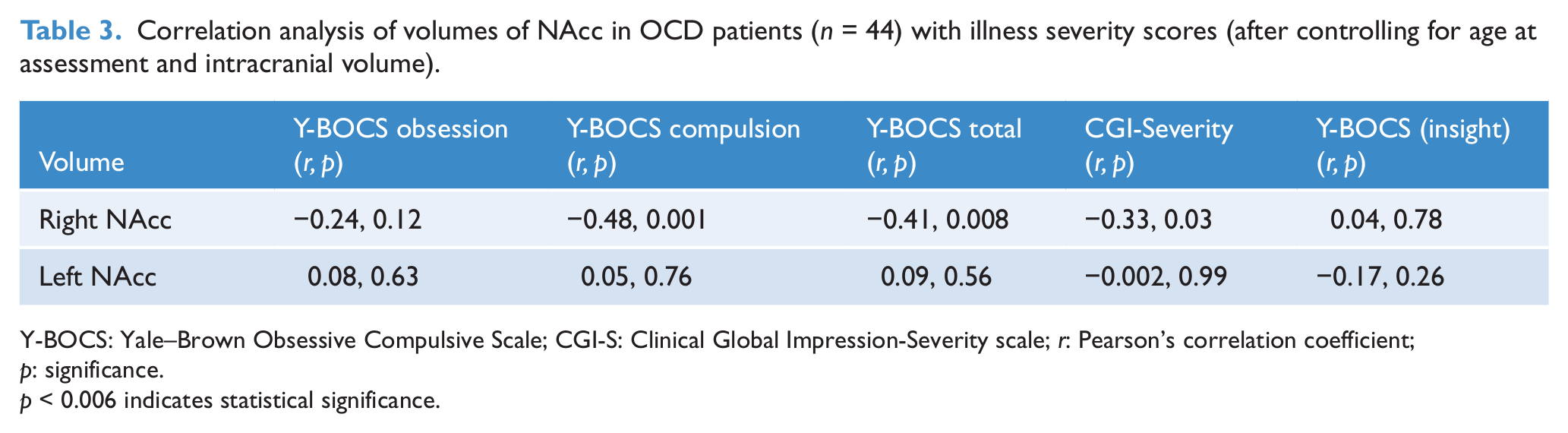
Y-BOCS: Yale–Brown Obsessive Compulsive Scale; CGI-S: Clinical Global Impression-Severity scale; r: Pearson’s correlation coefficient; p: significance.
p < 0.006 indicates statistical significance.
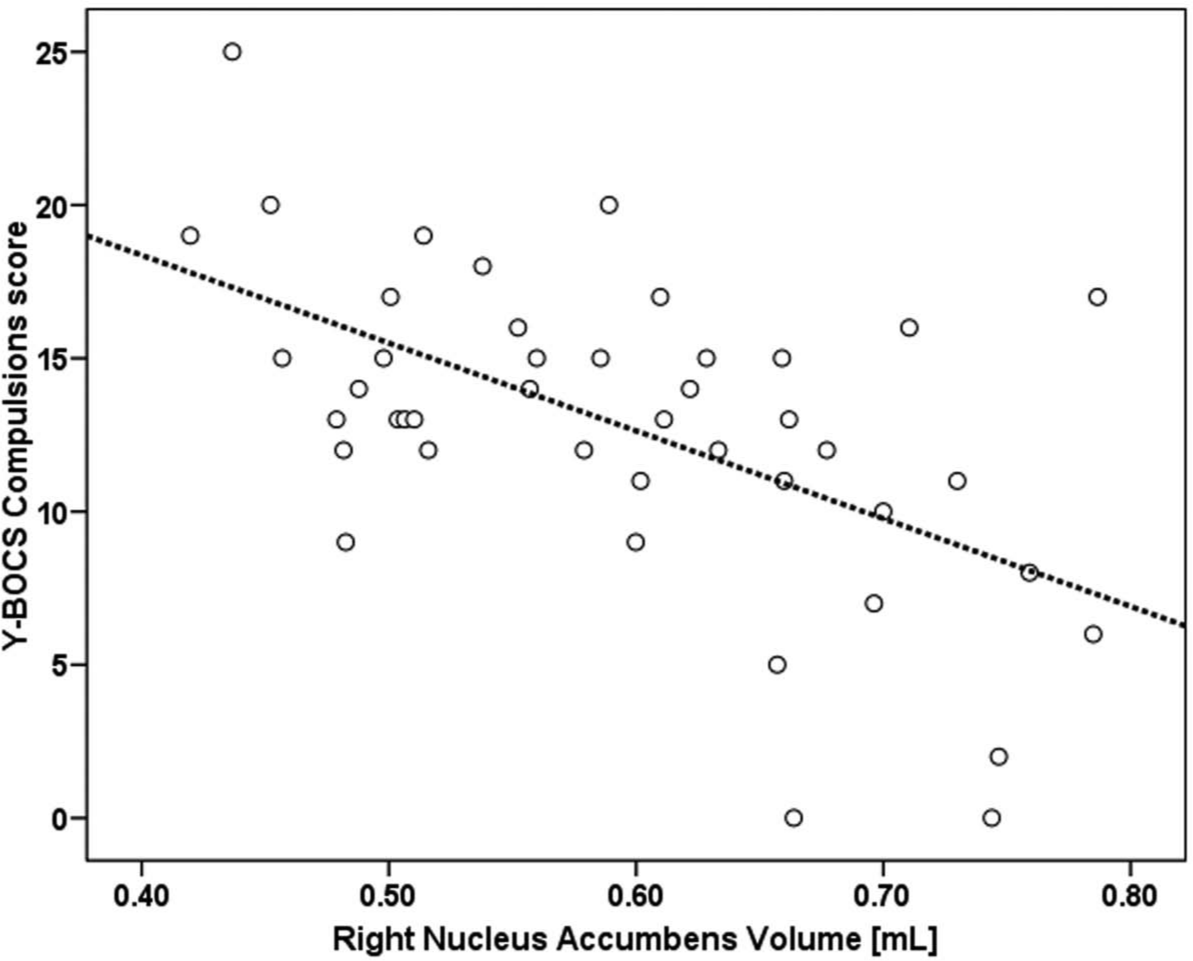
Relationship between right nucleus accumbens volume and YBOCS compulsions score in OCD patients (n = 44)
Discussion
We found a significant correlation between the right NAcc volume and the severity of compulsions. However, there was no significant volume difference of NAcc between subjects with OCD and HCs.
NAcc is a brain structure which is strongly implicated in incentive motivation, particularly in relation to its dopamine innervation from the ventral tegmental area (Berridge, 1996). Dopaminergic transmission in this region is important for maintaining response in conditions of intermittent reward (Salamone et al., 2003). As mentioned earlier in this article, reward processing abnormalities may underlie the symptoms in OCD. Reward processing depends on the integrity of the Orbitofrontal Cortex and basal ganglia, including the ventral striatum, especially the NAcc (Martin-Soelch et al., 2001). Since this is an important part of the frontostriatal circuit involved in the pathophysiology of OCD, it can be assumed that OCD is characterized by compromised functioning of this system. The structural and functional abnormalities in the ventral striatum may contribute to deficits in brain reward mechanisms (Aouizerate et al., 2004; Huey et al., 2008). Based on neuropsychological performance, there is evidence for impairment in reward processing in OCD (Cavedini et al., 2002, 2006). Similarly, neuroimaging studies have reported functional and structural deficits of the basal ganglia (especially