Abstract
Background
Triglyceride-rich, low-density lipoproteins (TG-rich LDL) have been reported as an oxidized lipoprotein species in patients with severe liver disease. Using TG-rich LDL as an immunogen, we obtained a monoclonal antibody (G11-6) that reacted with TG-rich LDL from patients with liver disease and with metal-oxidized LDL only in the early process of the oxidation reaction. This study determined the G11-6-reactive lipoproteins in hypertriglyceridemic serum.
Methods
Serum samples from healthy volunteers (n = 12) and hypertriglyceridemic patients (n = 9) were fractionated by gel filtration and subjected to a sandwich enzyme-linked immunosorbent assay (ELISA) using G11-6 and polyclonal anti-apolipoprotein B antibodies.
Results
Small LDL and larger lipoproteins reacted with G11-6. G11-6-reactive small LDL was identified in both the healthy subjects and hypertriglyceridemic patients, whereas G11-6-reactive larger lipoproteins were found only in the hypertriglyceridemic patients.
Conclusions
G11-6 is a useful tool for detecting increased large oxidized lipoproteins in hypertriglyceridemic patients.
Introduction
The oxidation of lipids and lipoproteins plays a key role in early atherogenesis and is involved in various diseases.1,2 Among lipoprotein species, the small, dense, low-density lipoproteins (LDL) are more susceptible to oxidation than larger, buoyant LDL. 3 Many studies have reported on the relationship between the concentration of small dense LDL and the development of atherosclerosis.3–5 However, there are few reports of serum oxidized lipoproteins other than small dense LDL, except for a report on the presence of phosphatidylcholine hydroperoxides in isolated remnant lipoproteins. 6
We developed a new monoclonal antibody, called G11-6, by immunizing mice with triglyceride (TG)-rich LDL isolated from the serum of a cholestatic patient with severe liver disease. 7 A sandwich enzyme-linked immunosorbent assay (ELISA) using G11-6 with polyclonal anti-apolipoprotein B (apoB) antibodies detected copper-oxidized LDL only in the early process of the oxidation reaction, while G11-6 reacted with neither native lipoproteins nor copper-oxidized LDL in the late process of the oxidation reaction, supporting the specificity of G11-6 for weakly oxidized lipoproteins. 7 In our previous study, G11-6 reacted with TG-rich LDL in patients with liver disease and with small LDL in normal controls. 7 To further understand weakly oxidized lipoproteins in serum, the present study examined the lipoprotein species in hypertriglyceridemic serum that reacted with G11-6.
Materials and methods
Blood was drawn from 12 healthy volunteers (controls) and nine patients with hypertriglyceridemia after an overnight fast. Hypertriglyceridemia was diagnosed according to the 2007 Guidelines of the Japan Atherosclerosis Society: LDL-cholesterol (LDL-C) ≥ 140 mg/dL (3.64 mmol/L), high-density lipoprotein-cholesterol (HDL-C) < 40 mg/dL (1.04 mmol/L), or TG ≥ 150 mg/dL (1.65 mmol/L). Serum was separated from the blood by centrifugation at 2000 g for 10 min and then stored at 4℃ until use.
Serum samples (0.3 mL each) were subjected to gel filtration chromatography on a Superose 6 column (GE Healthcare, Little Chalfont, England) in a liquid chromatography apparatus (Shimadzu, Kyoto, Japan), and 0.5-mL fractions were collected, as described previously. 7 The lipoprotein fractions were stored at 4℃ and analysed for lipids and lipoproteins within one day, as described below.
To confirm the elution position of each lipoprotein fraction in this chromatographic system, a serum sample was obtained from a hypertriglyceridemic patient, and then separated into three fractions by ultracentrifugation: chylomicron (CM)-intermediate-density lipoprotein (IDL) (d < 1.019 kg/L), LDL (1.019 < d < 1.063 kg/L), and HDL plus other serum proteins (d > 1.063 kg/L), as reported previously. 7 Each fraction was applied to the same column system, and its elution position was determined by measuring total cholesterol (TC) and TG in the eluates (data not shown).
Phospholipids (PL), TC, TG, LDL-C, and HDL-C were measured using automated enzymatic methods and commercial kits (Sekisui Medical, Tokyo, Japan). Malondialdehyde (MDA)-modified LDL was detected using a MDA-LDL ELISA kit (Sekisui Medical) with a commercial monoclonal antibody (ML25; Sekisui Medical). The lipoproteins detected by this kit are designated as MDA-LDL here.
