Abstract
An 81-year-old man was admitted to hospital with symptomatic coronavirus disease (COVID-19) infection. He had a background of progressive chronic inflammatory demyelinating polyneuropathy associated with Waldenstrom’s macroglobulinaemia. His plasma creatinine on four separate samples was inconceivably low (all ≤13 μmol/L), as measured by a Beckman Coulter enzymatic assay) after being 72 μmol/L 3 months earlier. On further investigation, his serum immunoglobulin M (IgM) was 15.4 g/L and his plasma creatinine measured by Roche enzymatic and Roche Jaffe methods was 62 μmol/L and 64 μmol/L, respectively. This was consistent with results post dilution studies and polyethylene glycol (PEG) precipitation on the Beckman Coulter assay. There was no evidence of similar interference when reviewing creatinine results from 10 other patients with IgM paraproteinaemia who had been tested in our laboratory. Clinicians and laboratorians are reminded that enzymatic creatinine is not free from interferences. IgM paraprotein negative interference of enzymatic creatinine is rare and specific to a patient’s IgM and assay combination, but should be considered in patients with an unexplained low enzymatic creatinine result. Useful investigations to identify an interference include dilution studies, PEG precipitation and measuring creatinine on an alternative method such as Jaffe, mass spectrometry or an enzymatic method from a different platform.
Case presentation
An 81-year-old man was admitted to hospital with 3 days of nausea, vomiting and general malaise. His wife had recently contracted coronavirus disease (COVID-19). COVID-19 infection in the patient was confirmed by polymerase chain reaction (PCR); he was started on nirmatrelvir/ritonavir and discharged after 3 days. His past medical history included chronic inflammatory demyelinating polyneuropathy, Waldenstrom’s macroglobulinaemia, epilepsy, Paget’s disease, atrial fibrillation, ischaemic heart disease and hypertension.
His plasma creatinine, as measured by a Beckman Coulter AU5822 enzymatic assay, was consistently low during the admission, with four results all reported as ≤13 μmol/L, strikingly low results. This was considered inconsistent with a result of 72 μmol/L, performed less than 3 months prior in the community laboratory using a Roche enzymatic assay and was inconsistent with plasma urea results of 4.0–6.6 mmol/L measured on the same four specimens during this admission. Interference was thought likely and the history of Waldenstrom’s macroglobulinaemia, with immunoglobulin M (IgM) kappa paraprotein, prompted investigation for suspected IgM paraprotein interference.
Investigations for suspected interference
Investigation results for a single sample for plasma creatinine (μmol/L).

PEG: polyethylene glycol.
The Beckman Coulter enzymatic creatinine assay involves addition of creatininase, creatinase and sarcosine oxidase, presumed to be in reagent 1 (R1), leading to the conversion of endogenous creatinine to glycine, formaldehyde and hydrogen peroxide. The addition of peroxidase converts 4-aminoantipyrine, presumed to be in reagent 2 (R2), into a blue pigment through a trinder reaction. The absorbance prior to addition of R2 is subtracted from the final absorbance, which relates to the concentration of creatinine. The primary wavelength is measured at 600 nm where the blue pigment is highly absorbant, while a secondary wavelength at 700 nm has minimal absorbance by the blue pigment formed and corrects for any variations in light intensity throughout the reaction.
