Abstract
Although enzymatic creatinine methods are subject to fewer interferences than traditional Jaffe creatinine methods, every method in clinical chemistry has limitations. We report, for the first time in the literature, a case of an immunoglobulin M (IgM) paraproteinaemia causing an undetectably low creatinine result on the Roche enzymatic assay. This interference did not occur with other enzymatic creatinine methods produced by Abbott and Siemens or the Roche Jaffe, VITROS dry slide and liquid chromatography with tandem mass spectrometry (LC-MS/MS) creatinine methods. IgM interference was confirmed as patient serum precipitated with polyethylene glycol (PEG) and anti-IgM antiserum yielded detectable Roche enzymatic creatinine results comparable to unaffected methods. The patient’s serum formed an obvious precipitate when mixed with reagent one of the Roche enzymatic creatinine method. This is in contrast to a report of positive interference from IgM paraproteinaemia in a different enzymatic creatinine method, which showed that a precipitate formed when mixing blood with reagent two. As each patient’s paraprotein has a unique structure, it is possible that there are variations in the chemical characteristics of IgM paraproteins between patients. This, as well as IgM-class antibodies’ tendency to form multimers and aggregates, can lead to unpredictable assay interferences and precipitation tendencies between different manufacturers of enzymatic creatinine reagents and their incubation steps. This case highlights the importance of continuing to question and investigates results that do not fit the clinical picture, especially as more laboratories switch from primarily using traditional Jaffe creatinine methods to enzymatic creatinine methods.
Introduction
Creatinine is one of the most important analytes measured in clinical chemistry due to its utility as a non-invasive, and relatively cheap, biomarker of renal function. Specifically, blood creatinine concentrations are commonly used in equations to calculate the estimated glomerular filtration rate (eGFR). 1 Although eGFR has recognised and well-documented limitations, 2 its increased convenience and accessibility over measured glomerular filtration rate has contributed to its widespread application in the diagnosis and monitoring of chronic kidney disease (CKD). Additionally, an increase in creatinine of as small as 26 μmol/L can be used to diagnose acute kidney injury (AKI), 3 which requires urgent medical treatment. The timely determination of precise and accurate creatinine results for the appropriate management of CKD and AKI patients is therefore vital.
Historically, many clinical laboratories primarily measured creatinine using the Jaffe method, which is based on an orange-red colour change reaction of creatinine with picric acid in an alkaline environment. 4 Despite the cost effectiveness of Jaffe methods, they are subject to well-documented positive interferences from glucose, acetoacetate, β-hydroxybutyrate, high total protein, ascorbic acid and cephalosporins, and significant negative interference from bilirubin.5,6,7 As well as being subject to fewer and less clinically significant interferences than Jaffe methods, 5 enzymatic creatinine methods produce more accurate and precise results for use in equations to calculate eGFR, allowing for more reliable diagnosis and monitoring of CKD.2,8 This has resulted in Kidney Disease: Improving Global Outcomes (KDIGO) recommending that clinical laboratories transition from the traditional use of Jaffe methods to more robust, accurate and precise enzymatic methods. 9 As more laboratories across the world adopt this recommendation, it is important to communicate factors that may affect enzymatic creatinine results to help prevent possible inappropriate patient management. We report, as far as we know, a currently unrecognised negative interference on an enzymatic creatinine method.
Case report
A 77-year-old male consulted with his GP for the annual monitoring of his medical conditions including hypertension, hypercholesterolaemia, bronchiectasis, non-diabetic hyperglycaemia, oesophagitis, benign prostatic hyperplasia, Meniere’s disease, psoriasis, myasthenia gravis, and immunoglobulin M (IgM) paraproteinaemia. His regular medications included losartan, rosuvastatin, budesonide with formoterol inhaler, lansoprazole, tamsulosin and betahistine. Regular monitoring blood tests included urea, creatinine and electrolytes (UEs), serum and urine protein electrophoresis, and others. The laboratory information system contained UEs results from April 2017 to September 2019. Throughout this period, the patient’s serum creatinine fluctuated between 90 and 102 μmol/L, with a calculated Modification of Diet in Renal Disease (MDRD) eGFR of > 60 mL/min/1.732. Serum samples were received in June, July and November 2020 where the serum creatinine results were suddenly < 5 μmol/L, rendering the MDRD eGFR incalculable. No other significant changes in clinical status or blood test results, including urea, were noted.
