Abstract
Atherosclerotic cardiovascular disease remains a major cause of premature death in the United Kingdom. Lipid testing is a key tool used to assess cardiovascular risk and guide clinical management decisions. There are currently no national guidelines to provide evidence-based recommendations on lipid testing and reporting for UK laboratories and clinicians. Here we present consensus guidance, following a review of published evidence by a multidisciplinary group of UK experts across a range of laboratory and clinical services. Recommendations include the composition of a standard lipid profile; indications for, and composition of, an enhanced lipid profile including apolipoprotein B and lipoprotein (a); use of the Sampson-NIH calculation for LDL-c estimation and guidance on when to flag abnormal results. This consensus guidance on lipid testing and reporting in the United Kingdom has been endorsed by HEART UK and The Association for Laboratory Medicine.
Introduction
Atherosclerotic cardiovascular disease (ASCVD) remains the leading cause of death worldwide and, in the United Kingdom, accounts for a quarter of all premature deaths. 1 Small apolipoprotein B (ApoB)-containing lipoproteins can cross the vascular endothelial barrier and accumulate in the arterial wall, leading to atheromatous plaque formation which is a precursor to subsequent blood vessel blockage and the clinical sequelae of myocardial infarction, stroke or other vascular disease. 2 Excess pro-atherogenic lipids causally contribute to an increased risk of ASCVD and this risk can be quantified and predicted by measuring the blood concentrations of pro-atherogenic lipid particles or their cholesterol content, most commonly expressed as calculated low density lipoprotein cholesterol (LDL-c) but also non-high density lipoprotein cholesterol (non-HDL-c) and/or ApoB concentrations.3–5 Importantly, optimisation and reduction of these pro-atherogenic lipids reduces the future risk of both primary and secondary cardiovascular events.6,7
For many years, LDL-c, as calculated using the Friedewald equation (FE), has been the focus of lipid reporting and cardiovascular risk management globally. LDL-c continues to be important both due to its proven causal role in atherosclerosis as well as the consistent relationship found between LDL-c reduction and observed cardiovascular risk reduction.8–10 As such, it has been an entry criterion and primary or secondary endpoint of many clinical trials for lipid lowering medications, is accepted as a surrogate endpoint for the purpose of regulatory approval of new drugs, and remains a key management target in many guidelines.11,12 Additionally, in clinical practice, healthcare professionals, particularly in primary care, may be more familiar with its use. However, it is acknowledged that there are important limitations to the use of LDL-c as a measure of lipid-associated risk and indeed to the FE from which it is most commonly estimated. 13 The FE assumes a constant relationship between the measured concentration of serum triglycerides and the cholesterol content of VLDL (very low density lipoprotein cholesterol or VLDL-c), which must be subtracted from the non-HDL-c to obtain the estimated LDL-c. Consequently, FE has a requirement for a fasting sample (to eliminate chylomicrons), can be inaccurate at low LDL-c concentrations and has limited use with raised triglycerides, a problem seen increasingly in clinical practice as obesity and diabetes-related dyslipidaemia have become more prevalent. 14 Moreover, despite apparent optimal lowering of FE calculated LDL-c, ASCVD events still occur frequently. 15 There is therefore a clinical need for alternative measures which are proven to be reliable for use in cardiovascular risk management, such as non-HDL-c and, in certain instances, ApoB and Lp(a) to estimate residual risk. 16 Most recently, the development of improved equations to calculate LDL-c appear to offer greater accuracy in particular in those with hypertriglyceridaemia or normal or low LDL-c or those already on a lipid lowering medication. 17
However, the use of these measures in current clinical practice is inconsistent and, whilst there are well-established national guidelines to assist clinicians with assessing and managing ASCVD risk,18,19 recommendations for laboratory testing of lipids and reporting in the United Kingdom are lacking. This article therefore reviews the current evidence for lipid testing in the context of ASCVD risk assessment. It contains evidence-based recommendations on the composition of a standard and enhanced lipid profile along with guidance on when and how to test and when to alert the requesting clinician at key decision limits (summarised in a recommendations table, Appendix 1 and ‘At a glance’ guidance in Appendix 2). It is beyond the scope of these recommendations to fully address in depth genomic testing, paediatric testing or diagnostic investigations for rare disorders of lipoprotein metabolism (e.g. lipodystrophy) which are all undertaken within lipidology clinics. These topics are referenced in brief where relevant in this guidance and there are several resources cited here that address these areas.20,21
Summary of current guidance on lipid testing from NICE including use of LDL-c and non-HDL-c
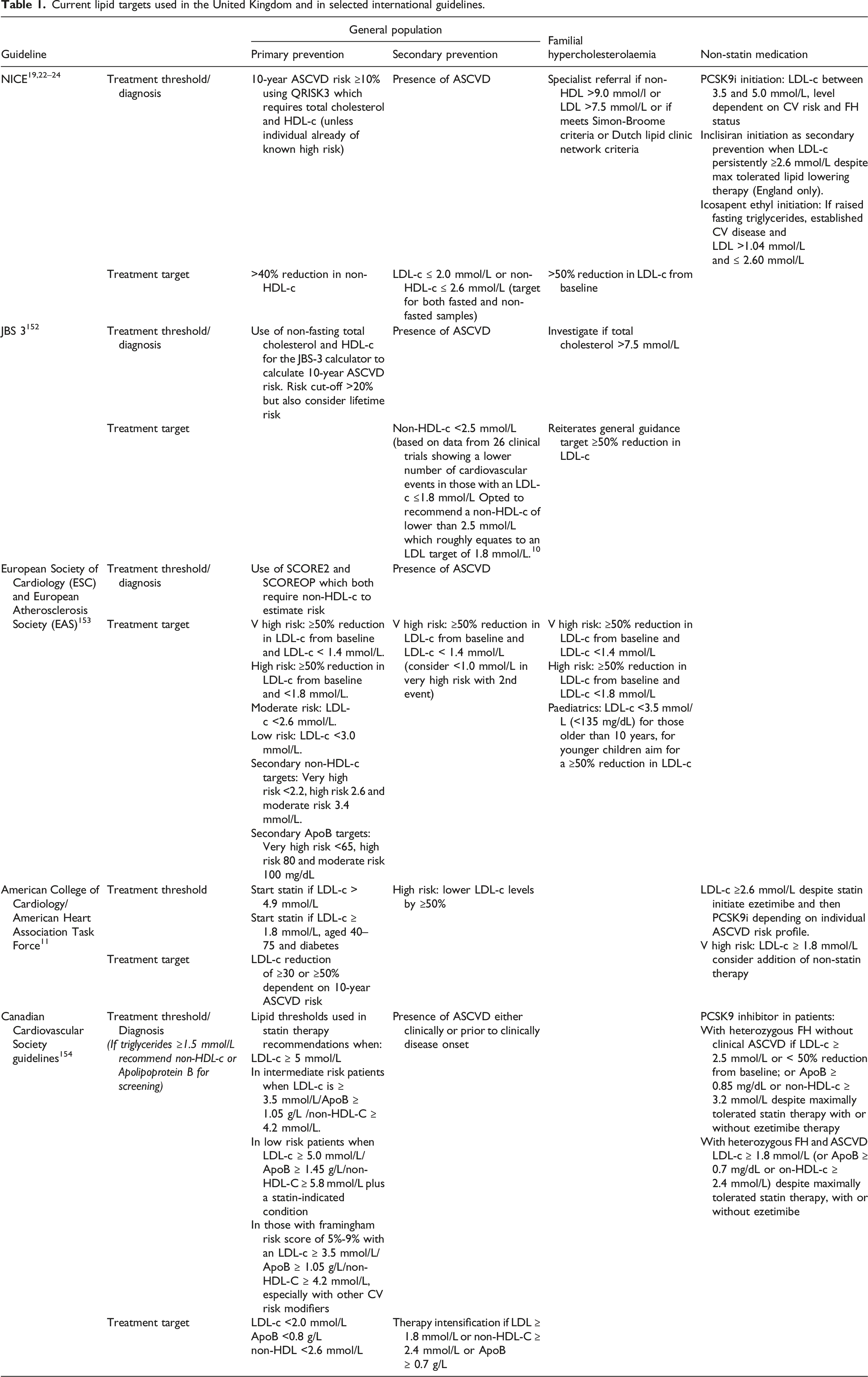
Current lipid targets used in the United Kingdom and in selected international guidelines.
Standard lipid profile
To harmonise lipid testing across the United Kingdom, included here is guidance for the composition of a basic lipid profile which is adequate in most ‘standard’ cases and an ‘enhanced’ profile in cases where more detail is required to accurately assess cardiovascular risk. The type of testing required may be dependent on where the test is requested, where along the patient journey it is performed and whether there are any specific clinical indications. For example, the reasons for testing may differ depending on whether the test is requested in primary care versus a specialist lipid clinic. Whilst the focus in primary care may be to screen for dyslipidaemia, for example, to estimate ASCVD in primary prevention or to assess initial treatment response, in a specialist lipid clinic assessment for suspected genetic dyslipidaemia, severe dyslipidaemias and medication intolerance may be more common. Specific patient factors that led to the testing being initiated may determine the type of profile required such as family history, clinical signs (e.g. xanthomata or other stigmata of hyperlipidaemia) and recurrent cardiovascular events despite reaching LDL-c or non-HDL-c targets. Furthermore, analytical factors such as raised triglycerides which may impact on the interpretation of a standard profile should also be considered. For these reasons, a standard and enhanced profile has been included in these recommendations.
Composition of lipid profile
The standard profile should include the following analytes: total cholesterol, triglycerides, HDL cholesterol (measured) and calculated non-HDL-c, LDL-c (see Section 9, recommendation 2 for formula) and total cholesterol/HDL-c ratio. This is in agreement with both current NICE guidance and the European Federation for Laboratory Medicine (EFLM) guidance. Reporting of the profile should include documentation of whether it was a fasting or non-fasting sample, details of which should be provided by the clinician at the time of the request, in addition to whether testing was requested in primary or secondary prevention to allow appropriate comments to be appended. See also Supplement 1 for guidance on standard units and decimal places to be reported.
