Abstract
Myocardial infarction remains a significant cause of mortality globally. High-sensitivity cardiac troponin is an essential criterion in the fourth universal definition of myocardial infarction. Our understanding of the structure and release mechanisms of troponin has been updated over the last decade, facilitated by ever more sensitive assays. This review initially outlines the structure and function of the troponin complex, then details the currently proposed mechanisms of release and elimination of troponin. It concludes by using this updated understanding to critique the current universal definition of myocardial infarction and injury.
Keywords
Introduction
The leading cause of death worldwide is cardiovascular disease (CVD). Within CVD, myocardial infarction (MI) is the main cause of mortality globally.1,2 MI results in cardiomyocyte necrosis, due to prolonged ischaemia.3,4 Early detection allows timely initiation of treatment, proven to reduce morbidity and mortality. 5
First discovered in 1965, with an immunoassay developed in 1987, cardiac troponin (cTn) has revolutionized the diagnosis of MI. 6 Its specificity for cardiomyocyte injury is superior to previously used biomarkers.6,7 Gradual refinement in the analytical performance of the assays has led to the development of high-sensitivity cTn (hs-cTn) assays, first commercially available in 2010. The International Federation of Clinical Chemistry and Laboratory Medicine defined the criteria for high sensitivity. Firstly, the concentration with a 10% coefficient of variation (CV) must be below the 99th percentile for the assay, and concentrations should be measurable in >50% of healthy individuals.8,9
An elevated hs-cTn is an essential criterion in the fourth Universal Definition of MI (UDMI). 10 Detecting lower concentrations of cTn has both improved sensitivity but reduced specificity for type 1 MI (T1MI). This has led to diagnostic challenges for those who manage acutely unwell patients in Emergency Departments.
Our understanding of how cTn is released into the extracellular space and bloodstream has been updated in the last decade.11–13 The previous notion that all cTn release indicated cardiomyocyte necrosis made diagnosis more straight forward. However, we now know this was an artificial reality caused by less sensitive assays. Multiple mechanisms of troponin release broaden the potential diagnoses in response to temporal changes in cTn concentrations. Clinicians need to understand the pathophysiology underlying all mechanisms, in addition to necrosis, that cause cTn release. This will ensure correct interpretation, leading to appropriate management of each patient.
The purpose of this review is to outline the currently understood structure and function of the troponin complex and discuss the proposed mechanisms for cTn release and elimination into and from the circulation. This will be followed by a review of the clinical utility of these release mechanisms in the context of MI and cardiomyocyte injury.
Troponin and its place in the sarcomere
The cTn complex is a functional part of the sarcomere.
14
This is characterized by parallel repeats of actin, myosin and titin (Figure 1) forming the thin, thick and elastic filament system, respectively.15,16 All are anchored laterally by actinin containing Z-discs, which form the borders of each sarcomere.15,17–19 Sarcomere with its components and regions.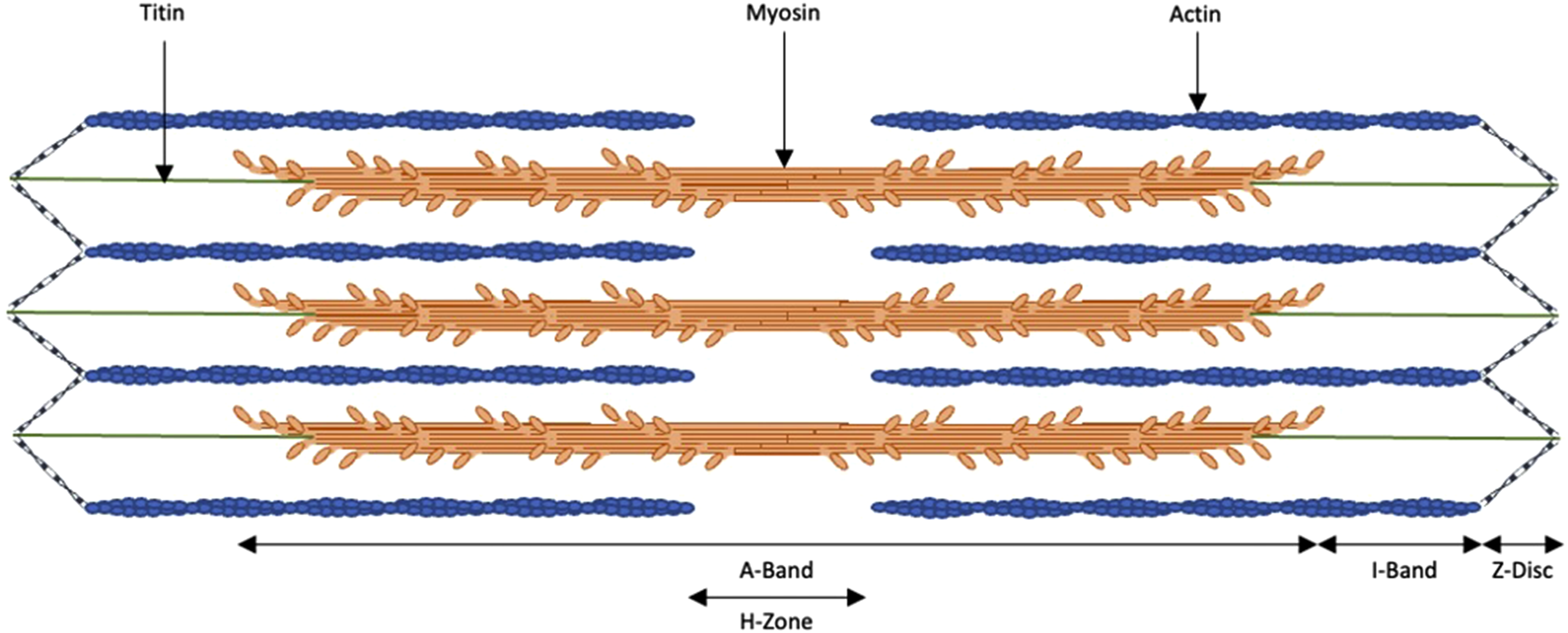
Myocardial contraction is achieved by the formation of cross-bridges from the globular head of myosin to the myosin-binding-site (MBS) on actin, allowing both filaments to slide over each other, causing contraction.17,20,21
The cTn complex is a key player in this process and forms part of the thin filament (Figure 2). It is made up of actin, tropomyosin (Tm) and cTn in a 7:1:1 ratio.21–25 The main structure of the thin filament is formed of polymerized globular actin producing a two-stranded right-handed helix.17,21,24 Wrapped around in a helical fashion but overlying actin sits Tm, the backbone of the thin filament.
26
Tm is an alpha-helical, coiled-coil protein, that exists in a long head-to-tail polymeric line of homo or heterodimers.
