Abstract
Background
Albumin–bilirubin (ALBI) grade is an index of liver function based on total bilirubin (T-BIL) and albumin levels, and its usefulness has been widely reported. This study aimed to investigate the effect of different methods of measuring T-BIL and albumin levels on the ALBI grade in patients with liver disease.
Methods
In total, 170 patients from our hospital were included in this study. Correlations between T-BIL levels measured using the vanadate oxidation and enzymatic methods were analysed. Similarly, a correlation analysis of albumin levels between the bromocresol green (BCG) and modified bromocresol purple (BCP) methods was performed. Additionally, the ALBI grade was calculated for patients with liver disease, and the differences between each method of albumin measurement were compared.
Results
No differences were observed in the measured T-BIL values between the two methods. Contrastingly, the albumin levels of 100 random samples and 70 liver disease patients obtained using the modified BCP method were significantly lower than those measured using the BCG method. The rate of change in the modified ALBI grade between the BCG and BCP methods was 25.7%.
Conclusions
Caution should be taken when comparing ALBI grades with those measured by other facilities because the method of albumin measurement can affect the ALBI grade. Standardization of albumin measurement is needed worldwide.
Keywords
Introduction
Most patients with hepatocellular carcinoma (HCC) have liver disease. Therefore, it is important to assess liver function when selecting treatment options. Although the Child-Pugh classification (C-P classification)1,2 has been widely used to evaluate liver function,3,4 it has many limitations, including the subjectivity of the clinical evaluation of hepatic encephalopathy and ascites. 5
Recent advances in various treatments have led to marked improvements in liver functional reserve in patients with HCC, and most of these patients have C-P class A. 6 Therefore, more detailed criteria are needed for patients with good liver reserves. In 2014, the albumin–bilirubin (ALBI) grade, 7 a simple and objective liver functional reserve assessment index based only on albumin and total bilirubin (T-BIL) levels, was proposed and its usefulness has been widely reported.5,8 Recent review article has shown that the granularity of the ALBI score permitted to detect the early deterioration of liver function in patients with C-P class A. 9
There are two widely used albumin measurement methods: the bromocresol green (BCG) 10 and modified bromocresol purple (BCP) methods. 11 Laboratories in many countries use the BCG method; however, most laboratories in Japan are shifting to the modified BCP method because it does not cross-react with globulin, which has high specificity for albumin. 11 As reported from Japan, the modified BCP method reflected the true value than the BCG method. 12 Particularly, at albumin concentrations below 35 g/L, there is a marked difference between the two methods.12,13 Therefore, it has been hypothesized that the method used may affect the ALBI grade determination;6,14 however, this remains unclear.
This study aimed to investigate the effect of different methods of measuring T-BIL and albumin levels on the ALBI grade in patients with liver disease.
Methods
Patients
This study included 170 patients treated at the Sapporo Medical University Hospital between December 2019 and June 2020. Of the 170 patients, 100 were randomly selected for correlation analysis, and 70 had liver diseases for which the ALBI grade was requested. The diagnosis of HCC was based on typical radiological imaging, according to the clinical practice guideline 4 or pathological findings. The exclusion criteria were as follows: 1) patients with a C-reactive protein (CRP) level of 10,000 µg/L or higher were excluded because high inflammation has been reported to interfere with the BCG method 15 ; and 2) patients receiving high-dose penicillin G treatment were excluded because it interferes with the modified BCP method. 16
Laboratory tests
Total bilirubin was measured using the vanadate oxidation 17 (Total Bilirubin E-HA Test Wako; FUJIFILM Wako Pure Chemical Corporation Ltd., Osaka, Japan) and enzymatic18,19 (Nescort VL T-BIL; Alfresa Pharma Corporation Ltd., Osaka, Japan) methods. Similarly, albumin was measured using the BCG 10 (Albumin-II HA Test Wako; FUJIFILM Wako Pure Chemical Corporation Ltd.) and modified BCP 11 (L-Type Wako BCP-ALB; FUJIFILM Wako Pure Chemical Corporation Ltd.) methods. Total protein (TP), immunoglobulin G (IgG), immunoglobulin A (IgA), immunoglobulin M (IgM), aspartate aminotransferase (AST), alanine aminotransferase (ALT), CRP, and platelet count (PLT) were measured in our hospital laboratory using samples collected at the same time as those used to measure the T-BIL and albumin levels. The albumin and TP values were used to calculate the albumin–globulin ratio (AGR; albumin (modified BCP method)/(TP − albumin [modified BCP method]). T-BIL, albumin, TP, IgG, IgA, IgM, AST, ALT, and CRP were measured by using LABOSPECT 008 (Hitachi High-Tech Corporation Ltd. Tokyo, Japan), and PLT was measured by using XN-3000 (Sysmex Corporation Ltd. Hyogo, Japan). All the parameters for the sample measurement were specified by the manufacturer. In addition, calibration was performed using a standard solution specified by the manufacturer before sample measurement.