An enhanced profile is required in selected clinical situations and may include measurements of ApoB and Lp(a) which should be measured where clinically indicated. Lp(a), in most instances, needs to only be measured on a single occasion (see Sections on ApoB and Lp(a) for further details).
Figure 1 highlights the lipids that are captured by analytes within the standard and enhanced lipid profiles in fasting and non-fasting settings. (a) Composition of lipoprotein particles. (b) Underlying composition of analytes measured or calculated in a lipid profile in a fasting and non-fasting state.*HDL subclasses include HDL-2a, HDL-2b, HDL-3a, HDL-3b, HDL-3c, pre-beta1-HDL and pre-beta2-HDL. **ApoB48 can cross react with ApoB assay but since the levels of these particles are much lower in concentration than ApoB 100-containing lipoproteins, the major contributors to an ApoB result are Lp(a), LDL, VLDL and IDL. *** IDL is not a significant contributor to a standard triglyceride measurement but can be an important particle measured in the hypertriglyceridaemia seen with dysbetalipoproteinaemia. HDL-c – high density lipoprotein cholesterol, Lp(a) – lipoprotein (a), LDL-c – low density lipoprotein cholesterol, refers to a calculated LDL, IDL – intermediate density lipoprotein, VLDL – very low density lipoprotein, CM – chylomicron, CM remnants – chylomicron remnants.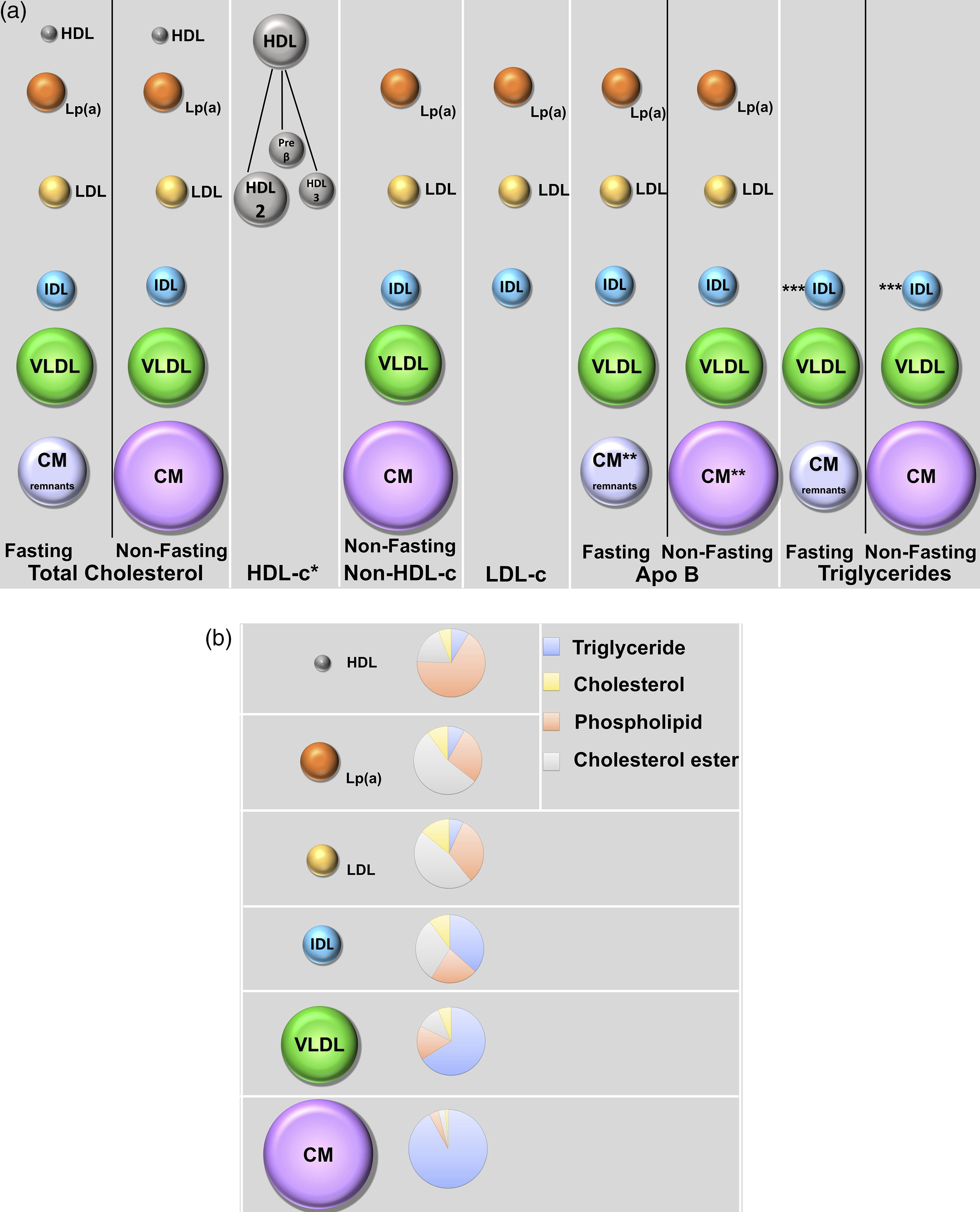
Fasting versus non-fasting lipid profiles
Whilst historically most lipid profiles were performed after a 10–12 h fast, current NICE guidance does not mandate a fasting sample and a non-fasting profile is actively endorsed by EFLM guidance.19,29 However, there is still marked heterogeneity in what laboratories offer, with only one in three European laboratories using a fasting sample as a first line investigation. 30
Non-fasting samples are easier and more convenient for patients, clinicians and laboratories. For laboratories and phlebotomy services, it avoids a bottleneck of patients requiring early morning blood tests. For patients, it allows them to book a blood test at a more convenient time and avoids unnecessary fasting in patients in whom it may present a risk or who find it particularly difficult, such as those with diabetes on hypoglycaemic medications or children. In addition, a non-fasting sample may more accurately reflect a patient’s normal metabolic state since most time is spent in the post-prandial state and several studies have suggested that at a population level cardiovascular risk can be assessed adequately from a non-fasting sample.5,29,31,32 Moreover, when fasting and non-fasting samples were measured in the same individuals, there was high concordance in risk classification of individuals for ASCVD and incident coronary events. 33
Instances when a fasting sample should be considered. 84 For Sampson calculated LDL-c, fasting and non-fasting samples can be used. Fasting is preferred but values may be reported where TG <9.0 mmol/L.

Pre-analytical considerations
Pre-analytical factors to consider when performing a lipid profile.
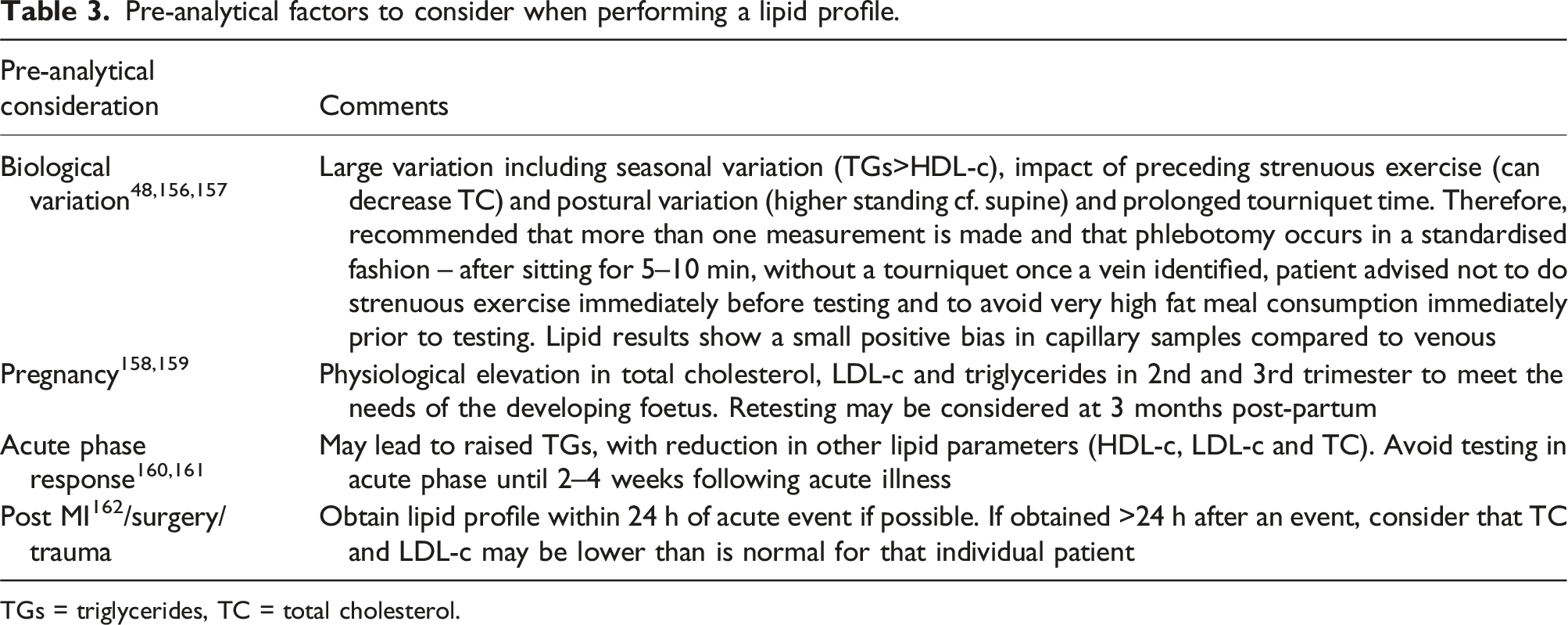
TGs = triglycerides, TC = total cholesterol.
Secondary causes of dyslipidaemias to be considered when performing a lipid profile.

