Abstract
Introduction
Cryoproteins are proteins which precipitate at temperatures below 37°C. Each cryoprotein is unique in terms of its thermal profile (temperature of precipitation), appearance and protein composition; however, precipitation of cryoproteins is generally reversible. The proteins which can cryoprecipitate are the immunoglobulins (IgG, IgA and IgM) and fibrinogen. Cryoprecipitating immunoglobulins will precipitate from both serum and plasma whereas cryofibrinogen, which consists of fibrinogen-fibrin complexes, precipitates only from plasma. 1 The term cryoglobulin is often used for cryoprecipitating immunoglobulins.
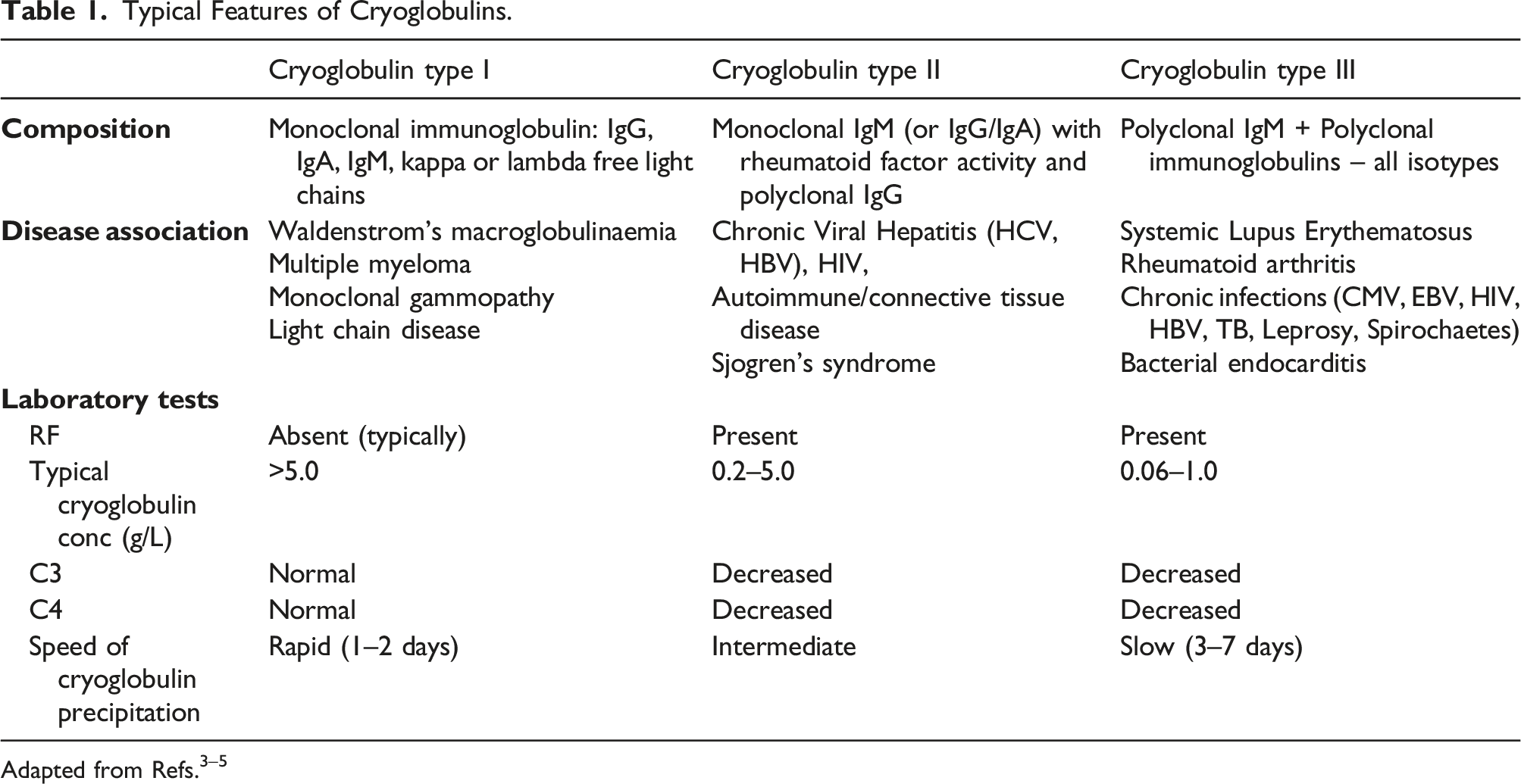
Typical Features of Cryoglobulins.
