Abstract
Background
Zinc deficiency is a global concern, particularly in low-income countries, but also among vulnerable groups in Western countries, such as children. Diagnosing mild or moderate zinc deficiency is however challenging because of nonspecific symptoms and due to circulating zinc showing only subtle changes, requiring high accuracy in measurement. Challenges to accurate measurement include variations from choice of analytical instrument, analysis performance, and preanalytical factors such as choice of sample matrices and delayed blood sample processing. This study aimed to examine the stability of zinc in plasma and serum, measured by the recommended inductively coupled plasma optical emission spectrometry (ICP-OES) method and a direct colourimetric assay on the fully automated Roche Cobas c702 analyzer.
Methods
A total of 245 whole blood samples were stored at room temperature for 0–8 h after blood sampling, then centrifuged for 10 min at 2000 g (serum) or 5 min at 2650 g (plasma), frozen at −20°C, and analysed, respectively, on ICP-OES and Cobas, the latter with the colourimetric kit from Sentinel diagnostics.
Results
Serum zinc concentrations measured on Cobas and ICP-OES showed no statistically significant change up to 6 h and never exceeded acceptable limits. Plasma zinc concentrations increased steadily over time, exceeding acceptable limits after 6 h. There were statistically significant differences between zinc measurements on ICP-OES and Cobas in both serum and plasma.
Conclusions
Zinc is stable for at least 8 h in serum and up to 6 h in plasma when measured by either Sentinel diagnostic colourimetric method on Cobas or ICP-OES.
Introduction
Zinc (Zn) is a vital micronutrient and as such is the second most abundant of the essential trace elements in the human body, second only to iron. 1 Its abundance is evident through its widespread involvement in different tissues and biological functions, reflected in the fact that an estimated 10% of the human genome encodes for Zn-relevant proteins.
While toxic at higher concentrations (>50 mg/day), the main health consideration is securing adequate intake and preventing deficiency. As a type II nutrient, Zn is essential to general metabolism, and Zn deficiency therefore affects critical biological functions such as the immune system, gastrointestinal function, and growth in general.2,3
Avoiding Zn deficiency is therefore a major international concern. Deficiency is prevalent in both low- and middle-income countries, leading to increasing susceptibility to, and severity of, infections as well as impaired development and growth, and as such constitutes a serious public health problem. Prevalence has also been found high in specific vulnerable patient groups living in high-income Western countries, especially newborn and young children, as well as vegetarians and patients suffering from metabolic and/or gastrointestinal diseases.2,4
Unfortunately, Zn deficiency is hard to diagnose. Being a type II nutrient, Zn deficiency quickly halts growth but generally results in a few and nonspecific symptoms without characteristic clinical signs because of its ubiquitous and widespread involvement in different tissues and functions.2,3,5 Furthermore, these tissues mostly retain their pre-deficiency Zn concentrations, evident by the narrow homoeostatic concentration range of circulating blood zinc, resulting in only discreet changes in circulating Zn concentrations even in chronic moderate depletion.6–8 Hence, high analytical accuracy and precision are necessary to detect these small, but clinically significant changes.
Analytical accuracy and precision depend on several factors, some of which are of special importance when measuring Zn: While both serum and plasma have been deemed acceptable materials for Zn measurement, the literature is still undecided about which matrix is superior. 6 Likewise, how long centrifugation and separation acceptably can be delayed lacks consensus or recommendations. 6 Studies mostly using a limited range of ‘gold standard’ analytical instruments have shown Zn to increase with delay of centrifugation in both serum and plasma, but often only examining few and largely spaced time intervals, making it difficult to apply these results to specific local laboratory requirements.6,14–16
Atomic absorption spectrometry (AAS) or inductively coupled plasma (ICP) instruments are recommended for Zn assessment.2,9 Less specialized analytical instruments also allow for Zn measurement using third party reagent kits, and these might be available to more laboratories than the former, provided that the precision and correctness are acceptable. 9
Therefore, we examined the impact of delayed centrifugation on serum and plasma samples when measured on both ICP-OES and with a direct colourimetric assay on the Roche Cobas c702 clinical chemistry analyzer.
Methods
Analysis
Zn was measured using two different instruments.
Inductively coupled plasma optical emission spectrometry (ICP-OES) (ICAP-7000, Thermo Fisher Scientific, Waltham, MA) was calibrated using calibrators traceable to National Institute of Standards and Technology (NIST) reference materials. The method used was accredited under ISO 15189 and employed quality control using internal standards (Trace elements serum controls, UTAK, Valencia, Santa Clarita, CA, USA) and external controls (TEQAS, UK NEQAS, Sheffield, UK).