G11-6 was used as the solid-phase antibody in combination with polyclonal anti-apolipoprotein B antibodies as the detecting antibody in a sandwich ELISA, which is referred to as the G11-6 ELISA here. 7 Serum samples were diluted 20-fold with 50 mmol/L phosphate buffer (pH 7.4) containing 150 mmol/L sodium chloride and 1 mmol/L ethylenediaminetetraacetic acid before the G11-6 ELISA. Serum G11-6-reactive lipoprotein concentrations were defined as the ratio of the absorbance of each serum sample to that of the control serum obtained from a patient with liver disease. To evaluate the reproducibility of the G11-6 ELISA, serum samples with high or low G11-6-reactive lipoprotein concentrations were measured 10 times to evaluate the within-run variation and four times on four consecutive days to evaluate the between-run variation.
To investigate the possible interaction between G11-6 and lipoprotein(a) [Lp(a)], Lp(a)-deficient serum was prepared as follows. A polyclonal goat anti-Lp(a) antibody (Abcam, Cambridge, MA) was covalently bound to magnetic tosylactivated Dynabeads (Invitrogen), according to the manufacturer’s instructions. Magnetic beads bound to non-immune goat IgG (Sigma-Aldrich, St. Louis, MO) were used as a control. The antibody-bound beads were incubated with serum from a hypertriglyceridemic patient overnight at 4℃. After magnetic separation, the resulting Lp(a)-depleted serum was chromatographed on a Superose 6 column, as described above. The eluted fractions were subjected to the G11-6 ELISA and were analysed for Lp(a) content using a latex-enhanced turbidimetric immunoassay [Lp(a)-Latex SEIKEN Kit; Denka Seiken, Tokyo, Japan].
The serum G11-6-reactive lipoprotein concentrations and clinical parameters of the groups, except sex, were compared using the non-parametric Mann–Whitney U-test. The influence of sex differences was analysed using Fisher’s exact probability test. The statistical analyses were performed using Statcel (OMS, Saitama, Japan). The relationships among the elution positions of G11-6 - or ML25-reactive lipoproteins were analysed using the split plot design analysis of variance (ANOVA) on log-transformed concentrations of G11-6-reactive lipoproteins for G11-6 ELISA and MDA-LDL for MDA-LDL ELISA. The statistical analysis was performed using StatFlex (Artech, Osaka, Japan). Values of P < 0.05 was considered to indicate statistical significance.
Results
Clinical characteristics
Clinical parameters and serum lipids in studied groups.
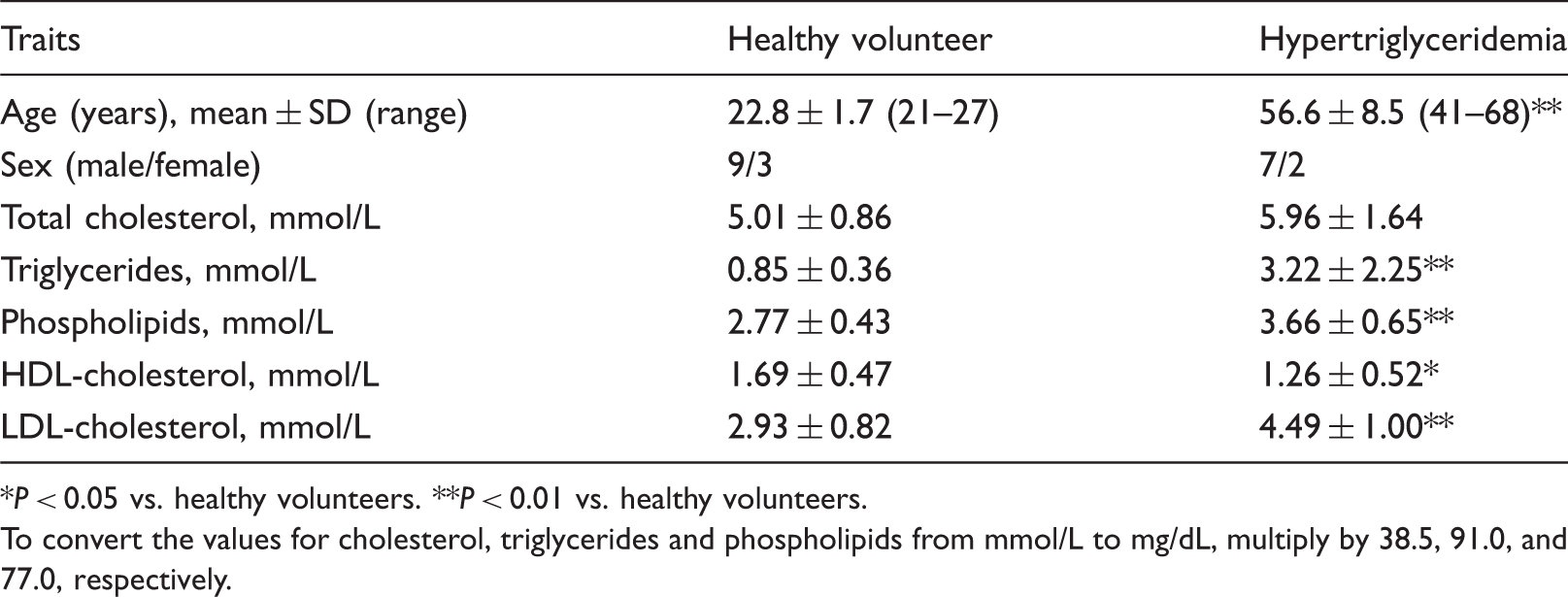
P < 0.05 vs. healthy volunteers. **P < 0.01 vs. healthy volunteers.