Analytical variation
Whilst biological variation can have an important impact on a patient’s results, analytical variation should also be considered. As with other testing, it is preferable for repeat or follow-up testing to be completed using the same method and for clinicians to be alerted to any method change. In view of total variation (i.e. biological plus analytical variation), these recommendations suggest that a minimum of two measurements are made to determine an individual’s lipid status. 35
Testing intervals
The evidence base for recommendations on lipid testing intervals is weak. 36 Therefore, these recommendations are informed, in the most part, by other national guidance. Minimum retesting interval guidelines produced jointly by the Royal College of Pathologists (RCPath) and Association for Laboratory Medicine (previously known as The Association for Clinical Biochemistry and Laboratory Medicine) suggests a minimum interval of 3 years for those at low risk of ischaemic heart disease and yearly for higher risk cases or those stable on treatment. A study of lipid testing intervals for ∼9000 patients with previous coronary heart disease on pravastatin suggests that, in those who are stable on treatment and below target, testing intervals for lipids could be lengthened to more than a year in view of the size of the combined biological and analytical variation as compared to longer term small fluctuations in cholesterol. However, since other clinical follow-up most commonly occur at this timing interval, it seems prudent to continue to recommend yearly testing. 37 If starting or modifying treatment, three monthly testing is suggested. More frequent measurements may be required in hypertriglyceridaemia, specifically at a one week interval if assessing response to dietary modification or alcohol restriction in severe hypertriglyceridemia or daily in those on total parenteral nutrition or those with hypertriglyceridemia pancreatitis. 38 NICE recommendations include repeat lipid testing within 3 months after treatment initiation and annually as part of a medication review in primary and secondary prevention. In those with severe hypertriglyceridaemia (10.0–20.0 mmol/L) NICE suggest repeat fasting measurements at 5–14 days.
Proposed testing intervals for lipid profiles.
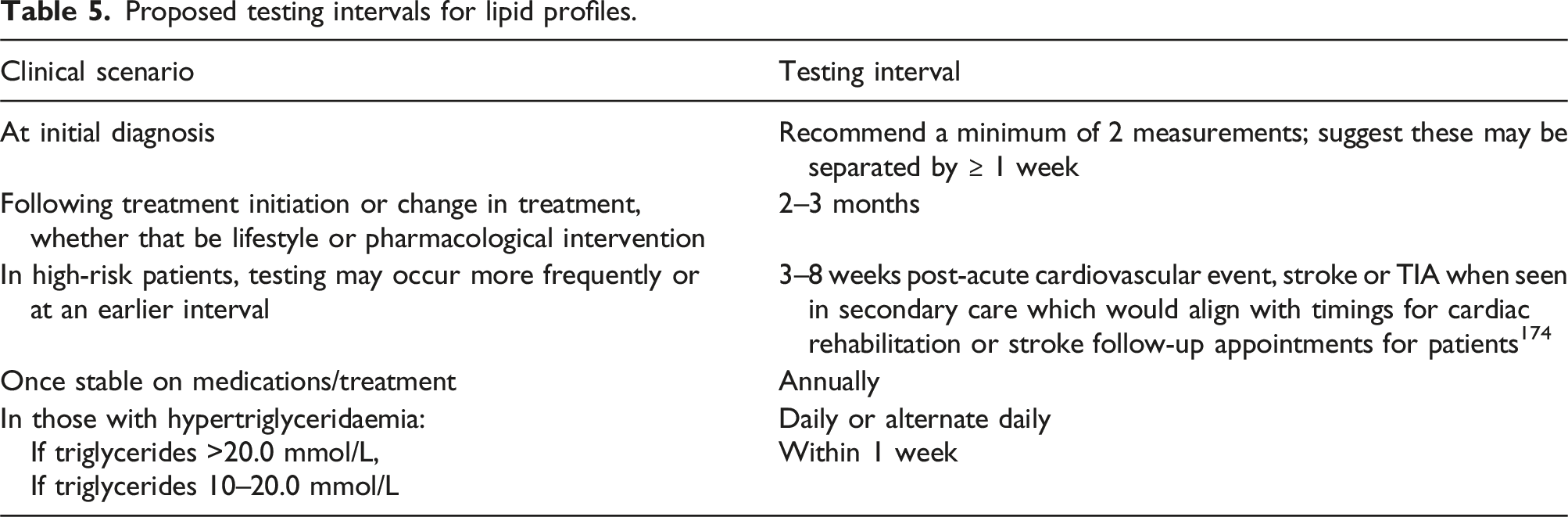
A standard profile should include total cholesterol, HDL-c, triglycerides and a calculation of non-HDL-c, LDL-c and total cholesterol/HDL-c ratio. An enhanced profile may include ApoB and Lp(a). Patients should not routinely be required to fast prior to lipid profile. However, laboratories should offer both options of fasting and non-fasting as there are circumstances when a fasting lipid profile may be necessary. Fasting status should be documented on results. Clinicians should be alerted to pre-analytical factors that may influence lipid result interpretation either directly or via an easily accessible source such as laboratory websites (see Appendix 2). Lipid profile measurement should be performed at least twice initially in view of biological variation. Repeat lipid profiles are suggested at 2–3 months following treatment change or initiation, 3–8 weeks post-acute cardiovascular event, stroke or TIA and annually once a patient is stable on treatment. Repeat measurement should be preferably performed using the same analytical method. More frequent testing may be required whilst managing severe hypertriglyceridaemia.
The wording used in the following and subsequent recommendations denotes the current level of evidence to support that recommendation as per the 2016 ACC/AHA Clinical Guideline Recommendation Classification System. 39
Total cholesterol
Total cholesterol (TC) is a key component of any standard lipid profile; it encompasses the cholesterol carried by LDL, intermediate density lipoprotein (IDL), HDL, Lp(a), VLDL and chylomicrons, see Figure 1, and is correlated with cardiovascular risk.
40
It is required for calculations of non-HDL-c, LDL-c
Total cholesterol can be significantly elevated in secondary dyslipidaemias, see Table 4 (such as hypothyroidism, nephrotic syndrome, cholestatic liver disease, uncontrolled diabetes mellitus and drug causes). Although a further discussion of these is beyond this review, relevant further references are cited here.41,42 However, as a single test, it is not adequate to diagnose the cause of hypercholesterolaemia and, therefore, it is used with other analytes in the lipid profile to further delineate type and cause of dyslipidaemia.
Laboratory methods for cholesterol measurement are standardised and traceable to the National Reference System for Cholesterol (NRS/CHOL) for which the NIST-certified pure cholesterol standard (SRM911b), measured by the NIST isotope-dilution mass spectrometry (IDMS) definitive method provides the accuracy base, and the Centers for Disease Control (CDC) reference method remains the standard which underpins clinical cholesterol testing (Myers 2000). In the CDC reference method, cholesterol ester is extracted first using potassium hydroxide and subsequently hexane and a chromophore is measured after addition of Liebermann-Burchard reagent.43,44 Routinely, total cholesterol is easily and cheaply measured on automated platforms in serum and plasma using enzymatic and colorimetric (CHOD-PAP) methods and reliable point of care methods also available, although laboratory testing is suggested to guide treatment decision.45–47 It is also possible to test, in selected clinical circumstances, using home fingerprick testing.
48
Total allowable error in the US-derived National Cholesterol Education Programme guidance for total cholesterol is 8.9 %, with estimated biological variation contributing 5.2% to this.49,50
Total cholesterol (TC) should be included in all standard and enhanced lipid profiles. Consider a flag to clinicians when TC meets criteria for familial hypercholesterolaemia. It is advisable to comment on the need to initially rule out secondary causes of dyslipidaemia. TC measurement should not be used in isolation for clinical assessment or monitoring of dyslipidaemia.
HDL cholesterol
HDL-c, often referred to as ‘good cholesterol’, is considered anti-atherogenic, although there remains debate about whether it has a causal role in reducing atherosclerosis and Mendelian randomisation studies have not supported this. 51 Its anti-atherogenic or athero-protective potential is, in part, thought to be due to the pivotal role it has in reverse cholesterol transport, returning cholesterol from cells in the periphery to the liver where it is then either re-used or excreted. It has also been attributed direct anti-oxidant, antithrombotic and anti-inflammatory actions.52,53 However, it must also be noted that inflammatory conditions, such as obesity and type 2 diabetes, reduce the concentration of HDL-c complicating interpretation of its anti-inflammatory role. Compared to other lipoproteins, HDL is smallest in size with the highest ratio of protein: lipid giving it the highest density. 54 Its major apolipoprotein, Apo AI, is synthesised by the liver and to a lesser extent the small intestine. After its synthesis, phospholipid and unesterified cholesterol is added to form nascent HDL. Subsequent lecithin-cholesterol acyltransferase (LCAT)-mediated cholesterol esterification and addition of core lipids convert this to mature spherical HDL composed of cholesterol, triglycerides and apolipoproteins. Whilst Apo AI is the major apolipoprotein that forms HDL, others including Apo AII, IV, V, Apo CI, Apo AIII and Apo E are present in some of the HDL subclasses. It is, therefore, important to note that serum HDL-c represents total HDL and refers to multiple subclasses with some differences in their roles and composition. Thus, serum HDL-c is not a direct measure of the anti-atherogenic potential of HDL, the metabolism of which, not yet fully understood, is complex and involves the interaction of multiple apolipoproteins, enzymes and cell surface receptors which ultimately determine its concentration. 55
Although it is still not clear if HDL itself can protect against atherosclerosis, there is now a large body of evidence for its use in predicting ASCVD risk. A wealth of epidemiological evidence has shown that higher HDL-c is associated with lower risk of ASCVD.56,57 Whilst it has not consistently been seen to predict cardiovascular events in those already known to have ASCVD, new meta-analysis level data supports a predictive role in this group.58,59 What is lacking, however, is evidence that therapeutic intervention to increase HDL-c can reduce risk of ASCVD.60–62 Moreover, there is discussion as to whether ‘HDL dysfunction’ exists in those with atherosclerotic disease. However, available functional assays that can assess this have yet to reach clinical practice. Additionally, whilst an inverse relationship between HDL-c and ASCVD exists, this is clearly non-linear at higher values; it plateaus at levels above ∼1.5 mmol with a paradoxical increase in risk of all-cause mortality seen at the upper extremes of HDL-c (approximately 2.4 mmol/L in male and 3.0 mmol/L in female).5,63–65 It is important for laboratories to flag those patients with very low HDL-c to requesting clinicians as they may require further investigation, after exclusion of secondary causes, for inborn errors of metabolism such as hypoalphalipoproteinaemia, as may be caused by Tangier, Fish Eye disease or Apo AI gene mutations such as ApoA1 Milano and very high levels seen in hyperalphalipoproteinaemia.66,67
Clinical decision thresholds for HDL.

