Abstract
Keywords
Introduction
Bipolar disorder (BD) and borderline personality disorder (BPD) share features that make them difficult to distinguish, raising the question of whether BPD is part of the bipolar spectrum (Berrocal et al., 2008; Brieger et al., 2003). Whilst research has predominantly focused on the clinical symptoms and pharmacological management of the two disorders, the degree to which the two compare in terms of childhood aetiological factors, self-schemas, and neurobiological substrates with respect to neuropsychological and neuroimaging probes, requires more extensive investigation.
The primary aim of this paper was to compare and contrast BD and BPD across clinical, behavioural, aetiological and neurobiological domains, so as to clarify diagnostic issues, and identify new avenues for future research.
Methods
An electronic literature search was conducted using MEDLINE and PubMed databases to identify studies that have directly compared, as well as researched the independent areas of BD and BPD in terms of emotional dysregulation, impulsivity, neurobiological substrates, and potential aetiological factors including a history of childhood trauma. These domains are widely recognised as having relevance to both these disorders and were therefore identified as being of potential interest. Keywords included ‘bipolar disorder’, ‘bipolar spectrum’, ‘borderline personality disorder’, ‘emotional (dys)regulation’, ‘impulsivity’, ‘childhood trauma’, ‘abuse’, ‘cognitive/neuropsychology/neuropsychological’, ‘neuroimaging/fMRI/PET/SPECT’ and ‘brain region’.
The paper is structured as follows: it begins with a brief description of BPD and the bipolar spectrum, and how the two overlap in clinical practice; specifically, the common core characteristics of emotional dysregulation and impulsivity, and childhood factors that influence behavioural outcomes and the course of illness, followed by an overview of the putative neurobiological substrates of each disorder gleaned from neuropsychological and neuroimaging studies. It then describes an integrated behavioural, aetiological and neurobiological model in which patients with BD and BPD are directly compared in the same methodological design.
BPD and the bipolar spectrum
Diagnostic overview and the common core characteristics of emotional dysregulation and impulsivity
BPD is a chronic syndrome characterised by enduring instability of affect and marked impulsivity, with associated problems in interpersonal relationships and poor self-image (American Psychiatric Association, 2000). BPD patients experience highly reactive moods that can change quickly between ‘highs’ and ‘lows’ several times a day. They also display a range of negative affect including tension, rage, sorrow, panic, and feelings of loneliness and emptiness, all of which they have difficulty in regulating, thereby prompting them to resort to maladaptive coping strategies. As a consequence of this affective instability, impulsivity in BPD typically constitutes deliberate and self-destructive behaviours that facilitate maladaptive coping strategies, including self-mutilation, substance abuse and eating disorders (Bazanis et al., 2002; Zanarini et al., 2006), compelling one in 10 patients to commit suicide (Lieb et al., 2004).
Controversies exist around the diagnosis of BPD, with some argument that it is incorrectly classified as a personality disorder, and that instead it is better placed within the mood disorders spectrum. Based on a cluster analysis of data sets containing personality traits between 1976 and 1978, before BPD became a fashionable term, Tyrer (2009) determined that the BPD symptom as opposed to trait profile, its failure to coalesce as a coherent persistent personality group, and its treatment-demanding propensities, set it apart from almost all other personality disorders. Further, there is insufficient evidence for BPD to remain in a group of disorders where it fundamentally does not belong, and that it is more appropriately classified as a condition of recurrent, rapidly changing unstable mood and behaviour.
BD is defined as a recurrent disabling illness that is characterised by periods of mania or hypomania, emerging usually in the context of prior depressive episodes. Symptoms of mania comprise elevated, expansive or irritable mood lasting at least one week (or less if hospitalisation is required), along with inflated self-esteem or grandiosity, decreased need for sleep, pressure of speech, flight of ideas, distractibility, increased involvement in goal-directed activities or psychomotor agitation, and excessive involvement in pleasurable activities (e.g. gambling, sexual indiscretions) with a high potential for adverse consequences (American Psychiatric Association, 2000). In practice, the diagnosis is often difficult (Malhi et al., 2010), and many of these symptoms can be conceptualised in terms of emotional dysregulation and impulsivity. Further, akin to BPD, interpersonal problems often feature in BD (Perugi et al., 2003).
Diagnostically, according to DSM-IV, BD is partitioned into bipolar I disorder (BD I) and bipolar II disorder (BD II) (American Psychiatric Association, 2000). The bipolar spectrum, although not formally recognised, ranges from BD I at one end to major depressive disorder (MDD) at the other. The efficacy of lithium for the acute treatment of mania, and its prevention of relapse, has been robustly demonstrated, and its specificity for classic manic depressive illness (Gershon et al., 2009) creates a clear distinction between BD I and the remainder of the bipolar spectrum (Malhi et al., 2009). In comparison, the definition of BD II which is based on milder forms of mania (i.e. hypomania) is much less precise. Consequently, in comparison to BD I, mood stabilisers produce less consistent effects (Malhi et al., 2010).
BD II blends into the ‘softer’ end of the bipolar spectrum where clinicians can find it difficult to diagnostically distinguish between the emotional dysregulation and impulsivity symptoms of BD and BPD, and this may explain why some BD II patients do not respond to mood stabilisers as effectively as those with BD I. This is evidenced by the fact that in recent research (Ruggero et al., 2010), 51% of BD patients had five or more traits that were deemed to be more typical of BPD, suggesting a risk of misdiagnosis, and BD II patients with co-morbid personality disturbances do not respond to medication to the same extent (George et al., 2003; Vieta et al., 1999).
In the past decade, the bipolar spectrum has been widened further to include bipolar III and bipolar IV disorder ‘subtypes’. Bipolar III disorder denotes a widely observed clinical phenomenon in which hypomania is induced by antidepressant treatment, but bipolar IV disorder, also termed ‘ultra-rapid cycling disorder’, in which mood changes from day to day and sometimes even more frequently, remains controversial (Paris, 2009). Fundamentally, it is unclear whether ultra-rapid cycling represents a form of mania, or is driven by a different pathophysiology. This makes it even more problematic to define BPD in relation to the bipolar spectrum (Akiskal et al., 2003; Paris, 2009), and in distinguishing BD from personality disorders.