Correlation of T-BIL values measured by the vanadate oxidation and enzymatic methods
Correlation analysis between the vanadate oxidation and enzymatic methods was performed using the serum or lithium–heparin plasma of patients. Difference in measured values between the two methods was evaluated using a residual plot.
Correlation of albumin values measured by the BCG and modified BCP methods
Correlation analysis between the BCG and modified BCP methods was performed on the same 100 samples used to measure the T-BIL levels and 70 samples from the patients with liver disease. A residual plot was used to visualize the differences in the measured values between the two methods.
ALBI grade
Albumin from the BCG or modified BCP method and T-BIL from the enzymatic method were used to calculate the ALBI score based on the following equation: (log10 T-BIL [μmol/L] × 0.66) + (albumin [g/L] × −0.0852). Since T-BIL was calculated in mg/dL, it was multiplied by 17.1 and converted to μmol/L before substitution into the formula. These scores were classified into three ALBI grades (1, 2, and 3). The ALBI grade 1, 2, and 3 scores were ≤ −2.60, > −2.60 to −1.39, and > −1.39, respectively. 7 Albumin–bilirubin grade 2 was further classified into two subgroups using an ALBI score of −2.270 as the cutoff value (the modified ALBI grade). 20 Correlation of ALBI score between the BCG and modified BCP methods was performed on the 70 samples from the patients with liver disease. A residual plot was used to visualize the differences in the measured values between the two methods.
Statistical analysis
Where appropriate, continuous variables were expressed as medians and interquartile ranges and analysed using the Wilcoxon signed-rank test. Fisher’s exact test was performed to investigate whether differences in aetiology affected the ALBI grade. Statistical significance was set at p < 0.05, and p-values were two-sided. In addition, Spearman’s rank correlation test was used to assess the association between various measurements and difference in ALBI score calculated by the BCG and modified BCP methods. For statistical analyses, we used EZR, 21 a graphical user interface for R (The R Foundation for Statistical Computing, Vienna, Austria) and the R software version 4.0.3 (R Foundation for Statistical Computing).
