Abstract
Background
Vitamin B12 status is assessed primarily by measuring total serum B12 using competitive binding methods. The lack of availability of a standard material and high-level reference measurement procedure affect the trueness of B12 results; this results in variation between methods. This study aimed to determine the reference intervals for vitamin B12 on three routine analytical platforms.
Method
A prospective reference population of healthy individuals was recruited according to the IFCC CRIDL criteria. Vitamin B12 samples were measured on Roche, Beckman and Siemens analytical platforms.
Results
In total, 300 adult subjects were recruited; the central 95th centile values for B12 for Roche (190–678 ng/mL) and Siemens (181–562 ng/mL) analytical platforms were in a close agreement. Beckman DXi, however, showed a significantly lower reference limit (110–562 ng/mL). All reference intervals are in keeping with previously published data but some are not in agreement with manufacturer provided reference interval.
Conclusion
As the quality of the reference intervals plays a significant role in clinical outcome, it is of great importance that laboratories use a method-specific reference interval and if possible, locally derived reference intervals until further method standardization occurs.
Introduction
Vitamin B12 can be routinely and inexpensively measured on most biochemistry analytical platforms. Though alternative biochemical markers have been proposed (e.g. active B12 and methyl malonic acid (MMA)), 1 the routine assessment of vitamin B12 status is most widely performed using total vitamin B12 measurement. Vitamin B12 is a complex molecule that exists in serum in multiple forms. It binds tightly to protein (transcobalamin) and is present in a very low concentration in serum. For these characteristics, defining the measurand and establishing traceability for vitamin B12 molecule has proved to be a difficult task and up-to-date, a purified standard does not exist. 2 Harmonization advances have been made by using World Health Organization (WHO) International Reference Reagent 03/178, with a vitamin B12 value assigned by a consensus approach. 2 At present, despite a WHO standard, there is no agreed high reference procedure and consequently there is variation between methods. 3 This study aimed to determine the reference intervals for vitamin B12 in a healthy population within Yorkshire, for Beckman DXi, Roche Cobas and Siemens Centaur assays. These were subsequently compared with published reference intervals to assess the level of standardization between vitamin B12 assays.
Materials and methods
A prospective reference population of healthy individuals was recruited, using an International Federation of Clinical Chemistry Committee on Reference Intervals and Decision Limits (IFCC-CRIDL)-approved health status questionnaire, for a worldwide reference interval study. 3 Ineligible candidates included inter alia, pregnant or lactating women, blood donors and individuals on medication or with long-term conditions such as diabetes. Sample collection and pre-analytical processes were standardized according to an IFCC-CRIDL protocol. The population consisted of white Caucasians of 252 (84.3%) and non-Caucasian ethnic groups mainly composed of black, Asian and mixed race participants 48 (15.7%) (African and Caribbean). Participants’ average age was 42 years (range 18–80), and the studied population was female 186 (62.3%) and 114 (38%) male. The individuals were recruited via advertisements in the Leeds Teaching Hospitals, Hull Royal Infirmary and Harrogate District Hospital. This study was approved by the National Research Ethics Committee (Ref 11/H/1302/5).4,5
The serum samples were all taken into a gel separator vacutainer (VacuetteGreiner, Stonehouse, UK). They were centrifuged within 6 h for 10 min at 3000 g. Serum samples were aliquoted into storage tubes and then frozen at –80C for a period of 8 weeks. Serum samples were transported frozen (on dry ice) to the participating laboratories. Each laboratory received an aliquot. On arrival, samples were thawed immediately at room temperature for 1 h. Homogenization was achieved by inverting samples 10 times and analysis undertaken within 4 hours from the start of thawing.
Vitamin B12 assay performance.

CV: coefficient of variation.