Comparing and contrasting BPD and BD in emotional dysregulation and impulsivity
Applying formal diagnostic criteria, BPD can theoretically be distinguished from BD II on the basis of duration of mood states necessary to meet criteria for hypomania. A DSM-IV episode of hypomania must last at least four days, however, its assessment is reliant on accurate retrospective self-report as to how long specific mood states have lasted, and whether or not they have been stable for the duration of this period. Ratings of hypomania are therefore often unreliable (Akiskal, 1996; Dunner and Tay, 1993). Thus, exploration of the possible aetiological differences that could drive variations in behavioural symptoms within the realms of emotional dysregulation and impulsivity may offer greater success.
Affective instability has not been well examined in BD (Henry et al., 2007), and only one study to date has directly compared BPD and BD patients on this dimension (Henry et al., 2001). This research established that both disorders had a significant level of emotional dysregulation, irrespective of current mood state, but that its nature was different between the two groups. Specifically, BD patients were prone to shifts in affect from euthymia to elation or depression, or from elation directly into depression, whereas BPD patients experienced shifts from euthymia to anger or anxiety. Corroborating this, another study of personality disorders (Koenigsberg et al., 2002) found that BPD patients exhibited greater fluctuations into anger and anxiety, and experienced oscillations between depression and anxiety. Further, mood swings between elation and depression that are typical of BD were not significantly associated with BPD.
The differential tendency of emotional shifts towards anger and anxiety in BPD as opposed to depression in BD may influence the manner in which different types of impulsive behaviours manifest. When directly comparing BD and BPD patients using identical measures, the impulsive behaviours are distinguishable (Wilson et al., 2007). For example, BD II is more closely associated with attentional impulsiveness which includes a range of internal cognitive processes, such as reduced attention and concentration, distractibility and racing thoughts. Conversely, BPD is associated more with motor and non-planning impulsiveness which involves rash actions without consideration of the consequences. BPD patients have also been shown to score highly on measures of hostility, resentment and guilt which, combined with tendencies toward anger, feelings of worthlessness, and self-loathing, may lead them to attack themselves and others in order to control the situation or escape psychological pain. Such behaviours include self-harm, suicide attempts and substance abuse, or escalation of interpersonal problems with outbursts of anger (Chapman et al., 2011; Conklin et al., 2006; Ferraz et al., 2009).
It is important to note, however, that BD patients also demonstrate aspects of non-planning and motor impulsivity (Peluso et al., 2007; Swann et al., 2008), and impulsivity in BD can result in high levels of substance abuse (Swann et al., 2004), suicidal behaviours (Pompili et al., 2008; Swann et al., 2005) and self-mutilation (Joyce et al., 2010). Moreover, manic individuals often manipulate the self-esteem of others, exploit areas of vulnerability, test interpersonal limits, and project responsibility or blame onto others – behaviours typically witnessed in BPD. In theory, manic patients exhibit these behaviours because of their mood state, whereas BPD patients do so unremittingly (Mackinnon and Pies, 2006); and when compared to BPD, these self-destructive aspects of impulsivity are less evident and severe in BD. Recent research found impulsivity to be a greater indicator of suicidality in BPD patients when co-morbid BD was accounted for (Rihmer and Benazzi, 2010).
Aetiological factors: Childhood trauma and maladaptive self- schemas
In terms of possible aetiological factors that influence the emergence of BD versus BPD, adverse early childhood events have been proven to be strong predictors of the latter, and it has been suggested that these events, which impact on attachment later in life, may contribute to the psychopathology of BPD (Lieb et al., 2004; Sadikaj et al., 2010).
Between 30% and 70% of BPD patients have a history of childhood maltreatment, with one study reporting up to 90% of patients having experienced emotional, physical and/or sexual abuse (Zanarini et al., 1997). The severity of childhood abuse (defined by any one or combination of physical neglect or abuse, emotional neglect or abuse, and sexual abuse) correlates to the expression of BPD symptoms including dissociative symptoms, interpersonal problems, increased number of suicide attempts, and self-mutilation (Brodsky et al., 1995; Joyce et al., 2003, 2010).
With respect to BD, childhood abuse is also an important risk factor (Etain et al., 2010). More than half of BD patients experience some form of neglect or abuse (Etain et al., 2010; Garno et al., 2005), especially women (Brown and Anderson, 1991), and emotional abuse is the main form of trauma associated with bipolar illness (Etain et al., 2010). Notably, a history of severe childhood trauma involving physical or sexual abuse impacts the course and associated features of bipolar illness. In a large-scale study of 631 outpatients with BD I or BD II (Leverich et al., 2002), those who endorsed a history of child or adolescent physical or sexual abuse, compared to those who did not, had an earlier onset of their illness and an increased number of Axis I, II and III co-morbid disorders. These included drug and alcohol abuse, a higher rate of suicide attempts and, of particular significance, rapid and ultra-rapid cycling of mood. The association between severe childhood abuse and rapid-cycling BD, its earlier onset, suicidal behaviour, and substance misuse has been substantiated by a number of studies (Etain et al., 2008, 2010; Garno et al., 2005; Leverich and Post, 2006). From a neurobiological standpoint, the change in course of bipolar illness which stems from childhood trauma may be attributed to altered hypothalamic-pituitary-adrenal (HPA) axis functioning. Specifically, childhood abuse and early stressful life events produce blunting of cortisol responses to psychosocial stress (Carpenter et al., 2011), and suppression of plasma cortisol is associated with greater impulsivity (Basurte et al., 2004). Consequently, it could be argued that the symptoms of emotional dysregulation and impulsivity exhibited in BD patients with a history of childhood trauma are linked to altered HPA functioning and co-morbid personality dimensions that are shaped by these early life events, as opposed to the product of intrinsic bipolar pathophysiology per se.