27
It sits in a groove on actin and wraps helically around its core.23,26 Tm and cTn sit on and block myosin’s interaction with the MBS when the sarcomere is in its resting state.17,21 Thin filament.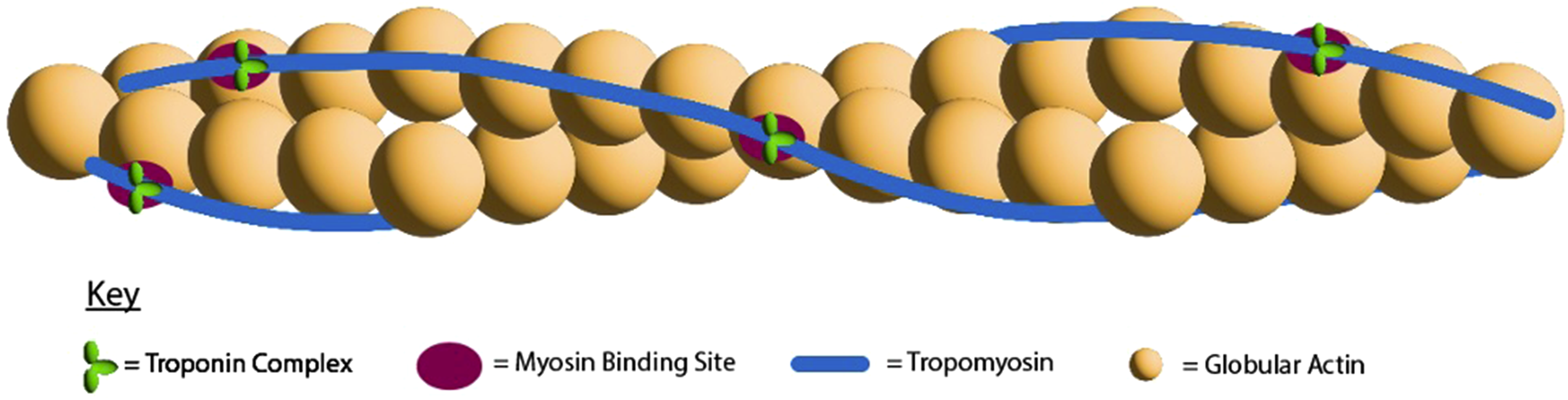
Cardiac muscle contraction
The contraction of cardiomyocytes is dependent on the movement of Tm in relation to actin. There are three positions Tm can occupy, as per the three-state model of thin filament activation: ‘blocked’, ‘closed and ‘open’.28–30
In a resting state, with an absence of calcium, Tm occupies the ‘blocked’ position preventing myosin accessing the MBS on actin. 29 Upon depolarization of pacemaker cells, an action potential is propagated along cardiomyocytes. This change in membrane potential triggers a small amount of calcium influx through dihydropyridine receptors, L-type voltage-gated calcium channels. 27 This small increase in intracellular calcium causes a mass release from the ryanodine receptor on the sarcoplasmic reticulum (SR), into the cytoplasm. This process is known as calcium-induced calcium release.27,31
At high concentrations, calcium binds to cTn, resulting in a conformational change that moves Tm from a ‘blocked’ to ‘closed’ position. In this process, Tm moves laterally 25° on actin and exposes several binding sites between actin and myosin. Once the myosin head binds to these sites, Tm moves a further 10° laterally, into an ‘open’ position, exposing the final binding sites between myosin and actin.29,30 Subsequent hydrolysis of ATP by its ATPase domain results in the myosin head sliding actin inwards, thus causing contraction.17,25,28,32
During relaxation, calcium is removed from the cytosol into the SR by calcium-ATPase and phospholamban, both of which are found on the SR membrane.27,31 When calcium is removed from the cytosol, it is simultaneously unbound from cTn. Thus, Tm relocates onto actin, once again blocking the MBS from forming cross bridges until the process repeats. 17
The troponin complex
The cTn complex is a heterotrimeric protein, with an approximate relative molecular mass of 80 kDa. Each of the three subunits has distinct structure and functions.12,22,33
Troponin T
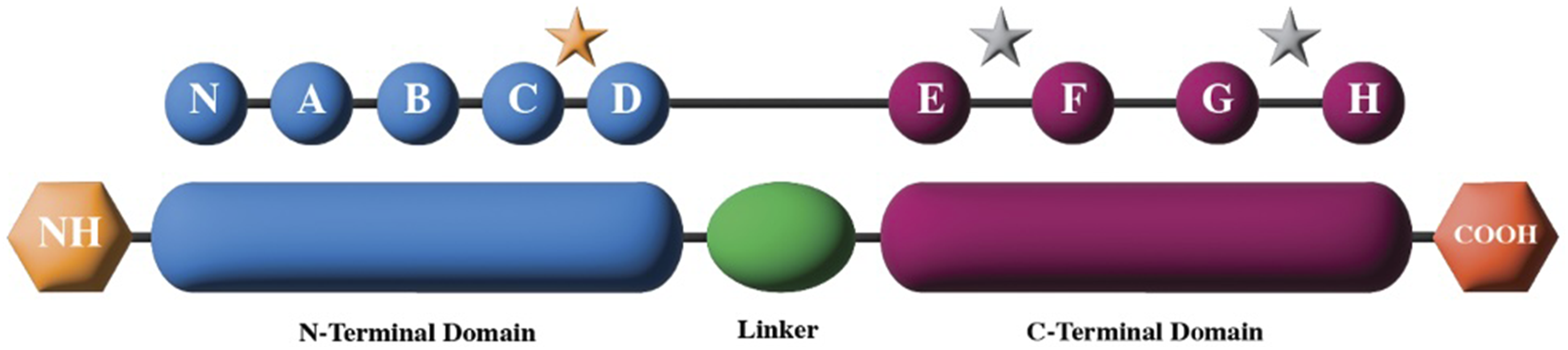
Troponin T is expressed in three different tissues in humans: slow and fast skeletal and cardiac, and the cardiac isoform is coded on the TNNT2 gene. This cardiac isoform is the 35.9 kDa cTnT3 and is 287 amino acids long. It is transiently expressed in some skeletal muscle during fetal development. This is resolved following birth and from that point it is the only cTnT isoform expressed in adult cardiomyocytes.24,34 It is the primary structural component of the cTn complex. By binding with Tm, it is secured to the actin filament. 35
The cTnT molecule is composed of four regions (Figure 3). The N-terminal domain, which is partly responsible for the binding of cTnT to Tm, independent of Ca2+ concentration.
36
The cardiac isoform has a 32-amino acid sequence not present in any of the skeletal isoforms of TnT. This makes it an attractive region for cardiac diagnostics.
24
Initially, studies suggested this highly polar region did not interact with other proteins of the thin filament. More recent findings suggest this region influences the shape of the complex and has a regulatory function.24,37 Structure of cTnT. Demonstrating domain organization, positioning of alpha-helices (H1 and H2) and location of tropomyosin-binding sites (T1 and T2). Adapted from Katrukha et al.
23
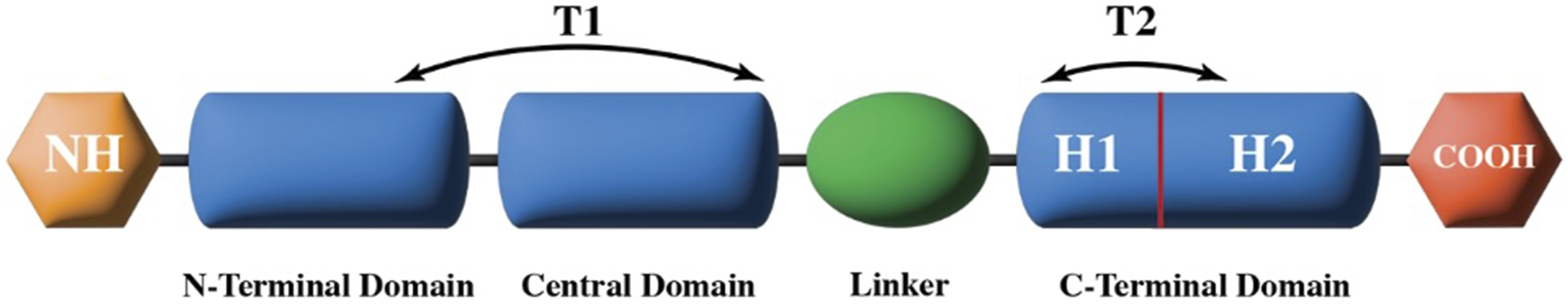
The central domain of cTnT forms part of the first binding site of cTnT to Tm, designated as T1, along with part of the N-terminus.24,35 The interaction of cTnT and Tm at T1 appears to be relatively calcium insensitive, and therefore stable. 23 After the T1 region of the central domain there is a flexible linker, connecting the central to the C-terminal domain. 24
This C-terminal domain contains two α-helices: H1 and H2.23,24 H1 contains part of T2 and is the second binding site for cTnT to Tm. The interaction of T2 with Tm, in contrast to T1, changes with different calcium concentrations, binding more strongly at lower calcium concentrations. 23 T2 also contains a portion of H2. The α-helix of H2 creates a coiled-coil with the α-helix of cTnI and links with the calcium-binding loop of the C-terminal domain of cTnC. 24
Troponin I
Troponin I is also expressed in three tissues: fast and slow skeletal and a specific cardiac isoform. The gene coding for the cardiac isoform is TNNI3.