Statistical analysis
Statistical analysis was performed using the Analyse-it add-in package (version 2.10) for Microsoft Excel (www.analyse-it.com). Outliers were identified using a Dixon test. Data distributions were shown to be non-normally distributed (Anderson–Darling test P < 0.05) Figure 1. Reference intervals were calculated using the quantile function method as per IFCC recommendation. A flow chart of population demographics and exclusion process.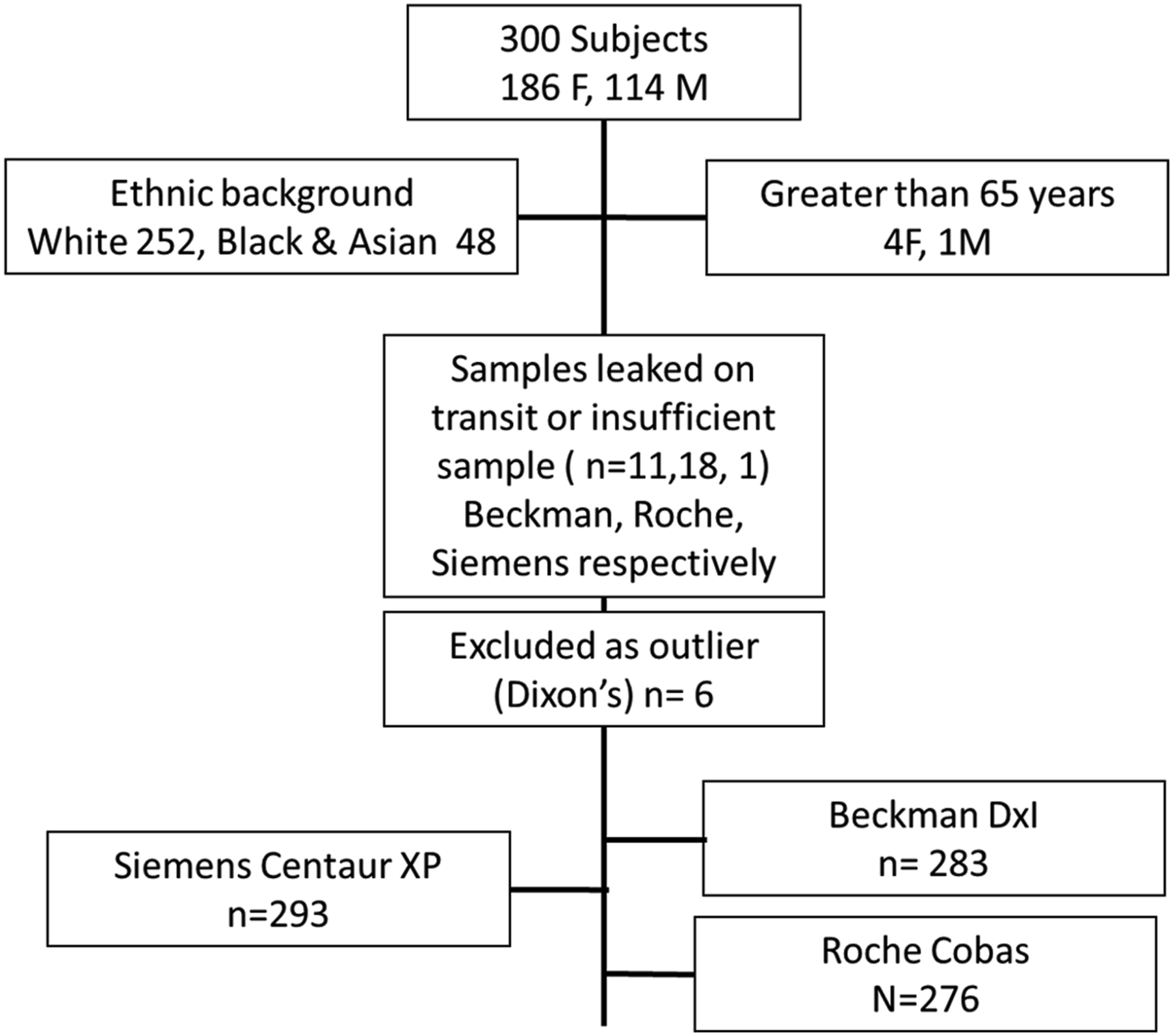
Results
Vitamin B12 reference intervals from this study, manufacturers and the literature.