Limit of detection was 0.4 μmol/L. Coefficient of variation (CV%) was within local analytical performance specifications of 4.7%, evaluated at three concentrations with CV% equal to 4.7% at 4.4 μmol/L, 3.1% at 11.0 μmol/L, and 2.8% at 39.7 μmol/L. Likewise, bias were within the chosen limit of 10%, evaluated by measuring a total of 22 external controls throughout a 1-year period resulting in an average bias of 3.6%. The ICP-OES was equipped with standard glass concentric nebulizer, glass cyclonic type spray chamber, and Cetac Autosampler ESI SC4DX with Fast Valve technology. Qtegra software (version 2.4.1800.96 [64 bit]) was used. Samples were prepared by 1:11 dilution in buffer containing 0.5% HNO3, 0.1% Triton X-100, and 5 mg/L Yttrium, the latter as internal standard. Zn was measured by using a spectrometer at a wavelength of 202.5 nm, and concentrations were determined by using standard addition calibration curves. To ensure accuracy, high and low calibrators were analysed at the beginning of each analytical run, as well as at every 20 samples (alternating between high and low). Furthermore, high- and low-quality controls were analysed both at the beginning and at the end of every analytical run. Validation of results was done using the multi-rule Shewhart principle. 10
Zn measurement was also performed on the Roche Cobas c702 modular analyzer instrument (Roche Diagnostics, Rotkreuz, Switzerland) using a third party Zn assay by Sentinel Diagnostics (reagent: 17640H; Sentinel Diagnostics, Milano, Italy), a direct colourimetric method without a deproteinization step for measurement in serum or plasma samples. The limit of detection was 0.8 μmol/L. Coefficient of variation (CV%) was within local analytical performance specifications of 6.4%, evaluated at two concentrations with CV% equal to 5.5% at 14.3 μmol/L and 4.2 % at 21.4 μmol/L. Likewise, bias was within the chosen limit of 10%. Analyses were performed according to the specifications and recommendations of the supplier. The supplier announces no specific difference between serum and plasma, conversely stating similar stability and reference range. The assay is CE-marked and was calibrated using calibrators traceable to NIST reference materials, as well as employed quality control using internal controls (SERO AS, Billingstad, Norway) and external controls (Labquality Helsinki, Finland).
Study design
Seven tubes were drawn from each of 35 healthy volunteers, 4 mL serum tubes (BD Vacutainer Serum tubes, PET tubes with clot activator, cat. no. 369032) from 20 volunteers, and 3 mL plasma tubes (BD Vacutainer lithium-heparin tubes, PET tubes, cat. no. 368884) from another 15 volunteers. All blood draws were performed by venipuncture in accordance with CLSI recommendations (GP41 7th ed.) 25 using either a Greiner Bio-One Vacuette Quickshield Complete Plus 21G x1″ or Vacuette Safety Blood Collection Set. Serum samples were collected at the ICP-OES laboratory, whereas plasma samples were collected at the Cobas laboratory.
No volunteers supplied both plasma and serum samples concurrently, and therefore no direct comparisons between serum and plasma Zn concentrations were made in this study.
All participants provided their informed consent prior to enrolment in the study. All data obtained were completely anonymized and hence, according to local laws, permissions from the Danish National Committee on Health Research Ethics were not required. 11 After collection, the seven blood samples were placed at ambient room temperature (18°C–24°C) for 0.5, 1, 2, 3, 4, 6, and 8 h, respectively, before being centrifuged, aliquoted, and frozen at −20°C until analysis. Serum samples were centrifuged for 10 min at 2000 g and aliquoted into two polystyrene tubes (Sarsted, order number 55.475), whereas plasma samples were centrifuged for 5 min at 2650 g and aliquoted into two Secondary Tube Multiplex PET (Vacuette, order number 459011), according to the local routine procedures. Other studies show Zn to be unaffected by such freeze–thaw cycles. 27
Samples stored for 0.5 h simulated normal handling of serum samples, allowing 30 min of coagulation time prior to centrifugation, and concentrations measured in those samples were therefore considered baseline values. Plasma samples were treated equally for comparison. Haemolysis was evaluated visually and if suspected was confirmed by Haemoglobin (H) Index measurement on Roche Cobas c702. Analysis on ICP-OES was in two replications and performed in multiple batches. On Cobas c702, analysis was performed in a single batch without replications.
