Abstract
Background
Vitamin B12 deficiency frequently appears in phenylketonuria patients having a diet poor in natural protein. The aims of this study were to evaluate vitamin B12 status in phenylketonuria patients by using combined indicator of vitamin B12 status (cB12) as well as methylmalonic acid and homocysteine, more specific and sensitive markers, in comparison with healthy controls.
Methods
Fifty-three children and adolescents with phenylketonuria under dietary treatment and 30 healthy controls were assessed cross-sectionally. Serum vitamin B12 and folate concentrations were analysed by chemiluminescence immunoassay. Plasma methylmalonic acid and total homocysteine concentrations were measured by liquid chromatography-tandem mass spectrometry and liquid chromatography, respectively. cB12 was calculated by using a formula involving blood parameters.
Results
Methylmalonic acid and folate concentrations in phenylketonuria group were higher compared with controls. Methylmalonic acid concentrations were high in 56.5% of the patients and 26.7% of the controls with normal vitamin B12 concentrations. Based on cB12, a significant difference within the normal values was detected between the groups. However, although 24.5% of phenylketonuria patients and 13.3% of controls had decreased vitamin B12 status according to cB12, there was no significant difference.
Conclusion
Children and adolescents with phenylketonuria having a strict diet can be at risk of functional vitamin B12 deficiency. This deficiency can be accurately determined by measuring methylmalonic acid concentrations. Calculation of cB12 as a biochemical index did not provide additional information compared with the measurement of methylmalonic acid alone, but may be helpful for classification of some patients with increased methylmalonic acid as having adequate vitamin B12 status.
Keywords
Introduction
Phenylketonuria (PKU) (OMIM 261600) is an inborn error of phenylalanine metabolism caused by hepatic phenylalanine hydroxylase (PAH; EC 1.14.16.1) deficiency, resulting in an increase in blood phenylalanine (Phe) concentrations. 1 The incidence of PKU varies among ethnic groups and geographic regions worldwide and ranges from 1:4500 in Ireland to 1:200,000 in Finland. Although the exact incidence is unknown, it is relatively common in Turkey due to the high consanguinity within the population. 2 The widely used traditional method for the treatment of PKU is dietary restriction of natural proteins rich in Phe.3,4 Thus, the PKU diet is mostly supplemented by vitamins/minerals or amino acid mixtures including vitamins and other micronutrients necessary for growth and development.1,5 However, this strategy does not always preclude micronutrient deficiency in those patients.3,6,7 Vitamin B12 deficiency is relatively common in patients with PKU compared with other micronutrient deficiencies. This is because the main source of vitamin B12 is protein-rich food, which is restricted in the PKU diet.7–9 Vitamin B12 deficiency is mainly reported in adolescent and adult PKU patients who do not regularly take Phe-free amino acid supplements including vitamins.3,6–13 It is known that compliance with the restricted diet treatment is better in childhood. 1 However, studies on children and adolescents with PKU have revealed conflicting findings about vitamin B12 deficiency and compliance with the diet. While some studies have shown high vitamin B12 concentrations in patients having the restricted diet,14,15 one study showed low vitamin B12 concentrations. 16
There is currently no gold standard for assessment of vitamin B12 deficiency. 17 The standard routine diagnostic test for vitamin B12 deficiency is serum vitamin B12 assay. Although measuring serum vitamin B12 concentrations is the first-line test, it has a low sensitivity.18,19 In addition, although serum vitamin B12 concentrations of >148 pmol/L are considered sufficient, it is emphasized that subclinical deficiency (with no haematological and neurological changes) can appear when vitamin B12 concentrations are from 148 pmol/L to 221 pmol/L, defined as grey zone or low reference ranges.20–23 Therefore, it is not enough to measure serum concentrations of vitamin B12 to make the diagnosis of its deficiency. 22 It is recommended that normal and especially low-normal serum B12 concentrations should be evaluated together with the second-line tests (methylmalonic acid [MMA], homocysteine [Hcy] and/or holotranscobalamin [HoloTC]) to make a more reliable diagnosis.18,19,24,25 Functional vitamin B12 deficiency is defined as elevated circulatory levels of MMA and/or Hcy even if serum vitamin B12 concentrations are within the reference values.6,26 MMA is a more sensitive and specific biomarker than Hcy for evaluation of vitamin B12 deficiency because Hcy also accumulates in the absence of other nutrients, such as folate and vitamin B6. These functional biomarkers are useful since they reflect early changes in tissue vitamin B12 status.27,28 In recent years, combined indicator of vitamin B12 status (cB12), calculated by using different combinations of four parameters (vitamin B12, HoloTC, MMA and Hcy) (in case of one or two missing markers), has become prominent in terms of evaluation of vitamin B12 status. 29
The aims of the present study were to evaluate vitamin B12 status in children and adolescents with PKU under dietary treatment by using conventional serum vitamin B12 assay in addition to functional biomarkers plasma MMA and Hcy, and cB12 as a biochemical index, in comparison with healthy controls and to evaluate vitamin B12 deficiency in PKU patients in terms of their compliance with the diet by using these parameters.