Results
Correlation and residual plot for T-BIL, albumin, and ALBI score
The T-BIL levels of 100 random samples measured using the vanadate oxidation method correlated well with the levels measured using the enzymatic method (Figure 1(a)). Contrastingly, the albumin levels of 100 random samples, as measured using the modified BCP method, were significantly lower than those measured using the BCG method (p < 0.001, Figure 1(b)). Greater differences were observed between the two methods for the lower albumin levels. Correlation and residual plots for total bilirubin (T-BIL) and albumin. (a) The result of the vanadate oxidation method (x) and the enzymatic method (y) in 100 randomly selected cases; r = 0.999, y = 0.96 x + 0.04. (b) Results of the bromocresol green (BCG) method (x) and the modified bromocresol purple (BCP) method (y) in 100 randomly selected cases; r = 0.992, y = 1.17 x − 0.97.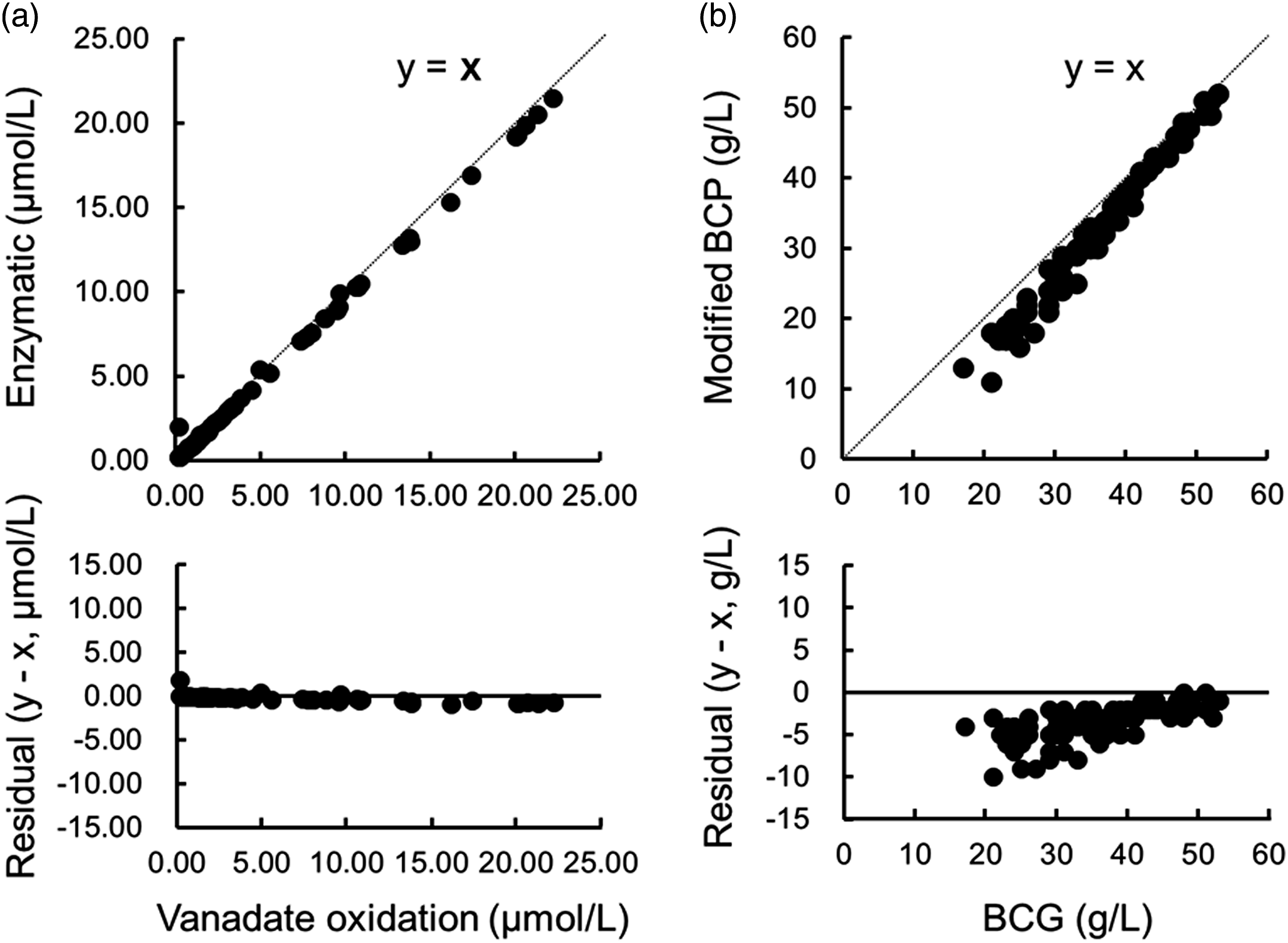
Clinical characteristics in patients with liver disease (n = 70).