Statistical analysis
Mean, absolute, and % difference from baseline values at each sequential time point were calculated. Statistical analysis was performed using Analyse-it (Analyse-it Software Ltd, Leeds, UK) and Prism 9 (GraphPad Software, Inc). Outlier analysis was done using Grubbs’ test. Normality was assessed by Anderson–Darling, Shapiro–Wilk as well as Kolmogorov–Smirnov tests. One-way ANOVA with multiple comparison as well as Dunnett’s post-hoc test was performed to analyse for statistically significant differences between baseline values and sequential values. P < .05 was considered statistically significant.
Differences between ICP-OES and Cobas measurements at each individual time point were evaluated by Wilcoxon signed-rank test as well as by Passing–Bablok regression and Bland–Altman plot (not shown).
Differences from baseline to sequential time points and between ICP-OES and Cobas were calculated for serum and plasma sample measurements, respectively.
Changes were assessed by Acceptable Change Limits (ACLs) (ACL = 1.96 × √2 × CVa), using Westgard’s desirable specification for imprecision for serum and plasma corresponding to an ACL of 13.0% for serum and 15.2% for plasma. 12
Results
Zn concentrations in serum measured on ICP-OES and Cobas did not change significantly over time from baseline, with the single exception of measurement at 8 h on ICP-OES (see Figure 1(a) and (c)). Likewise, the relative change of serum Zn concentrations stayed well within the ACL with a maximum mean %-change of 6.9 (3.0 to 10.8) % measured at 8 h on ICP-OES (see Figure 1(b) and (d)). All serum values from one volunteer were identified as outliers by outlier analysis and therefore excluded. Data was found to be normally distributed. Measured Zn concentrations at baseline (30 min) and following delayed time points before centrifugation. (a, b) Serum samples measured on ICP-OES. (c, d) Serum samples measured on Cobas. (e, f) Plasma samples measured on ICP-OES. (g, h) Plasma samples measured on Cobas. Lines indicate