To convert the values for cholesterol, triglycerides and phospholipids from mmol/L to mg/dL, multiply by 38.5, 91.0, and 77.0, respectively.
G11-6-reactive lipoprotein concentrations in normal and hypertriglyceridemic serum
The serum G11-6-reactive lipoprotein concentrations did not differ significantly between the healthy subjects and hypertriglyceridemic patients (0.32 ± 0.31 vs. 0.36 ± 0.70, mean ± SD; P = 0.337). In our assay, the within-assay coefficient of variation was 4.1% for high concentrations (1.72 ± 0.07) and 7.7% for low concentrations (0.52 ± 0.04). The between-assay coefficient of variation was 9.7% for high concentrations (1.61 ± 0.16) and 13.7% for low concentrations (0.35 ± 0.05).
Gel filtration of serum fractions isolated by ultracentrifugation
The CM-IDL fraction that was isolated by ultracentrifugation eluted in fractions 5–16, and TC and TG peaked in fractions 5 and 10 (data not shown). The LDL fraction eluted in fractions 11–16; TC peaked in fractions 13–14, and low TG peaks were observed in fractions 5 and 13. The fraction containing HDL plus other serum proteins eluted in fractions 19–27, and TC and TG peaked in fractions 21–23.
Gel filtration of normal and hypertriglyceridemic serum
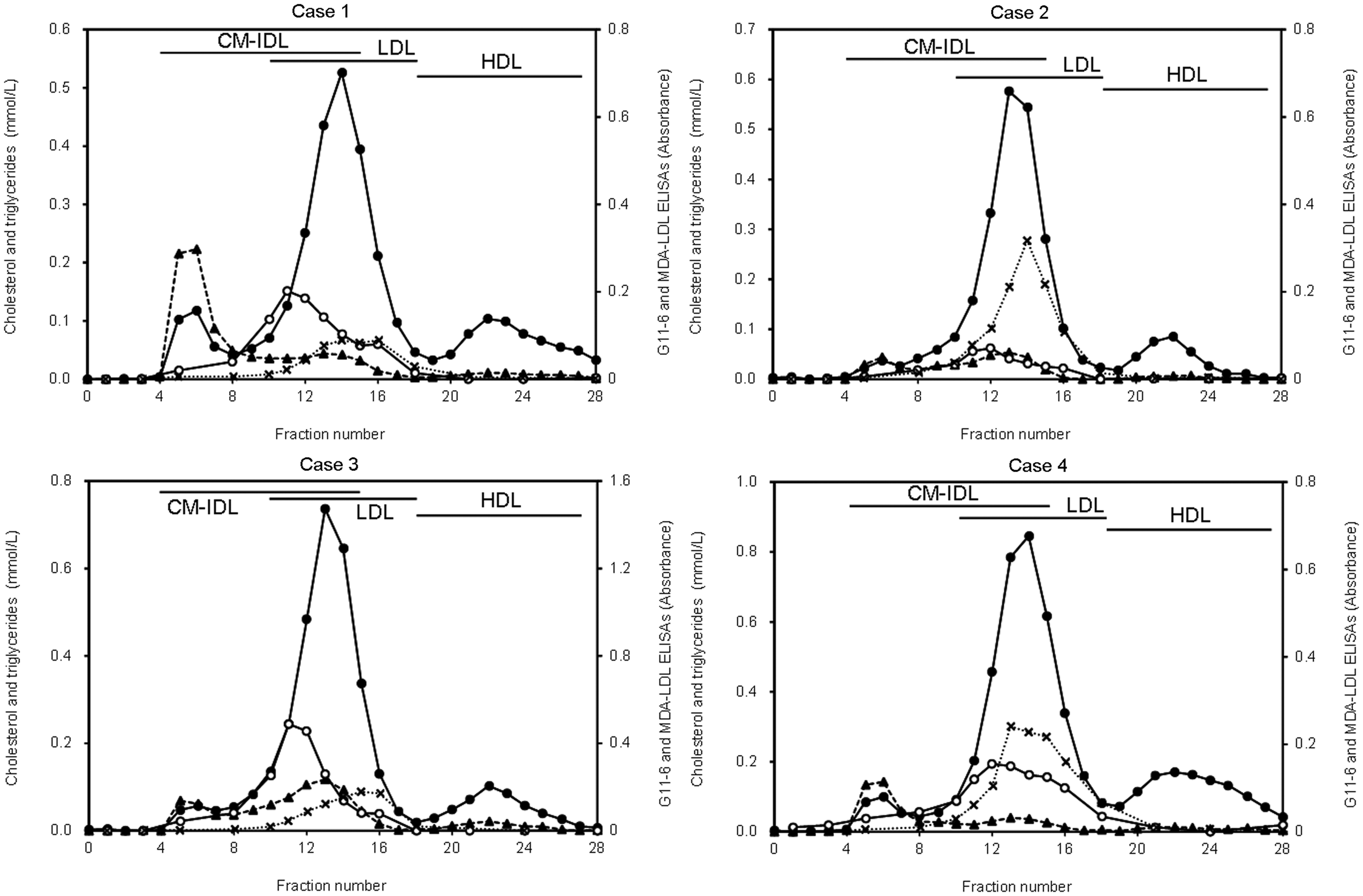
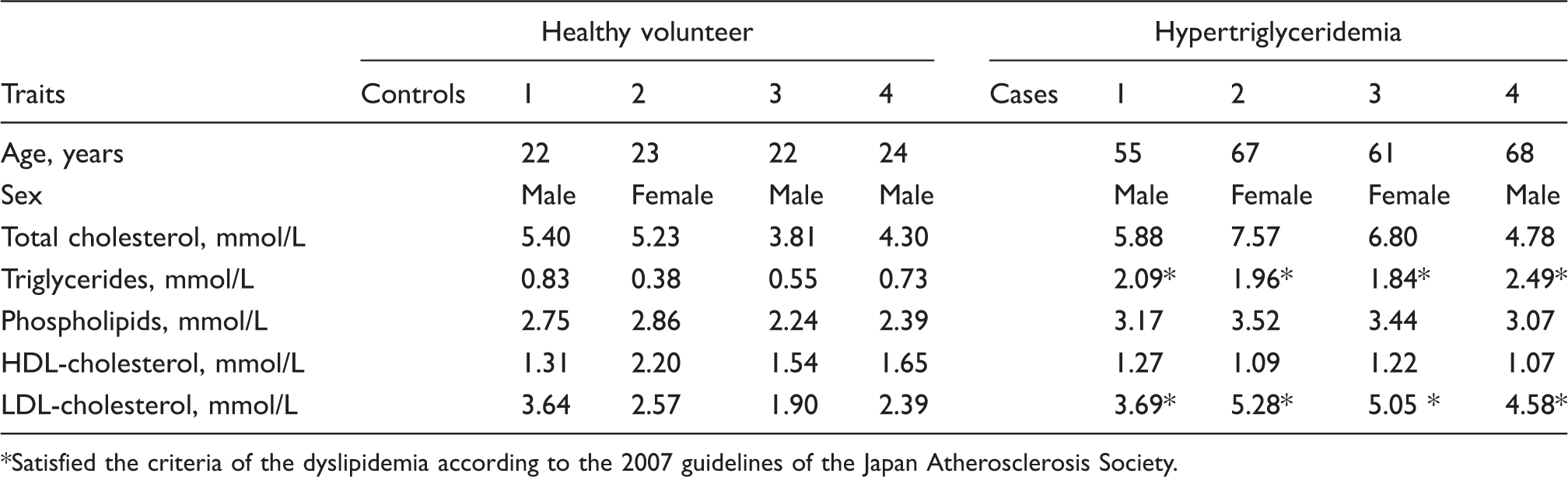
Figures 1 and 2 illustrate the gel filtration results for four of the 12 healthy subjects (control) and four of the nine hypertriglyceridemic patients, respectively. They were selected randomly from each group and represented the typical elution profile for each group. Their clinical data are summarized in Table 2. Cases 1–4 had elevated TG and LDL-C.
