Abstract
Determination of plasma vitamin B12 (B12) is a frequently requested laboratory analysis, mainly employed to establish B12 deficiency. However, an increased level of B12 is a common unexpected finding that may be related to an increased concentration of one of the B12 binding proteins, haptocorrin or transcobalamin. This paper describes the extensive laboratory evaluation of a patient with an elevated level of plasma B12 with various well-established assays. Initial studies suggested the presence of a macromolecule consisting of haptocorrin bound B12. Specific determinations of the B12-binding proteins revealed normal amounts of haptocorrin but a markedly increase in both total and B12 saturated transcobalamin (holo-TC). The results are in accord with the presence of macro-transcobalamin. These experiments reveal that determination of the nature of the B12-macromolecules is troublesome due to differences in assays applied to measure these proteins. In addition, this publication creates awareness of macro-holo-TC as a cause of an unexplained increased B12 level.
Introduction
Vitamin B12 (B12) is a water-soluble vitamin only found in food of animal origin. The uptake and transportation of B12 is a complex multistep process. 1 In summary, after ingestion, B12 is released by pepsin and gastric acid and bound to the glycosylated 60–70 kDa haptocorrin (HC) produced by the salivary glands. In the duodenum, this complex is dissolved by pancreatic proteolytic enzymes and the liberated B12 is bound to intrinsic factor, produced by the gastric parietal cells. This complex will bind to cubilin, a receptor present in the terminal ileum. After traversing the brush border B12 is dissociated and released into the circulation. In the circulation 6–20% of the B12 is bound to the non-glycosylated 43 kDa transcobalamin (TC: holo-TC if saturated with B12 and apo-TC if not), whereas 80–94% is bound to HC (holo-HC if saturated with B12 and apo-HC if not).
Holo-TC is the biological active fraction of B12 which can be taken up by cells. 2 In the cytosol B12 is converted to methyl-B12, a coenzyme for the delivery of a methyl group to convert homocysteine to methionine. This reaction is involved in enzymatic processes that are essential in the formation of purines and pyrimidines, and thereby steps that regulates protein and DNA synthesis. In the mitochondria, the adenosyl-B12 derivate acts as a cofactor to methylmalonic-CoA-mutase which regulates the conversion from methylmalonic-CoA to succinyl-CoA.
Determination of total plasma B12 concentrations can be troublesome due to variations in the various commercial assays. 3 All assays involve a step of releasing B12 from its binding proteins followed by a competitive assay of the liberated B12. The assays cover a measurement range of around 50–1500 pmol/L. There are numerous reviews on the clinical features and diagnostic work-up in patients with low B12 levels.4–6 The literature on the management of patients with elevated plasma B12 levels, however, is limited. 7 Notwithstanding, B12 levels above the normal measurement range in individuals without supplementation are reported to occur in around 10% of the tested samples in large surveys.7–10
A spontaneously elevated concentration of B12 is caused by elevation of the HC level or far less frequently of the TC level. 11 Though often unexplained, an increased level of B12 may be related with the presence of malignancy. High levels of HC are responsible for the elevated B12 levels in a variety of solid tumours and in various haematological malignancies including chronic myeloid leukaemia and polycythaemia vera.7,12 Also, administration of granulocyte colony-stimulating factor for stem cell mobilisation may lead to HC-mediated elevation of the B12 level. 13 In addition, an increased level of TC has been related to lymphatic malignancies. 14 A number of infectious and autoimmune disorders yields elevated B12 levels caused by an increased HC or TC production. In hepatic diseases elevated B12 may result from release of B12 or HC out of damaged hepatocytes or by decreased clearance of HC. In patients with renal failure diminished renal clearance of predominantly TC-bound B12 is associated with elevated B12 levels in a significant number of patients. 8 Notably, falsely elevated B12 levels can occur in patients with pernicious anaemia due to assay interferences. 15
An elevation of B12 levels related to a decrease in turnover rate because of the presence of macro forms of the binding proteins has been described in a limited number of studies, namely 69 cases in 10 studies.9,16–24 Macromolecules in blood are high molecular complexes of proteins, caused by either self-polymerisation or by association with other proteins or plasma components, mostly immunoglobulins, as described in the case of prolactin. 25 Taken together the various causes for an increased level of plasma B12 warrants further examinations in patients with an unexpected increased level of the vitamin.
Aim
The aim of this publication is to demonstrate a case-based approach of the analysis of an unexpected elevated B12, and the troublesome, complex interpretation of vitamin B12 binding protein assays in a suggested presence of a macromolecule.