the Acceptable Change Limit (ACL). Results of one-way ANOVA with Dunnett’s post-hoc test are indicated with ns = not significant, * = p < 0.05, ** = p < 0.01, *** = p < 0.001, and **** = p < 0.0001. Time points are shown categorically and not relative to elapsed time.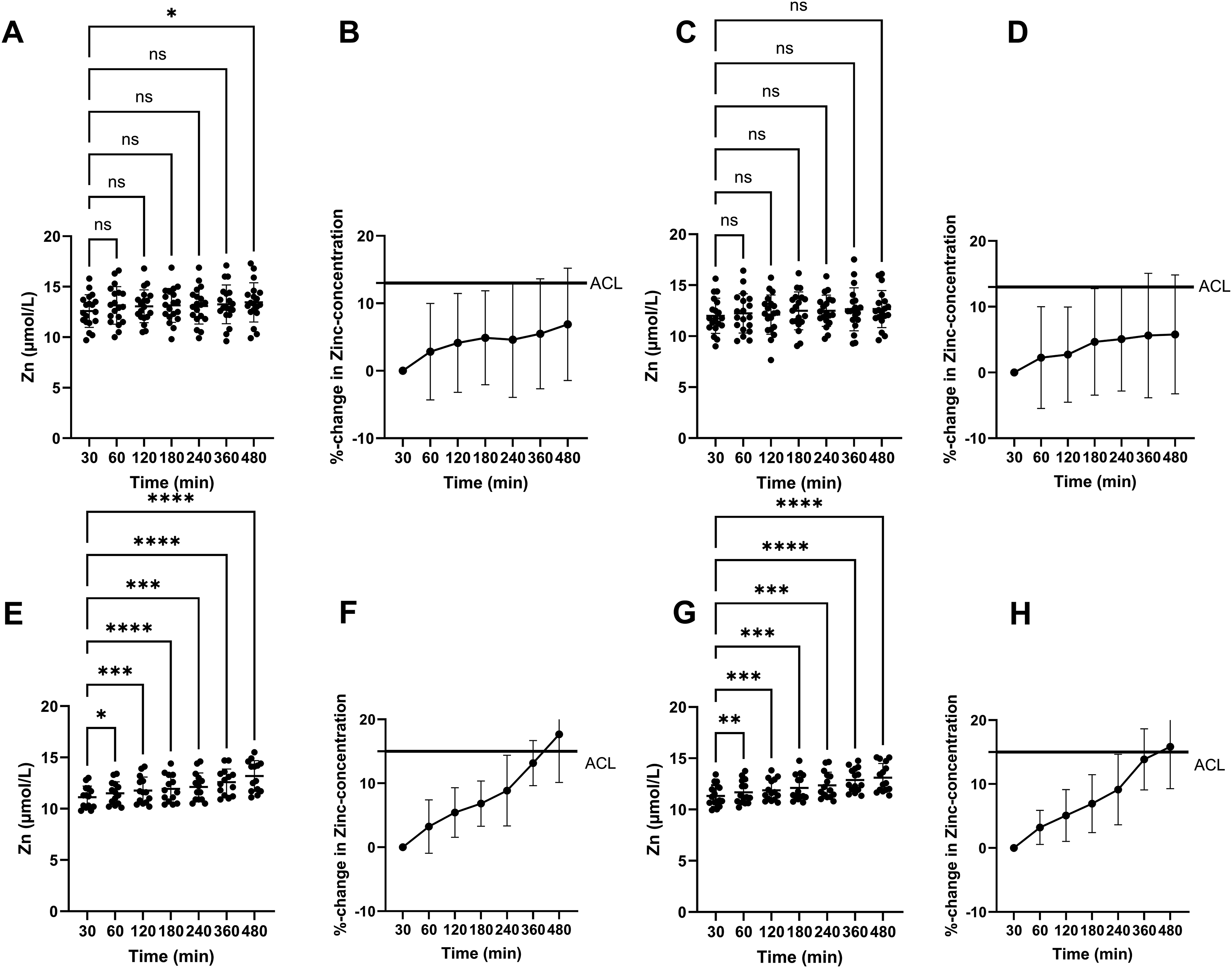
Zn concentrations in plasma, however, increased steadily over time on both analytical instruments, showing statistically significant elevated values at all time points compared to baseline and exceeding the ACL at 8 h (see Figure 1(e)–(h)).
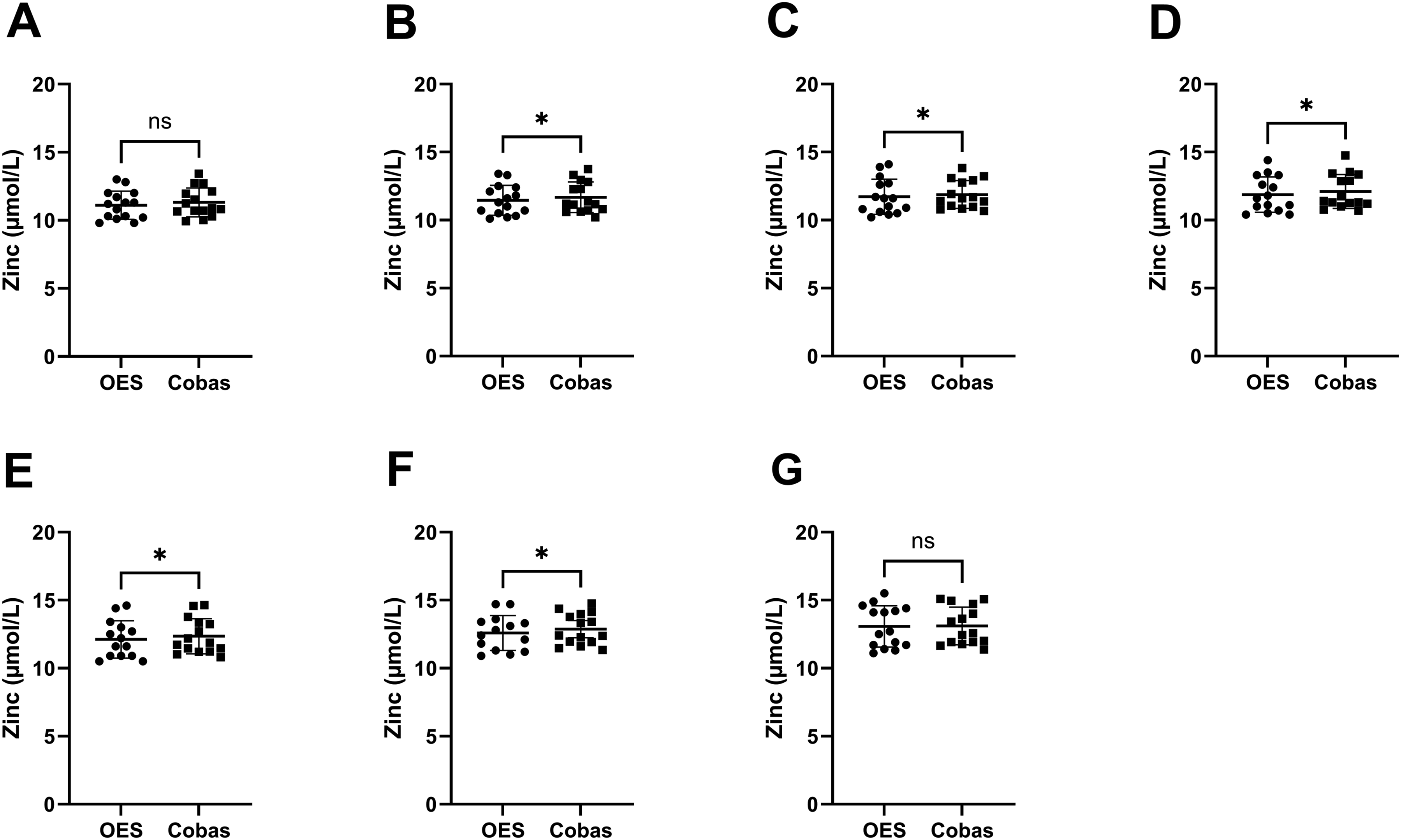
Statistical analysis found serum and plasma Zn measurements differed significantly between instruments at all time points, except for plasma at baseline and 8 h (see Figure 2 for serum, and Figure 3 for plasma). Measurements of Zn concentrations in serum samples by ICP-OES versus Cobas at different time points of delayed centrifugation. (a) Baseline (30 min). (b) 60 min. (c) 120 min. (d) 180 min. (e) 240 min. (f) 360 min. (g) 