Material and methods
Study population
The study group included 53 PKU patients aged 5–18 years with a median age of 9.5 years (20 females and 33 males), being on a Phe-restricted diet, having blood Phe concentrations of 120–900 μmol/L and followed by the Division of Pediatric Metabolism and Nutrition at the Dokuz Eylul University Hospital. All the patients were diagnosed as classical PKU (Phe concentration >1200 μmol/L) by the Turkish Newborn Screening Program, and their treatments were initiated immediately after their diagnosis in the neonatal period. They were treated with a diet restricted in terms of natural protein and supplemented with a Phe-free amino acid mixture enriched in vitamins and minerals. Patients with any chronic diseases other than PKU (celiac disease, hypothyroidism, growth hormone deficiency, obesity or malnutrition) and severe mental retardation or taking multivitamin supplements were excluded. Patients treated with sapropterin dihydrochloride and large neutral amino acids also were not included in the study.
PKU patients were divided into ‘low adherence’ and ‘high adherence’ groups according to their mean plasma Phe concentrations obtained in the last 12 months. High adherence to diet was defined as mean plasma Phe concentration <360 μmol/L for patients under six years of age, <480 μmol/L for children aged between 6 and 10 years and ≤600 μmol/L for older patients. 30
The control group comprised of 30 healthy children aged 5.0–17.5 years with a median age of 8.59 years (16 females and 14 males). People using multivitamin supplements or drugs and having growth and developmental retardation, anaemia, obesity and inborn errors of MMA and Hcy metabolism were excluded from the study. None of the PKU patients and controls had a known renal, hepatic or cardiovascular disease. Demographic and anthropometric data including age, gender, body weight, height, weight for height of the PKU and control groups were recorded.
Biochemical parameters
Serum and plasma samples were collected from the PKU and control groups after overnight fasting. Concentrations of Phe, vitamin B12, folate, ferritin and plasma total Hcy and full blood count were measured in Central Laboratory of Dokuz Eylul University Hospital. Serum samples were centrifuged at 3000 r/min for 10 min (Hettich, Rotina 38, Germany) after coagulation for 20 min at room temperature. Plasma Phe concentrations were determined with a fluorometric assay (Shimadzu, RF-5301PC, USA). 31 Serum vitamin B12, folate and ferritin were evaluated by the chemiluminescence immunoassay method (ABBOTT, Architect i200, USA). Plasma samples for Hcy measurements were centrifuged at 4°C and 3000 r/min for 10 min (Hettich, Rotina 35 R, Germany). Plasma total Hcy (albumin bound and oxidized forms) concentrations were measured by high-performance liquid chromatography (HPLC) with a fluorescence detector using a commercial kit (ImmuChrom, IC2801, Germany). 32 The HPLC (LC-20AT, Shimadzu, Japan) separation was carried out on a reversed phase column (MZ Inertsil ODS-2, 125 mm × 4 mm; 5 μm) by isocratic elution. The injection volume was 20 μL and the total analysis time was 5 min. Excitation and emission wavelengths of the fluorescence detector (RF-10A XL, Shimadzu, Japan) were 385 nm and 515 nm, respectively. Full blood count was performed with an autoanalyser (Beckman Coulter, LH 780 Analyzer, USA). Reference ranges used by our centre were 148–618 pmol/L for vitamin B12, 4.09–20.45 nmol/L for folate and 5–15 μmol/L for Hcy.
Measurement of MMA by LC-MS/MS
Plasma MMA concentrations were determined in the Medical Biochemistry Department of Dokuz Eylul University. Plasma samples were collected in tubes containing EDTA and centrifuged immediately at 4°C and 2000 ×
The MMA method was optimized for source- and compound-dependent parameters and performed for validation of the analytical method. The precision of the method was assessed by determining the coefficient of variations using intraday precision (
Statistical analysis
Statistical analyses of the data obtained were performed by using SPSS 15.0. The results were expressed in median and minimum–maximum values or numbers (percentages). Normality of the variables was assessed by using a combination of Shapiro-Wilk test, histogram, normal Q-Q plot, values of skewness, kurtosis and coefficient of variation. Since data about many parameters did not have a normal distribution, comparisons between two groups were made by using Mann–Whitney U test and Kruskal-Wallis test.