Accuracy in measurement is clearly important for several reasons: HDL-c is used to calculate other parameters, so any error in HDL-c directly also impacts non-HDL-c, TC: non-HDL ratio used to calculate CV risk and calculated LDL-c. Furthermore, decision points for increased CV risk are at the lower end of the range where small errors may have large impact on risk calculation. Methods for quantification of HDL-c include cholesterol measurement after precipitation of ApoB-containing lipoproteins, combined with ultracentrifugation as used in the CDC reference measurement procedure (RMP).
68
The reference method is ultracentrifugation.
69
In most laboratories in the United Kingdom, a homogeneous enzymatic colorimetric ‘direct’ HDL-c assay is used and it is important for clinicians to be aware there are differences in measurement between manufacturers and therefore they should be alerted to any change in method and be advised to do follow-up measurements in the same laboratory.
70
Whist functional assays are being developed, they are not yet at the stage where they are routinely employed in clinical practice. In addition, particle number measured by NMR has shown some promise at predicting CV risk but, again, its use is currently limited to research settings.
71
HDL cholesterol should be included in all lipid profiles (standard and enhanced). It should be used to calculate non-HDL-c in all lipid profiles. Suggest very low levels (<0.5 mmol/l) and very high levels (>2.5 mmol/l) are flagged to alert clinicians to the potential need to assess for secondary causes and inherited metabolic diseases (see Section 15).
Triglycerides
Measurement of serum triglycerides encompasses both the liver-derived, triglyceride-rich lipoproteins, VLDL and IDL and chylomicrons and their remnants originating from dietary fat absorbed in the intestine (see Figure 1). A small amount of triglyceride is also carried in HDL and LDL. Circulating triglyceride concentration is dictated by the balance between the production of these lipoproteins and their removal, which is mostly executed by lipoprotein lipase. Genetic mutations in this enzyme are an important cause of familial chylomicronaemia syndrome (FCS). However, whilst FCS is a very rare cause of hypertriglyceridaemia, multifactorial chylomicronaemia syndrome is a much more prevalent, likely polygenic, clinical entity. 72 Chylomicron remnants are mostly cleared by the liver whilst VLDL undergoes some direct hepatic clearance but is also converted, by hepatic triglyceride lipase, to IDL and LDL.
Whilst triglyceride measurement has an important role in the calculation of LDL-c, it is also considered a risk factor for ASCVD. The role of triglycerides, or the residual cholesterol within triglyceride-rich lipoproteins, in ASCVD has recently gained more acceptance but has remained controversial for many years despite several supportive epidemiological studies. In particular, it has been difficult delineate an independent role for triglycerides in view of the interplay between triglyceride concentration and other lipoproteins including the inverse correlation with HDL-c and the concomitant elevation in other non-HDL-c particles seen with hypertriglyceridaemia. However, there is now some evidence of a causal role in coronary heart disease from Mendelian randomisation studies.73–76 Furthermore, a large recent meta-regression of 25 randomised control trials would suggest that reduction of triglyceride concentration leads to a lowering of cardiovascular risk. 77 The REDUCE-IT study, in which icosapent ethyl was used to reduce triglycerides, led to a 25% risk reduction in cardiovascular events and informed its recommendation by NICE. 78 In addition to ASCVD, increased triglycerides are a well-established and significant risk factor for acute pancreatitis. 79 Hypertriglyceridaemia has become increasingly common due to the increased prevalence of dyslipidaemia and insulin resistance associated with overweight and obesity. Rare causes should not be forgotten, such as lipodystrophy, which represents an extreme phenotype of insulin resistance and is thus also associated with hypertriglyceridaemia in combination with low HDL-c.
Until recently triglyceride measurement was recommended to be performed fasting in view of the impact of food intake, with a mean maximal increase of 0.3 mmol/L one to 6 h after eating.29,80 Although many clinical trials continue to use a fasting sample which informs the targets for new drugs that are approved, in addition to the greater convenience of non-fasting samples for patients, two important considerations have informed the many ASCVD guidelines that now recommend lipid profile measurement in the non-fasting state. Firstly, in most of the population, the post-prandial state predominates and thus a non-fasting sample may more accurately reflect the habitual metabolic state and secondly, numerous studies suggest that non-fasting triglycerides may be a better predictor of both cardiovascular and pancreatitis risk.29,81–83 Nonetheless, there clearly remain instances where their fasting measurement is still important as been detailed by Nordestgaard et al see Table 2 in Section 3. 84
Triglyceride measurement is offered routinely in automated clinical laboratories, most commonly using enzymatic colorimetric methods. The current reference method is an isotope-dilution gas chromatography mass spectrometry method which measures total glycerides mirroring what is measured in automated laboratories in most instances. 85 Hypertriglyceridaemia can also interfere with the measurement of other analytes most notably sodium causing pseudohyponatraemia, and amylase leading to spuriously normal values in cases of pancreatitis but other analytes can also be affected. 86 Laboratories routinely obtain an automated lipaemic index on samples as an estimate of sample lipaemia. This is weakly correlated to triglyceride levels although it can be an important tool at high lipaemic index values for identifying hypertriglyceridaemic samples. 87 We would therefore suggest that laboratories consider reflex testing of a lipid profile above a locally agreed cut-off to capture undiagnosed, potentially clinically significant hypertriglyceridaemia. 88 Very high triglyceride concentrations, exceeding the laboratory measurement range, should be remeasured at dilution to provide a meaningful baseline for management.
Clinical decision thresholds for triglycerides. Fasting target (F) and non-fasting target (NF). In those with diabetes and a typical picture of raised triglycerides and low HDL, there may be benefit to additional triglyceride lowering over and above simply statin therapy alone. 12

Finally, pancreatitis risk correlates with the level of hypertriglyceridemia and the highest risk is conferred with those with very severe hypertriglyceridaemia. However, even those with a single one-off measurement with severe hypertriglyceridaemia are at high risk of intermittent increases to very severely elevated concentrations. Extreme hypertriglyceridemia >20.0 mmol/L is associated with pancreatitis and increased morbidity and mortality.79,90,91
Triglycerides should be included in all standard and enhanced lipid profiles, regardless of fasting status. Laboratories should offer both fasting and non-fasting requesting options and aim to apply different interpretive comments and flags on reports depending on fasting status. Laboratories may consider introducing a locally-derived raised lipaemic index cut-off for reflex lipid profile testing to identify previously undiagnosed hypertriglyceridaemia. We suggest newly diagnosed hypertriglyceridaemia >20.0 mmol/L should prompt an urgent alert to the requesting clinician including recommendation for referral to a specialist and investigation into secondary and genetic causes (if not related to suboptimal glycaemic control or alcohol excess).
LDL-c
The role of LDL, an atherogenic lipoprotein which carries apolipoprotein B100, in causing ASCVD is supported by a very strong body of evidence, although of course, other risk factors are known to also contribute 8 Following endothelial damage, the LDL particle enters the intima of blood vessel walls. Macrophage uptake of LDL leads to foam cell formation. Subsequent smooth muscle migration and fibrous fatty plaque formation leads to vessel narrowing or occlusion from plaque growth or rupture and the clinical sequelae of ASCVD including stroke and myocardial infarction. 92 Genetic evidence of a causal role for LDL-c in atherosclerosis comes from loss of function mutations in PCSK9 which lead to both very low LDL-c levels and very low risk of ASCVD along with other Mendelian randomisation studies.93,94 Randomised controlled trials and epidemiological studies consistently show a log linear relationship between LDL-c and ASCVD risk. 12 The corollary is that, for every mmol/L reduction in LDL-c in large clinical trials, there is a 22% reduction in cardiovascular mortality.6,7 LDL-c remains a prominent target and risk biomarker in national and international guidance and many clinical trial endpoints are based on a calculated LDL-c in view of the consistent relationship between LDL-c reduction and ASCVD risk. This includes not only statins and ezetimibe but also newer therapies such as bempedoic acid and inclisiran, a small interfering RNA molecule, as well as the more established PCSK9 monoclonal antibodies. Therefore, ongoing measurement and calculation of LDL-c continues to be of relevance. However, despite optimal LDL-c-directed treatment, ASCVD events still occur indicating that it is not the only atherogenic particle necessary to measure. 95
How should LDL-c be calculated?
The reference method for LDL-c measurement is beta quantification. Using this technique, triglyceride-rich lipoproteins (d <1.006) are separated by ultracentrifugation, physically removing VLDL-c; subsequently cholesterol in ApoB-containing particles is determined after subtraction of measured HDL-c. Although accurate, this analysis is both expensive and slow. 70 In most laboratories, LDL-c is calculated using the FE, which is total cholesterol minus HDL-c and estimated VLDL-c where VLDL-c is estimated by dividing the triglyceride concentration by a constant. 96 The FE was developed over 50 years ago, in the pre-statin era, from a small cohort of predominantly dyslipidaemic patients, none of whom were receiving lipid lowering therapy. The equation has several well-known important limitations: firstly, its use is limited to those with triglycerides ≤4.5 mmol/L as it underestimates LDL-c in hypertriglyceridaemia. In addition, at low LDL-c levels, the equation can underestimate LDL-c with the potential risk of undertreatment of high-risk patients. The original cohort excluded those with an LDL-c <1.8 mmol/L and as it is calculated from the measurement of three analytes (total cholesterol, triglycerides and HDL cholesterol), the bias of these three measurements results in inaccuracy at low concentrations. It was validated using a fasting sample and requires fasting to ensure that chylomicrons don’t negatively impact performance by leading to an overestimation of VLDL and has not been validated in individuals administered statins.13,97 Hypertriglyceridemia is predicted to be more of an issue facing laboratories due to an increased prevalence of non-fasting samples and dyslipidaemia associated with overweight and obesity. Recommended targets for LDL-c, summarised in Table 1, show that clinical decisions are often at the lower end of the LDL-c range such that accuracy at these concentrations is important.