480 min. Paired, non-parametric t-test Wilcoxon signed rank test with a significance level α = 0.05 was used, and results are indicated with * = p < 0.05, ** = p < 0.01, *** = p < 0.001, and **** = p < 0.0001. Measurements of Zn concentrations in plasma samples by ICP-OES versus Cobas at different time points of delayed centrifugation. (a) Baseline (30 min). (b) 60 min. (c) 120 min. (d) 180 min. (e) 240 min. (f) 360 min. (g) 480 min. Paired, non-parametric t-test Wilcoxon signed rank test with a significance level α = 0.05 was used, and results are indicated with * = p < 0.05, ** = p < 0.01, *** = p < 0.001, and **** = p < 0.0001.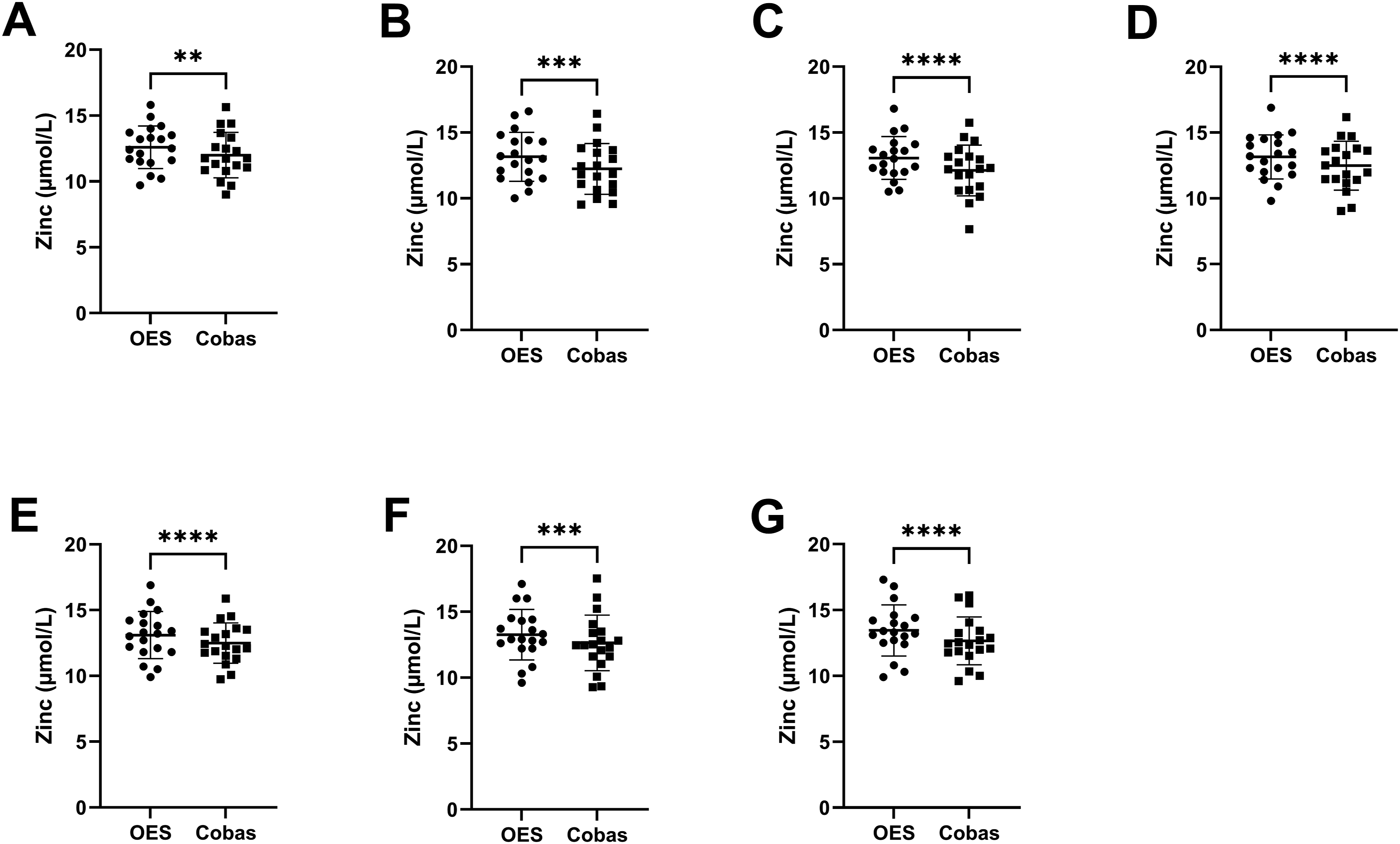
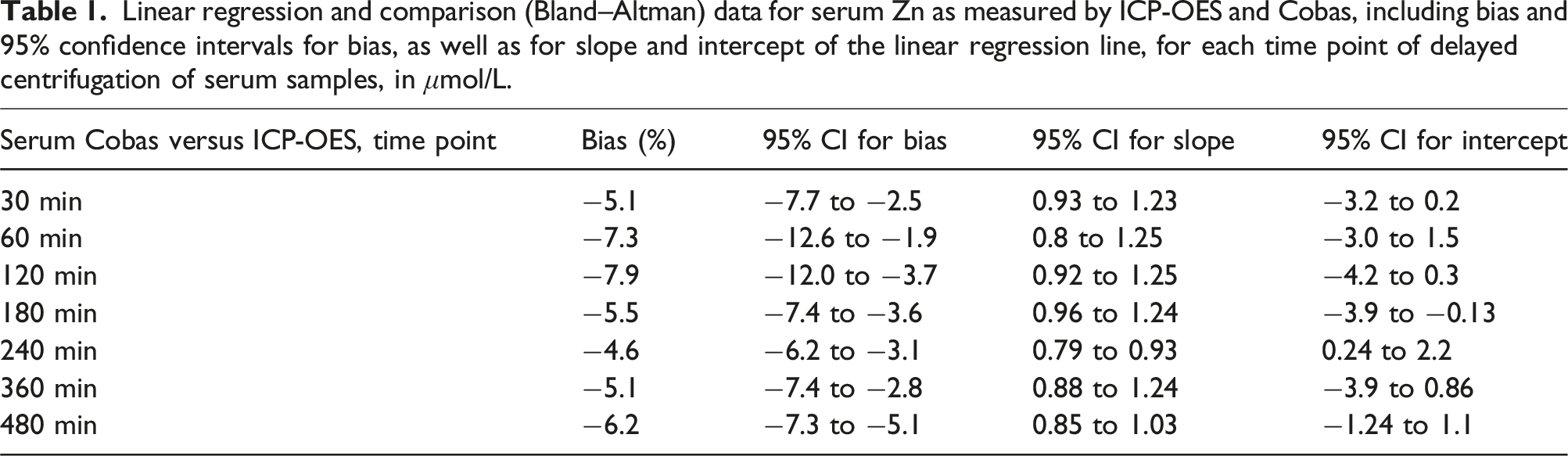
Linear regression and comparison (Bland–Altman) data for serum Zn as measured by ICP-OES and Cobas, including bias and 95% confidence intervals for bias, as well as for slope and intercept of the linear regression line, for each time point of delayed centrifugation of serum samples, in μmol/L.