24
This is the inhibitory component of the complex and blocks interactions between actin and myosin. The protein itself is made up of five domains (Figure 4). It has multiple calcium-dependent interactions with cTnC, cTnT, Tm and actin.22,24,38 Structure of cTnI. Demonstrating domain organization and the positioning of alpha-helices (H1–H4). ID indicates inhibitory domain, and RD indicates regulatory domain. Adapted from Katrukha et al.
23
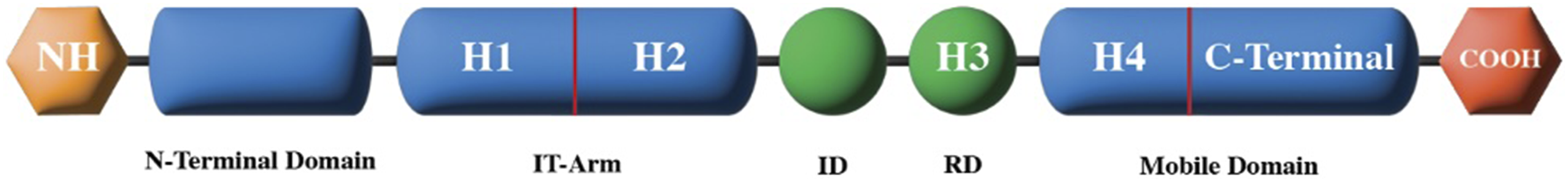
The N-terminal domain is present exclusively on the cardiac isoform. This makes it a target for diagnostics, much like cTnT. 24 It is composed of an acidic and Xaa-Pro region. Within the latter there are two adjacent serine residues, 22 and 23, which serve as potential phosphorylation sites.38,39 These sites influence the interaction of the N-terminal domain of cTnI with the N-terminal domain of cTnC. Under the influence of stress or exercise, adrenaline and noradrenaline increase activation of protein kinase A which induces phosphorylation. This decreases cTnC’s affinity for calcium and so dissociation of calcium in the relaxation phase occurs quicker allowing for a faster relaxation phase and thus a faster heart rate.25,39 The exact mechanism by which phosphorylation decreases calcium sensitivity and increases the cross-bridge kinetics of cardiac myofilaments remains poorly understood. 37
The next domain is a coiled coil formed by cTnI and cTnT, named the IT-arm. 40 This is a long asymmetrical structure and does not move in response to calcium. It gains stability through two key interactions with other subunits.22,24 The first is a α-helical coiled-coil formed between the C-Terminal domain of cTnT (H2(T2)) and the middle portion of cTnI (H2(I)). The second is an amphiphilic α-helix of cTnI (H1(I)) and its interaction with the C-domain of cTnC via polar and Van der Waals forces.22,23 The IT-arm acts as a scaffold that binds cTnI to the N-terminal section of cTnC and the H2 (T2) helix of cTnT.23,40
The third domain is the inhibitory region. This has the role of anchoring the cTn-Tm complex to the MBS on actin, therefore preventing the actin–myosin interaction in low calcium concentrations.25,41 The structure of the inhibitory region remains difficult to solve.23,25 Proposed structures are either an α-helix or a three-hairpin. 25
The regulatory domain, or switch-peptide region, is another region of cTnI with significant functional importance. The domain includes another short α-helix (H3), which at high calcium concentrations interacts with the N-terminal portion of cTnC. This interaction causes the inhibitory region to release from actin and move Tm, allowing ATP hydrolysis and the formation of the actin–myosin cross bridge.23–25
Finally, the mobile domain is made up of the H4 α-helix and the C-terminus.24,25 H4 does not appear to have any direct contact with any of the rest of the complex. 23 The exact structure of the C-terminal domain is not completely known. A study, where the troponin complex was crystallized in a calcium-saturated state, was unable to define its structure. 23 This leads to the theory that this area is highly mobile in the presence of calcium. 23 Animal models show that the C-terminal domain interacts directly with Tm and the actin filament. At low calcium concentrations, it is thought this region contributes to stabilization of cTn on the surface of the thin filament.23,24
Troponin C
The cTnC subunit is the calcium-binding subunit of the troponin complex. Unlike cTnT and I, it has only two different isoforms: slow skeletal/cardiac and fast skeletal. The gene coding for the slow skeletal/cardiac isoform is TNNC1.
24
With a molecular weight of around 18 kDa, it is a member of the EF-hand family of calcium-binding proteins, which consist of two α-helices named ‘E’ and ‘F’. The former is made up of two globular domains joined by a flexible linker (Figure 5).25,39 Each domain consists of two EF-hand motifs, containing two α-helices and an interconnecting loop. The loop has negatively charged residues that can bind and organize divalent cation.25,39,41 In total there are nine α-helices in the cTnC subunit, the first labelled N and then subsequently labelled alphabetically (A–H). The N-terminal and C-terminal domains contain helices N-D and E-H, respectively. Structure of cTnC. Spheres display alpha-helices. Silver stars demonstrate areas of cation binding at low concentrations. The gold star represents the calcium-binding region at only high concentrations (regulatory region). Adapted from Katrukha et al.
23
The N-terminal domain is the regulatory domain. 42 This houses EF-hands I and II. In contrast to skeletal troponin, only EF-hand II can bind cations in cTnC. It has a low affinity but is highly selective for calcium, meaning no calcium is bound at low concentrations.25,32,39
The second domain is the C-terminal, which keeps cTnC anchored to the rest of the cTn complex. It is made up of two EF-hands (III and IV) and only four α-helices (E-H).32,39 In contrast to the N-terminus, both EF-hands bind divalent cations and do so with greater affinity. In low calcium concentrations and physiological conditions, they bind calcium or magnesium. Therefore, a rise in calcium concentration does not alter the C-terminal structure. 39
Both cTnI and cTnT have different amino acid sequences for the cardiac isoform compared with the skeletal fast and slow twitch isoforms. This contrasts with cTnC, in which both isoforms have an identical structure. This is the reason cTnC has not been targeted for use in cardiac diagnostics. 12
Troponin’s role in myocyte contraction
The inhibitory region of cTnI anchors Tm to actin in the ‘blocked’ state and prevents Tm moving away from the myosin-binding sites at low calcium concentrations.39,42
As calcium concentrations rise, in response to membrane depolarization, calcium binds to the interconnecting loop of EF-hand II on the N-terminus of cTnC. EF hands I and II, on the N-terminus, move closer together and form a hydrogen-bonded beta-sheet motif. Opening of a hydrophobic patch allows binding of the cTnI switch peptide.39,42 This moves the cTnI inhibitory region off its binding site with actin. This allows the lateral movement of Tm and permits myosin access to the myosin-binding site, causing contraction.