Creatinine results on the same serum and lithium heparin plasma samples analysed by different creatinine methods.
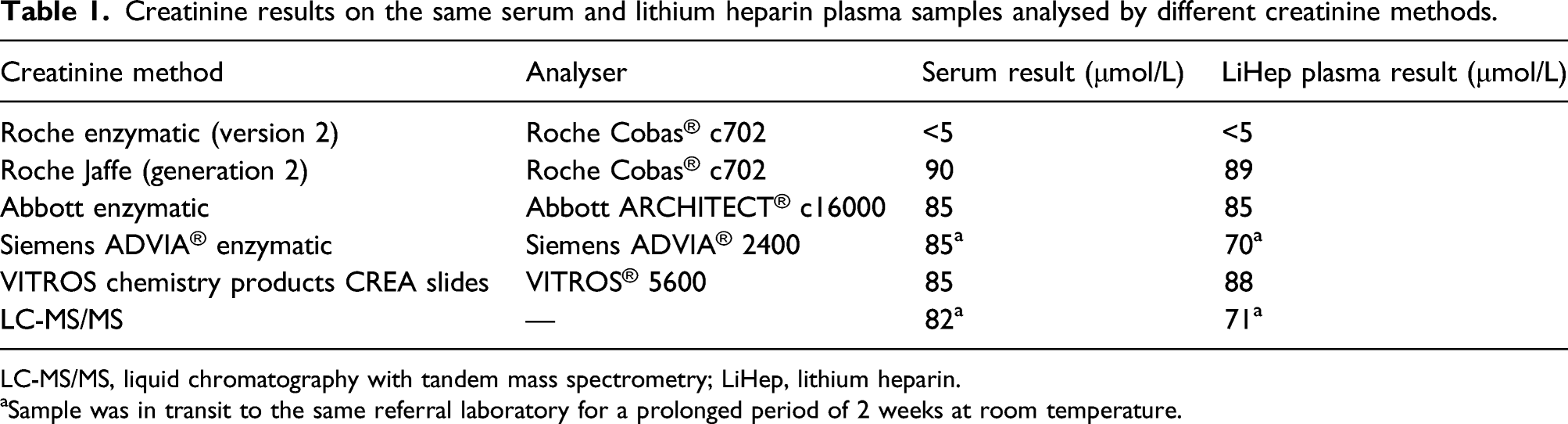
LC-MS/MS, liquid chromatography with tandem mass spectrometry; LiHep, lithium heparin.
aSample was in transit to the same referral laboratory for a prolonged period of 2 weeks at room temperature.
Interference investigations
Roche enzymatic creatinine (version 2) results before and after PEG 6000 precipitation of the patient’s serum and plasma, and control serum samples from different patients with and without detectable paraproteinaemia.
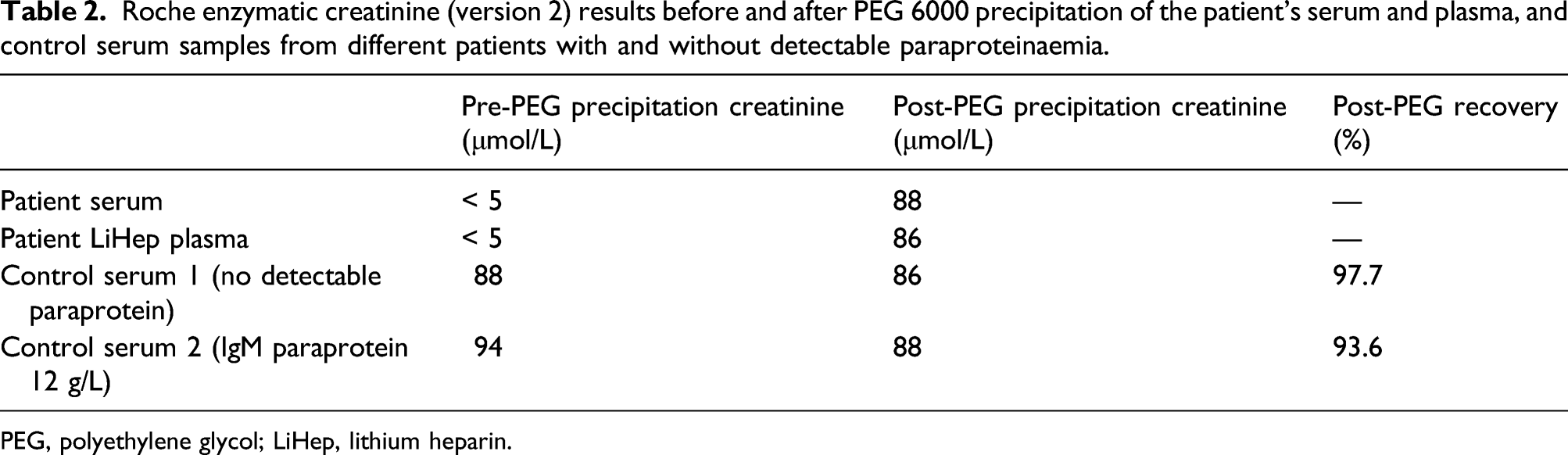
PEG, polyethylene glycol; LiHep, lithium heparin.