Reaction curves for this patient demonstrated a high baseline for enzymatic creatinine after the addition of R1, which resulted in minimal change in absorbance after addition of R2 in under usual conditions (Figure 1(a)). This resulted in artefactually low creatinine. With the addition of 25% PEG in 1 in 2 dilution (Figure 1(b)), the shape of the reaction curve more closely resembled a normal profile (Figure 1(c)), with relatively flat baseline after addition of R1, before addition of R2. However, with addition of 9% PEG in 1 in 2 dilution (Figure 1(d)), there was a more gradual rise in absorbance after addition of R1 compared to the undiluted sample, which appeared to continue even after addition of R2, leading to artefactually high creatinine under these conditions. Furthermore, for the current patient, the undiluted sample and the sample at 1 in 2 dilution in 9% PEG 6000 had rising absorbance at both the primary wavelength of 600 nm and secondary wavelength of 700 nm, whereas for the normal profile or current patient sample diluted in 25% PEG 6000 there was minimal absorbance at the secondary wavelength of 700 nm. Reaction curves for creatinine for: (a) current patient; (b) current patient with addition of 25% PEG 6000 at 1 in 2 dilution; (c) a patient where no interference was suspected; (d) current patient with addition of 9% PEG 6000 at 1 in 2 dilution.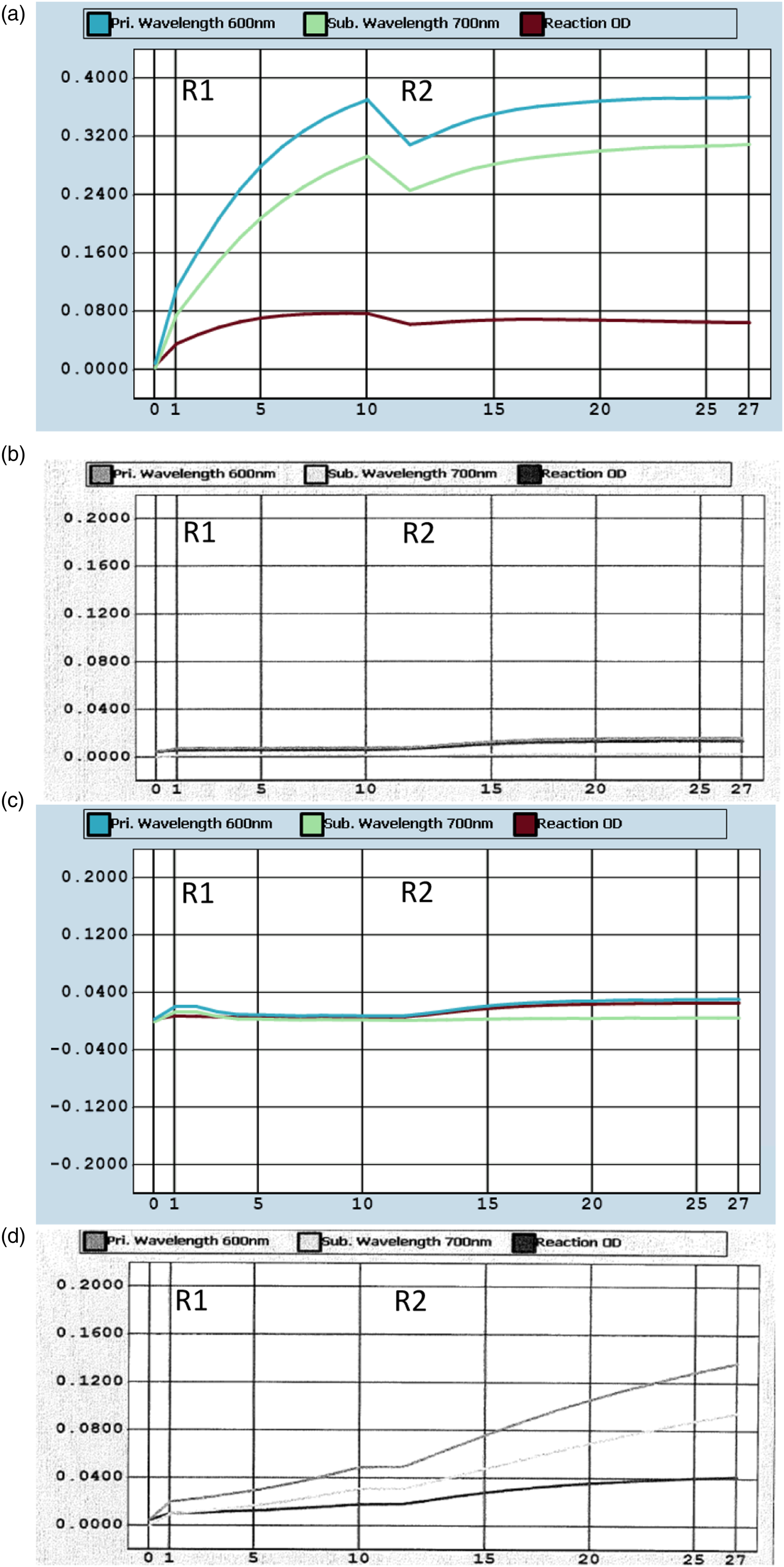
Historic samples from ten patients with IgM paraprotein (each with total IgM >10 g/L) measured in our laboratory were retrospectively reviewed to determine whether the interference was an isolated event. Results from Beckman Coulter enzymatic, Roche enzymatic and Abbott Jaffe creatinine from serial monitoring at different time points were compared for these patients. After accounting for expected analytical and biological variation, none of these patients had evidence of IgM paraprotein interference with either the Beckman Coulter or Roche enzymatic methods. This implies that the IgM paraprotein negative interference seen in the current patient is specific to the combination of the patient’s specific IgM paraprotein with the Beckman Coulter enzymatic creatinine method, rather than a generalised IgM interference with the Beckman Coulter enzymatic creatinine method.