Troponin release mechanisms
Within a cardiomyocyte, cTnT and cTnI are stored in two pools. First, 5% of cTnI and 8% of cTnT are free within the cytosol of the cardiomyocyte. This location is known as the ‘early release pool’.11,43,44 The second is the ‘structural pool’ and accounts for cTn bound to the thin filament in the sarcomere. This cTn is only thought to be accessible following necrosis or apoptosis of cardiomyocytes.11,13,43,45
When the first troponin assays were developed, cTn release and detection was a surrogate marker for cardiomyocyte necrosis.3,4 Current hs-cTn assays are sensitive enough to detect fluctuating concentrations in healthy individuals, including diurnal variations, indicating there are physiological as well as pathological mechanisms of cTn release.46–50 Multiple different release mechanisms of cTn have been described (Figure 6). Understanding these allows clinicians to make informed decisions when interpreting cTn results. Mechanisms of cTn release. ROS indicates reactive oxygen species; cTn indicates cardiac troponin; MMP-2 indicates matrix-metalloproteinase-2.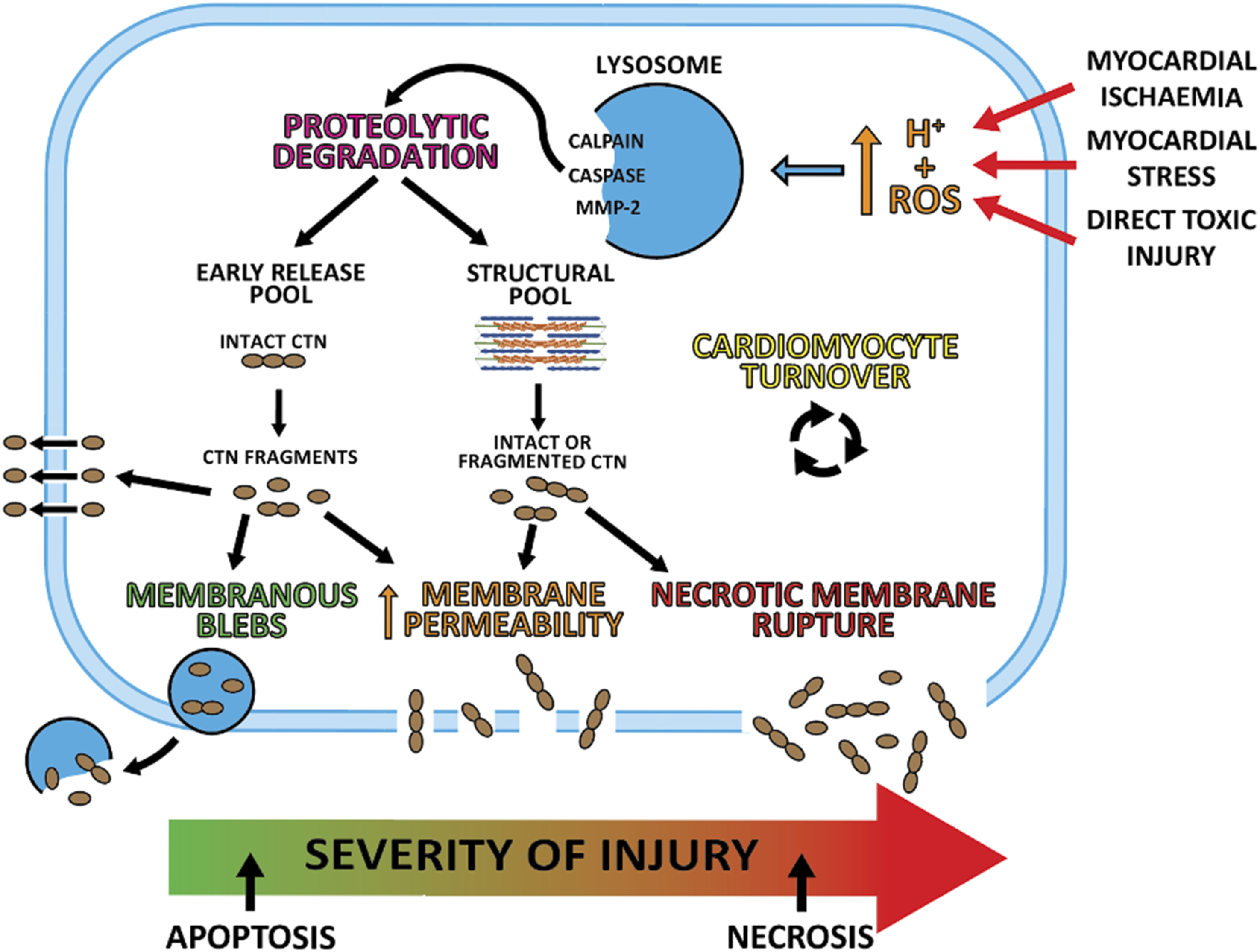
Necrosis
Necrosis is the most established and clinically important cause of cTn release. Necrosis has multiple causes, from trauma to toxins, but the most clinically relevant cause is prolonged ischaemia in MI (Figure 7).
3
Complete or partial occlusion of a coronary vessel prevents or reduces supply of oxygen to downstream myocardium. This hypoxia causes ATP synthesis to switch to anaerobic mechanisms, resulting in an accumulation of H+ within the cytosol.3,4 The H+/Na+ transporter removes H+ ions from the cytosol in exchange for Na+, to raise intracellular pH. Through osmosis, there is an accumulation of intracellular water and cell swelling. To prevent this, the Na+/Ca2+ exchanger works in reverse and brings Ca2+ into the cell and takes Na+ out.3,4 High cytosolic Ca2+ concentration results in an increase in the mitochondrial matrix Ca2+ concentration, via the mitochondrial Ca2+ uniporter. A plateau in the mitochondrial membrane potential, combined with an increase in reactive oxygen species (ROS), results in the opening of the mitochondrial permeability transition pore, leading to necrosis.3,4 Process from the onset of ischaemia to necrosis of a cardiomyocyte. MMP indicates mitochondrial membrane potential, and MPTP indicates mitochondrial permeability transition pore. Adapted from Chiong et al.