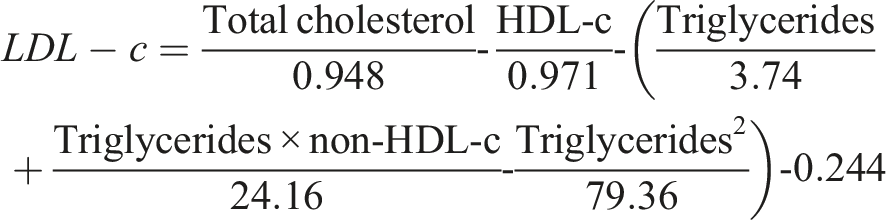
Several newer equations have been developed that may address some of the limitations of the FE, including the Martins-Hopkins in 2013, and its subsequent extended version, and the Sampson-NIH equation in 2020.27,28,98 Prior to the publication of the Sampson-NIH equation, both the EFLM/EAS and the AHA recommended use of the Martin equation in specific cases: in mild hypertriglyceridaemia (2.0–4.5 mmol/L) and in low LDL-c <1.8 mmol/L respectively. However, the original Martin equation was developed from vertical spin density-gradient ultracentrifugation, rather than comparison to the beta quantification reference method and in its original form was not validated in hypertriglyceridaemia, although the recently published extended equation has been developed to allow its use up to 9.0 mmol/L. 98
Using over 18,000 LDL-c results tested using the reference method of beta quantification, Sampson et al developed a formula that outperforms the Friedewald and the original Martin equations in those with hypertriglyceridaemia up to 9.0 mmol/L (800 mg/dL), in patients with low LDL-c and is equally good in those with normal triglyceride concentrations. 28 In addition, the Sampson-NIH equation may be used in non-fasting samples; when non-fasting results were compared to a Roche direct LDL measurement, there was a good correlation (correlation coefficients of 0.95 and 0.93 for samples from females and males respectively). The Sampson-NIH equation is not without limitations; a paper published by Sajja et al suggested that it could underestimate LDL-c at lower levels. However, this study is limited by the fact it did not use a reference method to measure LDL-c and additionally the LDL-c concentrations at which it suggested there may be an issue were below commonly used clinical decision targets (1.03 mmol/L). 99 A further study retrospectively compared ∼7000 samples measured using ultracentrifugation and calculated values using FE, Sampson-NIH and Martin-Hopkins equations – this showed there was still inaccuracy in these newer equations above triglycerides of 4.5 mmol/L , although they both out-performed the FE. 100
In summary, whilst in most cases the extended Martin-Hopkins and Sampson-NIH equation produce similar results, our recommendation is for UK laboratories to institute the Sampson-NIH equation for three main reasons (see Box 1). The first is that, unlike the Martin equation, Sampson-NIH equation is a single equation that is relatively easy to employ with laboratory information systems as opposed to requiring multiple equations dependent on the triglyceride and HDL-c results. Secondly, it was developed using the reference method and may have potentially better performance in the hypertriglyceridaemic patient. Finally, despite being available since 2013, the Martin equation was not taken up by laboratories as it was initially proprietary. As with any change in method, it is important that laboratory users are informed.
Direct measurement of LDL-c has been used as an alternative option to calculation by formula and, whilst it can be used in a non-fasted sample, there are concerns about its relationship to outcome data. 101 Furthermore, it is more expensive than a calculated LDL-c and its performance is method dependent and lacks standardisation between laboratories. 70 Although it has a possible role to quantify LDL-c with significant hypertriglyceridaemia (>9.0 mmol/L), when direct LDL-c measurement was compared to the CDC reference method, marked and clinically relevant bias was seen. 102 For these reasons, this guideline does not recommend using direct LDL-c in hypertriglyceridaemic samples and suggests measurement of ApoB as an alternative in these circumstances.
Since Lp(a)-associated cholesterol will be measured as part of LDL-c there is international guidance suggesting an LDL-c correction factor should be used for those with suspected or known raised Lp(a).84,103 However, this is not included in this guidance due to the significant variability in cholesterol content of Lp(a) (6–58%) and, moreover, correction has not been validated for use in routine clinical practice.
104
LDL-c cholesterol should be calculated in all standard lipid profiles where TG <9.0 mmol/L. Consider non-HDL-c or ApoB where not possible. Use of the Sampson equation is preferable for calculation of LDL-c in fasting and non-fasting samples. Fasting is preferred but values may be reported where TG <9.0 mmol/L. The Sampson equation has a lower reporting limit of 0.5 mmol/L. It is recommended that laboratories flag results according to guideline-based thresholds (see Section 15). Correction of LDL-c for Lp(a)-associated cholesterol is not advocated in current routine clinical practice.
Non-HDL-c
Using the simple calculation of total cholesterol (mmol/L) minus HDL cholesterol (mmol/L), non-HDL-c provides an estimate of pro-atherogenic ApoB-containing lipoproteins: LDL, IDL, VLDL and Lp(a) and, in non-fasted samples, chylomicrons and their remnants (see Figure 1). Its measurement plays a significant role in NICE guidance for assessment of statin therapy and is included in both European and US guidance. Within the European guidelines, non-HDL-c is used for risk calculation within SCORE2 and SCOREOP,105,106 whilst in American guidance it is noted as a risk-enhancing factor for ASCVD likely due to primary hypercholesterolaemia when 4.9–5.6 mmol/L and the cut-off for abnormal levels in childhood are ≥3.7 mmol/L, although insufficient evidence was noted for non-HDL-c treatment targets.
A meta-analysis by Robinson et al showed that for each 1% reduction in non-HDL-c an equivalent reduction was seen in risk of coronary heart disease. 107 There is evidence that it may predict CV risk more accurately than LDL-c or ApoB, although data is conflicting here and, as expected, its measurement is very highly correlated with both LDL-c and ApoB.5,108 It can be calculated in non-fasting samples and has been found to be more predictive of CV risk in those on statins when compared to LDL-c and ApoB. 109 However, it has rarely been used as a primary endpoint in clinical trials and targets are often an estimated equivalent value to LDL-c, typically 0.8 mmol/L higher based on the estimated VLDL concentration, as discussed by Nordestgaard et al. 84 Furthermore, like calculated LDL-c, it relies on the ability to reliably measure HDL-c, which is limited at high triglyceride concentrations (>10 mmol/L). In addition, amongst national and international guidance there are differences in how non-HDL-c targets are estimated from the original LDL-c targets. For example, the Canadian guidance uses 2.4 mmol/L versus 2.5 mmol/L used by JBS to equate to a LDL-c of 1.8 mmol/L. This exemplifies the issue of the lack of standardisation of this conversion and the need for specific, evidence-based non-HDL-c targets.
In summary, non-fasting, non-HDL-c is sufficient to assess response to routine lipid lowering therapy, but the evidence for treatment targets is significantly less than for LDL-c. Although non-HDL-c is a convenient alternative option to LDL-c when it cannot be calculated, in circumstances where triglycerides are elevated, measurement of ApoB should be considered.
Non-HDL cholesterol should be reported within a standard and enhanced lipid profile and calculated as total cholesterol (mmol/L) – HDL cholesterol (mmol/L). Non-fasting, non-HDL-c is sufficient to assess response to routine lipid lowering therapy. It is recommended that laboratories flag results according to guideline-based thresholds (see Section 15).
Total cholesterol/ HDL-c ratio
Total cholesterol/ HDL-c ratio is required for the calculation of 10-year ASCVD risk using QRISK3 and QRISK3-lifetime. However, it is important to note that this ratio should be interpreted with particular caution as it may be reassuringly normal due to a high HDL-c even though a patient has a high non-HDL-c and LDL-c. In patients with very high HDL-c (>2.5 mmol/l), risk may be underestimated.
Total cholesterol: HDL-c should be reported by labs to allow risk calculation in QRISK3 and QRISK3-lifetime. A normal ratio should be interpreted with caution when this is related to a very high HDL-c (>2.5 mmol/L). Under these circumstances, it is recommended laboratories append a comment to advise interpreting TC/HDL ratios with caution as they may underestimate risk.
Lipoprotein(a)
Lipoprotein(a), an LDL-like particle with pro-atherogenic and proinflammatory effects, is an independent major risk factor for ASCVD and calcific aortic valve stenosis.110,111 A Lp(a) of approximately 250 nmol/L nearly doubles the risk of ASCVD irrespective of other risk factors and patients with very high levels of Lp(a) (>430 nmol/L) have a similar ASCVD risk as those with untreated heterozygous familial hypercholesterolaemia (HeFH). 111 Compelling evidence for Lp(a) as a causal risk factor for ASCVD has led to development of novel Lp(a) lowering therapies which are currently in phase III trials.
Lp(a) concentration is mainly (>90%) genetically determined with an autosomal co-dominant inheritance and, unlike other lipoproteins, levels are not reduced by diet, exercise or common lipid lowering treatments like statins. It is possible that statins may slightly increase Lp(a) but this increase is not clinically significant.
112
Because Lp(a) concentrations remain relatively stable throughout life, a single measurement of Lp(a) is sufficient in most patients unless a secondary cause of elevated Lp(a) is suspected such as untreated overt hypothyroidism, chronic kidney disease, end stage renal failure on dialysis, nephrotic syndrome, autoimmune disorders and treatment with growth hormone. Twofold increases in Lp(a) levels can also be seen in pregnancy.111,113,114 It can also increase post-menopausally.