Materials and method
Patient
Experiments conducted in this paper are based on a case of a 56-year-old woman who was referred to the internal medicine department for the diagnostic work-up of persistent fatigue. The patient had no specific complaints and physical examination was unremarkable. Laboratory examination revealed a strongly elevated vitamin B12 level, but otherwise normal haematologic parameters and an extensive biochemical profile and autoimmune serology (including autoantibodies against intrinsic factor and parietal cells), were within the normal range.
Biochemical testing
Extensive research into vitamin B12 and the binding proteins has been conducted in two different accredited laboratories. The department of clinical chemistry of the University Medical Center Rotterdam in the Netherlands will be further appointed as Laboratory I. The department of Clinical Biochemistry of the Aarhus University Hospital in Denmark will be further appointed as Laboratory II.
In Laboratory I, total B12 was measured with an immunoassay on the Roche Cobas 6000 (Roche Diagnostics) platform. Holo-TC was measured with an immunoassay on the Abbott Architect platform. Total apo (apo-TC + apo-HC) was measured with an in-house radioassay as previously described. 26 In brief, 57Co-B12 with a known specific activity was added in excess to the sample followed by removal of unbound B12 by charcoal precipitation. The radionuclidic 57Co-B12 is a measure for the amount of Total apo. Subsequently, the sample was exposed to silicagel, that is known to precipitate TC bound B12 and 57Co-B12. 20 After precipitation apo-HC could be measured. Apo-TC, total TC, holo-HC and total HC were calculated.
In Laboratory II, total B12 was measured with an immunoassay on the ADVIA Centaur CP (Siemens Healthcare Diagnostics) platform. Holo-TC, total TC and total HC were measured with highly specific in-house ELISA assays for each of the proteins as previously described.27–29 Apo-TC, holo-HC, apo-HC and total apo were calculated.
PEG precipitation
PEG precipitation was performed with Polyethylene Glycol (PEG) 6000 20 as was described earlier. In brief an equal quantity of 25% PEG was added to 200 μL of serum from the patient and serum from an anonymous control. After mixing, the serum was centrifuged for 5 min at 1600 × g and B12 was measured in the supernatant. As control IgG and ASAT were measured.
Gel filtration chromatography
GFC was performed on the AKTA Prime Plus (GE Healthcare) equipped with a 1.6 × 60 cm Sephacryl-S300 column (GE Healthcare). The column was equilibrated with 10 m
The void volume (V0) determination and calibration were performed using blue dextran (1 g/L) and globular protein standards (3–4 g/L) in running buffer; 0.5 mL was loaded on the column; everything was performed in duplicate. V0 was determined to be 40 mL.
For serum samples, 1.0–2.0 mL was loaded on the GFC column. Serum albumin (67 kDa) and serum B12 were used as internal retention markers in the patients’ sera. For each sample loaded, fractions of 1.25 mL were collected, kept on ice until biochemical analysis measurements (<1 h) and subsequently stored at −80°C.
Results
Initial biochemical results
Vitamin B12 levels were persistent elevated in repeated measurements, that was >1000 pmol/L (reference interval 171–684 pmol/L). Laboratory examination revealed normal haematologic parameters and an extensive biochemical profile and autoimmune serology (including autoantibodies against intrinsic factor and parietal cells) were within the normal range. Additional tests to evaluate the B12-associated metabolic state revealed a normal folate (immunoassay, DxI 600 Unicel, Beckman Coulter) concentration of 19 nmol/L (reference interval 6–30 nmol/L). Methylmalonic acid (LC-MS/MS) and homocysteine (enzymatic assay, DxC 800 Unicel, Beckman Coulter) were 0.17 μmol/L (reference value <0.44 μmol/L) and 10 μmol/L (reference value <14 μmol/L), respectively, and indicated no functional deficiency of B12.
Distribution of B12 and the binding proteins in plasma and the analytical methods for laboratory determinations measured at laboratory I and at laboratory II. Concentrations in bold reflect elevations above reference range.