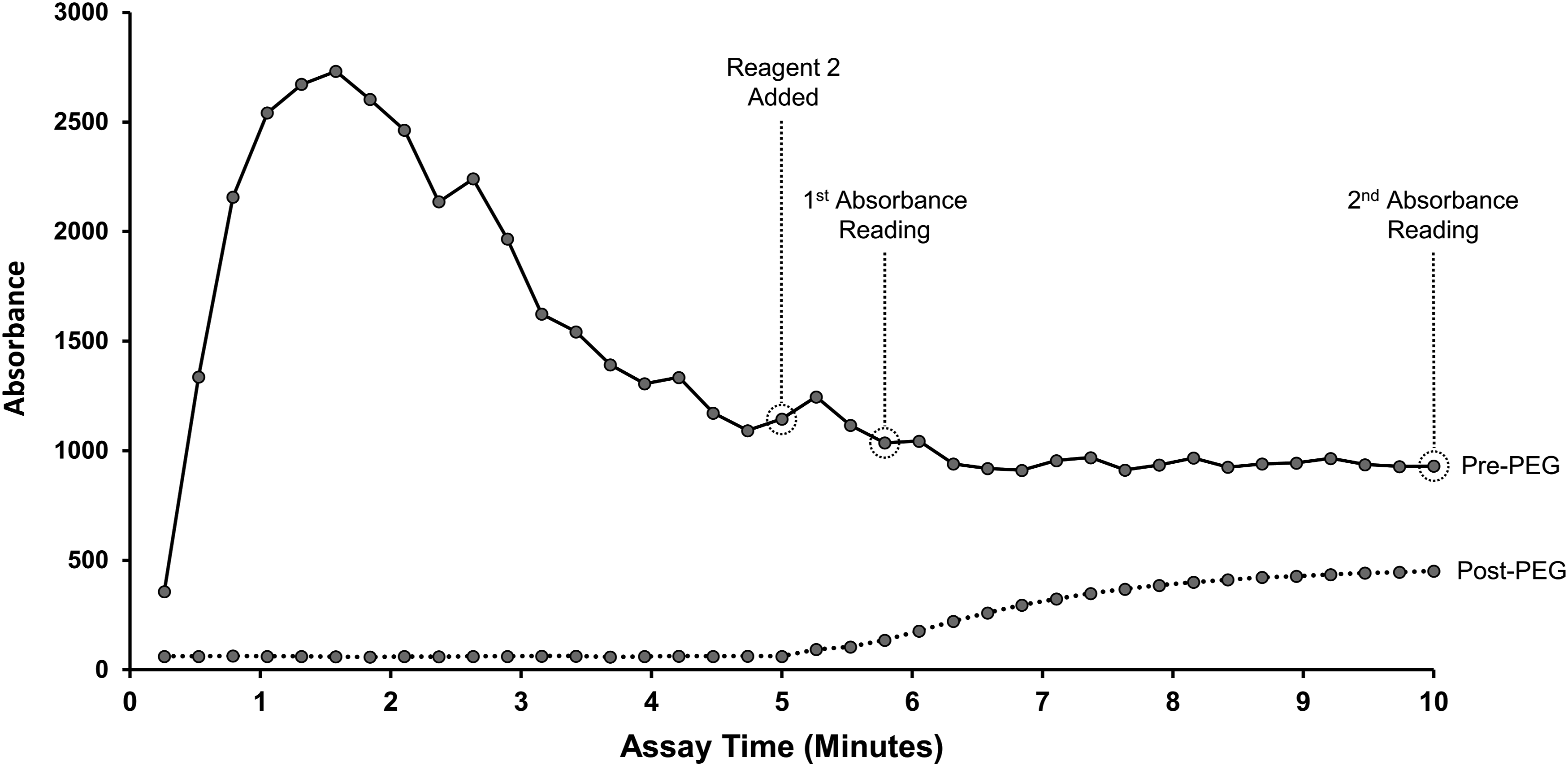
Reaction curves of the patient’s creatinine measured on a Roche Cobas c702 analyser using the Roche enzymatic method (version 2) on a pre-PEG precipitation serum sample (solid line) and a post-PEG precipitation serum sample (dashed line). Reagent one is added at the beginning of the reaction (absorbance not measurable), and reagent two is added at the five-min point. PEG, polyethylene glycol.