There is emerging evidence for distinct cognitive styles between BD and BPD which stem from experiences and relationships with significant others during childhood. Nilsson et al. (2010) compared BD and BPD patients who did not concurrently fulfil criteria for the alternate diagnosis on the Young Schema Questionnaire, a primary measure of maladaptive self-schemas (Young, 2005). Compared to BD patients, BPD patients scored significantly higher on most of the schemas, suggesting that unique mental processes arise in BPD when these maladaptive self-schemas are triggered, and that these manifest as affective instability and impulsivity under specific circumstances. For example, a recent empirical study showed that BPD patients display more negative affect for a longer period of time, and that this is particularly poignant in situations of abandonment and rejection (Sadikaj et al., 2010).
This research raises the interesting possibility that rapid-cycling BD encompasses individuals with BD and BPD traits, and that it is a distinct subtype of BD. Consequently, BD and BPD may be separate disorders, and rapid-cycling BD represents those who have developed symptoms of both conditions because of similar lifetime influences and neurobiological precursors. However, research is still needed to further elucidate the impact of early childhood trauma on maladaptive self-schemas and the subsequent development of BD and BPD.
Neurobiological substrates of BD and BPD
Emotional dysregulation and impulsivity
Emotion regulatory processes are goal-directed behaviours, functioning to modify dynamic features of emotion such as the magnitude and duration of behavioural (i.e. expressive), experiential and physiological responses (Gross and Thompson, 2007). In healthy individuals, Functional Magnetic Resonance Imaging (fMRI) studies investigating the cognitive regulation of emotion have shown increased activation of the lateral and medial prefrontal regions, and decreased activation of the amygdala and medial orbitofrontal cortex (OFC), supporting the hypothesis that the prefrontal cortex and its connections with limbic structures construct reappraisal strategies that can modulate activity in multiple emotion-processing systems (Ochsner et al., 2002). Given the central role of prefrontal regions and their connections with other cortical and subcortical-limbic structures, successful emotional regulation draws heavily on a wide range of cognitive domains including attention, executive abilities and memory. The range of attention and executive functions in particular that are pertinent to emotional regulation include working memory, inhibition, problem solving, planning, and cognitive flexibility (Gyurak et al., 2009).
Impulse control is also an important function of the frontal-subcortical executive system that allows the suppression, interruption or delay of an activated behaviour or cognitive course of action (Aron et al., 2004; Starkstein and Robinson, 1997). Commonly used neuropsychological measures of impulsivity primarily involve those of attention and executive functions, and include sustained attention, inhibition (i.e. the suppression of an activated motor or verbal response) (Dimoska-Di Marco et al., 2011) and decision making (Alvarez-Moya et al., 2011). It has also been suggested that processing speed problems may account for poor performance on tasks of impulse control (Mathias and Wheaton, 2007; Ponsford and Kinsella, 1992).
Studies that have investigated the neuropsychological and neuroanatomical profiles of BD and BPD patients have tended to compare each population against healthy controls, or in the case of BD, patients with BD I have been compared to BD II. There is no literature to date that has directly compared BD and BPD groups within the same methodological design. The results of neuropsychological evaluation across the domains of memory, executive functions, attention, and processing speed, and their neuroanatomical correlates from brain imaging research for each of the two groups, are discussed separately.
i. BPD
A meta-analysis of the neuropsychology of BPD literature performed by Ruocco (2005) identified 10 studies that compared BPD patients to healthy controls on a broad range of standardised, valid and reliable neuropsychological tests. The results found that BPD patients generally performed poorly compared to healthy controls across the four broad cognitive domains, with effect sizes spanning from small to large. Specifically, BPD patients performed worse on all facets of visual and verbal memory including learning and acquisition, spontaneous retrieval (short- and long- delay), and storage in long-term memory (recognition); a range of executive functions including indices of cognitive flexibility (such as simultaneous conceptual tracking, set-shifting, utilising error feedback, and verbal generativity), inhibition/suppression of over-learned responses, planning, and problem solving; aspects of attention including immediate attention span, working memory, sustained attention and selective attention; and speed of information processing. No data for premorbid IQ, general intellectual functioning or education has been provided for BPD.
In more recent studies, further support for impulsivity as a core characteristic in BPD stems from the finding that patients have impaired performance on behavioural tasks of response inhibition, even when confounds such as co-morbid substance abuse were accounted for (Coffey et al., 2011). While some studies have found that a negative emotional state may moderate performance in BPD (Chapman et al., 2008, 2010; Sprague and Verona, 2010), others have shown that BPD patients are unable to delay immediate gratification in return for a longer-term reward, and that this occurs irrespective of induced negative affect (Lawrence et al., 2010), suggesting that in BPD, impulsivity is more likely a trait characteristic rather than a state feature that is moderated by emotion.
Brain imaging studies in BPD have attempted to identify the neural substrates of these neuropsychological findings using a variety of neuroimaging modalities. Several studies have detected decreased metabolism and regional cerebral blood flow (rCBF) in a number of prefrontal regions including the dorsolateral prefrontal cortex (DLPFC), parts of the anterior cingulate cortex (ACC) (De la Fuente et al., 1997), OFC (Soloff et al., 2003), right fronto-polar cortex, and right ventrolateral prefrontal cortex (VLPFC) (Goethals et al., 2005). The decreased prefrontal activation found in these studies may underlie the increased impulsivity and aggression associated with BPD (Sripada and Silk, 2007). Specifically, Soloff et al. (2003) found decreased OFC activation which correlated with self-reports of impulsivity and aggression. Other authors have also linked impulsivity and social difficulties in BPD with OFC dysfunction (see Dinn et al. (2004)). Berlin et al. (2005) compared BPD patients against patients with OFC lesions and found that both groups showed similar dysfunctions in the areas of impulsivity and emotional instability. LeGris and van Reekum (2006) concluded that BPD patients exhibit impaired executive function and disinhibitory processes, and further linked this to DLPFC dysfunction, in particular, self-harming and impaired decision making.