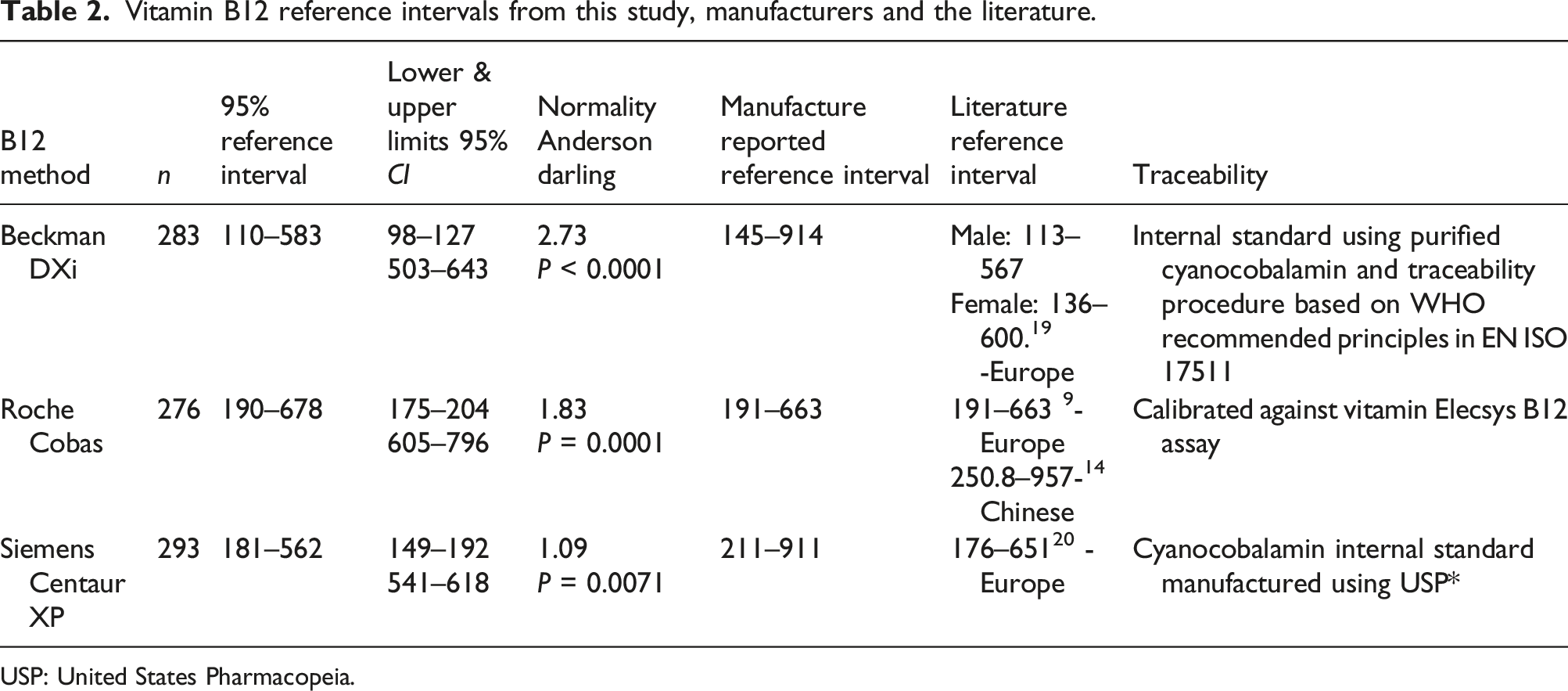
USP: United States Pharmacopeia.
Our locally derived reference intervals for both the lower (LRL) and upper (URL) limits are in good agreement with literature reported reference intervals, especially for those from European population studies (Table 2).
