Abstract
Background
The bromocresol green (BCG) and bromocresol purple (BCP) methods are widely used for albumin measurements in routine testing, but the BCG method is known to react with globulin fractions and to have low specificity for albumin. We evaluated a calibration method using different concentrations of human serum albumin standards (two-point calibration BCG method) with the aim of reducing the effect of globulin fractions on the BCG method in patients with hypoalbuminemia.
Method
In the two-point calibration BCG method, two concentrations of standard solutions and their calibration values are set based on the difference in albumin concentrations measured by the BCG method (BCG-HSA method) and the modified BCP (modified BCP-HSA method) calibrated with human serum albumin standard solution (HSA). Albumin concentrations were measured in 136 patient serum samples (healthy group: 52, hypoalbuminemic group: 84) by the two-point calibrated BCG method and compared with those obtained using the modified BCP-HSA method.
Results
The mean albumin concentrations obtained using the two-point calibrated BCG and modified BCP-HSA methods were 39.18 ± 3.42 g/L and 39.37 ± 3.14 g/L (healthy group) and 26.20 ± 6.23 g/L and 26.23 ± 5.67 g/L (hypoalbuminemia group), respectively. The results of the two-point calibration BCG method were in a close agreement over the entire concentration range tested compared to the modified BCP-HSA method.
Conclusions
Based on these results, this calibration method reduces the influence of the globulin fraction on the BCG method. In the hypoalbuminemic group, the calibration method was shown to provide results consistent with the BCP method, which is highly specific for albumin.
Keywords
Introduction
The dyes bromocresol green (BCG) and bromocresol purple (BCP) are widely used in dye-binding methods for routine serum albumin measurements. Both methods use sulfonphthalein dyes to act as pH indicators, but they differ in their specificity for albumin. The BCG method has problems with albumin specificity because it reacts with some globulins1,2 such as acute phase reactant proteins. 3 In response to this problem, the BCG method has been reported to avoid the effect of globulin by utilizing the difference in reaction time between albumin and globulin and shortening the measurement time, but this method remains inadequate.4,5 The BCP method does not react with globulin and is highly specific for albumin.6,7 However, the redox state of the thiol group of the albumin molecule causes differences in reactivity with the BCP method, and a modified BCP method was developed to reduce this effect. 8 This method is considered a highly accurate assay that provides results consistent with albumin levels measured by immunoassay. 9 In Japan, the modified BCP method is recommended and widely used in clinical laboratories. In contrast, the BCG method has a high prevalence in clinical laboratories internationally. It still has an important role in albumin measurement because it is used to measure albumin in animal serum and is not affected by drug-bound albumin due to penicillin G treatment. 10 In addition, routine testing is calibrated with a single concentration of protein standard serum based on human serum containing globulin fractions. This leads to inconsistent measurements that do not reflect the relationship between the albumin specificity of the two methods. 11 In this study, serum albumin concentrations were measured by the BCG (BCG-HSA) and modified BCP (modified BCP-HSA) methods calibrated using human serum albumin solution. The results showed that the BCG-HSA method provided higher values for albumin than the modified BCP-HSA method for all concentrations tested, reflecting the specificity of the BCG method for albumin. Furthermore, the difference in albumin measurements between the healthy group and hypoalbuminemic group was found to be due to differences in the amount of globulin fractionation between the two methods. Therefore, we evaluated a two-point calibration BCG method using human serum albumin standard solutions of different concentrations with the aim of reducing the effect of globulin fractionation on the BCG method in hypoalbuminemic areas of clinical significance.
Materials and Methods
Materials
Patient serum albumin measurement values less than 35.0 g/L as measured by the modified BCP method were set as the hypoalbuminemia group, and those greater than 35.0 g/L were set as the healthy group.