115
Lp(a) distribution varies with ethnicity with higher median levels in South Asian and Black individuals (Median 31 and 75 nmol/L, respectively) compared to the White population (median 19 nmol/L). Despite these differences, the linear relationship between Lp(a) concentrations and risk of major cardiovascular events remains consistent across different ethnicities.
111
As per guidance from HEART UK, Lp(a) measurement should be considered in patients with (a) A personal or family history of premature atherosclerotic cardiovascular disease. (b) First degree relatives with raised serum Lp(a). (c) Familial hypercholesterolaemia (FH), or other genetic dyslipidaemias. (d) Calcific aortic valve stenosis. (e) Moderate (10–15%) 10-year risk of cardiovascular event. A single measurement of Lp(a) is adequate in most patients unless a secondary cause for elevated Lp(a) is identified. Denka based assays with calibrators traceable in nmol/L to WHO/IFCC reference material are the only recommended assays at present. Results should be reported in nmol/L and conversion from mass to molar unit should be avoided.
Measurement – When and how to measure Lp(a)
Adapted from HEART UK recommendation for Lp(a) measurement in those with the following characteristics.

The risk of cardiovascular disease based on classified Lp(a) concentration.
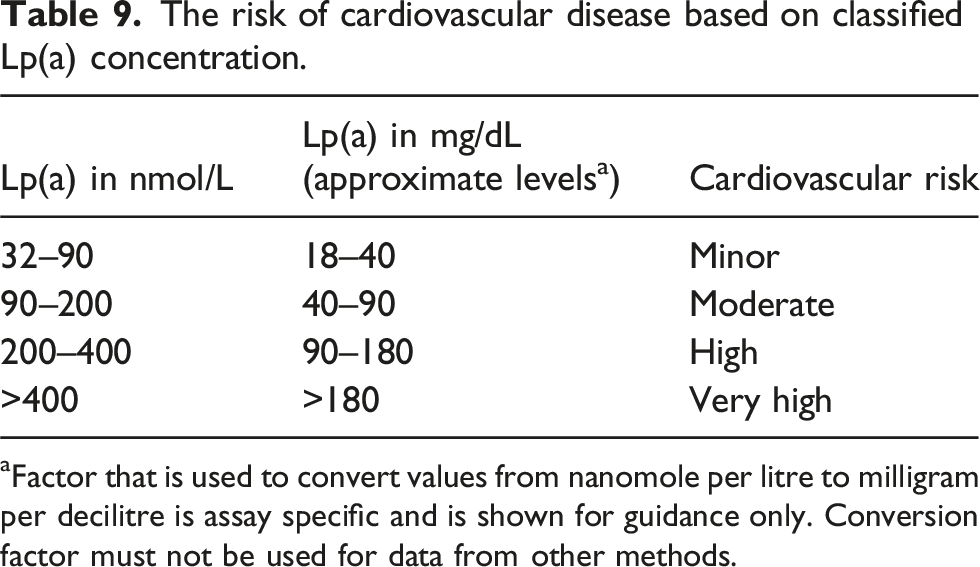
aFactor that is used to convert values from nanomole per litre to milligram per decilitre is assay specific and is shown for guidance only. Conversion factor must not be used for data from other methods.
Measurement of Lp(a) is challenging. This is due to significant heterogeneity in apo(a) sizes within and between individuals mainly as a result of huge variation in number of repeated Kringle IV type 2 (KIV2) domain in apo(a). 117 Available commercial immunoassays use polyclonal antibodies that cross react with KIV2. This leads to underestimation of Lp(a) in individuals with small apo(a) isoforms (lower number of KIV2 repeats) and overestimation of Lp(a) in those with larger isoforms.111,117 At present, immunoassays using Denka reagents are the most reliable method because they incorporate a range of calibrators covering different apo(a) sizes to partially address the isoform size issue; each calibrator is traceable in molar units (nmol/L) to the WHO/IFCC reference material. Future work should focus on developing truly isoform-insensitive commercial immunoassays.
Currently, most laboratories in the United Kingdom still use non-standardised assays and report Lp(a) in the mass unit (mg/dL). As these immunoassays measure the protein component of Lp(a) and not the entire particle, we recommend instead using an isoform-insensitive assay and reporting in molar unit which correctly reflects the particle numbers of Lp(a) binding to antibodies in isoform-insensitive assays. 117 Conversion of mass unit to molar unit and vice versa is not recommended as the ratio of mass to molecular weight is not constant.111,116,117
Clinical role of Lp(a) measurement
Failure to incorporate Lp(a) concentration in QRISK3 and other risk assessment tools significantly underestimates ASCVD risk in patients with elevated Lp(a). Recently, a risk calculator based on UK Biobank data, which includes Lp(a) in addition to other ASCVD risk factors, was introduced by European Atherosclerosis Society consensus statement (https://www.lpaclinicalguidance.com/). This calculator estimates lifetime CVD risk with and without Lp(a) concentration and highlights risk is underestimated considerably when elevated Lp(a) is not included. It also shows modifying other risk factors like LDL-c or blood pressure can reduce patient’s overall CV risk substantially even if Lp(a) is not changed. Whilst no specific Lp(a) lowering pharmacological treatment is available at present, using this calculator will help with more accurate risk stratification which is necessary for clinicians and patients to manage other modifiable risk factors more intensively.111,116
Once a patient is diagnosed with elevated Lp(a), aggressive management of lifestyle modifications, weight, blood pressure, glucose and dyslipidaemia are crucial. For management of dyslipidaemia in patients with Lp(a) > 90 nmol/L, achieving greater than 50% reduction in non-HDL-c, or alternatively non-HDL-c target of < 2.5 nmol/L (LDL-c <∼1.8 mmol/L), is recommended based on expert consensus opinion. 116
ApoB
ApoB has two isoforms: ApoB100 is a constituent part of LDL, IDL, VLDL and Lp(a) and ApoB48, a truncated form of ApoB100, binds to chylomicrons and chylomicron remnants. Whilst ApoB immunoassays measure both isoforms, ApoB100-containing lipoproteins predominate overwhelmingly, even in non-fasted samples where chylomicrons are less than one percent of the sample. Thus ApoB measurement in practice provides a measure of LDL, IDL, VLDL and Lp(a). 118 Importantly, a single ApoB molecule binds a single lipoprotein particle and, therefore, measurement of total ApoB provides a direct measure the number of atherogenic particle numbers as compared to the calculated parameter of non-HDL-c which estimates cholesterol content in all ApoB-containing particles. Similarly, ‘broad cut’ LDL-c, as estimated by beta quantification, upon which LDL-c calculations are based, is a measure of cholesterol content in IDL, LDL-c and Lp(a)-c but does not give any indication of particle number, which may be of relevance in those with a predominance of small dense LDL particles (see Figure 1). 119 Furthermore, there is evidence that, excepting Lp(a) and CM remnants, all ApoB-containing particles are equally atherogenic such that ApoB may be a superior estimate compared to LDL-c of atherosclerotic risk. Epidemiological studies have supported this with evidence that it is superior to LDL-c and non-HDL-c in risk prediction and of greater use in assessing and guiding lipid lowering therapy, particularly in those already on statins.120–122 Furthermore, when ApoB and LDL-c are discordant, the cardiovascular outcome has been found to be more likely to follow the ApoB result. 118 Thus, in assessing ASCVD risk, many lipid specialists consider measurement of ApoB to be more valuable than non-HDL-c or LDL-c. Furthermore, it can be measured with greater accuracy particularly at low concentrations. 12
However, there are several reasons why its use is not yet widespread, and it is not ubiquitously available in UK laboratories. There remains controversy over whether it offers added benefit over the cheaper measure of non-HDL-c and it currently lacks assessments of cost-effectiveness. Furthermore, it does not have validated decision thresholds as clinical trial endpoints are based on LDL-c, not ApoB and, as such, clinicians are less familiar with its use. Moreover, whilst it can be tested in non-fasting samples, assays may be limited due to cross-reactivity of triglycerides and light-scattering by chylomicrons and VLDL that can be seen at high concentrations of these particles. 123
In view of the clear advantages of this assay, however, it has already been introduced in selected instances into international guidance to date. It has been introduced as a secondary target in ESC guidance to direct therapy after LDL-c targets are reached (very high risk: ApoB <65 mg/dL, high risk: ApoB <80 mg/dL and moderate risk: ApoB <100 mg/dL) as well as being recommended as the best measure in those with hypertriglyceridemia, diabetes and obesity, metabolic syndrome or very low LDL-c because of the risk that direct or calculated LDL-c may underestimate both cholesterol within LDL but also the ApoB-containing lipoprotein burden. 12 Recent National Lipid Association consensus guidance has introduced ApoB thresholds to correspond to those for LDL-c and non-HDL-c (60 mg/dL in very high risk, 70 mg/dL in high risk and 90 mg/dL in those at borderline to intermediate risk for ASCVD). 124 An enhanced equation combining ApoB has also been developed to improve LDL-c estimates where the LDL concentration is in the lower range. 125
It is also suggested for use in diagnosing familial combined hyperlipidaemia (ApoB>120 mg/dL combined with triglycerides >1.5 mmol/L and family history). EFLM suggests using ApoB measurement as a secondary target in mild-moderate hypertrigylceridaemia (2.0–10.0 mmol/L), diabetes, obesity or metabolic syndrome as the use of ApoB can identify the presence of dyslipidaemia due to remnant particles and small dense LDL. The cut-off of >130 mg/dL, a concentration that is estimated to be equivalent to an LDL-c of >4.1 mmol/L is labelled a risk-enhancing factor in American Heart Association guidance and if triglycerides are ≥2.6 mmol/L, it is a relative indication to test ApoB. Furthermore, it has an increasingly important role in the diagnosis of familial dysbetalipoproteinaemia (FDBL or Type III), which has lipid parameters that may overlap with other lipid disorders, making diagnosis from a standard profile sometimes difficult. There have been several algorithms published to optimise its use in screening for this monogenic condition using either its ratio to non-HDL or a Sampson-NIH novel equation.126–128 A recent comparison of these diagnostic criteria undertaken in the UK Biobank found that the non-HDL-c/ApoB ratio >4.91, as proposed by Boot et al, showed the best diagnostic accuracy overall and identified a reasonable number of individuals that could benefit from APOE genotype testing to confirm a diagnosis of FDBL.129,130 Measurement of ApoB also has clear roles in hypobetalipoproteinaemia and abetalipoproteinaemia and, in those conditions associated with lipoprotein X, an abnormal and large lipoprotein lacking ApoB100, such as LCAT deficiency or primary biliary cirrhosis, where using the ratio of total cholesterol to ApoB can help to confirm the presence of lipoprotein X. 131
ApoB is measured most commonly by automated immunoassay (immunonepholometry or immunoturbidimetry). There is ongoing work led by the International Federation of Clinical Chemistry and Laboratory Medicine to standardise measurement and improve analytical performance. 132
In summary, whilst ApoB measurement cannot currently replace LDL-c and non-HDL-c, it is likely that its use will become more widespread as further evidence accumulates to inform thresholds and already there are particular clinical scenarios, in a specialist setting, when it would be of particular use including dysbetalipoproteinaemia, hypobetalipoproteinamia, abetalipoproteinaemia and dyslipidaemia associated with diabetes/obesity and conditions where lipoprotein X may be present.