3
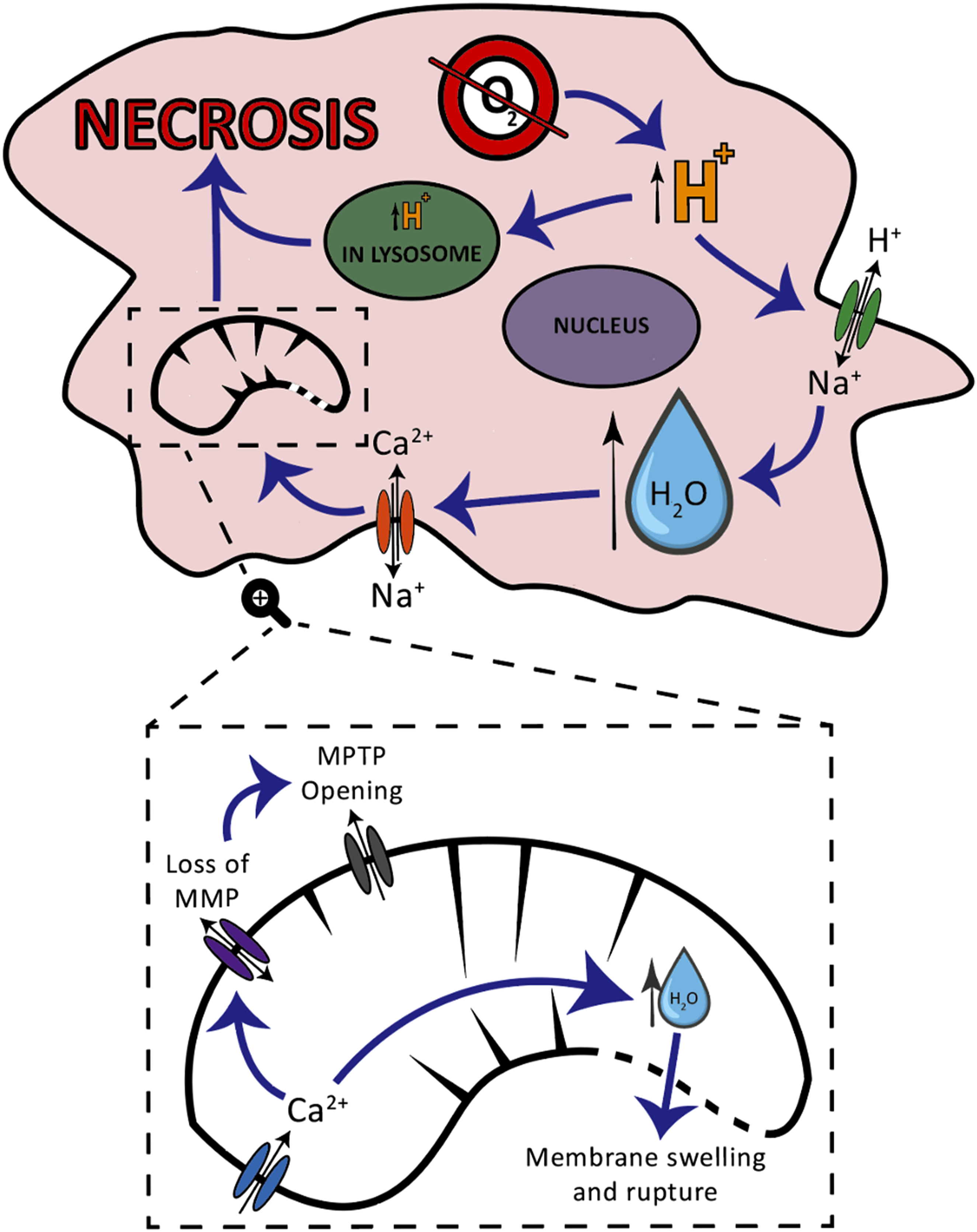
In addition, an increase in H+ ion concentration reduces lysosomal pH, which activates intracellular proteases. This, combined with increased intracellular water and the destabilizing nature of reactive oxygen species, results in rupture of the cellular membrane and release of cytosolic proteins into the interstitial space.3,4
Apoptosis
Apoptosis is a form of regulated programmed cell death which contributes to myocardial cell loss in MI. 51 Apoptosis increases caspase-8 enzyme activity, resulting in cell death despite relatively preserved cell membrane.3,12 Apoptotic cells are removed by macrophages, but disruption of membrane during this process could result in the release of the cytosolic proteins, including cTn, into circulation. 4
Ischaemia is a common trigger of apoptosis with cTn release occurring without necrosis. Experimental occlusion of the left anterior descending artery for 10 min led to large cTn release despite no evidence of necrosis on histological examination, whilst terminal deoxynucleotidyl transferase-mediated dUTP nick-end labelling (TUNEL) confirmed focal apoptosis. 52 Cardiomyocyte apoptosis results in fibrosis and heart failure with increased filling pressures and myocardial stretch triggering both acute and chronic cTn release. Chronic myocyte injury occurs in chronic heart failure while rapid cTn release follows acute left ventricular failure or tachyarrhythmia. The sudden increase in myocardial filling pressures or workload accelerates cardiomyocyte apoptosis and subsequent cTn release. 53
Sympathetic overactivation in acute heart failure amplifies beta-adrenergic-mediated cardiomyocyte apoptosis, likely explaining some of the clinical benefits of selective β-blockers in chronic heart failure.54,55 This was directly demonstrated in murine models. 56 Apoptotic release of cTn can be clinically relevant in other clinical states associated with myocardial stretch or sympathetic nervous system activation, such as acute pulmonary embolism, tachyarrhythmia, acute heart failure and atrial fibrillation.
Troponin release without cell death
Cardiomyocyte turnover
The heart was initially believed to be a post-mitotic organ. There is evidence now that cardiomyocytes (CMs) do regenerate with an annual rate of around 1% per year at the age of 25, reducing to 0.45% by age 75.11,13,45 It is however unlikely that this turnover will have any measurable impact on a patient’s cTn levels on a day-to-day basis, or in acute diagnostics. 45
Intracellular proteolytic degradation of troponin
The main proteolytic enzymes are calpain, caspase and matrix-metalloproteinase-2 (MMP-2).43,57 Following intracellular proteolysis of cTn, small fragments may be able to pass through an intact cell membrane.11,12
The ability of intracellular proteins to cross an intact cell membrane is directly related to their molecular size. 12 Following an insult, low-molecular weight (LMW) intracellular proteins, such as myoglobin, are detectable in the blood stream earlier than those with higher molecular weight, such as lactate dehydrogenase. 12
Ischaemia and increases in myocardial stretch have been shown to induce proteolytic degradation of cTn in multiple experimental studies.58,59 However, it is also known that these pathologies can also induce both necrosis and apoptosis. We currently do not know if proteolytic products are released in sufficient quantities to cause changes in cTn concentrations detected by high-sensitivity assays, without concomitant cell death.58,59
Increased cellular wall permeability
Proteolytic enzymes, released from lysosomes following injury, break down intracellular proteins and cause proteolysis of the cell membrane. 44 This increases membrane permeability and facilitates leakage of cTn and its fragments.3,12 It is not clear if this process can occur in milder insults that activate proteolysis of the membrane but do not trigger full blown apoptosis or necrosis.
It is hypothesized that mechanical stretch can result in alterations to the cardiomyocyte’s sarcolemma permeability and permit the release of cTn. 57 This mechanical stretch is sensed by integrins, transmembrane glycoprotein receptors, which link the extracellular matrix to the intracellular space. Stimulation of integrins results in the release of intact cTn from viable CMs in vitro. 57 This release was not associated with any evidence of necrosis, but the presence of apoptosis was not tested for. Therefore, as previously described, these results may be due to cTn release following apoptosis triggered by myocardial stretch. 53
Increased cellular wall permeability facilitates release of cTn from injured cardiomyocytes in vitro. However, further research is required to determine if this process can occur independently of cell death and whether it can cause meaningful changes in cTn concentration.
Formation and expulsion of membranous blebs
In response to ischaemia, vesicles form on the surface of hepatocytes and cardiomyocytes. In the latter these contain cytoplasmic proteins, such as fragmented cTn. These proteins are ejected from the ruptured vesicles at the cell membrane and released into the extracellular space.11,45 Schwartz et al. initially demonstrated this in ischaemic cultured cardiomyocytes in 1985. 60
During ischaemia, blebs are formed containing cytoplasmic proteins. Prolonged ischaemia causes release of blebs and rupture of the cell membrane. Theoretically, if the cell is re-oxygenated in time, blebs could be released prior to the fragmentation of the cell membrane. This was proposed by Hickman et al. and has been demonstrated in vitro. 61
The quantity of cTn within these blebs is unknown. In addition, this process is yet to be shown in cardiomyocytes in response to ischaemia in vivo. Further research is needed to understand how clinically relevant this mechanism is when interpreting hs-cTn results.