Typical gel filtration chromatography elution profile for serum from healthy subjects. Total cholesterol (•, solid line, left axis), triglycerides (▴, dashed line, left axis), ELISA using G11-6 (○, solid line, right axis), and MDA-LDL (×, dotted line, right axis). Typical gel filtration chromatography elution profile for serum from hypertriglyceridemic patients. Total cholesterol (•, solid line, left axis), triglycerides (▴, dashed line, left axis), G11-6 ELISA (○, solid line, right axis), and MDA-LDL (×, dotted line, right axis). Clinical characteristics of the controls and the cases. Satisfied the criteria of the dyslipidemia according to the 2007 guidelines of the Japan Atherosclerosis Society.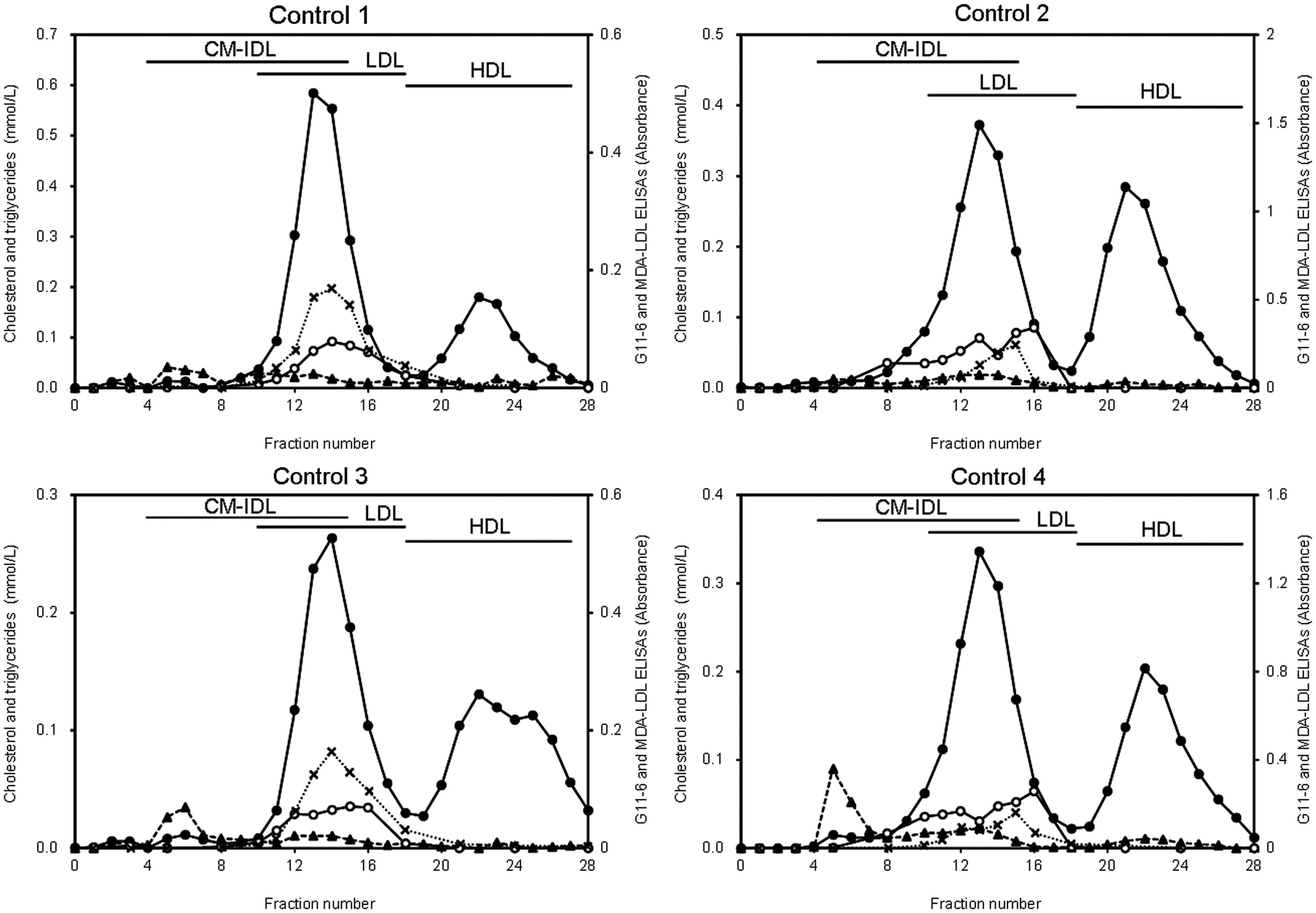
The elution profiles of the four controls were similar (Figure 1): LDL-C eluted in fractions 11–16 and peaked in fractions 13–14; HDL-C eluted in fractions 19–28 and peaked in fractions 22–23. MDA-LDL eluted in fractions 12–16 and peaked in fractions 13–15 in all four controls. G11-6-reactive lipoproteins eluted in fractions 10–16 and peaked in fractions 14–16 in all four controls.
The four cases also gave similar elution profiles (Figure 2): LDL-C eluted in fractions 11–16 and peaked in fractions 13–14; HDL-C eluted in fractions 19–28 and peaked in fractions 22–23. MDA-LDL eluted in fractions 12–16 and peaked in fractions 13–15 in all four cases. G11-6-reactive lipoproteins eluted in fractions 8–16 and peaked in fractions 10–13 in all four cases.