In terms of emotional dysregulation, in addition to fronto-limbic network alterations similar to those associated with impulsivity, patients with BPD also have reduced amygdala and hippocampal volume, and demonstrate enhanced amygdala responsiveness to emotional stimuli, such as negative scenes and facial expressions (Domes et al., 2009; Nunes et al., 2009). Specifically, Herpertz et al. (2001) found increased amygdala activation in BPD patients to negative or threatening stimuli. Similar results were found in another study which used fMRI to examine neural responses to neutral, happy, sad and fearful facial expressions (Donegan et al., 2003). Further, post-scan debriefing revealed that BPD patients had greater difficulty disambiguating neutral faces, or found them threatening, suggesting a threat and/or negativity bias in the assessment of ambiguous stimuli. Guitart-Masip et al. (2009) found a correlation between activity in the left posterior temporal cortex and impulsivity scores in their study of emotional discrimination of faces, in which BPD patients were less accurate in emotional discrimination of negative facial expressions, particularly those emotions related to disgust and fear. The authors found activations in the fusiform gyrus, insular, and left amygdala in all participants, but BPD patients showed higher activations in left medial and inferior temporal areas. These findings are in accordance with the suggestion that the temporal cortex sub-serves emotional discrimination and impulsivity in BPD patients (Nunes et al., 2009).
Whilst the predominant finding in BPD patients is that of decreased prefrontal activation, some studies have found increased activation in frontal and prefrontal areas (Domes et al., 2009) in relation to trauma, abandonment and unresolved conflict which are is often reported in the histories of BPD patients. Specifically, memories of abandonment were associated with an increase in blood flow in bilateral DLPFC and right cuneus, and a decrease in right ACC blood flow (Schmahl et al., 2003). Another study that compared BPD patients and healthy controls in terms of their activation patterns during recall of resolved versus unresolved conflicts (Beblo et al., 2006) found significant bilateral activation of fronto-temporal areas including the insula, amygdala, ACC, and the left posterior cingulate cortex in patients. The authors postulated that the activation of both the amygdala and prefrontal areas in BPD patients may reflect an increased, but insufficient, effortful attempt to control intense emotions during the recollection of unresolved life events.
In summary, neuroimaging studies which have attempted to localise the apparent neuropsychological deficits in BPD have identified both structural and functional changes that involve, in particular, the fronto-limbic network (Domes et al., 2009).
ii. BD
Relative to BPD, a much more substantial body of research has been devoted to the neuropsychology of BD. In recent years, three meta-analyses have been published. Two compared euthymic BD patients (subtype not specified) and healthy controls (Robinson et al., 2006; Torres et al., 2007) across 26 and 39 studies, respectively, whereas the third meta-analysis compared BD I and BD II patients across 11 studies, as well as BD II patients and healthy controls across nine studies (Bora et al., 2011).
Synthesising the findings, there is generally limited evidence for differences between BD and the normal population in terms of premorbid IQ, general intellectual functioning and education. BD patients (BD II and non-specified types) perform poorly compared to healthy controls on all facets of visual and verbal memory including learning and acquisition, spontaneous retrieval (short- and long-delay), and storage in long-term memory (recognition). Further, BD I patients are overall more impaired than BD II patients across all aspects of memory performance. In the domain of executive functions, BD patients (non-specified types) perform poorly compared to healthy controls on indices of cognitive flexibility (such as simultaneous conceptual tracking, set-shifting, utilising error feedback, and verbal generativity), and inhibition/suppression of over-learned responses. BD II patients also perform poorly compared to healthy controls across all areas of executive functioning, with the exceptions of set-shifting and utilising error feedback, for which there is inadequate research. Interestingly, comparing BD I and BD II patients, the former perform less well only on semantic fluency. Lastly, with respect to attention and information processing, BD patients (non-specified types) have a diminished immediate attention span and reduced working memory, sustained attention, and speed of information processing. BD II patients appear to have overall better processing speed and working memory compared to BD I patients, but compared to healthy controls, no significant differences in immediate attention have been found. Further, there is insufficient data on any contrasts between BD II patients and healthy controls on measures of sustained attention.
In terms of neuroanatomical abnormalities, investigators have found decreased prefrontal volumes in BD patients compared to controls (Arnone et al., 2009; Drevets et al., 1997; Sax et al., 1999). Using positron emission tomography (PET), one of these studies (Drevets et al., 1997) further found abnormally decreased activity in the prefrontal cortex ventral to the genu of the corpus callosum in BD patients. Within the prefrontal region, functional neuroimaging studies have also found decreased activation in the ventral prefrontal cortex (VPFC) when displaying affective stimuli (Malhi et al., 2005), and employing tasks of executive functions and inhibitory control (Blumberg et al., 2003). The latter study concluded that a lack of activity in an area of the brain usually involved in the inhibition of response may account for some of the disinhibition in BD.
Studies of other brain structures in BD have also identified abnormalities, but the results have been mixed. Based on a recent mega-analysis of structural MRI in BD (Hallahan et al., 2011), various studies have reported increased as well as reduced amygdala volume, and extensive heterogeneity appears to exist in several other brain regions, including the prefrontal cortex. The authors identified some key reasons for this heterogeneity including age and medication variables. Specifically, they found a differential effect of age on hippocampal volume, with BD patients showing a greater decrease with age compared to healthy controls, whilst patients treated with lithium had larger mean hippocampal and amygdala volumes than patients not treated with lithium, as well as healthy controls.
Hence, as in the case for BPD, these results indicate an underlying fronto-limbic network dysfunctionality in BD. In one study that employed a sustained attention task (Strakowski et al., 2005), BD patients showed significantly more activation in limbic brain regions including the parahippocampus and amygdala, suggesting a baseline limbic over-reactivity that contributes to the emotional lability seen in BD. In extending cognitive tasks to memory and encoding, BD patients have demonstrated blunted rCBF activation in the left DLPFC, and increases in the hippocampus and fusiform/parahippocampal gyri during the encoding stage (Deckersbach et al., 2006).