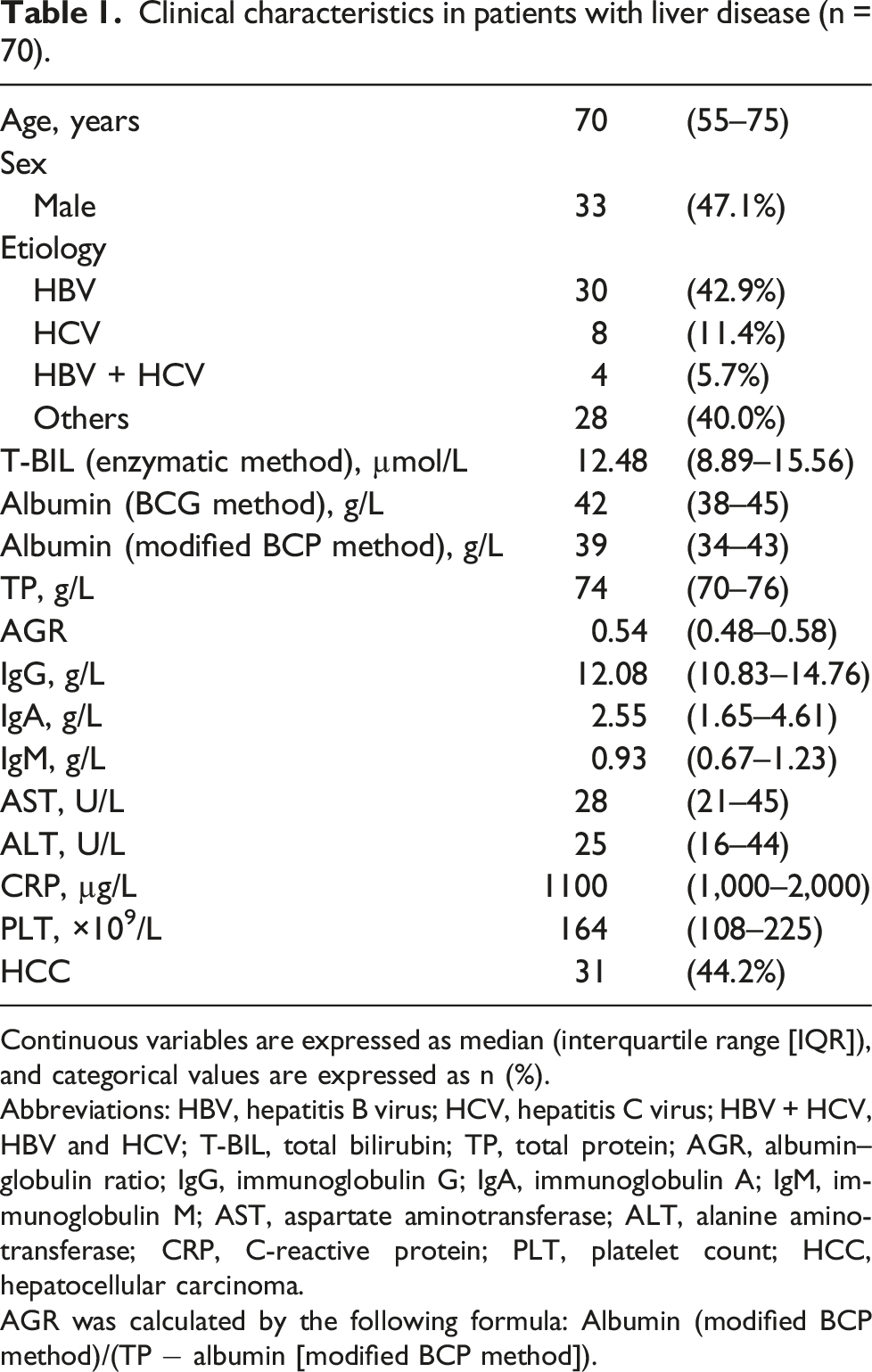
Continuous variables are expressed as median (interquartile range [IQR]), and categorical values are expressed as n (%).
Abbreviations: HBV, hepatitis B virus; HCV, hepatitis C virus; HBV + HCV, HBV and HCV; T-BIL, total bilirubin; TP, total protein; AGR, albumin–globulin ratio; IgG, immunoglobulin G; IgA, immunoglobulin A; IgM, immunoglobulin M; AST, aspartate aminotransferase; ALT, alanine aminotransferase; CRP, C-reactive protein; PLT, platelet count; HCC, hepatocellular carcinoma.