According to the elution position of the lipoproteins in the gel filtration chromatography, fractions 11–12, 13–14, and 15–16, which we named Fractions I, II, and III, respectively, appeared to correspond to the large TG-rich lipoproteins, large buoyant LDL, and small dense LDL, respectively. Fractions I, II, and III had the G11-6-reactive lipoprotein concentrations of 2.85 ± 1.56 (mean ± SD), 5.80 ± 4.00, and 7.25 ± 4.31, respectively, in the healthy subjects, and 1.84 ± 1.89, 1.30 ± 1.12, and 0.87 ± 0.82, respectively, in the hypertriglyceridemic patients (Figure 3(a)). According to the split plot design ANOVA, significant differences in the G11-6-reactive lipoprotein concentrations were detected between the groups of healthy subjects and hypertriglyceridemic patients (P = 0.0007) and among the fractions I to III (P = 0.0386). Additionally, there was a significant group × fraction interaction in the G11-6-reactive lipoprotein concentrations (P < 0.0001). On the other hand, MDA-LDL concentrations of Fractions I, II, and III were 28.6 ± 26.2 U/L, 111.0 ± 77.1 U/L, and 84.2 ± 4.69 U/L, respectively, in the healthy subjects, and 53.6 ± 42.4 U/L, 268.1 ± 239.6 U/L, and 194.1 ± 184.5 U/L, respectively, in the hypertriglyceridemic patients (Figure 3(b)). According to the split plot design ANOVA, significant differences in the MDA-LDL concentrations were detected among the fractions I to III (P < 0.0001), whereas no significant difference was found between the groups of healthy subjects and hypertriglyceridemic patients (P = 0.1506). Additionally, there was no significant group × fraction interaction in the MDA-LDL concentrations (P = 0.9580).
Distributions of G11-6-reactive lipoproteins and MDA-LDL classified by elution position in gel filtration: (a) G11-6 ELISA and (b) MDA-LDL ELISA. Note that log scale is used for the G11-6-reactive lipoprotein and MDA-LDL concentrations. G11-6-reactive lipoprotein concentrations were defined as the ratio of the absorbance of each sample to that of the control serum obtained from a patient with liver disease. According to the elution position of the lipoproteins in the gel filtration chromatography, fractions 11–12, 13–14, and 15–16, which we named Fractions I, II, and III, respectively, appeared to correspond to the large TG-rich lipoproteins, large buoyant LDL, and small dense LDL, respectively. Difference between the groups of healthy subjects and hypertriglyceridemic patients was significant (P = 0.0007) for the G11-6-reactive lipoprotein concentrations, but not significant for the MDA-LDL concentrations, according to the split plot design ANOVA. Additionally, group × fraction interaction was significant in the G11-6-reactive lipoprotein concentrations (P < 0.0001), but not significant in the MDA-LDL concentrations.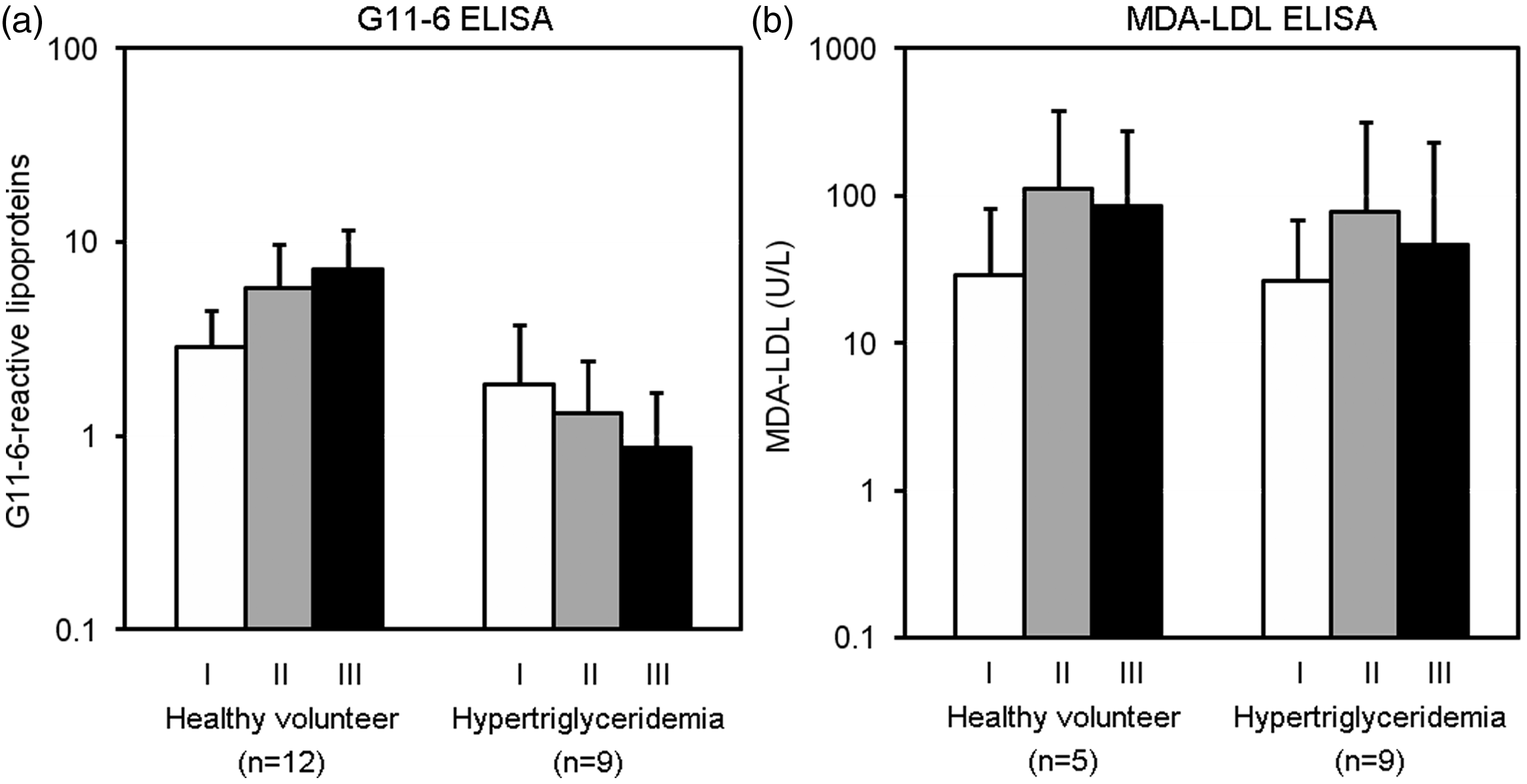
Lp(a) depletion experiment
The absence of Lp(a) in the eluates from the Superose 6 column loaded with Lp(a)-depleted hypertriglyceridemic serum was confirmed by turbidimetric measurement of Lp(a). The eluates had essentially the same elution profiles with and without Lp(a) depletion, indicating that G11-6 did not recognize Lp(a) (data not shown).
Discussion