Robust preanalytical and analytical processes are critical for the detection of cryoproteins. There is significant variation in practice in the detection, analysis and reporting. A survey in 2007 of 137 laboratories participating in the UK National External Quality Assessment Service for Immunology, Immunochemistry & Allergy (UK NEQAS IIA) quality control program showed significant variation in the laboratory processes which highlighted the need for standardisation of the detection, analysis and reporting of cryoglobulins.
In 2009, Sargur et al 3 proposed best practice guidance on cryoglobulin evaluation and emphasized the need for international and national consensus on the minimum standards for cryoglobulin evaluation to avoid missed diagnoses and to improve the comparability of results. Other groups have made similar recommendations. 6 Despite the clinical importance of cryoprotein testing, there is no external quality assurance scheme.
Due to the limitations of temperature controlled sample transportation UK NEQAS IIA has developed the first digital interpretative external quality assurance (Cryo-dEQA) scheme for cryoprotein investigation. This paper describes the findings of the pilot Cryo-dEQA scheme to date and results from a survey of scheme participants with respect to cryoprotein analysis. We propose a set of minimum laboratory requirements for cryoprotein analysis.
Rationale for a digital EQA scheme for cryoproteins
Every element of cryoprotein analysis lacks harmonisation.
Traditional EQA schemes involve distribution of patient samples to participating laboratories but this is not practical for cryoprotein samples. Patients with cryoproteins often have complex clinical issues so taking large volumes of sample for distribution would be dangerous. There are also potential issues with sample stability and the cost of global transportation of samples, particularly if temperature control is required. We developed an image based digital EQA scheme to examine the procedures currently in use for cryoprotein analysis and interpretation. We describe the results of the pilot phase and the lessons derived.
Methods
Digital Cryoprotein EQA scheme (Cryo-dEQA)
UK NEQAS IIA and the Protein Reference Unit at St George’s Hospital, London, collaborated to develop a digital EQA scheme for cryoproteins.
Each distribution consisted of a single case, presenting images of the cryoprotein samples after 7 days incubation at 4°C and 37°C, together with the clinical scenario. The participating laboratory used this initial information to decide whether further testing was required. The scheme had been designed so participants must commit to an interpretation to be able to see ‘downstream’ images of further investigation. Participants were expected, where appropriate, to define the type of cryoprotein present. Participants’ results and interpretation were compared to the target/designated response (DR; based on consensus results) and awarded a misclassification score (MIS) for each incorrect response within each element of the EQA pilot scheme. Consistency of reporting and variation in practice were monitored.
Pilot Cryo-dEQA scheme participant survey
In 2018, participating laboratories of the UK NEQAS IIA pilot Cryo-dEQA scheme were sent an electronic survey to describe their current practice in cryoprotein testing and identification. The survey reviewed sample transport and handling, laboratory tests undertaken, reporting methods of cryoproteins as well as staff training and the volume of work undertaken.
Cryoprotein workshops
Multiple face to face workshops were organised by UK NEQAS IIA. Laboratories participating in the pilot Cryo-dEQA scheme were invited to attend. The workshops included educational presentations, case-based discussion, review of the pilot Cryo-dEQA scheme to date and discussion on practical aspects of offering a cryoprotein laboratory service. This culminated in the development of minimum requirements for cryoprotein testing.
Results
Pilot Cryo-dEQA scheme results
The UK NEQAS IIA digital EQA scheme was the first scheme for cryoprotein investigation designed to understand current practice in cryoglobulin testing and identification, in addition to providing training and education in this complex field.