Reagents
Five different serum albumin measurements were performed: the BCG method, the BCG-HSA method, the two-point calibration BCG method, the modified BCP method, and the modified BCP-HSA method. The BCG method, the BCG-HSA method, and the two-point calibration BCG method used the Albumin II-HA Test Wako. For the modified BCP and modified BCP-HSA methods, L-type Wako ALB-BCP was used. For total protein measurement by the Biuret method, L-type Wako TP was used. Reagents for albumin and total protein were from FUJIFILM Wako Pure Chemical Corporation (Osaka, Japan). QuickGel SP (Helena Laboratories Corporation, Saitama, Japan) was used for protein fractionation testing by agarose gel electrophoresis.
Apparatus
All albumin and total protein measurements were performed using a Model 7180 Hitachi Automated Analyzer (Hitachi High-Tech Corporation, Tokyo, Japan). Serum protein fractionation was performed using an Epalyzer 2 junior (Helena Laboratories Corporation).
Analysis conditions
For the BCG method, BCG-HSA method, and two-point calibration BCG method, 2.5 μL of sample and 250 μL of BCG reagent were mixed and measured 590 s after mixing at 660 nm as the main wavelength and 700 nm as the secondary wavelength (one-point two-wavelength method). In the modified BCP and modified BCP-HSA methods, 3.0 μL of sample was mixed with 160 μL of the first reagent and measured at 600 nm as the main wavelength and 660 nm as the secondary wavelength 230 s after mixing, followed by mixing 80 μL of the second reagent 300 s later and measuring at 600 nm as the main wavelength and 660 nm as the secondary wavelength 590 s later (two-point two-wavelength method). The Biuret and agarose gel electrophoresis methods were conducted according to the respective instrument instruction manual.
Standard Solution
Protein standard serum
Calibration of the BCG, modified BCP, and Biuret methods was performed using a TP/ALB calibrator (FUJIFILM Wako Pure Corporation). The calibration values were those of the specified Biuret (70.9 g/L), BCG (47.4 g/L), and modified BCP (47.3 g/L) methods.
Human serum albumin standard solution 1
Human serum albumin A8763 (4.5 g, Sigma-Aldrich Japan, Tokyo, Japan) with a purity of 99% or higher was weighed on an electronic balance and dissolved in physiological saline solution to 100 mL. The albumin concentration of this solution was 45.0 g/L and was used as standard solution 1. The BCG-HSA method and the modified BCP-HSA method were calibrated using this standard solution 1, and its calibration value was set at 45.0 g/L.
Human serum albumin standard solution 2
A solution of 10 mL of the above standard solution 1 was mixed with 7.64 mL of physiological saline solution. The albumin concentration of this solution was 25.5 g/L and it was used as standard solution 2. This standard solution was used only for the two-point calibration BCG method.
Comparison of BCG, BCG-HSA, modified BCP, and modified BCP-HSA methods
The BCG, BCG-HSA, modified BCP, and modified BCP-HSA methods were used to measure albumin in sera from 60 patients (28 in the healthy group and 32 in the hypoalbuminemic group) and the results were compared.
Determination of calibration values for the two-point calibration BCG method
Calibration of the two-point calibration BCG method was performed using standard solutions 1 and 2. Therefore, the calibration values of standard solutions 1 and 2 in the two-point calibration BCG method were calculated from the differences in albumin values of the BCG-HSA method and the modified BCP-HSA method in section Comparison of BCG, BCG-HSA, modified BCP, and modified BCP-HSA methods. The calculated calibration values were 40.5 g/L for standard solution 1 and 18.6 g/L for standard solution 2.
Protein fractionation assay
Protein fractions were measured by agarose gel electrophoresis in sera from 60 patients (28 in the healthy group and 32 in the hypoalbuminemic group). The α-, β-, and γ-globulin fractions were calculated from the total protein value measured by the Biuret method.
Calibration effect of the two-point BCG method
The calibration effectiveness of the two-point calibration BCG method was compared with the modified BCP-HSA method for the healthy and hypoalbuminemic groups using sera samples from 136 patients (52 in the healthy group and 84 in the hypoalbuminemic group).
Ethical approval
Patient sera were residual sera from medical treatments at Higashimatsuyama Medical Association Hospital, and comprehensive consent was obtained. This study was approved by the Ethics Committee of Higashimatsuyama Medical Association Hospital (approval No.: 21-1). The experiments were conducted in compliance with the relevant national laws and regulations and in accordance with the tenets of the Declaration of Helsinki.