Recommended by Fedosov et al., ‘the combined indicator for vitamin B12 status (cB12)’ was calculated by using vitamin B12, MMA and Hcy concentrations according to the formula cB12 = log10[(vitamin B12)/(MMA×total Hcy)]–(age factor). cB12 values lower than <–0.5 indicate decreased vitamin B12 concentrations or vitamin B12 deficiency status.
29
Pearson’s Chi-squared tests were used to compare the frequency between the groups, but when any of the cells had an expected value smaller than five, Fisher’s exact test was used. Correlations between the two groups were determined by using Spearman correlation test. The values
Results
The results of demographic, anthropometric and biochemical parameters except that of vitamin B12 status are shown in Table 1. There were no significant differences between the PKU and control groups in terms of age, gender, weight, height, weight for height, full blood count and ferritin. When the PKU and control groups were compared with regard to serum B12 concentrations, no significant difference was found (Table 2). When functional vitamin B12 deficiency was examined, the median plasma MMA was significantly higher in the PKU group than in the control group (
Demographic, anthropometric and biochemical parameters of the PKU and control groups.
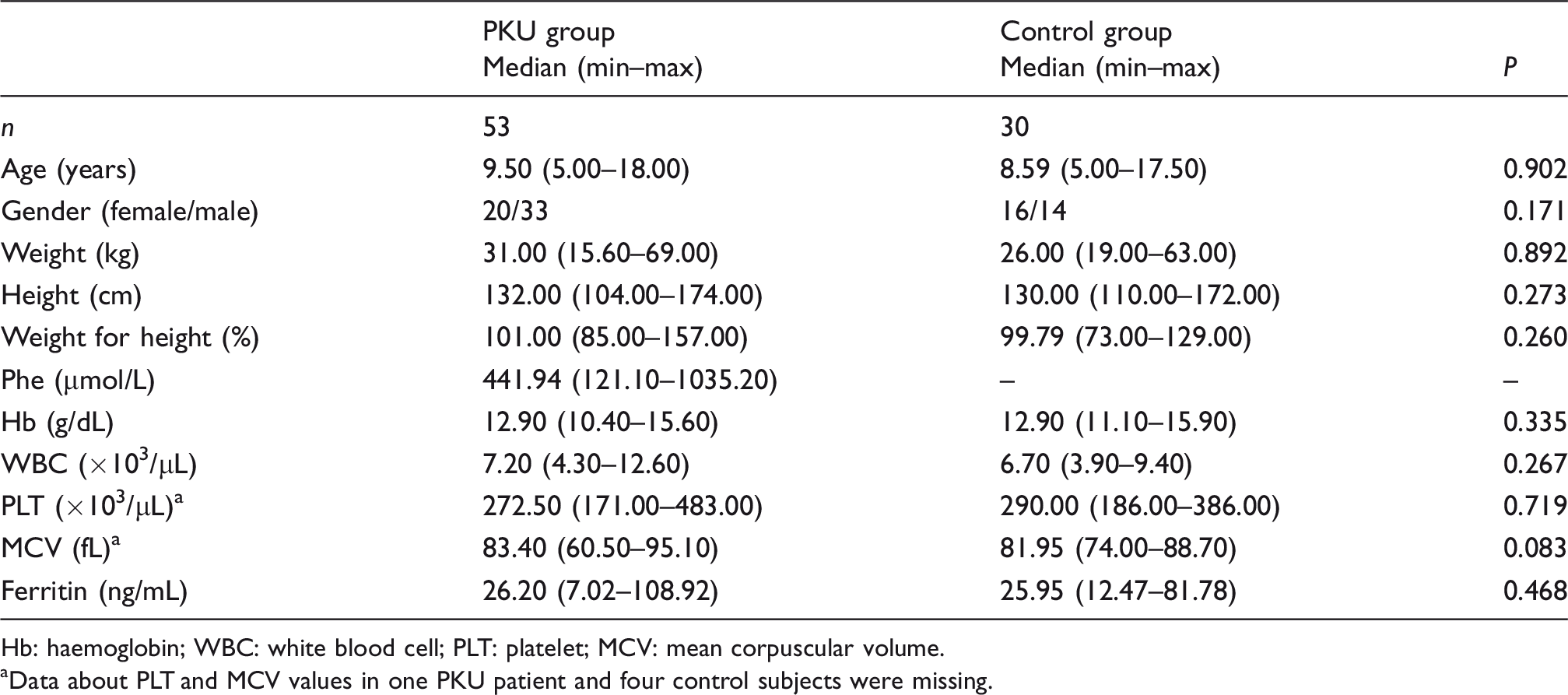
Hb: haemoglobin; WBC: white blood cell; PLT: platelet; MCV: mean corpuscular volume.
aData about PLT and MCV values in one PKU patient and four control subjects were missing.