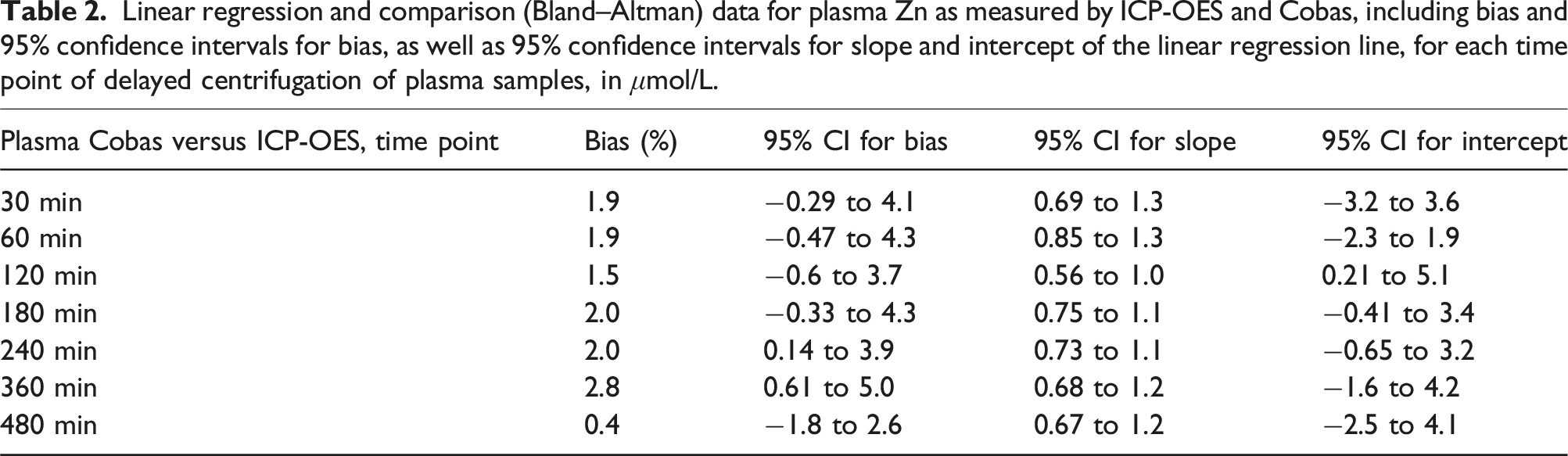
Linear regression and comparison (Bland–Altman) data for plasma Zn as measured by ICP-OES and Cobas, including bias and 95% confidence intervals for bias, as well as 95% confidence intervals for slope and intercept of the linear regression line, for each time point of delayed centrifugation of plasma samples, in μmol/L.
Discussion
We investigated the impact of delayed centrifugation on serum and plasma samples, respectively, when measured by two different assays, the ‘gold standard’ analysis ICP-OES and a third-party assay performed on a fully automated clinical chemistry analyzer,2,9 and found that serum samples were stable for 8 h, while plasma samples exceeded the allowable performance limits after 6 h. Also, we found statistically significant differences between instruments in both matrices, throughout the time period studied including at baseline.
Stability of analytes is an important preanalytical issue, especially for samples taken outside the hospital, for example, at general practitioners. It is therefore mandatory to perform stability studies to elicit this, not only to fulfil the ISO15189 demands but also to ensure patient safety. 13 Unfortunately, there is no firm consensus or recommendation on delayed centrifugation and separation of samples for Zn analyses. Our data shows that uncentrifuged serum samples incubated at room temperature remained stable for at least 8 hours, whether measured by using the ICP-OES or Cobas assay. This allows serum samples for Zn measurements to be drawn and transported from general practitioners, using standard stability and transport procedures similar to most other biochemical parameters. In contrast, plasma did not exhibit the same stability as serum, showing a steady increase over time and exceeding the ACL after 6 hours. Since samples from primary care are routinely kept at 20°C until and during transport to the laboratory, this implicates that the allowable transport time for plasma Zn cannot exceed 6 hours.
Other studies have also shown a significant increase of plasma Zn over time in uncentrifuged plasma samples incubated at room temperature and measured on ICP-MS.14–16 In contrast with our findings, a study using a different third party colourimetric Zn assay on a Roche Cobas c502 analyzer found that both serum and plasma Zn concentrations increased rapidly over time, and even that serum increased more than plasma. This could however be due to differences in study design, as they used a different colourimetric Zn assay (LT‐SYS in vitro diagnostic Zn assay) and did not include measurement by any ‘gold standard’ assay. Furthermore, they used gel tubes for both serum and plasma, whereas we avoided those due to local experiments leading us to suspect contamination of Zn from gel tubes. 1
Another study also found significant elevation in both serum and plasma Zn measurements by using ICP-OES at 4 h, albeit deviating most in plasma. 6 It should be noted that this study differed considerably from ours, in that their samples only were kept at room temperature for 1 h and then 3 h at 4°C. Furthermore, they examined EDTA plasma, whereas we used lithium-heparin plasma.