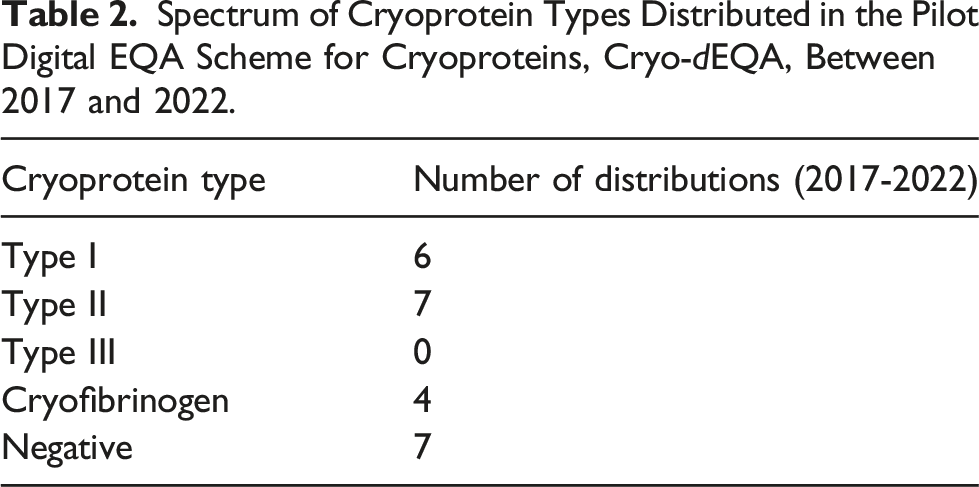
Spectrum of Cryoprotein Types Distributed in the Pilot Digital EQA Scheme for Cryoproteins, Cryo-dEQA, Between 2017 and 2022.
It was challenging to create an effective format for the Cryo-dEQA scheme due to the variation in practice between labs, such as whether laboratories tested serum and plasma samples, analysed immunoglobulins, C3, C4 and rheumatoid factor, and whether they determined the type of the cryoprotein when one was identified. The final format used a ‘gated’ approach, where participants were given images of samples incubated at 4°C and 37°C and asked to state whether or not they would investigate further (i.e. do the images show a cryoprotein or not), before the other analytical results were presented for interpretation. Participants who stated they would not investigate further were not shown any further material.
Concordance of identification of Type I cryoglobulins varied between 62–98%, with up to 29% of responses incorrectly identifying one case (Distribution 191) as a Type II cryoglobulin. Type II cryoglobulin identification varied between 71–92% with fa smaller percentage of laboratories misidentifying the cryoglobulin type (5–24%). Identical cases were circulated to monitor the consistency of reporting by participating laboratories as highlighted within Figure 1. It can be observed that the identification of cryofibrinogen samples improved during the time period from 91% in 2018 to 100% in 2022. Participant responses by cryoprotein type in the pilot UK NEQAS Cryoproteins scheme; Cryo-dEQA; participant responses are colour coded with type 1 in blue, type 2 in orange, type 3 in grey, cryofibrinogen in yellow and negative in green.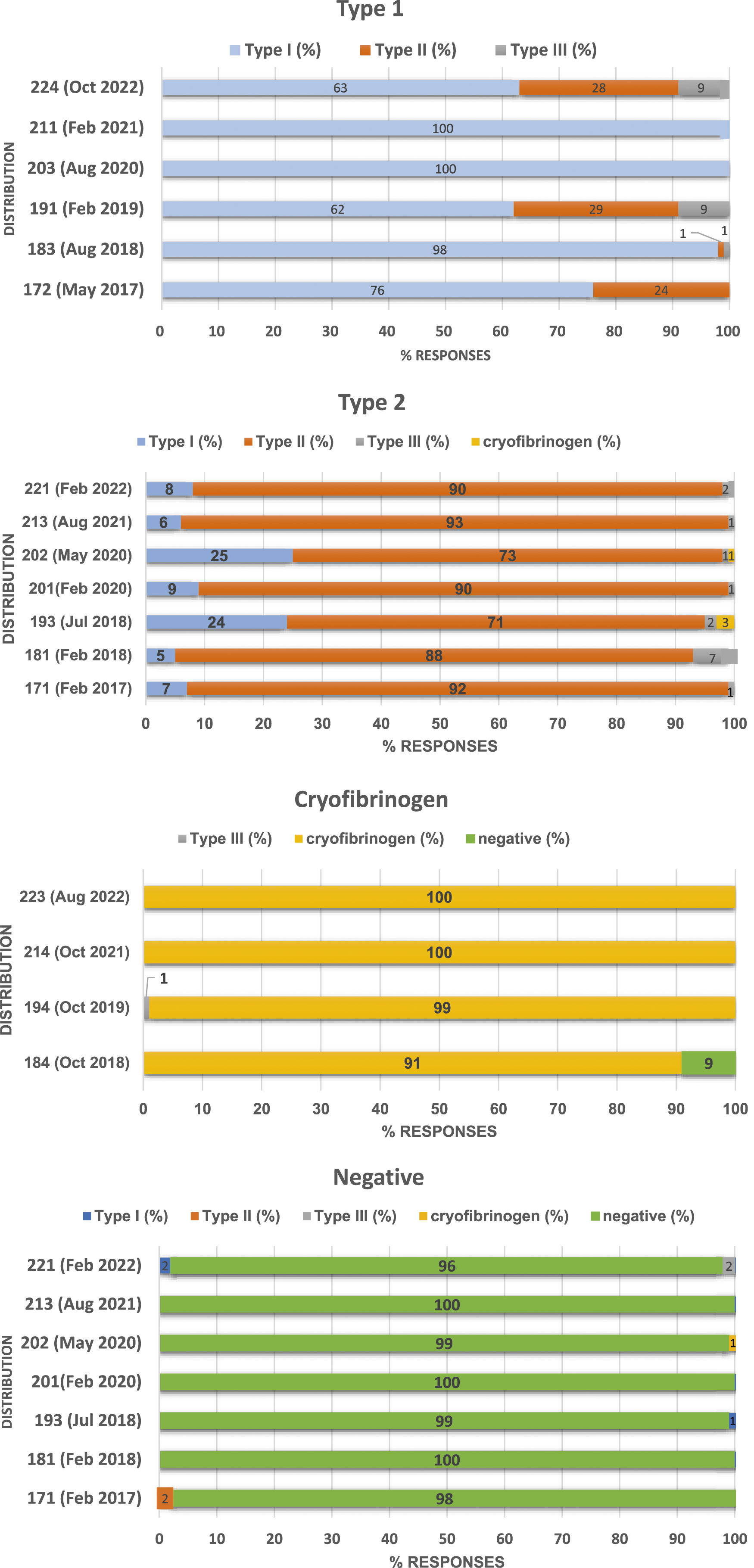
Cryoprotein EQA survey results
In 2018 participants within the pilot UK NEQAS IIA Cryoprotein scheme, Cryo-dEQA, were surveyed to obtain a better understanding of local practices undertaken for cryoprotein analysis. The response rate to the survey was 85% (137/192), with 40% of responses from participants located within the UK and 60% from non UK participants.
Sample types
From the 137 laboratories that submitted a response to the survey, 55 laboratories (40%) requested serum and plasma samples for cryoprotein testing and 74 laboratories (54%) requested serum samples only.
Preanalytical sample handling requirements
Results from the survey indicated that 21% (27/126) respondents did not pre warm the collection tubes to 37°C before the addition of patient blood.