Biochemical parameters associated with vitamin B12 status.
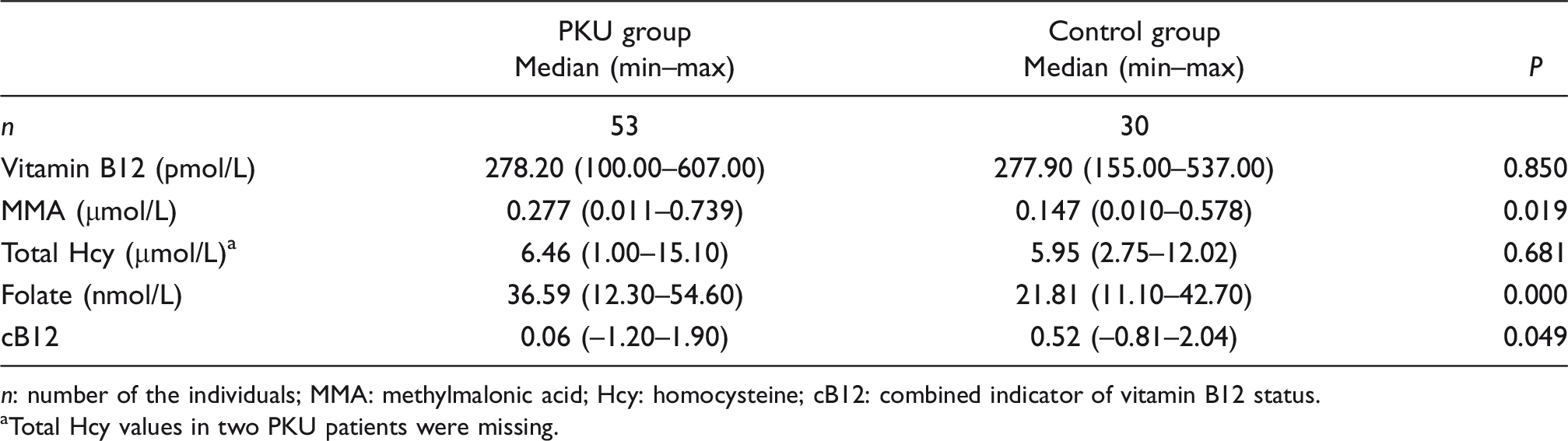
aTotal Hcy values in two PKU patients were missing.
When we divided the PKU group according to diet adherence based on Phe concentrations, 31 PKU patients (58.5%) had high adherence to the restricted special diet, while 22 patients (41.5%) were not strictly adherent to the diet (Table 3). There were no significant differences between the PKU subgroups and the control group in terms of age, gender, weight, height, full blood count and ferritin (data not shown). Phenylalanine concentrations differed significantly between the high adherence and low adherence subgroups (
Pairwise comparisons of biochemical parameters associated with vitamin B12 status in the PKU subgroups and the control group.