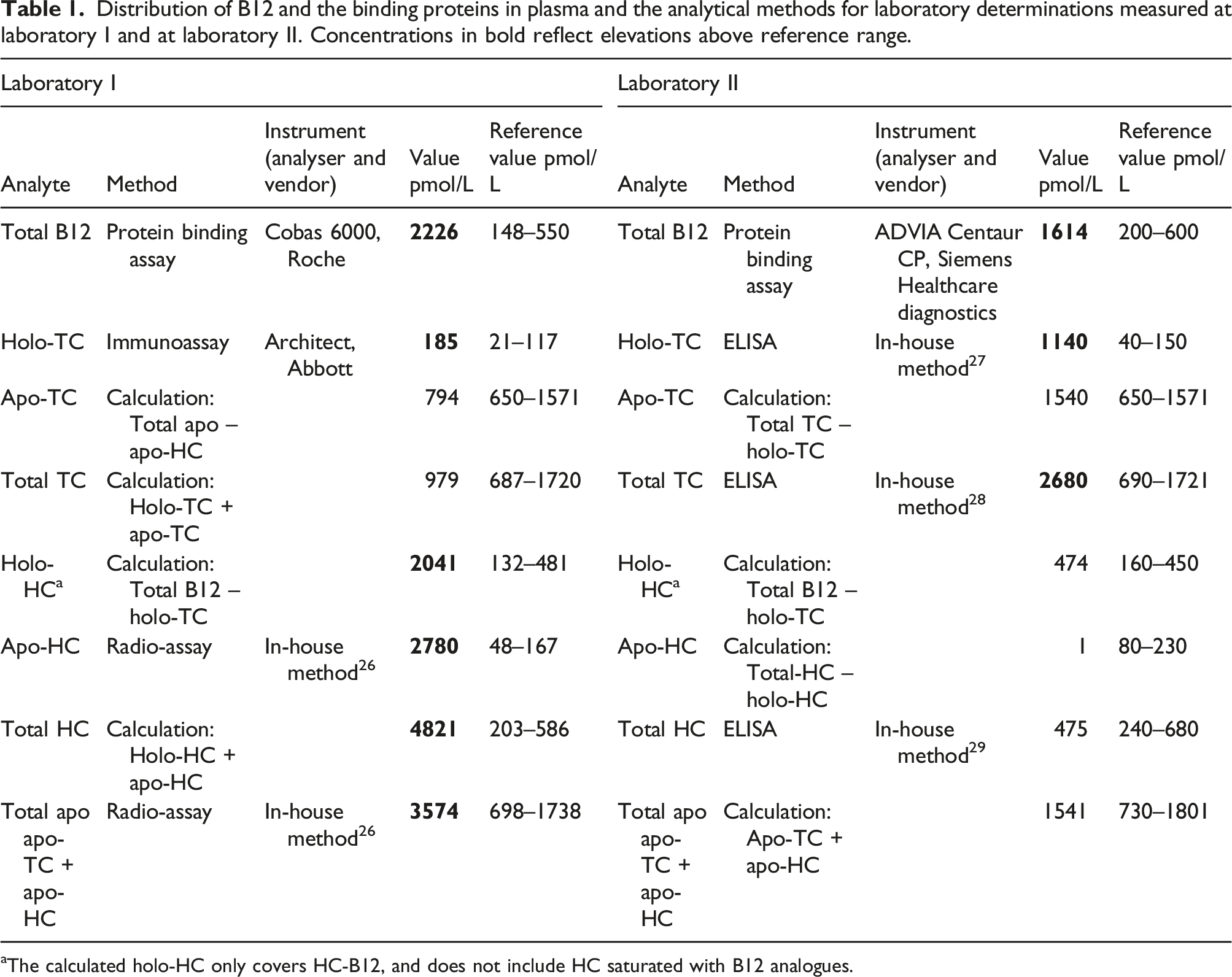
aThe calculated holo-HC only covers HC-B12, and does not include HC saturated with B12 analogues.
Interference studies
In order to determine whether the high plasma B12 was caused by interference with heterophilic antibodies the B12 concentration was measured before and after treatment with an heterophile blocking tube. 15 Since the concentration in the heterophile blocking tube (2500 pmol/L) and of the untreated sample (2000 pmol/L) were comparable, there was no indication of interference. Also, the linear relationship (y = 1711,1x – 25,833; r2 = 0.99) between the theoretical amounts of B12 (x) and the amounts found by the B12 assay (y) performed in diluted samples (1:1, 1:4, 1:8, and 1:16) of the original plasma confirmed the absence of interference.