A total of 79% (99/126) of respondents stated that they transported their samples to the laboratory under temperature-controlled conditions for cryoglobulin analysis. The method of maintaining the temperature was mainly using a pre-warmed flask filled with warmed water or sand; however, there was limited data to validate the temperature control. Thirteen laboratories (12%) make no attempt to maintain the samples at 37°C during collection and transport with three laboratories (3%) stating they transported their samples in their hands and 10 laboratories (9%) stating they transported their samples at room temperature. Collection and transportation of blood for cryoglobulin analysis at 37°C is crucial to prevent the possibility of false negative results.5,7,8 Furthermore, the lack of validation of the collection processes for maintaining the samples at 37°C is concerning.
The transportation temperature on receipt of samples was checked by 61% of laboratories (69/114) completing the survey question; most laboratories stated that they had a minimum threshold between 35–37°C and would accept samples within this temperature range. However, 39% of laboratories did not include any monitoring of the transportation temperature.
Once the samples had arrived in the laboratory 98% (115/117) of respondents incubated the samples at 37°C before centrifugation of the samples (two laboratories kept samples at room temperature prior to centrifugation) with 78% stating that the samples were incubated at 37°C for up to 4 hours before centrifugation. Most laboratories (95%, 111/117) had minimum acceptable criteria in place for monitoring incubator temperatures.
Summary of UK NEQAS Survey Results.

Analytical procedures – sample inspection
Following the temperature-controlled centrifugation of samples, the serum and/or plasma should be removed from the red cells and be aliquoted for incubation at 37°C and 4°C.
Visual inspection of the samples incubated at 37°C and 4°C is an essential component of cryoprotein analysis. In the absence of any precipitate in the samples after a 7-day incubation at 4°C, most laboratories (93%) would not do any further analysis on the samples. The remaining 7% of laboratories stated that they would continue to conduct further investigations, including serum electrophoresis, immunofixation, quantitation of immunoglobulins, rheumatoid factor, total protein and complements, despite the lack of a precipitate within the samples.