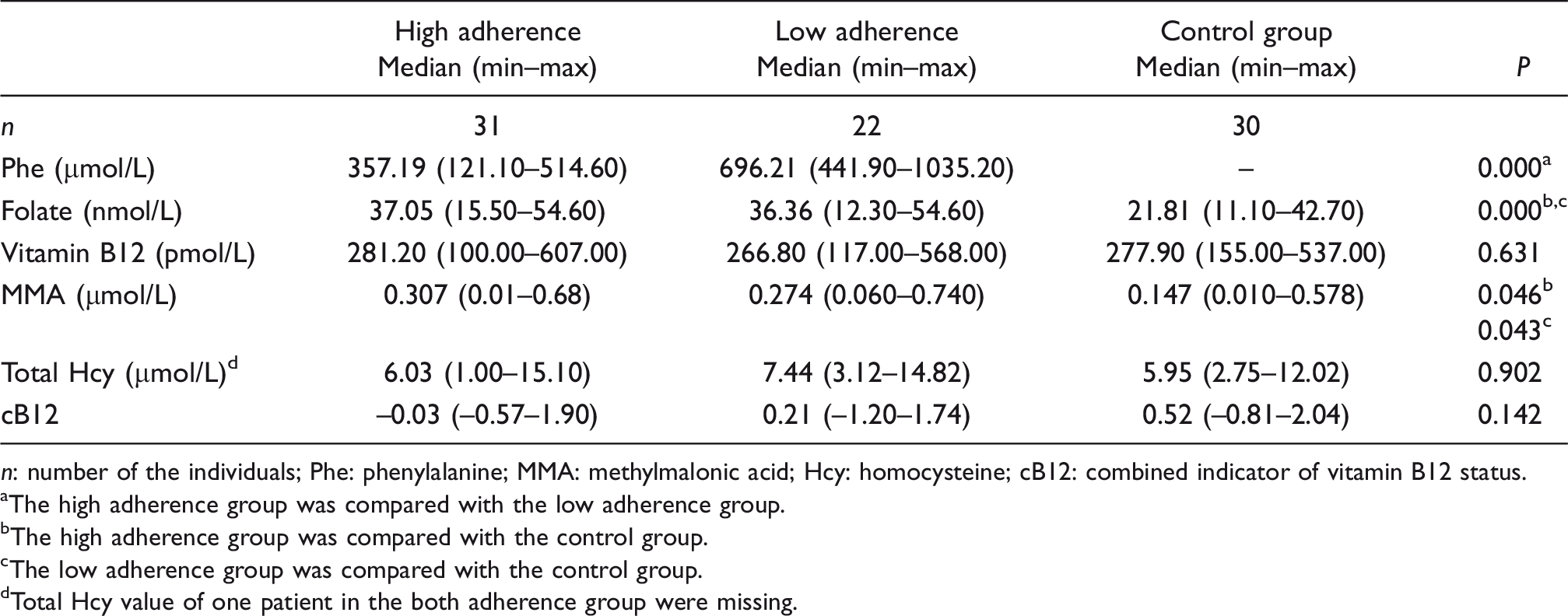
aThe high adherence group was compared with the low adherence group.
bThe high adherence group was compared with the control group.
cThe low adherence group was compared with the control group.
dTotal Hcy value of one patient in the both adherence group were missing.
Table 4 shows the number of the individuals with increased plasma MMA and/or total Hcy and with decreased serum vitamin B12 and cB12 in the PKU and control groups according to cut-off utilized to evaluate functional vitamin B12 deficiency. Thirty patients (56.6%) with PKU had elevated plasma MMA and one patient (2.0%) had high total Hcy concentrations. Of 46 PKU patients with normal vitamin B12 concentrations, 56.5% had high MMA concentrations and 19.6% had low cB12 values (<–0.5). In the control group, vitamin B12 and total Hcy concentrations were normal, but 26.7% of the controls had elevated MMA concentrations and 13.3% of the controls had a low cB12 value.
Cross-tabulation analysis of functional vitamin B12 deficiency in the PKU and control groups.
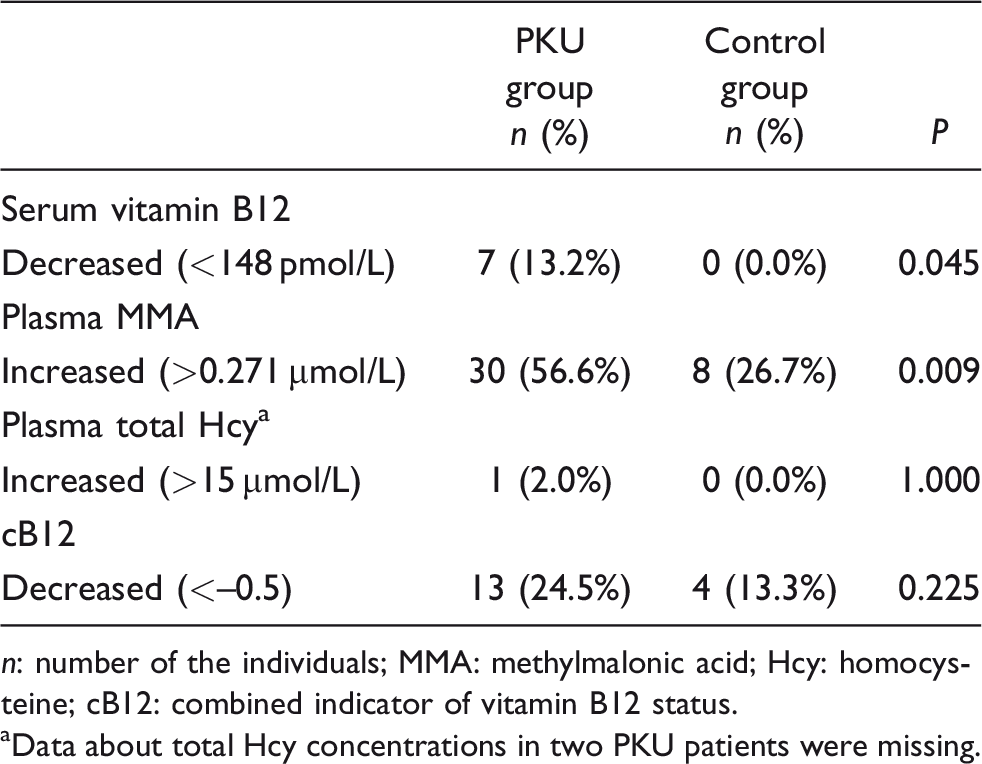
aData about total Hcy concentrations in two PKU patients were missing.