Next, we developed a protocol to more specifically precipitate the IgM paraprotein. The patient’s serum was mixed with anti-IgM antiserum from Helena Biosciences at a volume ratio of 1:7, and left to incubate at 4°C for 48 h. This ratio was chosen as this was the maximum relative volume of antisera that could be used which would not dilute the creatinine to below the limit of detection of 5 μmol/L. The solution was then centrifuged at 17,000 RCF for 5 min, and the supernatant was analysed for creatinine on a Roche Cobas® c702 analyser using the Roche enzymatic (version 2) method. Control serum one from the PEG precipitation experiment was also subjected to the same anti-IgM incubation and centrifugation process. The anti-IgM-incubated patient serum yielded a creatinine result of 64 μmol/L, and the reaction curve was very similar to the post-PEG precipitation sample in Figure 1. Control serum one incubated with anti-IgM yielded a creatinine result of 72 μmol/L (82% recovery).
Finally, the kinetics of the interference during the assay reaction were investigated. The patient’s serum was mixed with reagent one and reagent two from the Roche enzymatic creatinine (version 2) method separately, and then reagent one and two together, in test tubes in accordance with the manufacturer’s instructions. On visual inspection of the reaction, a white precipitate was observed within a few seconds of the patient’s serum being mixed with reagent one. The precipitate remained when reagent two was added. No precipitate formed when the patient serum was mixed with reagent two only. Control serum from a patient without detectable paraprotein and another patient with an IgM paraproteinaemia of 16 g/L were also mixed with the Roche enzymatic creatinine reagents in the same way. Neither of the two control serum formed a precipitate when mixed with the two reagents separately or together. The supernatants from the PEG and anti-IgM precipitation protocols of the patient’s serum did not form any precipitate when mixed with either reagent separately or together, confirming that these preparations no longer contained the interferent.
Discussion
The interference experiments we have described show that the patient’s IgM paraprotein is the likely negative interferent in this case. This was demonstrated by showing that PEG (Table 2) and anti-IgM antiserum precipitation of the patient’s serum, which had previously given unmeasurably low results, gave measurable creatinine values which were comparable to unaffected method results (Table 1) (with the exception of plasma that was delayed in the post at room temperature). This was unexpected as published cases reporting IgM interference in enzymatic creatinine methods suggest that this is usually a positive, rather than a negative, interference.12–17 As far as we are aware, this is the first published case of IgM paraproteinaemia causing falsely decreased enzymatic creatinine results.
The Roche, Abbott and Siemens enzymatic creatinine methods are based on similar reactions with two incubation periods. During the first incubation period, serum or plasma is added to reagent one, which contains creatinase, sarcosine oxidase and catalase to remove endogenous creatine. The second incubation period starts when reagent two, which contains creatininase and peroxidase, is added to the mixture. The formed peroxide then reacts with a manufacturer-dependent chromogen to initiate a colour change, the intensity of which is proportional to the concentration of creatinine in the sample. The similar assay design between the different enzymatic creatinine manufacturers does not provide an obvious explanation as to why only the Roche enzymatic method is negatively affected by this IgM paraproteinaemia. However, other researchers have reported varying susceptibilities of positive interference from IgM paraproteinaemia between different enzymatic creatinine manufacturers.13,15,16 This suggests that despite similar assay design, subtle differences in the pH, preservatives and/or ionic strengths of the reagents between manufacturers could cause some IgM paraproteins to precipitate in some reagent solutions and not others.