Statistical analysis
Data were tabulated in Microsoft Excel (Microsoft Corporation, Redmond, WA), and EZR 12 was used for all statistical analyses. EZR is statistical software that extends the capabilities of R and R Commander. Comparison of albumin levels by different standard solutions is expressed as mean ± SD, and correlation analysis was performed using Spearman’s rank correlation coefficient test. Comparison of albumin levels by the two-point BCG method and modified BCP-HSA method is expressed as mean ± SD, and correlation analysis was performed using Pearson’s test. Differences were considered significant at p < 0.05.
Results
Grading of protein standard serum and human serum albumin standard
Protein fractionation was performed by agarose gel electrophoresis of protein standard serum and standard solution 1. The results showed that the albumin fraction was 64.8%, α fraction was 11.2%, β fraction was 9.4%, and γ fraction was 14.8% in the protein standard serum, and that the albumin fraction was 100% in standard solution 1. Standard solutions 1 and 2 were also measured in triplicate using the Biuret method. The average values were 44.7 g/L for standard solution 1 and 25.3 g/L for standard solution 2, similar to the theoretical values for both adjusted samples (45.0 g/L for standard solution 1 and 25.5 g/L for standard solution 2).
Comparison of albumin concentration between the BCG and modified BCP methods due to differences in calibrators
Serum albumin measurements were made for serum samples from 60 patients using the BCG method calibrated with protein standard sera and human serum albumin standard solutions, and using the modified BCP method. Serum albumin concentrations (mean ± SD) for both methods calibrated with protein standard serum were 46.20 ± 3.27 g/L and 30.53 ± 4.56 g/L using the BCG method for the healthy and hypoalbuminemic groups, respectively, whereas the modified BCP method provided the values 45.07 ± 4.06 g/L and 25.60 ± 4.89 g/L, respectively. The results for the healthy group were almost identical for both methods, whereas the BCG method provided a mean value for the hypoalbuminemic group that was 5.08 g/L higher (Figure 1(a)). Serum albumin concentrations obtained using the BCG-HSA method calibrated with human serum albumin standards were 49.51 ± 3.24 g/L and 32.45 ± 5.01 g/L for the healthy and hypoalbuminemic groups, respectively, whereas the modified BCP-HSA method provided values of 45.03 ± 3.72 g/L and 25.54 ± 5.08 g/L. The BCG-HSA methods gave mean values for both the healthy and hypoalbuminemic groups