When the cut-off value for vitamin B12 concentrations was considered as 221 pmol/L, the upper limit of reference values, to evaluate subclinical vitamin B12 deficiency, 34% of the PKU group (
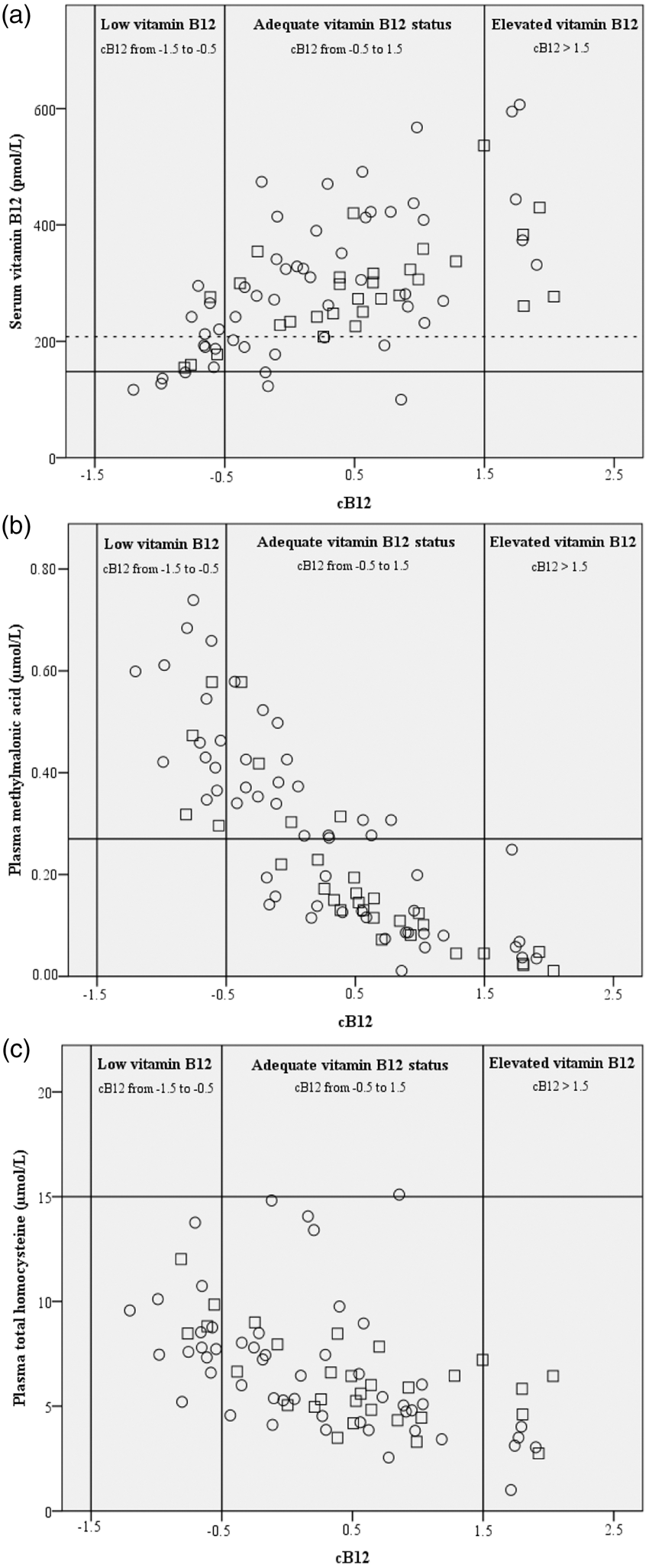
The relations between serum vitamin B12 (a), plasma MMA (b) and plasma total Hcy (c) values and cB12 in the PKU (○) and control (□) groups. Horizontal straight lines show cut-off values of parameters. Cut-off values are 148 pmol/L and 221 pmol/L (dashed line) for serum vitamin B12, 0.270
In the correlation analysis of parameters related to vitamin B12 deficiency, vitamin B12 had a significant positive correlation with cB12 in the PKU patients (r = 0.618,
Discussion
This is the first study to evaluate MMA, Hcy and cB12 as well as vitamin B12 to detect functional vitamin B12 deficiency in children and adolescents with classical PKU and to compare results with those from age and gender matched healthy controls. One important finding of this study was that the PKU patients had higher MMA concentrations than the controls, although vitamin B12 concentrations were within the reference range without a difference between the two groups. Several studies about vitamin B12 deficiency in a range of diseases showed superiority of MMA and Hcy, especially the former, over serum vitamin B12 analysis, as markers of functional deficiency, and suggested their evaluation in combination.19,24,26 However, in studies on PKU patients, vitamin B12 deficiency was generally evaluated by using serum vitamin B12 only8,10 or a combination with plasma Hcy.11,14–16 There have been only three studies using MMA measurement; however, PKU patients were not compared with healthy controls.6,12,13 In those studies including adults with PKU, there was no significant difference in MMA concentrations, but the patients with normal or low vitamin B12 concentrations had increased concentrations of MMA and/or Hcy6,12,13 or decreased concentrations of HoloTC.12,13 Although HoloTC is considered a relatively early biomarker of vitamin B12 deficiency, there is conflicting evidence for its diagnostic performance.21,22,34
HoloTC is an active form of serum vitamin B12 binding to transcobalamin and entering into cells. While several studies have shown that it is the most sensitive marker to detect vitamin B12 deficiency at the earliest time,35,36 other studies have revealed that it does not have a superiority over serum vitamin B12 test.17,37 There are great differences in evidence from the literature about its half-life, correlations with serum vitamin B12, sensitivity after absorption, cut-off values and measurement methods and effects of genetic variants.17,38–40 Although there has been an increase in the clinical use of HoloTC in developed countries like Australia, Germany and UK, its routine use is still limited due to costs of its measurement methods.36,38 There have been two studies measuring HoloTC in adult PKU patients. Whereas one of them showed no difference in HoloTC between the patients with vitamin B12 concentrations of over 200 pmol/L and those with vitamin B12 concentrations of below 200 pmol/L, 12 the other revealed that HoloTC remained in the reference values despite the presence of a difference. 