Based on the findings from these studies, a widely held view is that BD patients experience abnormally elevated activity in subcortical-limbic structures, with comparatively muted activity in prefrontal structures, areas that are associated with automatic and voluntary emotion regulation respectively (Hassel et al., 2008). However, other studies in BD patients suggest that the increased and decreased responses seen in limbic and frontal areas respectively are more reflective of a manic state. In studies where manic BD patients were recruited, results have shown increased activation in the left amygdala and decreased bilateral activation in the OFC in response to facial expressions (Altshuler et al., 2005); decreased activity in the VLPFC in response to both emotional and cognitive tasks (Chen et al., 2011); and increased amygdala activation during tasks of inhibitory control (Kaladjian et al., 2009a; Malhi et al., 2004). However, in a recent meta-analysis of fMRI studies in BD, decreased activity in the VLPFC was not observed in euthymic patients (Chen et al., 2011), and in a longitudinal paradigm where manic patients were reassessed during remission (Kaladjian et al., 2009a), the authors found that the left amygdala showed a time-dependent change in activation that was significantly different between BD patients and healthy subjects. Analyses revealed that this difference arose from the patient group, in which amygdala activation was decreased between mania and subsequent remission. In another study by the same authors which utilised the same task of inhibitory control (Kaladjian et al., 2009b), euthymic BD patients exhibited significantly decreased activation in the left fronto-polar cortex and bilateral dorsal amygdala compared with healthy subjects. Lack of amygdala activation has also been reported in other studies of euthymic BD I patients in response to emotional stimuli (Malhi et al., 2005).
When contrasting BD patients in manic and depressed mood states, further differences have been found in response to emotional stimuli in terms of a ‘mood-congruent bias’. In other words, depressed BD patients have demonstrated a bias towards negatively valenced stimuli (Lyon et al., 1999), whilst manic patients have demonstrated complementary behavioural biases toward positively valenced stimuli (Murphy et al., 1999) or a negative bias towards negative stimuli. For example, manic patients have shown a selective deficit in the recognition of fear and disgust (Lembke and Ketter, 2002), and in response to sad facial expressions, attenuated activation in the subgenual ACC and bilateral amygdala, with increased activation in the posterior cingulate and posterior insula (Lennox et al., 2004). In a single case design study where an ultra-rapid cycling BD patient was tested (George et al., 1998), the patient tended to interpret neutral faces as sad, and sad faces as very sad, when depressed, suggesting a state-dependent bias. However, other studies have found mood-incongruent effects, for example, one study (Chen et al., 2006) found that manic BD patients displayed overactivity in the fusiform gyrus in response to sad faces, whereas bipolar depressed patients displayed overactivity in fronto-striato-thalamic systems in response to happy faces. In euthymia, BD patients have been shown to be more emotionally reactive to positive stimuli, such as happy faces, but not to negative stimuli (Johnson et al., 2007).
Summary and conclusions
Tables 1, 2 and 3 respectively, provide a summary of the behavioural and childhood factors, neuropsychological profiles, and neuroimaging findings associated with BD and BPD outlined in this review. Whilst there are many similarities between the two disorders within each domain, there are also differences that appear to distinguish the two groups. However, given that BD and BPD patients have not, to date, been directly compared in the one study using the same design that encompasses the full breadth of measures pertaining to emotional dysregulation, impulsivity, childhood trauma, maladaptive self-schemas, neuropsychological functions and neuroimaging, the true extent of these differences, and whether they are meaningful, remains a matter of debate.
Summary of behavioural and childhood factors in bipolar disorder and borderline personality disorder.
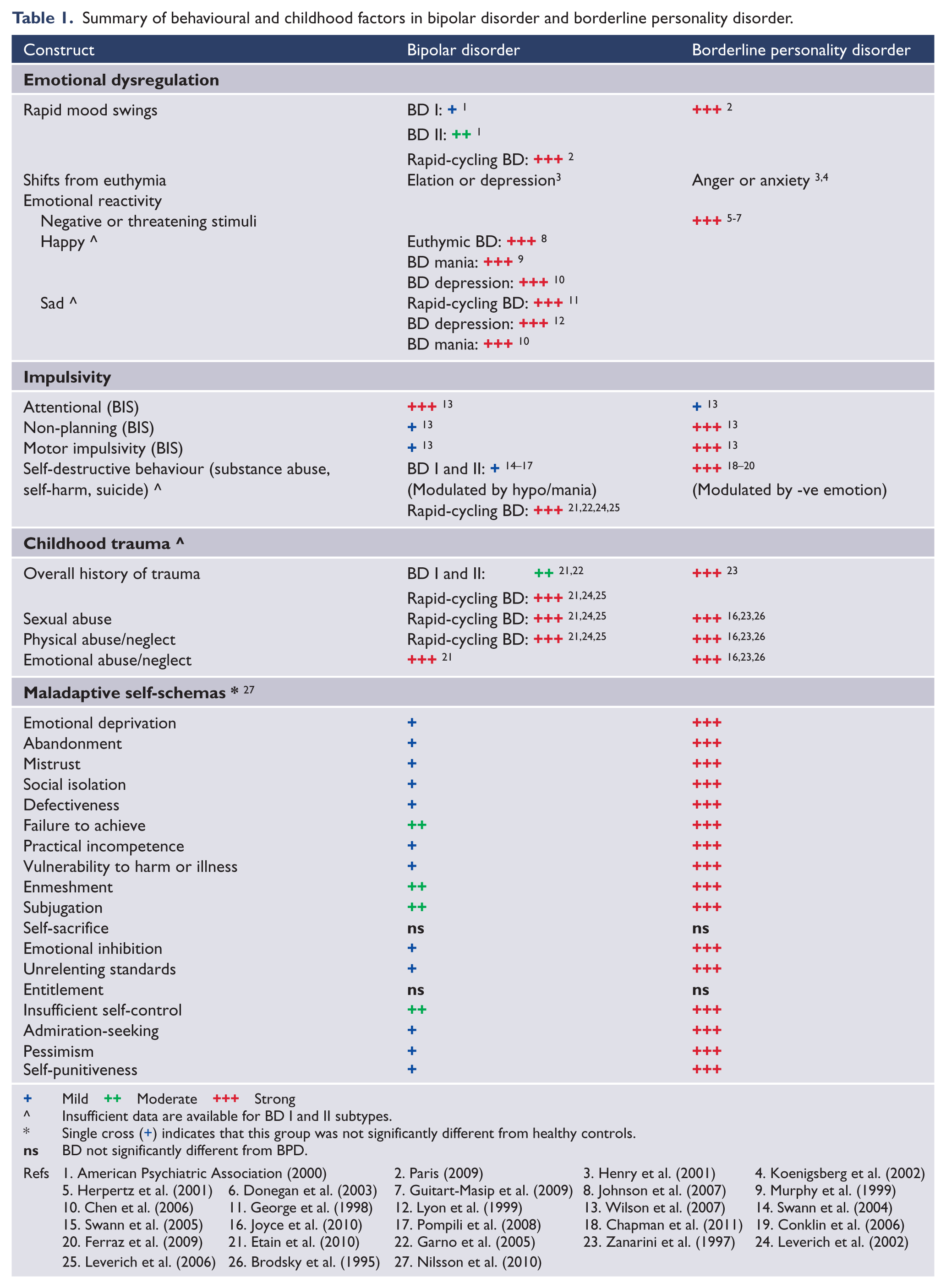
Mild ++ Moderate +++ Strong
Insufficient data are available for BD I and II subtypes.