When free radical-mediated oxidation of unsaturated fatty acids in LDL occurs, a chain reaction leads to the massive formation of PL hydroperoxides.6,8 The PL hydroperoxides undergo carbon–carbon bond cleavage via alkoxyl radicals in the presence of transition metals, forming short-chain unesterified aldehydes and short acyl-chain PL. 8 The short-chain unesterified aldehydes such as MDA and 4-hydroxy-2-nonenal bind to the positively charged amino groups of apoB.8,9 The MDA-modified apoB can be detected with monoclonal antibody ML-25 in a MDA-LDL ELISA. 9 By contrast, Itabe and Ueda10,11 reported that short acyl-chain PL is recognized by DLH3 antibody.
In our previous report, G11-6 reacted with copper-oxidized LDL during the early process of the oxidation reaction. 7 ML-25 and DLH3, however, showed time-courses different from that for G11-6. ML-25 reacted with copper-oxidized LDL in both the early and late processes of oxidation, and DLH3 reacted in the latest process of oxidation. 7 Furthermore, G11-6 did not react with artificially prepared MDA-modified LDL, in contrast to ML-25 and 4E6, another monoclonal antibody against oxidized LDL reported by Holvoet et al. 12 These data clearly show the unique immunological property of G11-6 among the reported antibodies to oxidized LDL. In addition, the amino acid sequence in the hypervariable region of G11-6 was not found in BLAST search (unpublished data). Given that no detergent was needed for the immunoreaction between G11-6 and oxidized LDL, the epitope of G11-6 must be exposed on the surface of LDL particles, although its identity remains to be elucidated.
The serum G11-6-reactive lipoprotein concentrations measured by G11-6 ELISA did not differ significantly between the healthy subjects and hypertriglyceridemic patients. In comparison, the G11-6-reactive lipoprotein elution profiles in gel filtration chromatography differed significantly between them: G11-6-reactive lipoproteins eluted most abundantly in Fraction III (small LDL) in the healthy subjects and in Fraction I (lipoproteins larger than LDL) in the hypertriglyceridemic patients (Figure 3). Many studies have reported the relevance of small LDL to oxidation. Small dense LDL is more oxidizable in vitro than large buoyant LDL4,5 and is able to enhance foam cell formation by THP-1 macrophages without copper-induced oxidation. 4 Furthermore, ML-25, or the antibody to MDA-LDL, has been reported to react with isolated small dense LDL. 9 LDL isolated from hypertriglyceridemic serum has reduced affinity for the LDL receptor 13 and a prolonged plasma half-life (3.2 days for hypertriglyceridemic LDL vs. 2.0 days for normal LDL), which may underlie the higher oxidizability in vivo of small LDL. 14
The elution position of the large G11-6-reactive lipoproteins coincided with that of the CM-IDL fraction. We speculate that the large G11-6-reactive lipoproteins observed in hypertriglyceridemic patients are oxidized remnant lipoproteins. Remnant lipoproteins are reported to have elevated thiobarbituric acid-reactive substance (TBARS) and greater oxidizability in vitro than very low-density lipoproteins (VLDL). 15 Since CM elute in the void volume with our column system (data not shown), they are not the G11-6-reactive lipoproteins. The presence of circulating oxidized Lp(a) in normal and hypertensive subjects has been reported. 16 With our gel filtration system, Lp(a) elutes at a position similar to that of remnant lipoproteins. However, G11-6 does not seem to react with Lp(a), according to the result of the Lp(a) depletion experiment.