13 These two studies were different in terms of measurement methods and the reference values used (>50 pmol/L and 19–119 pmol/L, respectively).
The second important finding of the present study, which is the first to calculate cB12 in PKU patients, was that the PKU group had a lower cB12 value than the control group even though within the reference values. On the other hand, one fourth of the PKU patients and more than one tenth of the healthy controls had decreased vitamin B12 status which requires vitamin B12 supplements.
Folate concentrations were also significantly higher in the PKU group (both high and low adherence) than in the control group. Some researchers also reported that PKU patients had higher folate concentrations. They attributed these high folate concentrations to the fact that a Phe-restricted diet includes vegetables to a great extent.11,13,14 Stølen et al. reported that PKU patients younger than 18 years had much higher folate concentrations than adults with PKU, which could cause risk for some diseases. 15
The Phe-restricted diet is difficult to comply with and has to be maintained for a lifetime.1,41 Patients have to avoid food rich in protein (food rich in vitamin B12) like meat, eggs and fish. PKU patients, especially adolescent and young patients, have insufficient adaptation to the diet. There have been three studies in which PKU patients have been categorized according to their compliance with the diet and compared with healthy controls. Robinson et al. 11 and Kose and Arslan 42 showed that the patients having low adherence to their diet and/or not receiving amino acid supplements had lower vitamin B12 concentrations than those having higher adherence to their diet or healthy controls, which is consistent with the results of the present study. Interestingly, Schulpis et al. reported that PKU patients with adherence to their diet had both lower vitamin B12 concentrations, lower dietary intake of vitamin B12 and in turn higher Hcy concentrations than those without adherence to their diet and healthy controls. 16 It was attributed to low vitamin B12 contents in amino acid supplements enriched in vitamins in the patients following a strict diet. MMA concentrations were not measured, and only Hcy concentrations were determined in the abovementioned studies.11,16 In the current study, although the high adherence and low adherence groups had normal vitamin B12 concentrations, they had higher MMA concentrations than the control group. These findings suggest that PKU patients, whether they comply with their diet or not, can be at risk of vitamin B12 deficiency.