Single cross (+) indicates that this group was not significantly different from healthy controls.
BD not significantly different from BPD.
Refs 1. American Psychiatric Association (2000) 2. Paris (2009) 3. Henry et al. (2001) 4. Koenigsberg et al. (2002)
5. Herpertz et al. (2001) 6. Donegan et al. (2003) 7. Guitart-Masip et al. (2009) 8. Johnson et al. (2007) 9. Murphy et al. (1999)
10. Chen et al. (2006) 11. George et al. (1998) 12. Lyon et al. (1999) 13. Wilson et al. (2007) 14. Swann et al. (2004)
15. Swann et al. (2005) 16. Joyce et al. (2010) 17. Pompili et al. (2008) 18. Chapman et al. (2011) 19. Conklin et al. (2006)
20. Ferraz et al. (2009) 21. Etain et al. (2010) 22. Garno et al. (2005) 23. Zanarini et al. (1997) 24. Leverich et al. (2002)
25. Leverich et al. (2006) 26. Brodsky et al. (1995) 27. Nilsson et al. (2010)
Summary of results from meta-analyses of neuropsychological findings in patients with bipolar disorder and borderline personality disorder versus healthy controls.
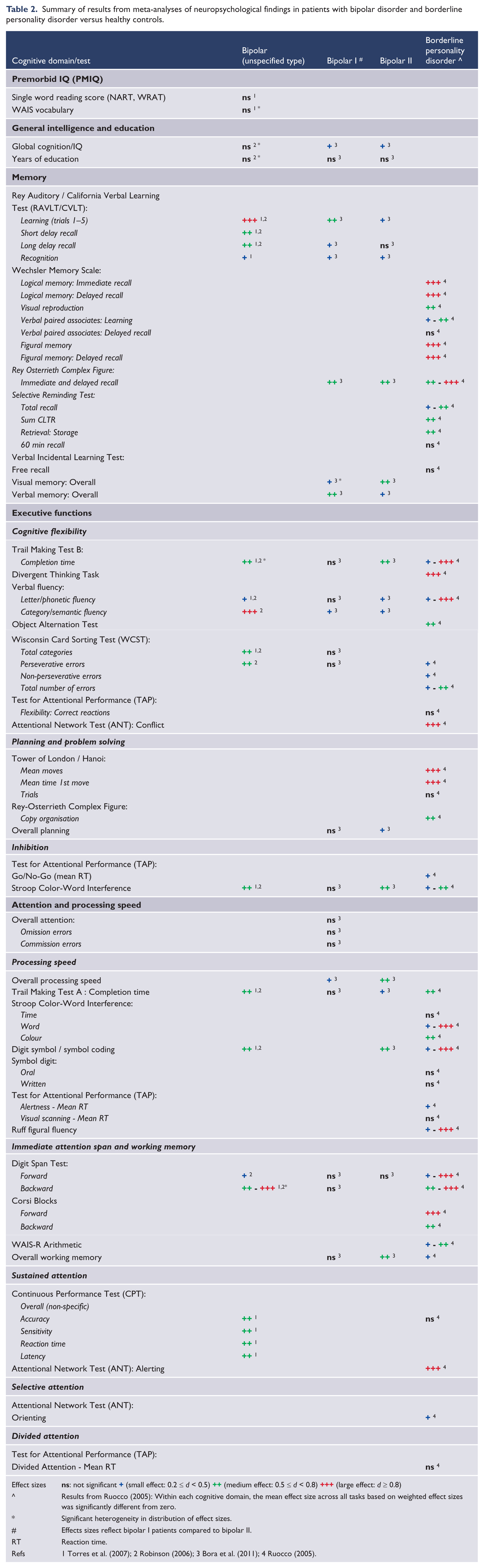
Effect sizes
Results from Ruocco (2005): Within each cognitive domain, the mean effect size across all tasks based on weighted effect sizes was significantly different from zero.
Significant heterogeneity in distribution of effect sizes.
Effects sizes reflect bipolar I patients compared to bipolar II.
RT Reaction time.
Refs 1 Torres et al. (2007); 2 Robinson (2006); 3 Bora et al. (2011); 4 Ruocco (2005).