caudate) in patients with OCD (Robinson et al., 1995; Rubin et al., 1992). However, no structural volumetric study of NAcc, which is the key region in ventral striatum, could be located on a thorough literature search of OCD. Recent studies using single-photon emission computed tomography have reported dopamine abnormalities in the basal ganglia of patients with OCD (Denys et al., 2004a; van der Wee et al., 2004). Similarly, OCD patients showed attenuated reward anticipation activity in the NAcc compared with healthy control subjects in a functional MRI paradigm. Reduced activity of the NAcc was more pronounced in OCD patients with contamination fear than in patients with high-risk assessment in that study (Figee et al., 2011). In a recent imaging study to investigate neural responses during anticipatory and consummatory phases of incentive processing, drug-naïve patients with OCD showed increased activation in the ventral striatum compared to healthy controls and a significant positive correlation between ventral striatal activation and compulsive symptom severity (Jung et al., 2011). The present study results also depict that volume deficit of NAcc on the right side correlates with increased severity of compulsions as measured by the Y-BOCS compulsion score (Figure 1). This underscores an important role of NAcc in the pathogenesis of obsessive compulsive symptoms, probably based on reward mechanisms. The precise mechanisms behind the volume changes and their relationship with compulsion severity are unclear. However, there is emerging evidence to suggest that striatal synaptic plasticity alterations, like changes in myelination and dendritic arborization, occur in many of the disorders that affect basal ganglia, including OCD (Kreitzer and Malenka, 2008).