Analytical procedures – cryoprotein isolation and identification
If a precipitate is present, 80% of laboratories stated that they wash the precipitate in cold saline for 3 cycles and centrifuge in a cold centrifuge (2-8°C). There was a variation in the incubation time with 25% of respondents re-dissolving any precipitates by an overnight incubation at 37°C, while 71% of respondents stated an incubation time of up to 4 hours. Many laboratories, 76%, stated that the redissolved cryoprecipitate is immunofixed to identify the precipitated protein.
Analytical procedures – quantification of the cryoprotein
45% of laboratories estimate the amount of protein precipitated for example in the serum 4°C by a cryocrit.
Post analytical practice
When reporting results, 63% indicated that the cryoglobulin type was reported but 37% of respondents provided a qualitative response with no indication of cryoglobulin type.
The final part of the survey reviewed the number of samples that were tested by laboratories for cryoproteins each month. 27% (36/137) test between 1–15 samples per month with 22 laboratories (16%) testing more than 30 samples per month for cryoproteins (Figure 2). Number of cryoglobulin requests per month.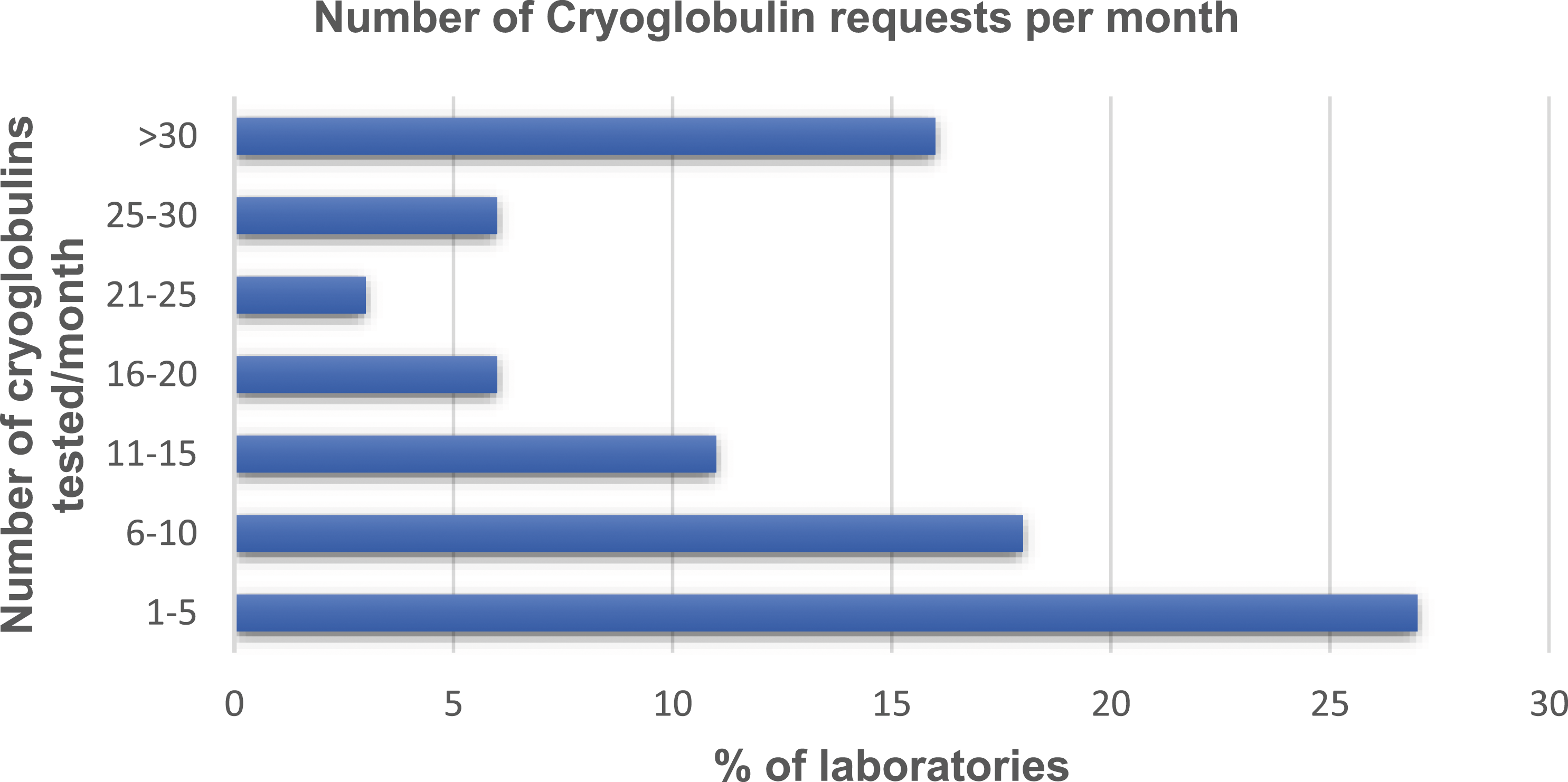
Cryoprotein workshops
Minimum Recommendations for Cryoprotein Testing.