that were 4.48 g/L and 6.91 g/L higher, respectively (Figure 1(b)). The modified BCP method matched albumin concentrations for both calibrators (Figure 1(c)). Table 1 shows the serum albumin concentrations obtained by the BCG and modified BCP methods for each calibrant. Comparison of albumin concentration obtained using the BCG method and the modified BCP method with different calibrators. The protein standard serum (a: y = 0.810x + 9.74, r = 0.977, p < 0.01) and human serum albumin standard (b: y = 0.874x + 10.07, r = 0.981, p < 0.01) were used as calibration standards. Protein standard serum and human serum albumin standard were used in the modified BCP method (c: y = 0.995x – 0.22, r = 0.987, p < 0.01). Correlation was evaluated using Spearman’s rank correlation coefficient test. Comparison of albumin levels by the two methods with different standard solutions using sera from healthy and hypoalbuminemia subjects.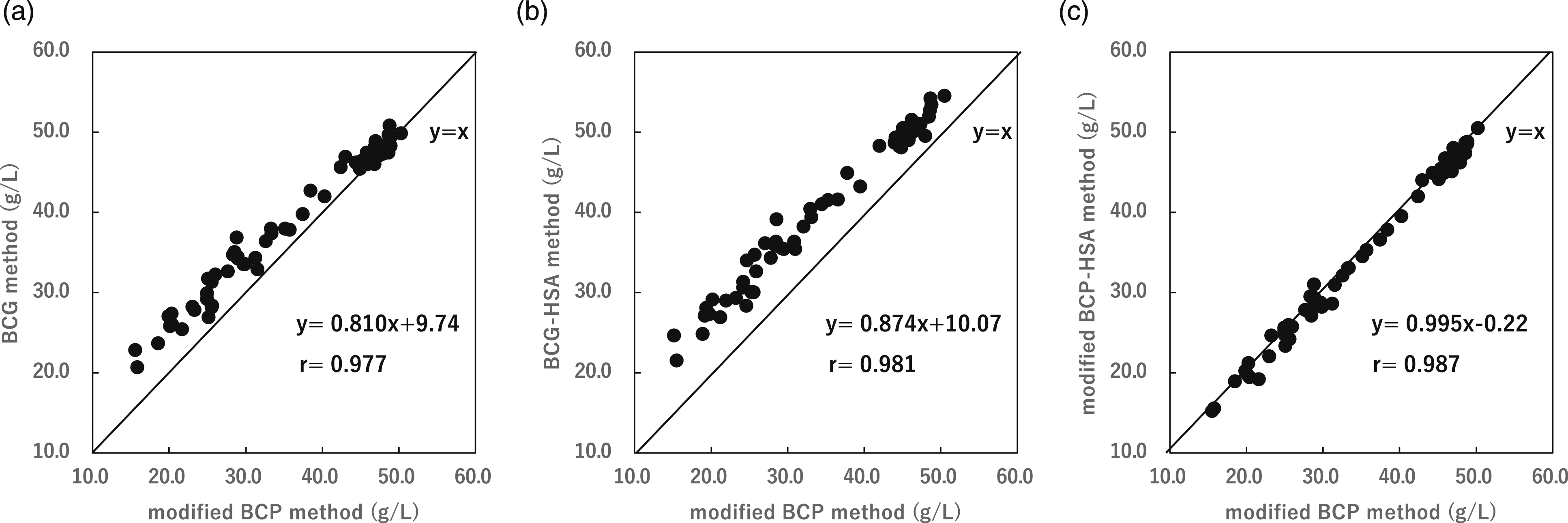

Correlation of differences in albumin concentration and globulin fractions between the BCG-HSA and modified BCP-HSA methods
Protein fractionation was performed by agarose gel electrophoresis on serum samples from 60 patients, and the correlation between the difference in albumin concentration and globulin fractionation was compared between the BCG-HSA and modified BCP-HSA methods. The results showed a strong correlation with the α fraction: r = 0.885, β fraction: r = −0.528, and γ fraction: r = 0.292 (Figure 2). Correlation coefficient for the difference between the BCG method and the modified BCP method and the α-globulin (a), the β-globulin (b), and the γ-globulin (c) fraction. (a) The correlation coefficient with the α-globulin fraction was r = 0.885, p < 0.01. (b) The correlation coefficient with the β-globulin fraction was r = −0.528, p < 0.01. (c) The correlation coefficient with the γ-globulin fraction was r = 0.259, p < 0.05. Correlation was evaluated using Pearson’s test.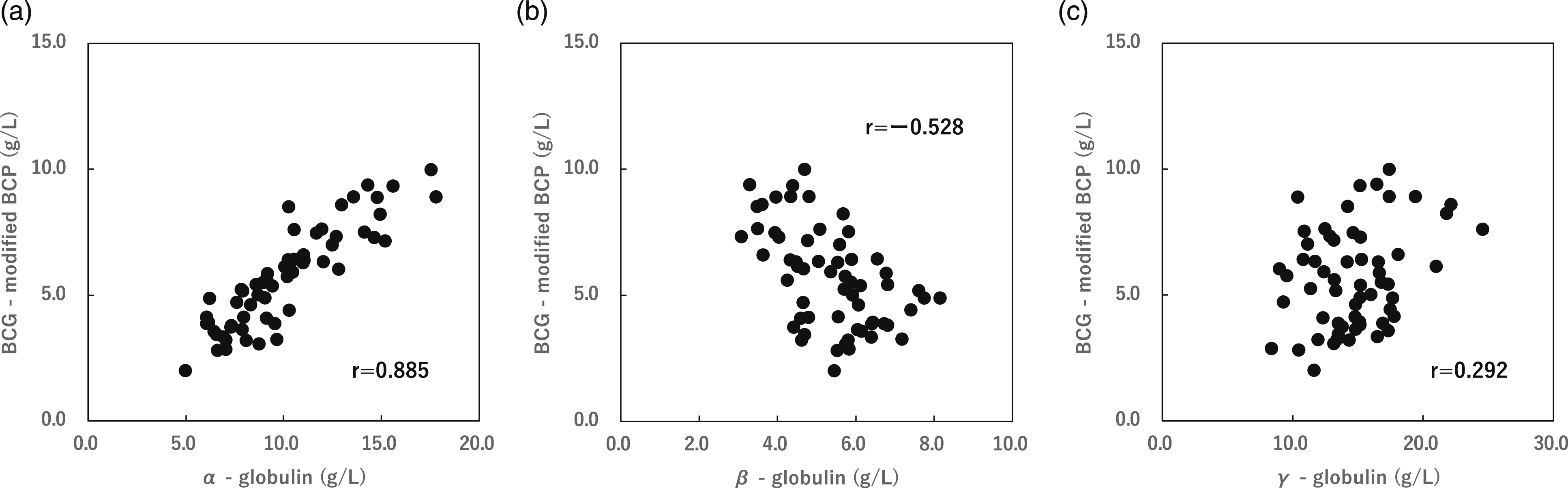
Determination of the calibration value for the two-point BCG-HSA method
Calibration materials and calibration values for the BCG method.

Comparison of the two-point calibration BCG method and the modified BCP-HSA method