It has been shown that patients with refractory OCD who received DBS at a region adjacent to the ventral striatum or NAcc showed significant improvement in symptoms (Denys et al., 2011; Greenberg et al., 2010; Huff et al., 2010; Sturm et al., 2003). In an experimental animal model, electrical modulation of NAcc core and shell activity via DBS reduced quinpirole-induced compulsive checking behavior in rats, therefore concluding that both the NAcc core and shell constitute potential target structures in the treatment of OCD (Mundt et al., 2009). The shell region of NAcc could thus represent a convergence zone for impulse-propagation from the amygdaloid complex to the basal ganglia, mediodorsal thalamus and prefrontal cortex, which in turn are the areas involved in the pathophysiology of OCD, as shown with functional imaging (Saxena and Rauch, 2000; Sturm et al., 2003). The blocking of this hypothetical pathological impulse-flow through chronic high-frequency stimulation of the shell of the NAcc could explain the good results in DBS of NAcc in OCD patients. It is also worth noting that unilateral stimulation on the right side was sufficient for the improvement of the DBS reports mentioned earlier. In tune with this, it is very interesting to highlight from our study, results of the role of right NAcc in the modulation of severity of compulsive behaviors – even though this needs further replication.
Our findings support the involvement of NAcc in the pathophysiology of OCD and add to the scant literature on the conceptualization of OCD as a disorder of reward processing similar to behavioral addiction. There appears to be an association between NAcc structural changes and severity of compulsions, indicating that this could serve as a marker of illness severity. The subjects were drug-naïve and had not received cognitive behavioral therapy when the imaging was performed, thus obviating the potential effects of medications and therapy on the brain structures. The duration of untreated illness could be a potential factor affecting the structural integrity of the brain. The illness duration in the present study is similar to that reported earlier in a large sample from the same population (Narayanaswamy et al., 2012) and this appears to be lesser when compared to the western literature (Catapano et al., 2006). However, as shown in the results, illness duration did not have a correlation with NAcc volumes on either side. In addition, the sample was chosen from a treatment-seeking population of a tertiary care center. This might limit its generalizability to a community level OCD population and could be a source of selection bias. The heterogeneity of symptom dimensions in the sample could also affect the results because the ventral striatal volume changes might be confined to specific dimensions and hence in certain subtypes of OCD. The effect of depression and anxiety comorbidity in the sample also needs to be kept in mind while interpreting the results, even though the depression rating score did not have a significant correlation with OCD severity scores.
Reward processing and its neural correlates in OCD may be important avenues for future research. Future studies should study the relationship between NAcc and various symptom dimensions of OCD as there is preliminary evidence that the involvement of reward circuitry (and NAcc) is somewhat specific to those with contamination fears (Figee et al., 2011). The specific relationship between the right NAcc volume deficit and severity of compulsions offers strong support for this region as a potential candidate for DBS treatment in OCD.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Declaration of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