Release of troponin from non-cardiac cells
Initial cTnT assays showed cross reaction with skeletal muscle TnT. 62 However, current hs-cTnT and cTnI assays appear to be truly cardiac specific.7,63 There have been clinical and experimental studies demonstrating the presence of a cross-reacting isoform of TnT, in diseased skeletal muscle.63–65 These studies have shown that diseased skeletal muscle expresses a cross-reacting isoform of TnT, as opposed to current assays detecting skeletal muscle TnT. Elevated values in those with skeletal myopathies can cause false positives. In addition, these concentrations can fluctuate, sometimes enough to fulfil rule-in criteria for MI in some diagnostic pathways. 64
The same studies have not found these issues with cTnI. Skeletal myopathies can alter detectable cTnT concentrations, and this consideration is important when this cohort present with suspected MI. For these patients, it will be necessary to take more than two samples to demonstrate continued rise or fall in cTnT concentrations. Alone, two samples could fulfil rule in criteria and lead to false positives.
Troponin in the blood pool
Degradation of troponin in the blood pool and blood samples
Once released into the plasma, the cTn complex is known to circulate in one of many forms.66–70 Immediately following release, it exists as a cTnI-cTnT-cTnC (ITC) complex. Subsequent to this the N-terminus and central domain of cTnT are cleaved resulting in LMW-ITC. Finally, the central domain of cTnT dissociates from the LMW-ITC complex to leave the IC complex, where the N- and C-termini of cTnI are readily cleaved. 70 The central domain of cTnT undergoes further degradation in the plasma in its free form, whilst there appears to be little detectable cTnI circulating in a free form and this is quickly degraded and eliminated. 70
This is clinically important because the time interval from MI to blood sampling will influence the proportion of expected cTn forms in the plasma. Therefore, cTnI assays must be specific to the 23 to 126 amino acid regions to ensure detection of all cTnI complexes. A cTnT assay must be able to detect both cTnT present in the ITC complex, in free fragments and be specific to the central domain. 70 Without this, assays would provide false negatives at a frequency dependant on the interval from MI to sampling.
Degradation of cTnI and cTnT occurs within the cytosol.11,36,43,57,71 There is evidence in vitro that this process also takes place directly in the blood pool, and the process of degradation of cTnI and cTnT differs post-MI.71–73
Following an MI, cTnT is present initially as an intact molecule (37 kDa) or in large fragments (29 kDa). Over time, there is increasing fragmentation of cTnT from the intact and large fragments into smaller fragments (15–20 kDa).74–76 In those with cTnT elevations in response to exercise or with chronic kidney disease (CKD), only small fragments are detected.77,78 Additional research is required to understand if this difference is due to increased proteolysis of cTn and membrane permeability, caused by prolonged physiological stress, allowing release of LMW fragments, without releasing large fragments because there is no necrosis or apoptosis-induced membrane rupture.
The exact mediators of cTnT and I degradation in the blood pool are poorly understood. One cause is thrombin, which has a significant effect on cTnT. Intraplaque rupture and cardiomyocyte necrosis result in increased thrombin activity in vivo.72,74,79 The amount of thrombin-associated degradation of cTnT depends on the blood sample matrix used in laboratory assessment. Serum samples generate abundant thrombin. Multiple studies have shown significant cTnT degradation in this matrix.71–73 This in vitro effect is likely to be less significant in vivo. When samples are taken using lithium heparin (LH) (which inhibits thrombin activity), there is specific cleavage of the central domain of cTnT.74,75 Apart from this specific site, no further proteolysis is observed when samples are incubated in LH. 74
Ethylenediaminetetraacetic acid (EDTA) prevents further in vitro generation of thrombin but will not inhibit the action of any thrombin already present in the plasma. 72 Degradation in this matrix does occur after around 4–8 h, as opposed to immediately in serum samples. 72 This indicates that there is likely to be some thrombin degradation in vivo.
In vitro degradation of cTnT in EDTA, coupled with inhibition of proteolysis in LH, suggests that thrombin is likely to be the primary cause of cTnT degradation in the blood pool. Despite this, the majority of proteolytic degradation of cTnT appears to occur in the cytosol of injured cardiomyocytes.72,74
In contrast to cTnT, free cTnI fragments are quickly degraded by proteolytic enzymes in either the necrotic cardiomyocytes or the blood into fragments that are not detectable by the monoclonal antibodies used in current assays.80,81 Thus, almost all detectable cTnI in the circulation is the stable central portion and is bound to cTnC in the IC complex. There are around 11 fragments detectable in the serum of patients post-ST-elevation MI (STEMI), with all these fragments derived from the N- or C-terminus. 81 The ratio of intact cTnI to fragments does not appear to change over time post-STEMI. This suggests that, similar to cTnT, the majority of proteolytic degradation of cTnI occurs in the injured cardiomyocyte, not in the blood pool. 81
In addition to degradation of the ITC complex to IC and free cTnT, it is also possible for the ITC complex to dissociate to component forms without degradation in vitro. In a recent experimental study, samples incubated at body temperature for 24 h had a gradual decrease in the amount of ITC complex. 79 This decrease could not be explained by degradation in heparin and citrate samples, as this was not detected by Western Blotting at 24 h. However, dissociation is unlikely to be clinically significant as it does not influence the amount of cTnI or cTnT detected by current high-sensitivity assays.
Persistently elevated cTnT in patients with chronic myocyte injury (CMI), particularly in those with CKD, reduces specificity for MI. This is often a result of persistent small cTn fragments in the blood. An exciting avenue of research is the development of cTnT assays that only detect intact cTnT or the large 29 kDa fragment. These are present in acute MI but not CKD and may provide greater specificity for MI in those with CKD and other causes of CMI in the future.
Elimination of troponin from the blood
Historically, the most disputed mechanism of elimination is the role of the kidney, which appears to have a different pharmacokinetic role for cTnT and cTnI. It would be expected that if cTn was excreted renally, it should be detectable in the urine. Interestingly, urine levels of both cTnI and cTnT are higher in those with CKD than in those with normal renal function.82,83 This implies that under normal conditions the glomerulus and the proximal convoluted tubule reabsorb cTn. 84 In diseased kidneys, these mechanisms may fail and produce leakage of a small amount of cTn into the urine. In patients with large acute cTnT release, such as following MI, cTnT is present in the urine of patients with normal kidney function. 82 This is a result of ‘overflow proteinuria’, saturation of the reabsorption/degradation mechanisms and subsequent overspill into the urine. 84
With reducing estimated glomerular filtration rate (eGFR), the amount of circulating cTnT increases.77,85 However, eGFR does not significantly affect the clearance of cTnT post-myocardial infarction. 86 Therefore, post-MI, the majority of cTn clearance from the blood must occur independent of the kidneys.