Roche manufacturer’s reference interval agreed well with the one developed from this study. However, Beckman and Siemens manufacturers ranges did not align with the intervals determined in the study. Beckman DXi in particular showed a LRL lower (by approx. 25%) than the manufacture’s reported reference interval and lower than the LRL for Roche and Siemens platforms (by approx. 40%) obtained from this study. The URLs for Beckman and Siemens were significantly higher than the one obtained from our healthy individuals (Figure 2). Data distribution of vitamin B12 for the included analytical platforms.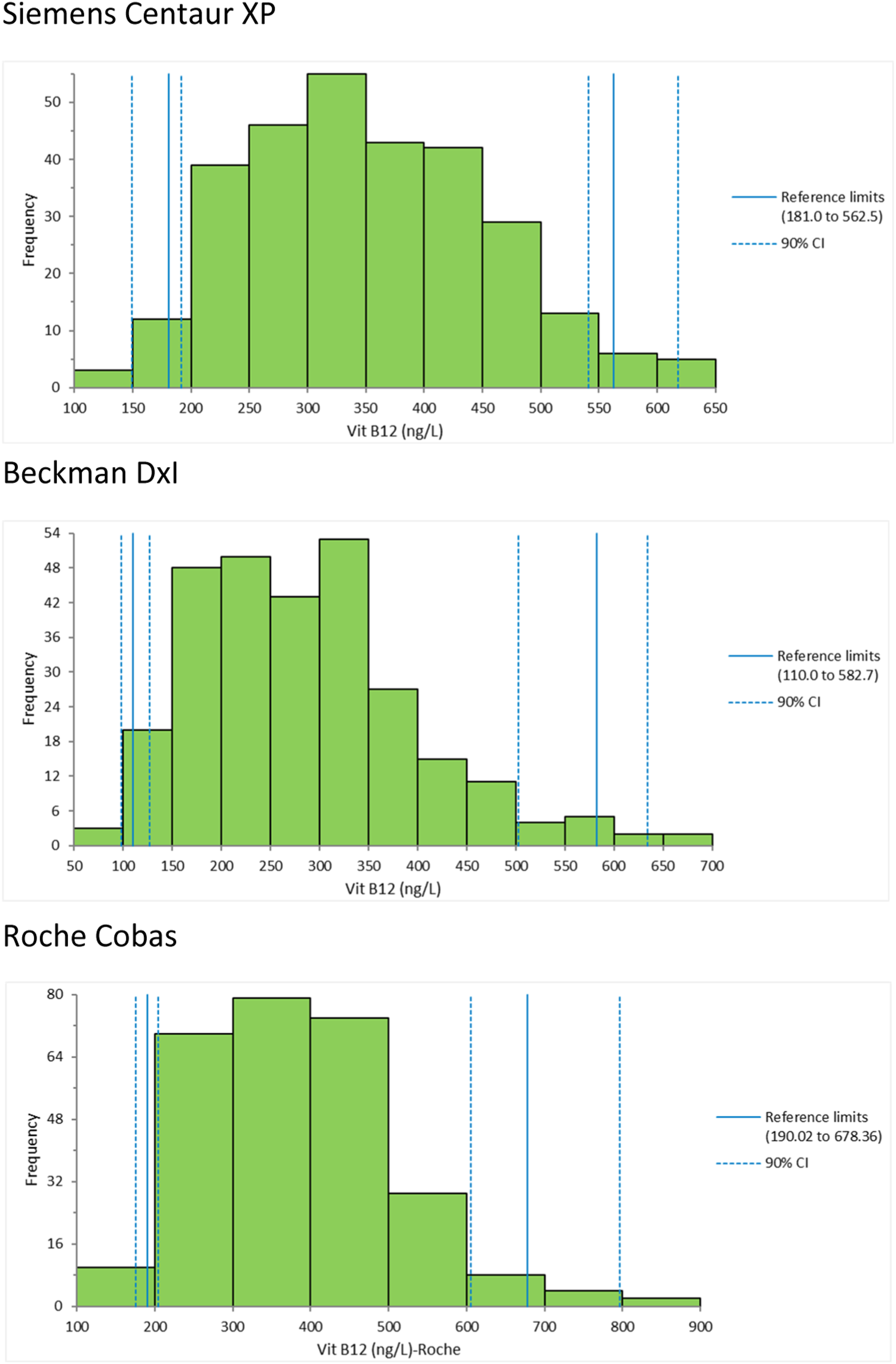
Discussion
While Vitamin B12 URL is of less diagnostic value and usually affected by local cultural factors such as dietary habits and the intake of vitamin B12 supplements, the LRL is used as a diagnostic cut off in the assessment of deficiency status. In case of Beckman DXi only, the LRL is significantly lower (approx. 25%) than the manufacture’s reported reference interval. Although Beckman DXi method is traceable to WHO EN ISO 17511 standard, data from UK External Quality Assessment Scheme (NEQAS) confirms that DXi gives low results and manufacturer reference interval do not reflect the magnitude of method negative bias (Table 2). 6
The three studied methods measure vitamin B12 by competitive protein binding with chemiluminescence detection. The variation in reference intervals is likely attributed to the fact that they are traceable to three different standards (Table 2). In fact, Roche Cobas assay has been re-calibrated against Elecsys B12 assay II with no documentation of the internal standard used. Nonetheless, Roche’s assay information sheet refers to Elecsys Vitamin B12 II assay high recovery (102%) against the WHO International Standard 03/178.