The albumin concentrations obtained using the two-point calibration BCG and BCG-HSA methods were compared with the modified BCP-HSA method using 136 newly collected patient serum samples. The correlation between the BCG-HSA method and the modified BCP-HSA method was y = 0.907x + 8.58, r = 0.978, p < 0.01 and that between the two-point calibration BCG method and the modified BCP-HSA method was y = 1.006x - 0.27, r = 0.978, p < 0.01 (Figure 3(a) and (b)). Serum albumin concentrations (mean ± SD) obtained using the two-point calibration BCG method and the modified BCP-HSA method were 39.18 ± 3.42 g/L and 26.20 ± 6.23 g/L in the healthy and hypoalbuminemic groups, respectively, whereas the corresponding values obtained using the modified BCP-HSA method were 39.37 ± 3.14 g/L and 26.23 ± 5.67 g/L. Over the entire concentration range, the two-point calibration BCG method gave results in a close agreement with those of the modified BCP-HSA method (Table 3). Correlation between the modified BCP-HSA method and the BCG-HSA method (a) and the two-point BCG method (b) and a plot of the differences. (a) The correlation with the BCG-HSA method was y = 0.907x + 8.58, r = 0.978, p < 0.01. (b) The correlation with the two-point BCG method was y = 1.006x − 0.27, r = 0.978, p < 0.01. Correlation was evaluated using Pearson’s test. Comparison of albumin levels by the two-point BCG method and the modified BCP-HSA method using sera from healthy and hypoalbuminemia subjects.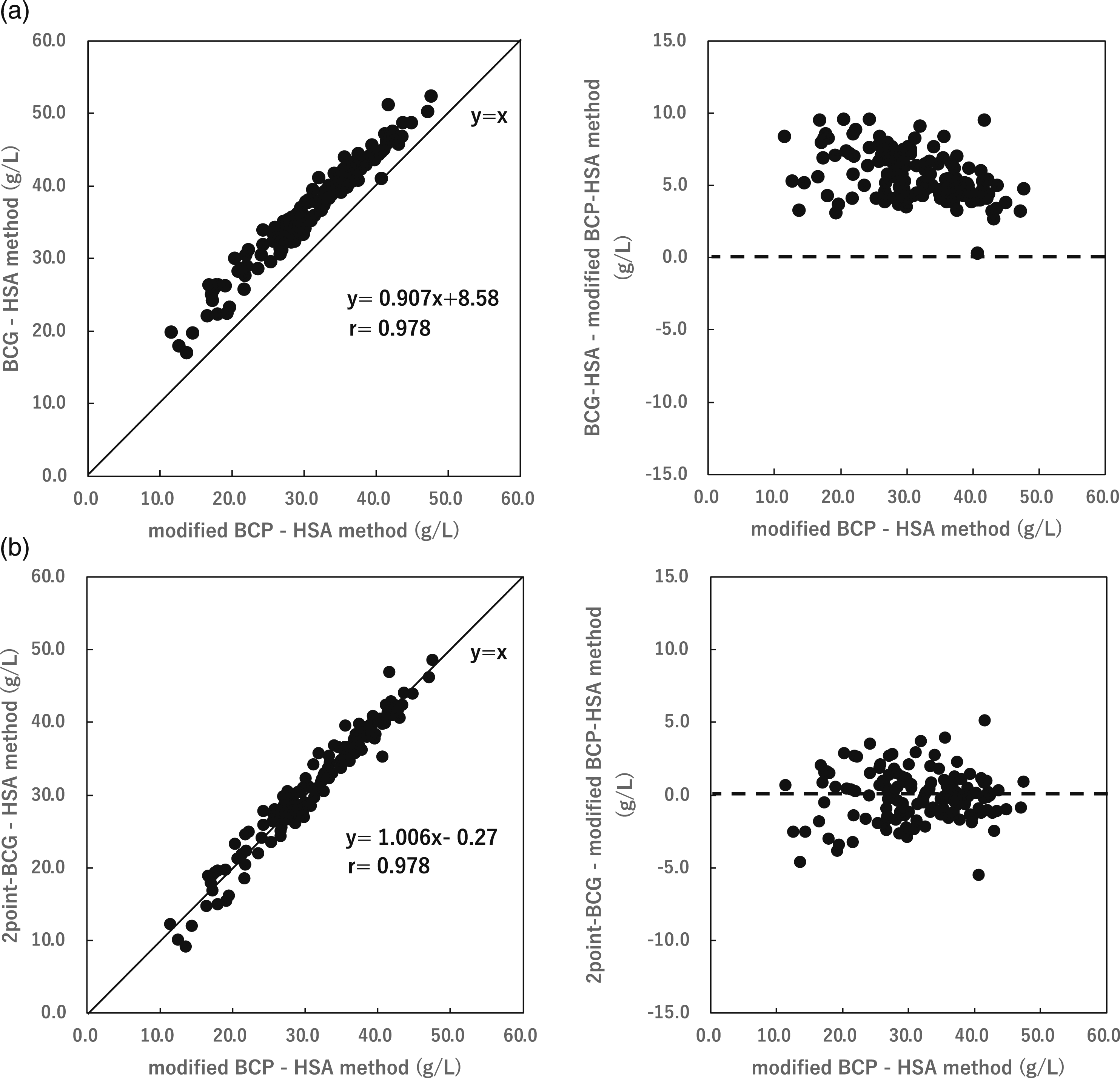

Discussion
The BCG and BCP methods, which are dye-binding methods used to measure albumin, differ in their specificity for albumin. This reportedly can affect clinical judgment, especially for hypoalbuminemic patients where the proportion of globulin fractions is increased.13-16 In routine testing, the two methods provided almost identical albumin level values in the healthy group, whereas the BCG method provided higher values for the hypoalbuminemic group, a contradictory result that does not reflect the specificity of the two methods for albumin. We previously evaluated the effects of calibration for both methods using human albumin and α- and β-globulin samples adjusted to different concentrations and reported that calibration with a protein standard serum based on human serum was responsible for the observed inconsistency in routine testing. 11 Given the above, we considered that the problems with the BCG method for albumin measurements are its low specificity for albumin and the fact that it is calibrated with a single concentration of protein standard serum. Single-point calibration is inadequate because the BCG method generates similar coloration for several globulins, such as α-globulin, regardless of whether the patient is healthy or hypoalbuminemic. To address these problems, we developed a two-point calibration BCG method using human serum albumin standards at different concentrations