It is known that the liver removes proteins like lactate dehydrogenase from the plasma by receptor-mediated endocytosis. This has been shown to be the case for cTn in murine models. 87
It appears that cTn elimination follows a first-order elimination model. Namely, the rate of elimination is higher the greater the concentration of serum cTn in the blood.88,89 Previous studies assessing the kinetics of cTn elimination in humans have shown the plasma half-life to be between 12 and 15 h for cTnI and around 17 h for cTnT. 90 These estimates are in patients in the early phase following an initial insult from an STEMI and are therefore confounded by the ongoing release of cTn.88,90
The first true assessment of pure cTn pharmacokinetics in the absence of ongoing release has recently been elegantly presented. 89 Participants were re-transfused their own cTn rich plasma 4–6 weeks following STEMI. Repeated samples were then taken over the following hours to assess elimination kinetics. These demonstrated a biphasic elimination of cTn, with distribution and elimination phases, in keeping with previous animal models.88,91
At high concentrations of cTn, the distribution phase predominates. This primarily involves scavenger receptor-mediated endocytosis in the liver, spleen and kidney. Following this, the elimination phase comprises renal excretion. In this study, the plasma half-life was 134 and 206 min for cTnT and cTnI, respectively. This is similar to previous publications using rats, dogs and ponies, where elimination of cTn was also assessed without simultaneous in vivo release from injured myocardium.88,91
When this evidence is applied clinically to those with acute MI or myocarditis, the cTn half-life will be closer to 12–17 h, due to ongoing cTn release of CMs. Prolonged release implies release from the ‘structural pool’ and may signify cell death via apoptosis or necrosis. In patients with less significant injury, not significant enough to cause cell death, one would expect a shorter half-life, closer to 4 h. It is known that the half-life of cTn elevations in exercise is less than in those with MI.
Clinical application of these mechanisms
The diagnosis of MI is dependent on the 4th UDMI.
10
Initially, the patient must have a cTn concentration above the assay’s 99th percentile upper reference limit (URL). Once this condition is fulfilled, the patient can either receive one of three diagnoses: CMI, acute non-ischaemic myocardial injury (ANIMI) and MI. Categorization is dependent on the presence of a temporal change in cardiac troponin concentration and then the presence/absence of ischaemia (Figure 8). Flow diagram to illustrate how to use and interpret the 4th Universal Definition of Myocardial Infarction.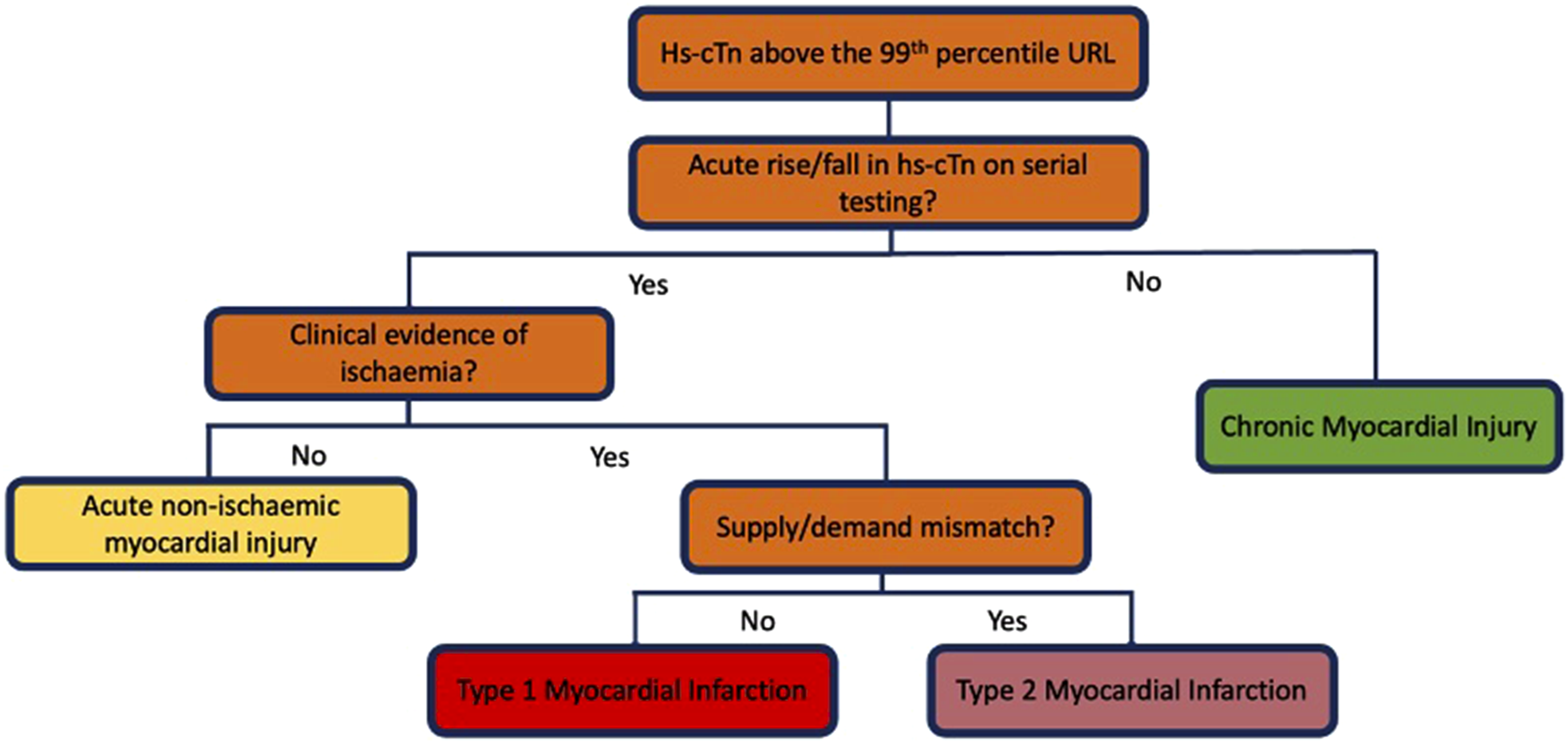
In clinical practice, the two most frequently encountered types of MI are T1MI and type 2 MI (T2MI). The former is acute athero-thrombosis in the coronary artery. Clinically, this can present as a STEMI or a non-STEMI (NSTEMI) based on the appearance of the patients ECG. 92 STEMI is associated with complete or near-complete occlusion of the coronary artery and without reperfusion results in transmural infarction. NSTEMI can also be caused by complete occlusion but more frequently is a result of non-occlusive thrombus, resulting in subendocardial infarction.10,93 T2MI is the presence of a mismatch between myocardial oxygen supply and the demand of the myocardium, leading to ischaemia and ultimately necrosis. 10
ANIMI is a term with significant heterogeneity. It comprises acute cardiovascular conditions, such as myocarditis and Takusubo cardiomyopathy, that are not related to myocardial blood supply. Additionally, there are conditions with injury secondary to increased myocardial strain due to acute illness without evidence of ischaemia. Finally, there can be physiological release of cTn in response to intense or prolonged exercise (Figure 9).88,90–92 Causes of T2MI and ANIMI. T2MI indicates type 2 myocardial infarction, and ANIMI indicates acute non-ischaemic myocardial injury.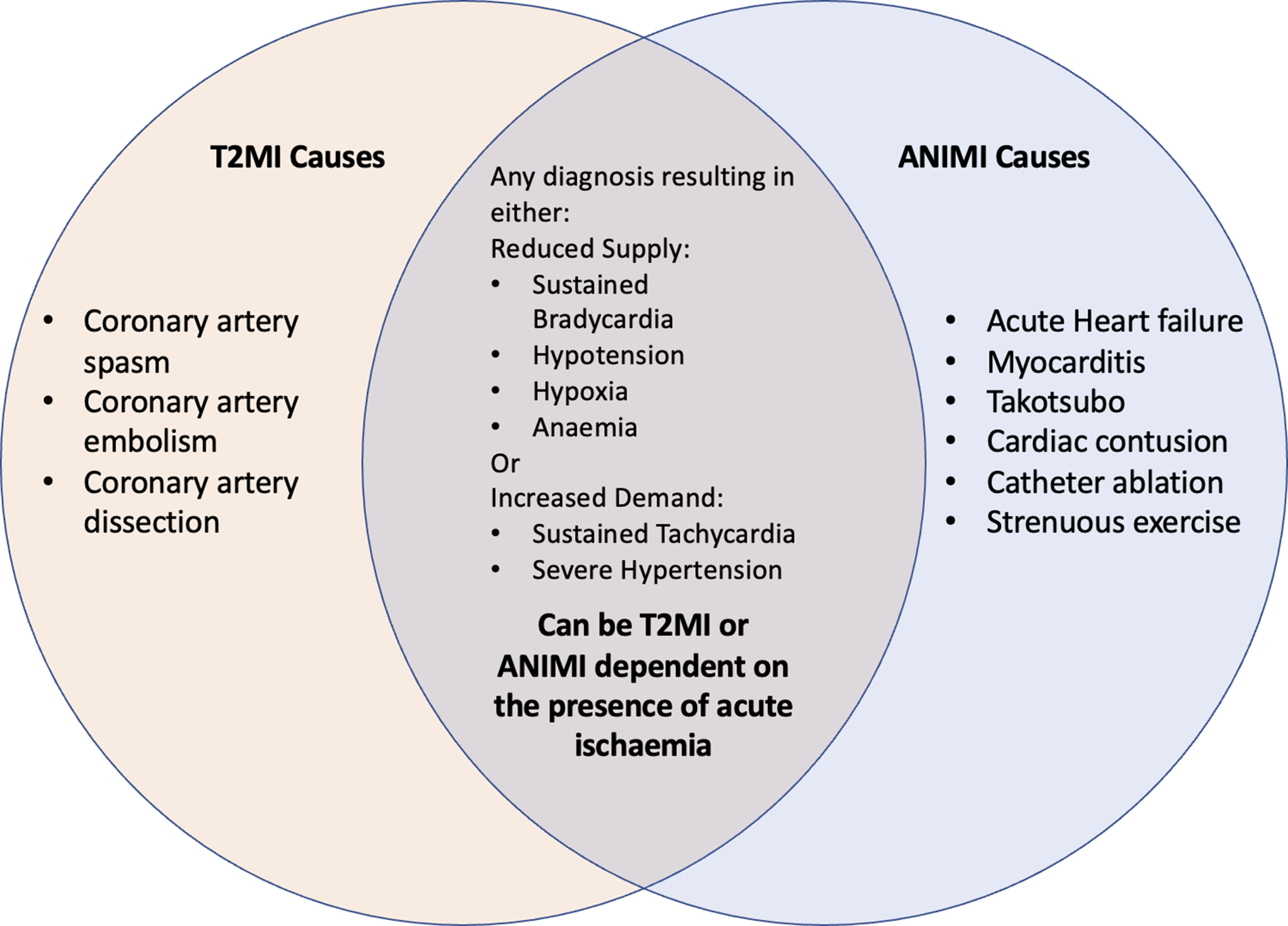
The 4th UDMI works on the premise that all measurable cTn release is a direct result of cardiomyocyte death. There are multiple proposed mechanisms of cTn release that could occur without cell death. Demonstrating this in a human model is problematic. It has been shown in humans that balloon occlusion for as little as 60 s caused sufficient acute release of cTn to fulfil the 4th UDMI. 94
The relative contribution of different mechanisms following vessel occlusion is currently unknown. It is possible that there may be gradation from reversible cTn release through to cell death. Similarly, how the relative contributions of all the possible mechanisms interplay, depending on the dominant pathophysiology and duration, remain unknown.