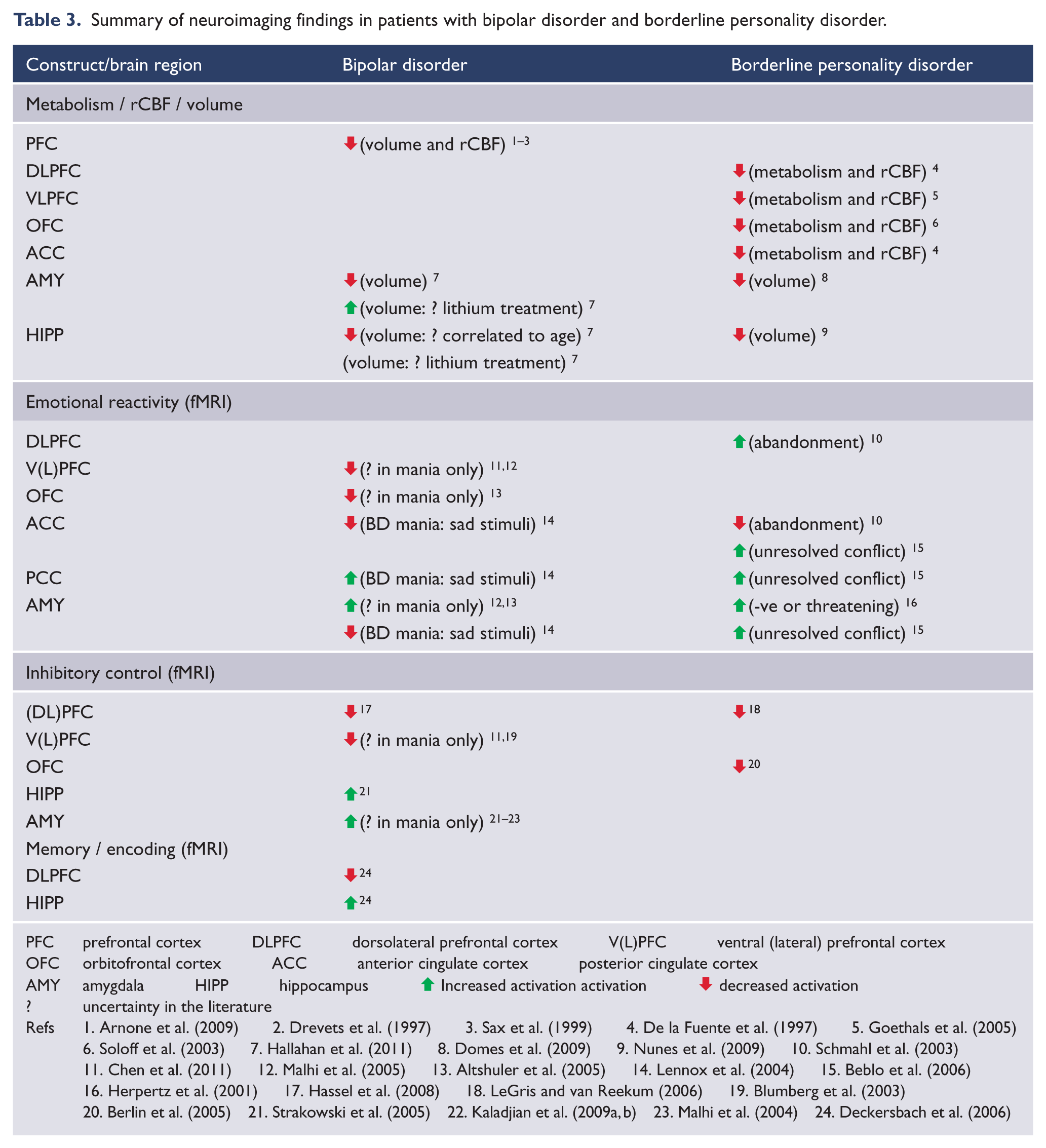
Summary of neuroimaging findings in patients with bipolar disorder and borderline personality disorder.
PFC prefrontal cortex DLPFC dorsolateral prefrontal cortex V(L)PFC ventral (lateral) prefrontal cortex
OFC orbitofrontal cortex ACC anterior cingulate cortex posterior cingulate cortex
AMY amygdala HIPP hippocampus ⬆ Increased activation activation ⬇ decreased activation
? uncertainty in the literature
Refs 1. Arnone et al. (2009) 2. Drevets et al. (1997) 3. Sax et al. (1999) 4. De la Fuente et al. (1997) 5. Goethals et al. (2005)
6. Soloff et al. (2003) 7. Hallahan et al. (2011) 8. Domes et al. (2009) 9. Nunes et al. (2009) 10. Schmahl et al. (2003)
11. Chen et al. (2011) 12. Malhi et al. (2005) 13. Altshuler et al. (2005) 14. Lennox et al. (2004) 15. Beblo et al. (2006)
16. Herpertz et al. (2001) 17. Hassel et al. (2008) 18. LeGris and van Reekum (2006) 19. Blumberg et al. (2003)
20. Berlin et al. (2005) 21. Strakowski et al. (2005) 22. Kaladjian et al. (2009a, b) 23. Malhi et al. (2004) 24. Deckersbach et al. (2006)
Summarising the findings, Table 1 indicates that the greatest overlap is between BPD and rapid-cycling BD. This is not surprising given that compared to ‘typical’ BD, both BPD and rapid-cycling BD patients exhibit more frequent shifts between mood states, a higher rate of self-destructive behaviour that is modulated predominantly by negative emotion in BPD (as opposed to hypo/mania in BD), and a more severe history of childhood trauma that includes sexual abuse and physical neglect/abuse (as opposed to a preferential prevalence of emotional abuse/neglect in BD). In addition, BPD patients tend to be selectively reactive to negative or threatening stimuli, especially those that invoke sentiments of abandonment or rejection, whilst BD patients can be equally reactive to negative/sad and positive stimuli; and BPD patients exhibit a higher degree of maladaptive self-schemas which theoretically fuel emotional and impulsive behavioural responses to negative or threatening situations. Interestingly, one study to date that compared BD and BPD patients on a measure of impulsivity also found that BD II was uniquely associated with attentional impulsiveness, whereas BPD was associated with a greater level of motor and non-planning impulsiveness.