Discussion
Results from the pilot UK NEQAS IIA Cryoproteins EQA scheme; Cryo-dEQA has highlighted that significant variability continues to exist in current laboratory practice for the detection of different types of cryoproteins as outlined in Table 1.
Participant responses showed significant disagreement, especially for cryofibrinogen cases in the early distributions, with improvement as participants became familiar with the EQA scheme and the gated approach which involved a uni directional approach to reviewing the information. (Figure 1). This was particularly challenging for laboratories who did not routinely determine cryoprotein type, although participants could select ‘refer to another laboratory’ as a valid answer.
Interpretation of cryofibrinogen cases proved particularly challenging for up to 60% of participants (86 laboratories). On further analysis, this was found to be mainly due to most laboratories not analysing plasma samples as part of their local protocols. Those that did analyse plasma samples produced between 91–100% concordance. The laboratories that do not request plasma samples would have limited experience in detecting cryofibrinogen. However, the presence of an obvious precipitate in the plasma sample incubated at 4°C that was not seen in the plasma 37°C or the serum at 4°C or 37°C should have alerted participants to an abnormality that required further investigation.
Pre analytical sample handling requirements
It is well recognised that the preanalytical requirements for cryoglobulin testing are fundamental to ensure that a reliable result is produced.6,8 Blood should be collected, transported and processed at 37°C in a controlled and monitored temperature environment and samples must not be allowed to cool before analysis. From the EQA survey, most laboratories were aware of the need to transport samples at 37°C to minimise false negative results but far fewer laboratories seemed to have a temperature-controlled transport system that was regulated and monitored throughout the whole transport process.
A surprising number of respondents (3%) stated that samples were not incubated, and samples were centrifuged straight away which raises concerns regarding sufficient time for the sample to clot before further analysis is undertaken. Ideally the samples should be transported to the laboratory within a short time period.
Most laboratories centrifuged the samples at 37°C although other laboratories without a temperature-controlled centrifuge left the samples in a temperature-controlled incubator for a prolonged time for clotting and separation to occur.
It is vital that cryoprotein samples are maintained at 37C during collection, clotting and sample separation, to minimise the chance of a cryoprotein precipitating and being lost with the clot/red cell pack. Without these precautions, there is a risk of underestimating the amount of cryoglobulin or even a false negative result. This is particularly important for Type 1 cryoglobulinaemia, where the paraprotein concentration is monitored. In addition, the procedures used to maintain samples at 37C must be verified, as for any other laboratory equipment, to demonstrate that the procedures are effective. 9
Analytical procedures
From the EQA survey, many laboratories run basic investigations on the serum sample that has been received and incubated at 37°C. This should include serum protein electrophoresis, immunofixation (if indicated), rheumatoid factor, total protein, IgG, IgA, and IgM and complement C3 and C4 quantification; these parameters would all contribute to the final, holistic interpretation of the results, including where relevant the classification of the cryoglobulin type (Table 1).