ApoB is recommended to form part of an enhanced lipid profile for the following indications: Initial investigation for familial dysbetalipoproteinaemia (non-HDL-c/ApoB). Hypo- and abetalipoproteinaemia diagnosis. For risk assessment in those with hypertriglyceridaemia. Initial investigation for presence of lipoprotein X when used in a ratio with total cholesterol.
ApoA1
ApoA1 is the major apolipoprotein that carries HDL and facilitates HDL binding to the cell surface receptor, ABCD1. 133 It is strongly correlated to HDL-c levels and, as with HDL-c, is predictive of a lower cardiovascular risk. 134 ApoA1 was an independent predictor of fatal and nonfatal MI in those with known coronary artery disease. 135 When used in a ratio with ApoB (ApoB:ApoA1), a higher ratio value is correlated with an increased risk fatal myocardial infarction. 136 However, since ApoA1 concentration is strongly correlated with that of HDL-c, there remains debate as to its use over and above HDL-c and other HDL-c calculated parameters alone. There is international standardisation 137 and it is measurable in an automated laboratory using immunoassay making measurement easy and quick, although it is not as cheap as other lipid profile components and not yet available in all routine clinical laboratories.
Therefore, while one role of ApoA1 may be its use in the ApoB:ApoA1 ratio as part of an additional work-up in patients at borderline ASCVD risk, there is not enough evidence that it is superior to HDL-c to recommend that it should form part of a standard or enhanced lipid profile. Of course, Apo A1 is important in the diagnosis of monogenic disorders such as Familial hypoalphalipoproteinaemia, Tangier disease, LCAT deficiency (familial LCAT deficiency and Fish Eye disease) and hyperalphalipoproteinaemia due to CETP deficiency, hepatic lipase deficiency, endothelial lipase deficiency or loss of function mutations in scavenger receptor, class B type 1 (SRB1).
138
Apolipoprotein A1 is not currently recommended as part of a routine or enhanced lipid profile. Apolipoprotein A1 is indicated for the investigation of possible hypo-or hyperalphalipoproteinaemia within specialist services.
Lipoprotein subfractions
Testing of the subclasses of lipoproteins, in particular LDL and HDL subclasses, has been considered by some to have clinical utility – for example, in the context of those with a predominance of atherogenic small dense LDL who are known to have an increased risk of coronary heart disease or those lower levels of HDL2.139,140 There are multiple techniques that have been used to determine the profile of lipoprotein particles such as nuclear magnetic resonance spectroscopy, electrophoresis, High Performance Liquid Chromatography and Vertical Auto Profile. However, there is a lack of standardisation of these assays regarding which particles are measured limiting the current use of this testing in clinical practice.
141
Furthermore, the impact of measuring lipoprotein subfractions on clinical outcome or cost-effectiveness data is lacking.
142
Therefore, whilst it is feasible that subfraction testing may have an important role to play in the future, in particular for refining cardiovascular risk measurements in those currently deemed non-high risk by traditional risk factors and current lipoprotein testing, currently there is not enough evidence to recommend their use for routine practice.
Testing of lipoprotein subfractions is not currently recommended in routine clinical practice.
Paediatrics
Dyslipidaemia amongst children is increasingly common due to the epidemic of diabetes and obesity within the United Kingdom.
143
Furthermore, genetic causes of dyslipidaemia such as heterozygous and homozygous familial hypercholesterolaemia are important to diagnose in the paediatric population to allow optimal early treatment.
144
In keeping with this, Lp(a) screening has been recommended in certain clinical circumstances by international guidance.
145
There are, as yet, no UK harmonised reference ranges for lipids in the paediatric population, although these guidelines would encourage that UK specific intervals are established. The Canadian CALIPER database is a vital resource that can be used by laboratories to inform specific references ranges for paediatric lipid profiles.146–148 There are a few references to paediatrics within international guidelines and diagnostic criteria; these include total cholesterol and LDL-c cut-offs for familial hypercholesterolaemia (>6.7 mmol/L and >4.0 mmol/L, respectively) and a table of abnormal values in American guidance which are mainly based on consensus opinion (TC ≥ 5.1 mmol/L, LDL-c, ≥3.4 mmol/L, non-HDL-c ≥3.7 mmol/L, HDL-c <1.0 mmol/L, triglycerides ≥ 1.1 mmol/L (0–9 years) and ≥ 1.4 mmol/L (10–19 years)). Further evidence is needed to inform recommendations in this area.
Use paediatric specific references ranges in children. Consider Lp(a) testing in those <18 years who have possible or definite familial hypercholesterolaemia, ischaemic stroke of unknown cause, or if there is a relevant family history of premature cardiovascular disease or very high Lp(a).
Flagging and critical results
A vital role that the laboratory plays is the alerting and interpretation of abnormal lipid results for requesting clinicians. This encompasses three main functions: firstly, the alerting of critical results that require urgent action; secondly, the interpretation of individual or a pattern of abnormal results that may require further investigation or management; and finally, the flagging of results that are around key decision limits that would affect patient management. With respect to lipid profiles, in common with EFLM guidance, we recommend that rather than reference interval limits, it is more clinically valuable to flag lipid values at key decision points. For laboratories to do this effectively, it is important for requesting clinicians to inform laboratories if the lipid profile is requested for primary or secondary prevention management. Furthermore, it is recommended that for paediatric testing, a local reference range should be derived.
Critical results
The current recommendations from the Royal College of Pathologists on communicating clinical results do not include any lipid parameters. 149 In practice, many laboratories will communicate urgently samples with severe hypertriglyceridaemia due to the well-known risk of pancreatitis as discussed in section ‘Triglycerides’. NICE guidance recommends urgent specialist review if triglycerides >20.0 mmol/L with a caveat that this is not secondary to poorly controlled glycaemia or alcohol excess. 19 EFLM suggests that triglycerides above 10.0 mmol/L should prompt the following interpretative comment ‘severe hypertriglyceridemia with high risk of acute pancreatitis’. 150 In view of the risk of pancreatitis, we suggest urgent alert (within 24 h) of a patient sample with triglycerides >20.0 mmol/L.
Flagging
Recommended thresholds for laboratories to flag results and suggested comments.
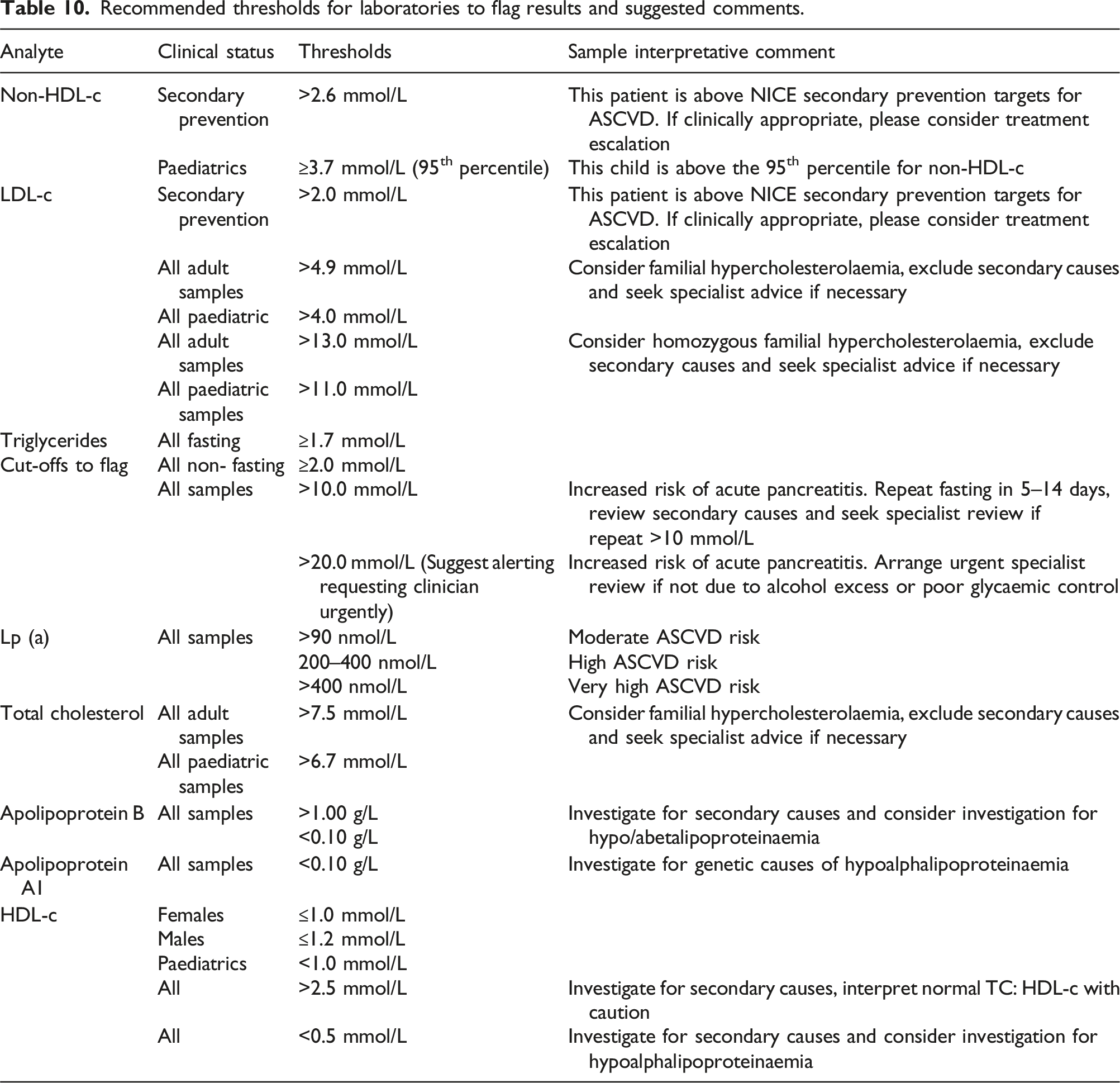
In practice, there are multiple targets internationally for LDL-c and non-HDL-c, but here we state those recommended by NICE. However, as per NHS England guidance, in secondary prevention, LDL-c and non-HDL-c should be reduced as much as possible.