Tables 2 and 3 report on the neurobiological underpinnings of BD and BPD separately. In terms of premorbid IQ, general intellectual functioning and education, there is limited evidence of any significant differences between BD and the normal population. There is insufficient data for these indices in BPD, but again, a significant difference is unlikely and would accord with clinical experience. In terms of their neuropsychological profiles, both BD (unspecified subtypes) and BPD patients are impaired relative to controls on all facets of visual and verbal memory, a range of executive functions, and aspects of attention and processing speed. BD II patients also perform poorly in comparison to healthy controls across all of these areas with the exceptions of set-shifting, utilising error feedback, and sustained attention, for which there is insufficient data. BPD patients are also compromised relative to healthy controls in the areas of selective attention, planning and problem solving, with inadequate data available for BD in these areas. Finally, BD I patients have been shown to be generally more impaired than BD II patients in all aspects of memory performance, semantic fluency, working memory, and overall processing speed. On the basis of these results, there appears to be a less apparent distinction between BD and BPD, although again, these results are inconclusive on the grounds that there are some substantial differences in cognitive measures that have been administered in studies of BD and BPD. For example, in the domain of memory, the Wechsler Memory Scale has been the primary measure of visual and verbal processes administered to BPD patients, whereas other visual and verbal tests of learning, short-delay and long-delay recall including the Rey Osterrieth Complex Figure Test and Rey Auditory Verbal Learning Test have been administered in studies of BD.
In linking neuropsychological dysfunctions to brain structural and functional regions, Table 3 shows that both BD and BPD are characterised by reduced metabolism, rCBF, and/or volume in frontal regions that correspond to impaired inhibitory control in both disorders, and in the case of BD, this extends to other cognitive functions including memory and encoding. Reduced amygdala and hippocampal volumes have also been identified in BD and BPD, although there is uncertainty around whether a smaller hippocampal volume in BD is a factor of age, and if increased volumes of the amygdala and hippocampi are related to lithium treatment. The heterogeneity of neuroanatomical abnormalities in BD could also partially relate to the lack of specific data on differences in brain structures between subtypes of BD including BD I, BD II and rapid-cycling BD. In any case, alterations in limbic structures, in conjunction with deficits in frontal regions, account for compromised fronto-limbic networks in both BD and BPD that contribute to problems in emotional regulation and inhibitory control. Table 3 illustrates the various degrees of increased and decreased activation in frontal and limbic regions for both BD and BPD patients, and the directions of activation appear to be a function of the valences of stimuli presented, as well as mood state in BD. For example, there are inconsistencies in the BD literature around whether or not increased or decreased activations in frontal- limbic areas are a function of mania, and whether the functional responses to the stimuli are mood-congruent or incongruent. Moreover, the range and valences of stimuli that have been presented to BD patients are very different to those that have been specifically designed for BPD patients, and so it is difficult to draw direct comparisons between the two groups.
In conclusion, these findings are far from definitive, largely because of insufficient data gathered across the areas of emotional dysregulation, impulsivity, childhood factors, and neurobiological indices for each of the populations of BD (I and II), rapid-cycling BD, and BPD; the absence of direct comparisons between BD and BPD; and other considerations which render the presentation and diagnosis of BD II unclear (Malhi et al., 2010). As previously mentioned, ratings of hypomania are often unreliable (Akiskal, 1996; Dunner and Tay, 1993), as assessment of hypomania often depends on accurate retrospective reporting by patients as to how long specific mood states have lasted, and whether or not they have been stable over a four-day period (Malhi et al., 2010). Furthermore, it is not always clear whether the anger and anxiety experienced by BPD patients are distinct in quality from the subjective moods experienced in an irritable or dysphoric manic state (Mackinnon and Pies, 2006).
A primary question that remains largely unanswered concerns the make-up of rapid-cycling BD, whether this condition is indeed a form of BD, or potentially represents BD individuals with co-morbid BPD traits. Based on empirical data, Tyrer (2009) has argued that BPD is more appropriately classified as a condition of recurrent, rapidly changing unstable mood and behaviour, as opposed to a personality disorder. In keeping with this view, it can be seen in Table 1 in particular, that the most common areas of overlap between the two disorders occur between BPD and rapid-cycling BD patients. Therefore, one perspective is that the resemblance of BPD to rapid-cycling BD is largely a function of a common underlying mood disorder with similar alterations in HPA functioning and co-morbid personality dimensions that have been shaped through early life events. The research by Nilsson et al. (2010) specifically targeted BD and BPD individuals who did not concurrently fulfil criteria for the alternate diagnosis, and the distinctions between the two groups in regards to self-schemas were dramatically marked. In the same vein, future research needs to focus on directly comparing BD and BPD individuals in one single comprehensive study that incorporates the full range of behavioural, childhood and neurobiological measures to determine if BPD is part of the bipolar spectrum. As there are significant health care and societal costs associated with the treatment of BPD in particular (Bender et al., 2001; van Asselt et al., 2007), with such costs substantially increased when misdiagnosis between the two illnesses occur (John and Sharma, 2009), this type of research would aid in diagnostic clarification of BD and BPD, which has important implications for current practice.
Footnotes
Acknowledgements
The authors would like to acknowledge Ms Kimberly Tufrey who assisted in the earlier drafts of this manuscript.
Funding
GSM has received grant/research support from the NHMRC, NSW Health, Astra Zeneca, Eli Lilly, Organon, Pfizer, Servier and Wyeth. The authors also acknowledge the NHMRC Program Grant (510135) for essential financial support.
Declaration of interest
GSM has been a speaker for Astra Zeneca, Eli Lilly, Janssen Cilag, Lundbeck, Pfizer, Ranbaxy, Servier and Wyeth, and a consultant for Astra Zeneca, Eli Lilly, Janssen Cilag, Lundbeck and Servier. RJP has received support for travel from Wyeth and Honoraria from Sanofi-Aventis.