It is well known that the standardization of serum vitamin B12 methods has not been achieved yet. The WHO Expert Committee on Biological Standardization established 03/178 an International Standard (IS) for serum vitamin B12. In 2007, Thorpe and colleagues evaluated the new standard material (03/178) using Abbott, Beckman, Roche and Siemens methods. While WHO standard material reduced inter methodology variability, significant bias exists and the Beckman method mean was negatively biased compared to the studied analytical platforms. 7 They concluded that the mean difference was due to the use of a non-commutable lyophilised human serum in the calibration process. Hence, method accuracy cannot reliably be obtained until a commutable reference material has been developed. A more recent study compared B12 sera on the most common routine platforms confirmed the finding from Thorpe et al and showed that mean for Beckman assay negatively deviated from Roche, Abbott and Siemens. 8 Another study showed relatively good agreement of results from Abbott, Siemens and Roche but excluded Beckman’s assay. 9 Recently, NICE guidelines for the management and treatment of anaemia used a cut off on 200 ng/L. The application of this cut off to Beckman users would mislabel asymptomatic and B12 sufficient individuals as deficient and consequently lead to unnecessary treatment or investigation. 10
A cut off for the diagnosis of vitamin B12 deficiency has been recently reviewed. 11 Aparicio- Ugarriza and colleagues included 69 studies, in which deficiency was defined using a range of biochemical markers (such as active B12, MMA, homocysteine, folate and haematological markers) and symptoms. The study found a wide scatter of cut offs ranged from 166 ng/L (123 pmol/L) to 473 ng/L (350 pmol/L) reflecting various diagnostic sensitivities. The authors related this scatter to factors such as method difference, age, gender and analytical precision at the chosen cut offs. Indeed, in this study, the uncertainty (UM) at the LRL of 110 ng/L (Beckman assay) is considerably wide (±26 ng/L) which affects the diagnostic certainty of vitamin B12 (Table 1) for this method users. The uncertainty around the LRL is another reason for the call of introduction of an intermediate range where further biochemical markers can be used to confirm or refute the diagnosis of vitamin B12 deficiency. 17 In relation to this study, it is worth noting that the impact of other factors such as ethnicity, age and gender on reference intervals has not been assessed, as we have not been able to partition our data by age or gender due to a small sample size (Figure 1). Table 2 showed that the URL for both Beckman and Siemens are higher than that obtained from this study. We believe the main reason for this finding is the inclusion of subjects on supplements in the population of those studies. In our study, however, the studied population is a group of healthy healthcare professionals in which the percentage of those on supplements is very small; therefore, a direct comparison to other studies is not possible.
Vitamin B12 measurement remains the first-line test to define deficiency. Due to the broad range of non-specific symptoms of vitamin B12 deficiency and the use of strategies reliant mainly on a single biochemical marker, accurate assessment of vitamin B12 status is problematic. This issue is complicated by the fact that vitamin B12 concentrations vary with age, gender, ethnicity and cultural and regional dietary habits.12,13 A recent Chinese study reported that a manufacturer’s original reference interval from a population of European descent was quite different from the reference interval determined from a local population. 14 A recent study in North India found 47% prevalence of vitamin B12 deficiency using European derived reference interval. 15 This may reflect different study population or indeed different dietary or ethnic attributes. Therefore, the non-standardized vitamin B12 assays and the use of published reference interval add an extra layer of complexity to an already troubled diagnostic tool.
The Institute of Standards and Technology (NIST) is currently developing a serum-based reference material, which consists of pools of frozen human serum for vitamin B12. 16 Until a full standardization of vitamin B12 assays is achieved, laboratories are recommended to use local population derived reference intervals, as some manufacturer quoted reference intervals may be inappropriate. Furthermore, until further methods performance improvement, including additional diagnostic strategies that combine intermediate vitamin B12 range plus a better marker of intracellular vitamin B12, such as MMA and/or homocysteine may be useful to minimise the effect of vitamin B12 method differences.17,18
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Approved by the National Research Ethics Committee (Ref 11/H/1302/5).
Guarantor
NJ.
Contributorship
TB, NJ and JHB wrote the first draft. JHB, AL, WM and NJ conducted the reference interval study.