151
It is advisable to decide locally a strategy for reflex testing where necessary. Laboratory systems should allow clinicians to input if the testing is requested for primary or secondary prevention, and if feasible, whether the patient is taking lipid lowering therapy.
Lipid profile flags should be based on thresholds related to increased ASCVD risk.
Conclusion
Lipid testing is a key tool in assessing and managing cardiovascular risk. This consensus guidance provides recommendations to standardise lipid testing and reporting in UK laboratories. Key recommendations include the change from Friedewald equations to using Sampson-NIH equations for calculation of LDL-c, that laboratories should offer fasting and non-fasting testing, recommendations for the composition of all standard lipid profiles and the indications for Lp(a) and ApoB in enhanced lipid profiles.
Supplemental Material
Supplemental Material - Standardising lipid testing and reporting in the United Kingdom; a joint statement by HEART UK and The Association for Laboratory Medicine
Supplemental Material for Standardising lipid testing and reporting in the United Kingdom; a joint statement by HEART UK and the Association for Laboratory Medicine by Julia S Kenkre, Tina Mazaheri, R Dermot G Neely, Handrean Soran, Dev Datta, Peter Penson, Paul Downie, Alexandra M Yates, Katharine Hayden, Mayur Patel and Jaimini Cegla in Annals of Clinical Biochemistry.
Footnotes
Acknowledgements
This article was prepared at the invitation of the Clinical Sciences Reviews Committee of the Association for Clinical Biochemistry and Laboratory Medicine.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DD: Advisory boards/ speaker fees over last 3 years – Amarin – Novartis – Daiichi-Sankyo – Chiesi – Ultragenyx – Lilly. PD has received Speaker/Consulting fees from the following: Amgen, Amarin, Besins Healthcare, Daiichi Sankyo, Sanofi and Sobi. PD has received financial support for travel and accommodation to attend national/international conferences from Amgen and Sanofi. JC has received speaker/consultancy fees or research grants from Amgen, Sanofi, Amryt, Pfizer, Novartis, Daiichi Sankyo, Akcea, Ultragenyx, Chiesi, Silence Therapeutics and Verve Therapeutics. PP owns four shares in AstraZeneca PLC.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
JC.
Contributorship
JC and MP came up with the initial concept for the article. JSK drafted the initial article with contribution from TM. All authors were involved in reviewing and editing the article.
Supplemental material
Supplemental material for this article is available online.
Appendix 1. Short summary of recommendations
Recommendations
Section 3.
Standard lipid profile1. A standard profile should include total cholesterol, HDL-c, triglycerides and a calculation of non-HDL-c, LDL-c and total cholesterol/HDL-c ratio.
2. An enhanced profile may include ApoB and Lp(a).
3. Patients should not routinely be required to fast prior to lipid profile. However, laboratories should offer both options of fasting and non-fasting as there are circumstances when a fasting lipid profile may be necessary. Fasting status should be documented on results.
4. Clinicians should be alerted to pre-analytical factors that may influence lipid result interpretation either directly or via an easily accessible source such as laboratory websites (see Appendix 2).
5. Lipid profile measurement should be performed at least twice initially in view of biological variation. Repeat lipid profiles are suggested at 2–3 months following treatment change or initiation, 3–8 weeks post-acute cardiovascular event, stroke or TIA and annually once a patient is stable on treatment. Repeat measurement should be preferably performed using the same analytical method. More frequent testing may be required whilst managing severe hypertriglyceridaemia.
Section 4.
Total cholesterol1. Total cholesterol (TC) should be included in all standard and enhanced lipid profiles.
2. Consider a flag to clinicians when TC meets criteria for familial hypercholesterolaemia. It is advisable to comment on the need to initially rule out secondary causes of dyslipidaemia.
3. TC measurement should not be used in isolation for clinical assessment or monitoring of dyslipidaemia.
Section 5.
HDL cholesterol1. HDL cholesterol should be included in all lipid profiles (standard and enhanced).
2. It should be used to calculate non-HDL-c in all lipid profiles.
3. Suggest very low levels (<0.5 mmol/l) and very high levels (>2.5 mmol/l) are flagged to alert clinicians to the potential need to assess for secondary causes and inherited metabolic diseases (see Section 15).
Section 6.
Triglycerides1. Triglycerides should be included in all standard and enhanced lipid profiles, regardless of fasting status.
2. Laboratories should offer both fasting and non-fasting requesting options and aim to apply different interpretive comments and flags on reports depending on fasting status.
3. Laboratories may consider introducing a locally-derived raised lipaemic index cut-off for reflex lipid profile testing to identify previously undiagnosed hypertriglyceridaemia.
4. We suggest newly diagnosed hypertriglyceridaemia >20.0 mmol/L should prompt an urgent alert to the requesting clinician including recommendation for referral to a specialist and investigation into secondary and genetic causes.
Section 7.
LDL-c1. LDL-c cholesterol should be calculated in all standard lipid profiles where TG <9.0 mmol/L. Consider non-HDL-c or ApoB where not possible.
2. Use of the Sampson-NIH equation is preferable for calculation of LDL-c in fasting and non-fasting samples. Fasting is preferred but values may be reported where TG <9.0 mmol/L. The Sampson-NIH equation has a lower reporting limit of 0.5 mmol/L.
3. It is recommended that laboratories flag results according to guideline-based thresholds (see Section 15).
4. Correction of LDL-c for Lp (a)-associated cholesterol is not advocated in current routine clinical practice.
Section 8.
Non-HDL cholesterol1. Non-HDL cholesterol should be reported within a standard and enhanced lipid profile and calculated as total cholesterol (mmol/L) – HDL cholesterol (mmol/L)
2. Non-fasting, non-HDL-c is sufficient to assess response to routine lipid lowering therapy.
3. It is recommended that laboratories flag results according to guideline-based thresholds.
Section 9.
Total cholesterol: HDL-c1. Total cholesterol: HDL-c should be reported by labs to allow risk calculation in QRISK3 and QRISK3-lifetime.
2. A normal ratio should be interpreted with caution when this is related to a very high HDL (>2.5 mmol/L). Under these circumstances, it is recommended laboratories append a comment to advise interpreting TC/HDL ratios with caution as they may underestimate risk.
Section 10.
Lipoprotein (a)1. As per guidance from HEART UK, Lp(a) measurement should be considered in patients with (a) A personal or family history of premature atherosclerotic cardiovascular disease. (b) First degree relatives with raised serum Lp(a). (c) Familial hypercholesterolaemia (FH), or other genetic dyslipidaemias. (d) Calcific aortic valve stenosis. (e) Moderate (10–15%) 10-year risk of cardiovascular event.
2. A single measurement of Lp(a) is adequate in most patients unless secondary cause for elevated Lp(a) is identified.
3. Denka based assays with calibrators traceable in nmol/L to WHO/IFCC reference material are the only recommended assays at present.
4. Results should be reported in nmol/L and conversion from mass to molar unit should be avoided.
Section 11.
Apolipoprotein B1. ApoB is recommended to form part of an enhanced lipid profile for the following indications:
a. Initial investigation for familial dysbetalipoproteinaemia (non-HDL-c/ApoB).
b. Hypo- and abetalipoproteinaemia diagnosis.
c. For risk assessment in those with hypertriglyceridaemia.
d. Initial investigation for presence of lipoprotein X when used in a ratio with total cholesterol.
Section 12.
Apolipoprotein A11. Apolipoprotein A1 is not currently recommended as part of a routine or enhanced lipid profile.
2. Apolipoprotein A1 is indicated for the investigation of possible hypo-or hyperalphalipoproteinaemia in a specialist setting.
Section 13.
Lipoprotein subfractions1. Testing of lipoprotein subfractions is not currently recommended in routine clinical practice.
Section 14.
Paediatrics1. Use paediatric specific references ranges in children.
2. Consider Lp(a) testing in those <18 years who have possible or definite familial hypercholesterolaemia, ischaemic stroke of unknown cause or if there is a relevant family history of premature cardiovascular disease or very high Lp(a).
Section 15.
Flagging and critical results1. Lipid profile flags should be based on thresholds related to increased ASCVD risk.
At a glance guidance for clinicians and laboratories
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