to calibrate the BCG method and evaluated this method using patient sera. First, patient serum was measured by the BCG-HSA and modified BCP-HSA methods calibrated with a human serum albumin standard. The mean difference between the two methods was 4.48 g/L for the healthy group and 6.91 g/L for the hypoalbuminemic group, with the BCG method providing higher values at all concentrations (Figure 1(b)). Protein fractionation of patient serum was performed by agarose gel electrophoresis to determine the relationship between the differences in the measurement results obtained using the two methods and the globulin fraction. The results showed a strong correlation coefficient of r = 0.885 with the α-fraction, and patient sera with low albumin concentrations had increased levels of the α-globulin fraction (Figure 2). When calibrated with a protein standard serum, the α- and β-globulins in the calibrant are also calibrated in a reactive state using the BCG method. In the healthy group, this calibration effect corrects for α- and β-globulin effects of the BCG method. However, calibration with a single concentration of protein standard serum assumes that the ratio of albumin to α- and β-globulins is identical to that of the protein standard serum at all albumin concentrations. Therefore, the BCG method can be considered to measure higher than the true values for the hypoalbuminemic group. In addition, since the amount of α-globulin is higher in the hypoalbuminemic group than in the healthy group, the albumin value measured by the BCG method is expected to be even higher than that measured by the BCP method. The human serum albumin standard used in this study does not contain a globulin fraction. Thus, the BCG method calibrated with the human serum albumin standard did not correct for the globulin fraction at all albumin concentrations, reflecting its specificity for albumin. The difference in albumin concentration between the BCG-HSA and modified BCP-HSA methods therefore reflects the globulin fractions in the patient’s serum, with a confirmed strong correlation with the amount of α-fraction. Given the difference in the amount of globulin fraction in sera from the healthy and hypoalbuminemic groups, two concentrations of standard solutions and their calibration values were established based on the difference in albumin measurements between the BCG-HSA method and the modified BCP-HSA method in the two groups. Furthermore, the modified BCP-HSA method and the two-point calibration BCG method, which subtract the amount of colored α-globulin