Following the centrifugation of samples in a temperature-controlled centrifuge, the samples should then be aliquoted for incubation at 37°C and 4°C. From the survey, most laboratories indicated that serum (and plasma) samples incubated at 37°C are compared with the comparable sample at 4°C and are checked for precipitation formation up to a maximum of seven days. If no precipitation is visible at seven days, appropriately, 93% (101/109) of laboratories conducted no further analysis. The length of time for precipitation formation can be crucial depending on the cryoglobulin type. It is well recognised that Type I monoclonal immunoglobulin cryoprecipitates generally appear quickly, usually within 1-2 days, but Type II and Type III cryoglobulins (mixed) can take up to a week to appear.7,8
The visual examination of the samples is fundamental to detecting any cryoprotein; however, every cryoprotein is different and there is no single image that would typify a cryoprotein. The cryoproteins can appear as clear gels, cloudy gels, samples that have completely gelled, fine cloudy layers or crystalline appearance on the bottom of the tubes or even unusual flocculence in the samples. The presence of any of these should generate further analysis to confirm the presence or absence and to classify any cryoprotein. If a precipitate is present, 80% laboratories (87/109) indicated that they wash the precipitate in cold saline for 3 cycles and centrifuge in a cold centrifuge (2-8°C). The washed precipitate is then dissolved at 37°C, ideally overnight, before further analysis is undertaken.
The redissolved cryoprecipitate would need further analysis to identify the protein content, and this should be done by immunofixation. It is important to remember that immunofixation for fibrinogen is only appropriate if there is a precipitate seen in the plasma sample that has been incubated at 4°C but not in the plasma sample at 37°C. The cryocrit, typically reported as percentage of the total volume of the sample can be difficult due to limitations in sample volume and the poor availability of graduated sample tubes that can be centrifuged. The cryocrit has limited use in the majority of cryoprotein cases; type 1 cryoproteins are typically monitored using the total paraprotein concentration in samples collected at 37°C. Type 2 and 3 cryoproteins are typically only a very small proportion of the sample so the cryocrit is unreliable. Instead the C4 concentration is a more useful way of monitoring disease activity. Photographs of the tubes after incubation are an invaluable piece of evidence for interpreting cryoproteins and for a long-term record of results.
Post analytical practice
Our survey showed a large variation in the reporting of results associated with cryoprotein analysis; some laboratories reported a qualitative response which included only whether cryoglobulin was positive or negative with no indication of cryoglobulin type while others reported all the quantitative analyses, commented on the visual inspection of the samples and offered interpretation on all the results. Specific identification of a cryoprotein AND interpretation of the results is important for all types of cryoprotein. It may be thought that type I cryoglobulins where concentrations are often >5 g/L 10 are easy to interpret but there is such a variety in even the appearance