A future experimental animal study, causing short-lived ischaemia via balloon occlusion, followed by measurement of cTn, histological examination to assess for necrosis and TUNEL for apoptosis, would provide valuable insights. If this demonstrated cTn release in the absence of necrosis or apoptosis, it would go some way to explain the heterogeneity in ANIMI.92,95–97
Kinetics of troponin in myocardial infarction and injury
The differences in cTn kinetics between T1MI, T2MI and ANIMI have been scarcely studied. The concentration at presentation appears to be higher in those with T1MI than T2MI or ANIMI.95–100 In addition, in those with T1MI there is a sharper change in concentration over the first hours of insult.98,99 However, in those who present acutely to hospital, there does not appear to be a significant difference in the release kinetics following T2MI or ANIMI.98,99 The kinetics of cTn alone is a poor differentiator between T1MI, T2MI and ANIMI.
Differentiation between T2MI and ANIMI
Patients diagnosed with T2MI and ANIMI are older, more likely to be female and more co-morbid than those with T1MI.95,96,98,99,101 Unsurprisingly, they have a higher risk of all-cause mortality and a worse prognosis. However, this effect is likely related to the differences in demographics between the populations. One study found equivalent all-cause mortality between the three diagnoses when adjusted for confounders like age, sex, renal function and history of prior MI. 97
There are diagnoses that cause T2MI that cannot result in ANIMI. Equally, there are those that cause ANIMI that cannot result in T2MI (Figure 9). Many patients sit in the middle, where differentiation between T2MI and ANIMI is dependent on the presence or absence of ischaemia (Figure 10). This is subjective and initially clinically irrelevant because treatment will first focus on the precipitating disease. Once the patient is stabilized, there are two avenues that could potentially generate therapeutic benefit. Both ANIMI and T2MI are at different ends of the spectrum of one clinical entity. Differentiation is often subjective and dependent on the amount of ischaemia caused by the insult. ANIMI indicates acute non-ischaemic myocardial injury, and T2MI indicates type 2 myocardial infarction.
Firstly, it is important to check for evidence of left ventricular dysfunction secondary to T2MI or ANIMI. If significant impairment is present, the patient would benefit from evidence-based medical therapy for heart failure with reduced ejection fraction. 54
Secondly, T2MI may unmask asymptomatic obstructive coronary artery disease that could benefit from preventative therapy. In 93 patients diagnosed with T2MI who underwent subsequent CT or invasive coronary angiography and cardiac magnetic resonance imaging (CMR) or echocardiography, the prevalence of coronary artery disease (CAD) was 68%, with 30% of these having obstructive CAD. 102 In the 68 patients who underwent CMR, 54% demonstrated late gadolinium enhancement, indicative of MI. Unfortunately, there was no comparative ANIMI group. It would be beneficial to understand if the diagnosis of T2MI carries a higher risk of obstructive CAD than ANIMI. This may enable clinicians to better target cardiac investigations.
There are currently no evidence-based guidelines on how patients with T2MI should be investigated. Nor is there evidence that preventative therapy in those with CAD uncovered by T2MI actually improves cardiovascular outcomes. There are trials ongoing to assess the optimal strategy for investigation of patients diagnosed with T2MI. 103
Conclusions
Improvements in the sensitivity of cTn assays have allowed the detection of more minor myocardial injury, reducing specificity for T1MI. Release of detectable cTn into the blood can result from necrosis and apoptosis. It is possible that some mechanisms could occur independently from cell death, but further research is required to confirm this hypothesis.
Release kinetics of cTn alone are unable to distinguish between T1MI, T2MI and ANIMI in clinical practice. Differentiation between T2MI and ANIMI can be challenging and in those with preserved left ventricular function may not be clinically relevant. Trials are ongoing to provide an evidence base of how best to investigate this heterogenous group of patients.
Footnotes
Acknowledgements
This article was prepared at the invitation of the Clinical Sciences Reviews Committee of the Association for Clinical Biochemistry and Laboratory Medicine.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Nil required.
Guarantor
AK.
Contributorship
JH undertook the literature review, wrote the main body of text and created all scientific figures. All other authors provided feedback and editing of the manuscript drafts.