Even though the number of studies about vitamin B12 deficiency in PKU patients is increasing, they are still low in number and have yielded conflicting results.6,8,10–16,42 While most of the studies reported presence of vitamin B12 deficiency in PKU patients, some studies showed that PKU patients had high vitamin B12 concentrations. The reasons for these conflicting findings can be explained by differences not only in age groups and sizes of study samples and adherence to diet but also cut-off values for MMA and Hcy, biomarkers for functional B12 deficiency. Besides, there is no agreement on cut-off values for each parameter. The most commonly used cut-off values, also used in the present study, were <148 pmol/L (200 pg/mL) for vitamin B12, recommended by the WHO as well,
23
>0.271
While MMA concentrations are a more sensitive and specific biomarker for the diagnosis of vitamin B12 deficiency, they can be affected by renal dysfunction; polymorphisms in the relevant biochemical pathways. 44 Serum/plasma creatinine concentrations were not measured in any of the studies about vitamin B12 deficiency in PKU. Only in a study by Prochazkova et al., urinary MMA and creatinine concentrations were measured, but no significant findings were reported. 13 In our study, there were not any individuals with a known renal impairment. Therefore, taking account of cB12 as a biochemical index in evaluation of vitamin B12 deficiency can minimize false-positive and false-negative results caused by different biomarkers and cut-off values of these biomarkers.
This study is the first to show a relation between cB12 and vitamin B12, MMA and Hcy in PKU patients. Vugteveen et al. 6 and Schulpis et al. 16 reported a negative relation between vitamin B12 and its functional markers. Stølen et al. and Schulpis et al. could not find a significant correlation between Phe and vitamin B12 deficiency-related parameters,15,16 which is compatible with the results of our study. However, Huemer et al. noted that there was a negative correlation between Phe and vitamin B12 and folate. 14 Similarly, Vugteveen et al. showed a negative relation between vitamin B12 and Phe when they took account of all age groups. 6
The current study has some limitations. One limitation is that HoloTC, a relatively new biomarker of vitamin B12 deficiency, could not be measured. The second limitation was that serum creatinine was not measured. Since MMA is affected by renal functions, this potential confounder was not accounted for. Although none of the individuals had renal impairment based on their self-reports, renal impairment could not be fully excluded in this study because serum creatinine could not be measured. The third limitation is that dietary contents could not be determined, and that neurological evaluations could not be made in the PKU patients. The last limitation is concerned with the analytical methods used. Phe measurements were performed with fluorimetry and Hcy measurements were made with HPLC with a fluorescence detector. At present, these measurements are carried out with more specific and sensitive mass spectrometric methods, which are even considered as reference tools.45,46 However, establishing the substructure for these methods in clinical laboratories is not still widespread, since it is costly and requires staff with special qualities. The methods used to measure Phe and Hcy in the present study were validated and were being routinely used at the time of the study.
Conclusion
This study revealed significantly increased plasma MMA concentrations in PKU patients compared with healthy controls, suggestive of functional vitamin B12 deficiency. However, other markers of vitamin B12 status (serum vitamin B12 concentration and plasma homocysteine concentration) were not significantly different between the PKU and control groups. Even children and adolescents with PKU following a strict dietary regimen can be at risk of functional vitamin B12 deficiency. Assuming the PKU patients had normal renal functions, the findings suggest that plasma MMA may be the most sensitive indicator of vitamin B12 status especially in the patients with low-normal vitamin B12 concentrations. The use of cB12 as a biochemical index did not provide additional information compared with the measurement of plasma MMA alone in present study and in fact the use of cB12 would have classified some patients with increased plasma MMA concentrations (functional vitamin B12 deficiency) as having adequate vitamin B12 status. The performance and usability of cB12 should be investigated in a larger sample of children and adolescents.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a grant from Dokuz Eylul University Scientific Research Projects Coordination Unit (Project number: 2009-KB-SAG-37).
Ethical approval
This study was approved by Ethics Committee of Dokuz Eylul University Faculty of Medicine (Approval date: 2 March 2011, Number: 88-GOA) and informed consent was obtained from all the participants.
Guarantor
MA.
Contriburorship
HI designed and supervised the study and provided funding acquisition. II, PTK, EK and NA performed the selection of appropriate participants and evaluated clinical data. MA and MK collected the samples and optimized the method for methylmalonic acid assay. MA carried out analysis of methylmalonic acid. MA and HI were responsible for statistical analysis and interpretation. NA contributed to data analysis. MA wrote the article. HI contributed to writing and editing of the article. All authors approved the final version of the article.