Albeit not consistently, Zn concentrations have been found to be slightly higher in serum than in plasma samples. 6 Other studies have discussed hypothetical mechanisms leading to difference in Zn concentrations in plasma and serum, suggesting such factors as increased cellular Zn release into serum, perhaps especially from platelets,28,29 loss of plasma proteins in serum, and potential interference from tube additives in plasma tubes. While our study cannot directly compare serum and plasma Zn values, one could hypothesize that factors like those mentioned caused elevated baseline serum Zn values with an analogous but delayed increase occurring in plasma samples, making the observed relative difference larger in plasma than in serum samples.
Our data showed statistically significant differences between the two instruments in both matrices when tested using Wilcoxon signed-rank test at nearly all time points of centrifugation delay (see Figures 2 and 3), as well as highly variable bias (see Tables 1 and 2) evaluated by Passing–Bablok regression. The difference including bias was most pronounced in serum samples, whereas the differences observed in plasma samples are unlikely to have clinical significance, especially when considering that diurnal variation of Zn can reach up to 20%. 17 Observed mean biases of plasma samples were all within desirable bias limits based on biological variation as suggested by EFLM, but the confidence intervals of mean bias were not. Mean bias of serum samples were not within even minimum bias limits. 26
Of note, studies of different colourimetric Zn assays have generally reported poor agreement with ‘gold standard’ methods, for example, FAAS and ICP-MS.9,18,19 Similar to us, these studies rarely observed constant or proportional differences of statistical significance, one study attributing this to low spread of their Zn data. 18 The poor agreement reported in previous studies therefore stemmed mainly from discordant zinc status classifications, particularly in detecting hypozincemia.9,18,19 Given that no Zn values in our study were below the reference range, we could not meaningfully evaluate the classification agreement between Cobas and ICP-OES when assessing zinc status.
A few studies have described the Sentinel Diagnostics assay (reagent: 17640H; Sentinel Diagnostics, Milano, Italy),20–22 and even fewer specifically using a Cobas 8000 analyzer23,24; to our knowledge, this is the first study describing stability on this assay alongside a ‘gold standard’ assay, here ICP-OES.
A limitation of our study is the use of healthy volunteers, which results in a very low variety in Zn values, mainly middle to high within the reference range. This is unfortunate when the primary indication for Zn analysis is to rule out deficiency, and the stability dynamics at low concentrations therefore are of major interest. Additionally, this restricted range limited our ability to compare analytical methods in the most clinically relevant zinc concentrations. Further studies should therefore include samples of patients and a wider concentration range. Furthermore, we did not use trace element-free tubes, otherwise common in the literature.1,4,14–16 We do however not consider this to introduce an analytical problem, but it could reduce the comparability with other studies using these tubes. Due to our study design, we cannot report whether or not serum continues to be stable past 8 h, although this information would be relevant to many international laboratories. Lastly, the centrifugation protocol of plasma and serum samples in our study was systematically different and hence may have contributed in some degree to our observed matrix differences. Pertinently, we did not include measurement of Haemolysis (H) Index with every Zn measurement; only visual inspection for haemolysis and therefore cannot rule out that unseen haemolysis affected our results, although we deem this unlikely.
In conclusion, we found Zn concentrations stable at room temperature prior to centrifugation for at least 8 h in serum and up to 6 h in plasma, when measured by using Sentinel assay on Cobas c702 or ICP-OES. We found that Cobas and ICP-OES Zn measurements differed significantly throughout the time period studied with the highest degree of difference observed in serum samples, whereas differences observed in plasma were likely inconsequential to clinical use.
Based on this, we cannot recommend Zn analysis performed in plasma samples transported at room temperature for longer than 6 h.
Footnotes
Acknowledgements
All the authors have accepted full responsibility for the entire content of this submitted manuscript and approved the submission. We would like to thank Birgitte Meyer Petersen, Christina Jørgensen, and their colleagues for their invaluable contribution to this study. AI writing assistance was used sparingly to optimize readability of the paper.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
According to local ethical guidelines, the completely anonymized data used in this study constitutes a quality project and does not require ethical approval.
Guarantor
Mads Nybo.
Contributorship
KVB, MNY, HGB, and LHJ: Contributed to design of the work, editing, and critical review of the manuscript. KVB: Researched literature and wrote paper draft. LHJ and HGB: Developed the protocol, performed the study, and obtained data. LHJ: Analysed data and performed the figures.
Data Availability Statement
The data underlying the results presented in the study are available from the corresponding author on reasonable request.