fraction, were compared. The reference method used was the modified BCP method, which provides results consistent with albumin levels measured by immunoassay9,17. The results were 39.18 ± 3.42 g/L and 26.20 ± 6.23 g/L for the two-point calibrated BCG method and 39.37 ± 3.14 g/L and 26.23 ± 5.67 g/L for the modified BCP-HSA method, respectively, for the healthy and hypoalbuminemic groups, and the results were similar across the entire concentration range tested (Table 3). These results indicate that the two-point calibration BCG method reduces the influence of α-globulin levels in the low albumin region of clinical significance, and avoids the influence of the calibration method using protein standard serum. The albumin assay should employ the modified BCP method, which is highly specific. If the BCG method is employed, it would be desirable to change to this calibration method that reduces the difference in albumin concentration from the modified BCP method.
A limitation of this study is that it did not consider globulin levels in diseases associated with hypoalbuminemia. Albumin concentrations may be overestimated and underestimated in patients with diseases involving increased globulin fractions other than the α-globulin fraction, and in hypoalbuminemia where globulin fractions are not increased. Therefore, it is necessary to proceed with the evaluation of individual cases and changes in globulin fractions as an issue for this method.
Conclusion
In this study, considering the differences in globulin fractions in the healthy and hypoalbuminemic groups, the two-point calibrated BCG method was developed using human serum albumin standard solutions at different concentrations, and compared with the modified BCP-HSA method. The results of the two-point calibration BCG method were in a close agreement over the entire concentration range tested compared to the modified BCP-HSA method. Based on these results, this calibration method reduces the influence of the globulin fraction on the BCG method even in hypoalbuminemic areas, which is of high clinical significance.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The study was approved by Higashimatsuyama Medical Association Hospital (approval no. 21-1).
Guarantor
MM.
Contributorship
TI and MM designed the study. MM and YH supervised the study. TI and KN carried out the research. TI and MM analyzed the data. TI and MM wrote the manuscript. MM and YH reviewed drafts of the manuscript. All authors have accepted responsibility for the entire content of this manuscript and approved its submission.