Discussion
Enzymatic creatinine assays are starting to replace the more traditional chromogen-based methods (Jaffe assays) due to a decreased susceptibility to interferences than the Jaffe method.1,2 However, clinicians and laboratorians are reminded that enzymatic creatinine methods are not free from interferences.
Although positive interference for enzymatic creatinine assays by IgM paraproteins have been recognised for some time,3–6 and IgM paraprotein interference of enzymatic creatinine assay is noted in the Beckman Coulter instructions for use as a rare interference, negative interferences have only recently been described. One case of negative interference has been described with two IgM kappa paraproteins on a Mindray BS-360e platform, 7 while another case has been described with IgM kappa paraprotein on a Roche enzymatic creatinine method. 8
In both cases of negative interference with enzymatic creatinine due to IgM paraprotein previously described as well as our current case, the paraprotein resulted in a high baseline after addition of R1, leading to a lower change in absorbance than usual after addition of R2. PEG precipitation was successful in removing the interference in all cases. In the case of the Roche enzymatic creatinine interference, precipitation was reportedly visible when R1 was mixed with the patient sample, consistent with the reaction curves. 8
Dilution studies were performed in this current case. When the sample was diluted sufficiently, the results were similar to those obtained after PEG precipitation. However, our current case had a paradoxical positive interference at 1 in 2 dilution, which was not present at 1 in 4 or 1 in 8 dilutions. This may have been present to a lesser extent in the previously described case on the Mindray BS-360e platform, with creatinine 134 μmol/L at 1 in 2 dilution, compared with 110 μmol/L at 1 in 5 dilution, or 108 μmol/L after PEG precipitation. 7 From inspection of the reaction curves, the most likely explanation is that small dilutions may not be adequate to completely eliminate the precipitation of a patient’s IgM paraprotein with R1. It appears that the background absorbance from the IgM with R1 proceeds more slowly than without dilution and continues to increase even after the addition of R2, resulting in a falsely high enzymatic creatinine calculated from the change in absorbance.
Positive interferences with enzymatic creatinine from IgM paraprotein are more widely reported than negative interferences.3–6 This may be due to precipitation occurring more with R2 than R1. However, as shown by our patient’s sample at 1 in 2 dilution, a positive interference may also happen with a slower precipitation with R1, which may conceivably occur in other patient samples with IgM paraprotein without dilution.
IgM paraprotein interference appears to be idiosyncratic to the combination of a patient’s IgM paraprotein with a specific platform. Our patient’s IgM affected the Beckman enzymatic creatinine method, while Roche enzymatic creatinine was unaffected. In contrast, a previously reported case demonstrated IgM paraprotein interference with Roche enzymatic creatinine but did not have any interference with enzymatic creatinine on Abbott, Siemens ADVIA or VITROS platforms. 8 Whilst the Abbott, Siemens, Roche and Beckman Coulter enzymatic creatinine assays have similar design, minor differences in pH, preservatives or concentrations of reagents may lead to IgM precipitation in one reagent and not other reagents.9,10 Our retrospective review indicates that this was an isolated and likely rare event which was not seen in other patients with elevated IgM seen in our laboratory.
Conclusion
Negative interference of enzymatic creatinine assays by IgM paraproteins is rare but should be suspected in patients with unexpectedly low enzymatic creatinine results. In this case the creatinine result was physiologically implausible and history of IgM paraprotein was already previously identified making it relatively easy to identify the interference. However, it is conceivable that a similar interference may reduce a truly elevated creatinine into the reference interval, thus masking a diagnosis of renal failure. Laboratory staff validating creatinine results should remain suspicious of interference if the observed result does not match the clinical picture or is out of keeping with other markers of renal function such as urea or cystatin C.
Useful investigations to confirm interference include dilution studies, PEG precipitation and measuring creatinine on an alternative method, such as Jaffe, mass spectrometry, or an enzymatic method from a different platform. If performing dilution studies, it is important to test at multiple dilutions to achieve concordant results and decrease the risk of a falsely high result due to inadequate dilution. Likewise, if using PEG precipitation, the PEG must be at high enough concentration to ensure precipitation of the interfering paraprotein.
Footnotes
Acknowledgements
Thanks to Canterbury Southern Community Laboratories for running creatinine on the Roche enzymatic method and LabPlus Auckland for running creatinine on the Roche Jaffe method.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Informed consent was obtained from the patient prior to submission.
Guarantor
BL.
Contributorship
BL, ST and CF initiated the investigation. BL wrote the first draft. KI ran experiments on the Beckman Coulter analyser. MD was the physician managing the patient’s hospital care. All authors reviewed and edited the article and approved the final version of the article.