Macro molecule studies
PEG precipitation was performed to explore a possible presence of macro B12. Figure 1 indicates that only 22% of B12 remained in the supernatant for the patient, while we have seen 98% (as depicted in the same figure) for the control. Both IgG and ASAT behaved alike for the patient and the control. Thus, these results strongly support the presence of macro-B12. PEG precipitation of plasma from the patient and a healthy control. The precipitation showed a recovery of 22% for total B12 for the patient. The recovery for total B12 for the control was 98%. The recovery of IgG was 3% in the patient and 1% in the control, indicating a successful precipitation. The recovery of ASAT was 89% in the patient and 74% in the control. Biochemical analysis of vitamin B12 and binding proteins.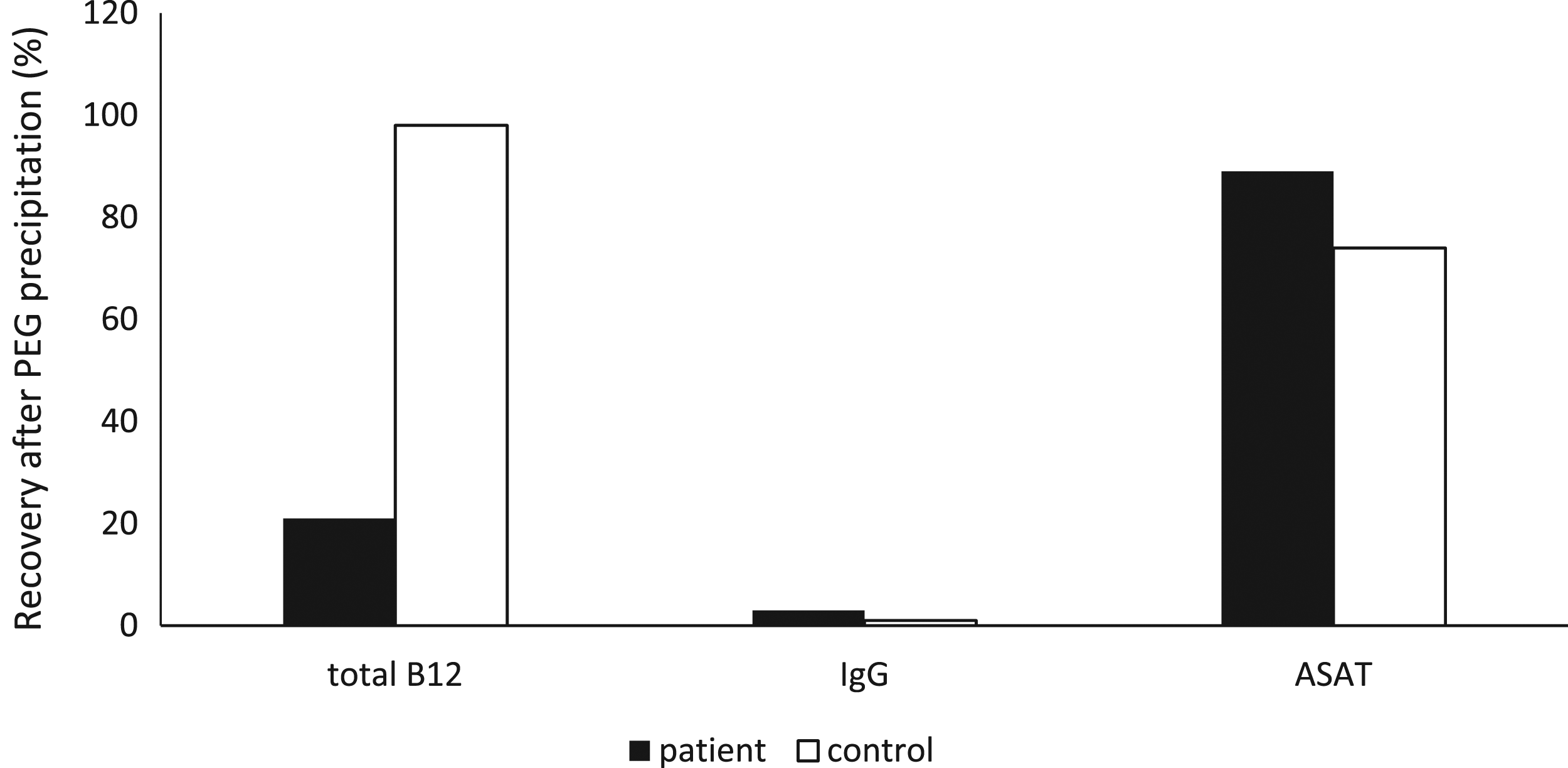
Further investigations into the assumed macromolecule in more detail by gel filtration chromatography were inconclusive. Nevertheless, the B12 elution profile demonstrated concurrent elution of B12 as a much larger molecule than does free HC and TC (data not shown).