Fraction I had different triglyceride contents in the healthy subjects and hypertriglyceridemic patients, as shown in Figures 1 and 2. Here, we defined Fraction I as the lipoprotein fraction with a density < 1.019 g/mL and elutes in gel filtration chromatography at the positions that G11-6-reactive lipoproteins are detected. Hence, Fraction I may be composed of IDL, VLDL, and possibly small contaminating amounts of LDL. In hypertriglyceridemic patients, IDL and VLDL should increase in Fraction I. Since these lipoproteins are rich in triglyceride and poor in cholesterol, their increase should result in the elevation of triglyceride in Fraction I.
Isolated remnant lipoproteins are reported to contain detectable amounts of phosphatidylcholine hydroperoxides. 17 In addition, the hypertriglyceridemic VLDL remnants induce cholesteryl ester accumulation in cultured macrophages as efficiently as oxidized LDL. 18 Hypertriglyceridemic remnant lipoproteins also induce the expression of proatherothrombogenic molecules such as intercellular adhesion molecule-1, vascular cell adhesion molecule-1, and tissue factor, in endothelial cells via a mechanism that is suppressed by antioxidants. 19 Hypertriglyceridemic remnant lipoproteins have an echinocytogenic effect on red blood cells, which is also suppressible by antioxidants. 16 Consequently, remnant lipoproteins have chemical and biochemical properties corresponding to those of oxidized lipoproteins. Thus the immunological change in remnant lipoproteins may have been caused by oxidation.
The observed increase in oxidized remnant lipoproteins in hypertriglyceridemic serum may be partly explained by delayed metabolism of remnant lipoproteins in hypertriglyceridemic patients. Cortner et al. 20 reported the prolonged clearance of CM remnant lipoproteins (CM-free d < 1.006 fraction), with half-times of 14.1 ± 9.7 and 50.7 ± 20.8 min in controls and hypertriglyceridemic patients, respectively. They speculated that this delay was largely due to the overproduction of VLDL particles in the liver and the consequent competition between VLDL and CM remnants for hepatic uptake via apoE receptor-mediated endocytosis. Although VLDL remnants were not mentioned in the literature, it is likely that their clearance is also delayed in hypertriglyceridemic patients.
Interestingly, ML-25 did not react with the large lipoproteins that were reactive with G11-6 (Figure 2). The oxidative change in this fraction might be too weak to be recognized by ML-25. We speculate that the clearance of TG-rich lipoproteins is more rapid than that of small dense LDL, 14 and therefore, little MDA-modification occurs in TG-rich lipoproteins.
The acquisition of an adequate quantity of standard substance for G11-6 ELISA remains to be solved. Although TG-rich LDL isolated from patients with advanced liver disease can be used as a standard substance in G11-6 ELISA, it is difficult to obtain a large volume of serum from patients. The use of copper-oxidized LDL might solve this problem, as reported by Kotani et al. 9 in their MDA-LDL ELISA.
In conclusion, G11-6 has the advantage of detecting large oxidized lipoproteins, probably oxidized remnant lipoproteins, which are increased in hypertriglyceridemic patients. G11-6 might be useful in elucidating the role of large oxidized lipoproteins in cardiovascular disease.
Footnotes
Acknowledgements
We are grateful to the Central Research Laboratory, Faculty of Health Sciences, Hokkaido University, for providing the work space and equipments. Toshihiro Sakurai is a Research Fellow of the Japan Society for the Promotion of Science.
Declaration of conflicting interests
None.
Funding
This research was supported by a Grant-in-Aid for Scientific Research from the Japan Society for the Promotion of Science and also by the Regional Innovation Strategy Support Program of the MEXT, Japan.
Ethical approval
The study was approved by the ethics review board at the Faculty of Health Sciences, Hokkaido University (approval number 08-57).
Guarantor
HC.
Contributorship
TS and HC researched literature and conceived the study. A Ichikawa, AIkuta, H Furumaki, S-PH, SJ, ST and H Fuda were involved in lipoprotein separations and lipid measurements. NW, YT, MF, CS and H Furukawa were involved in providing samples and patient recruitment. SK and TS were involved in the preparation and characterization of monoclonal antibodies. HN was involved in discussion concerning the role for abnormal lipoproteins. TS wrote the first draft of the manuscript. All authors reviewed and approved the final version of the manuscript.