of the precipitate that positively identifying it and correlating this with the paraprotein seen in the serum is vital. Type II and type III cryoglobulins (mixed) are more difficult to identify in the samples incubated at 4°C. The interpretation of the immunoglobulin and complement concentrations and the rheumatoid factor in conjunction with the sample appearance and the identification of the protein in the precipitate all contributed to the interpretation and the laboratory is best placed to consider all these components into one overarching report to inform the clinicians and aid diagnosis.
Investigation and reporting of cryoglobulins is complex in terms of both analysis and interpretation. There are several ‘best practice’ guidelines although the evidence for interpretation and reporting do not exist and considering the relative infrequency of cryoprotein requests it is hard to imagine a sound evidence base for such a document.
Discussions at the UK NEQAS IIA Cryoprotein Workshops, alongside the information gathered from the EQA survey and distributions revealed some of the reasons for the variation in laboratory practice, including financial constraints, low volume workload, controversy and limited literature around the clinical significance of cryofibrinogens, lack of published guidelines or laboratory benchmarks. To support laboratories in improving and harmonising processes, a set of minimum laboratory recommendations was created and reviewed (Figure 3). We hope this will stimulate further discussion and harmonisation and provides a tool that laboratories can audit against to guide future developments. Outline of recommended testing pathway for cryoproteins.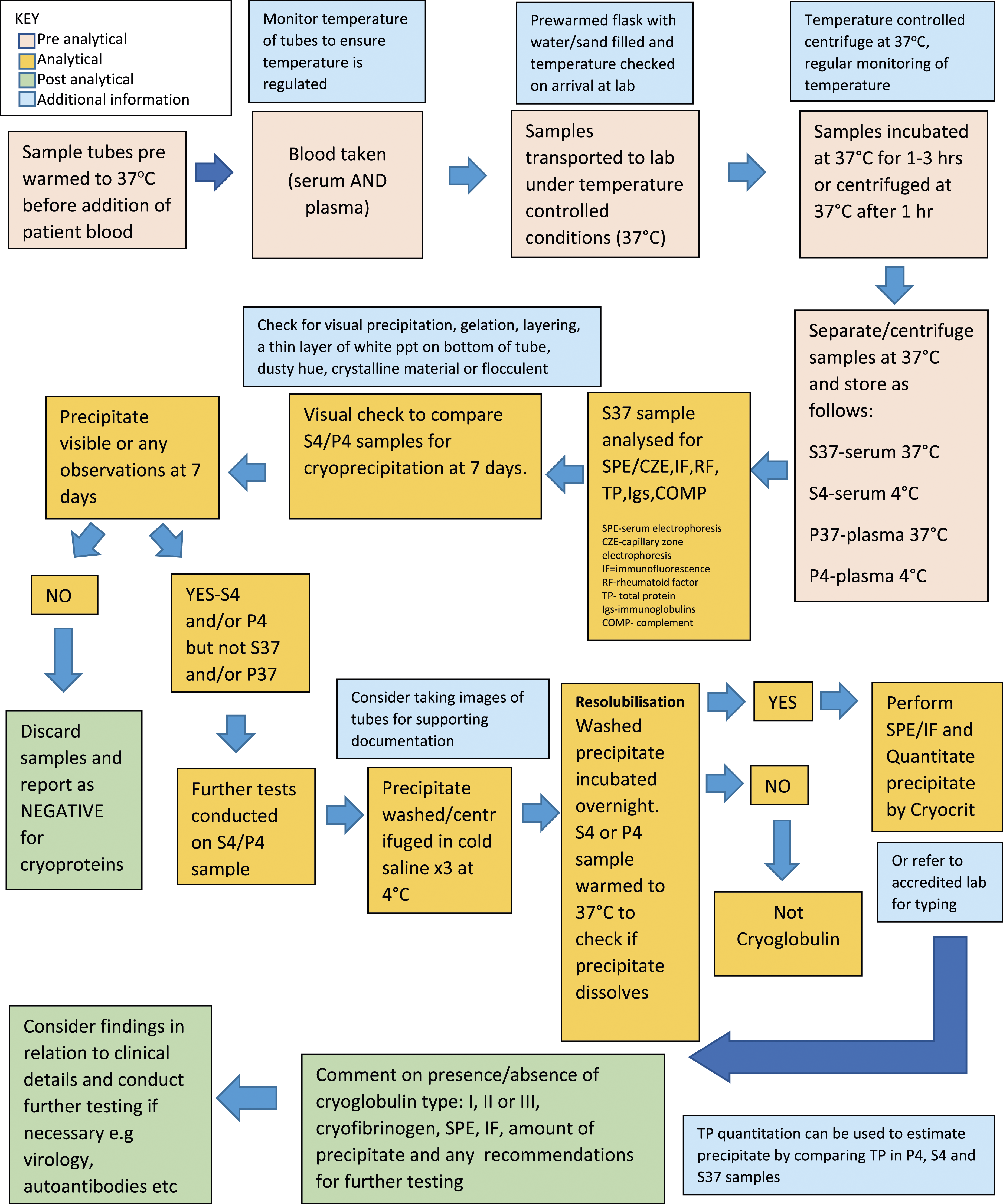
Laboratories investigating samples for cryoproteins (as with all laboratory testing) should hold ISO 15,189 accreditation. In addition, it is important to ensure that there are a sufficient number of samples passing through laboratories to provide adequate staff training opportunities and maintaining staff competence. Participation in an EQA scheme is an excellent way to assess the interpretative aspects of cryoprotein investigation but also to provide training and education opportunities in a diverse selection of cryoprotein samples.
Conclusions
Strict preanalytical and analytical conditions are critical for detection of cryoglobulins.
Several best practice guidelines and laboratory standards are currently available3,5,9,11,12 to ensure practice is optimised to benefit patient management but uptake by laboratories has not been successful.
The survey as well as initial results from the pilot UK NEQAS IIA Cryoproteins EQA scheme confirms that the laboratory procedures involved in testing cryoproteins vary enormously.
The first available EQA scheme aiming to harmonise practice for cryoprotein testing has been developed by UK NEQAS IIA and laboratories should participate in an appropriate EQA scheme to fulfil requirements for ISO accreditation against standard 15,189.
Laboratories need to standardise the pathway for cryoprotein testing across all three phases. This would provide a more robust mechanism for patient diagnosis and the resulting treatment and outcomes, regardless of the location of the patient or laboratory, and help harmonise future studies into cryoproteins.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