The potential presence of macro-B12 prompted us to reconsider the results obtained by the initial analysis showing an increased HC since it is unknown how macro-B12 behaves in this assay. In order to confirm or discard the finding of an increased level of B12 and HC in Laboratory I we analysed B12, holo-TC and the total concentration of TC and HC in Laboratory II. The results for both measured and calculated values by each laboratory are indicated in Table 1. In both laboratories an increased total B12 level was found using a protein binding assay. However, analysis of B12 binding proteins TC and HC showed no agreement between the two laboratories. While the investigations at Laboratory I using a radioassay yielded an increased HC level, an increased TC level was found in laboratory II using an ELISA.
Discussion
Main findings
Based on one case of an unexpected elevated level of vitamin B12 we show that macromolecules of B12 have significant influence on the performance of well-established assays and thereby the interpretation of B12 diagnostics.
Both laboratories used commercial assays – though from different companies – for measurement of B12, and both laboratories reported an increased B12 level. However the result obtained in Laboratory I was almost 40% higher than the value obtained in laboratory II. We have no definitive explanation for this difference, and thus cannot exclude that it could be caused by the presence of the macro-protein.
Laboratory I reported a marginal increase in holo-TC and a markedly increased concentration of HC, while laboratory II reported an increased level of both holo and total TC but a normal concentration of HC. We believe that the discrepancies can be explained by the presence of macro-TC.
The commercial assay used for measurement of holo-TC in laboratory I employs a monoclonal capture antibody specific for B12 saturated TC (33). A previous study has demonstrated that variant forms of TC may lead to spuriously low results (32). Our results strongly suggest that also the presence of macro-TC may interfere in this assay. Laboratory II used an assay that employs polyclonal antibodies and measures holo-TC after removal of apo-TC (27, 28). This assay is unlikely to be influenced by macro forms of the protein.
Laboratory I calculated holo-HC as the difference between total B12 and holo-TC, and an underestimation of holo-TC will therefor result in an overestimation of holo-HC. The result derived for apo-HC was based on precipitation of TC with silicagel leaving HC in the supernatant. However, a macro form of TC may not – like normal TC – be precipitated and therefore be mistakenly classified as HC. Thus, the conclusion of laboratory I concerning the presence of HC is likely to be caused by the presence of a macro-TC.
The laboratory determination of the essential cofactor B12 is frequently requested by both general practitioners and specialists within the hospital. It is interesting that, about 10% of patients referred for B12 measurement have high plasma levels.10,30 Recent studies show that one fifth of the cases is caused by the presence of a macromolecule, mainly as a complex between holo-TC and IgG. However, several studies also report the presence of IgA or IgM.9,16-20
Macromolecules are concurrently measured as an elevated concentration of that particular molecule. Macromolecules may be biologically less active, and therefore, a proper analysis for its presence is warranted. Gel filtration chromatography is the reference method. However, PEG precipitation is the most widely used technique for identification of a macromolecule.31,32
Earlier studies have shown that elevated B12 levels due to macromolecules are of limited clinical relevance. 9 Also, in our patient the presence of macro-TC did not interfere with its metabolic activity, as judged by the normal concentrations for methylmalonic acid and homocysteine.
Limitations and strengths
Our study has some limitations. First, this was a case-based laboratory investigation. Also due to lack of remnant patients’ serum, definitive gel filtration chromatography experiments to prove complex formation of B12 with other plasma proteins could not be conducted.
However, the strength of this paper is the thorough laboratory investigations that have been done and the comparison between well-established assay to measure the B12 binding proteins haptocorrin and transcobalamin.
Conclusion
In conclusion we report a patient with macro-TC initially misdiagnosed as macro-HC. The results underscores that a detailed knowledge of B12, HC and TC assays are crucial when further examination of patients with an increased level of B12 is warranted.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Written informed consent from the patient.
Guarantor
André P. van Rossum.
Contributorship
SD and LV equally contributed to this work, and both should be considered first authors. AR started the work and reviewed all drafts. SH and CR performed experiments in Rotterdam. EN performed experiments in Aarhus. SM and AM performed experiments in Maastricht. EN, DB, SH, AM and CR reviewed all drafts. All authors contributed towards the writing of this article.