The sample-reagent mixing experiments demonstrate why we obtained an undetectable creatinine result in this patient. We observed that a precipitate was formed when mixing patient serum with reagent one, but not reagent two, of the Roche enzymatic creatinine method. The consequence of this can be observed in the reaction curves of the pre- and post-PEG serum samples displayed in Figure 1. The IgM precipitation which occurred during incubation with reagent one resulted in increased sample turbidity, causing an absorbance peak several orders of magnitude greater than the absorbance peak of the post-PEG precipitation sample. This prevented a low and steady absorbance baseline being established before the addition of reagent two. Additionally, this precipitate remained after reagent two was added, likely affecting the subsequent absorbance readings. It is possible that these two factors together led to an undetectable creatinine result. Mase et al. 15 -who reported a positive interference from IgM paraproteinaemia – reported that a similar exaggerated absorbance response occurred due to a precipitate that formed when adding patient blood to reagent two of their enzymatic creatinine method. The exact mechanism of this inconsistent paraprotein precipitation is difficult to elucidate. However, it could be that the variable characteristics of IgM paraproteins between patients can cause them to precipitate in either reagent one or reagent two, causing a negative or positive interference, respectively. As every other published case (that we could find) reporting an effect of IgM paraproteinaemia on enzymatic creatinine methods state a positive interference, it is possible that IgM precipitation in reagent two is the more common occurrence.
When presented with questionably low enzymatic creatinine results, causes and interferences including low protein intake, low muscle mass, significant hyperbilirubinaemia, endogenous and therapeutic dopamine, phenindione, methyldopa and N-acetylcysteine treatment following paracetamol overdose should be ruled out.6,18,19 This case shows that the possibility of interference from an undetected paraprotein should also be considered. If an interferent is identified, several options exist for the continued monitoring of renal function. Urea (often referred to as blood urea nitrogen) and cystatin C 20 are alternative biomarkers of renal function. However, these biomarkers have their own limitations or are not yet well established in routine practice. If interference from a paraprotein is identified and creatinine is required urgently, we have shown that PEG precipitation could provide an acceptable approximation of the true creatinine concentration. However, caution is advised as it is uncertain if the precision and accuracy of the creatinine results generated after PEG precipitation is satisfactory for monitoring CKD or diagnosing AKI. In this case, we determined that the most appropriate and simple solution is to send this patient’s serum away for analysis via an alternative and hopefully unaffected enzymatic creatinine method.
Conclusion
Although the enzymatic creatinine method is a significant improvement over the traditional Jaffe creatinine method, every method utilised in clinical chemistry has limitations. We report a case of IgM paraproteinaemia causing a negative interference in the Roche enzymatic method, with enzymatic creatinine methods from other manufacturers, and the Roche Jaffe, VITROS dry slide and LC-MS/MS creatinine methods, being unaffected. As this negative interference has not been described in the literature before, this case highlights the importance of clinicians and laboratory scientists continuing to question and investigate results that do not fit the clinical picture.
Footnotes
Acknowledgements
We would like to thank the patient for giving us consent to publish this case, and the subscribers to the Association of Clinical Biochemistry and Laboratory Medicine mailbase for their invaluable insight during the early stages of this investigation. In particular, thank you to Thomas G Morris, Nick Flynn, Robyn Shea and Natalie Mullee for accepting samples for analysis at their laboratories.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author’s Note
This case and its relevant data has been reported to the Medicines and Healthcare products Regulatory Agency (MHRA) (Reference number: 2021/012/021/401/501) and directly to Roche Global as a patient safety concern.
Ethical approval
The patient provided written informed consent for the publication of this case report after reviewing the final version of the article.
Guarantor
KCF.
Contributorship
KCF, TC and JM developed the protocol. AL and KCF gained patient consent. AL and ET collated information for the case study section. FGB supervised the laboratory work. KCF wrote the first draft of the article. All authors were involved in data analysis, and reviewed the article and approved the final version.