AGR was calculated by the following formula: Albumin (modified BCP method)/(TP − albumin [modified BCP method]).
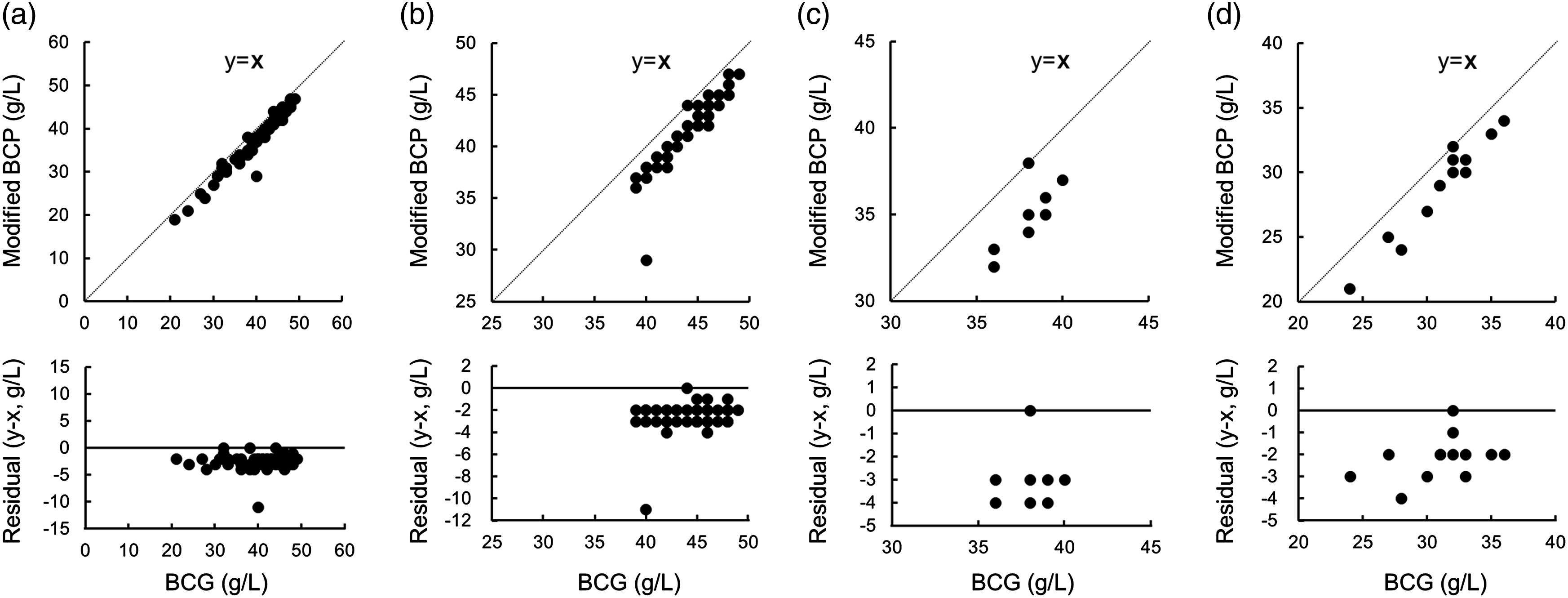
Correlation and residual plots of albumin measured by the BCG method (x) and the modified BCP method (y) in patients with liver disease. (a) Seventy cases with liver disease; r = 0.992, y = 1.03 x − 0.36. (b) Forty-eight cases of modified albumin–bilirubin (ALBI) grade 1; r = 0.910, y = 1.23 x − 1.27. (c) Eight cases of modified ALBI grade 2a; r = 0.758, y = 1.41 x − 1.87. (d) Thirteen cases of modified ALBI grade 2b; r = 0.970, y = 1.15 x − 0.6.
The correlation between the ALBI scores calculated by the modified BCP method and the BCG method also suggests that the former is higher, and the ALBI score of the modified BCP method for all cases was not lower than that of the BCG method (Figure 3). Correlation and residual plots of albumin–bilirubin (ALBI) score by the BCG method (x) and the modified BCP method (y) in patients with liver disease; r = 0.980, y = 0.99 x + 0.21.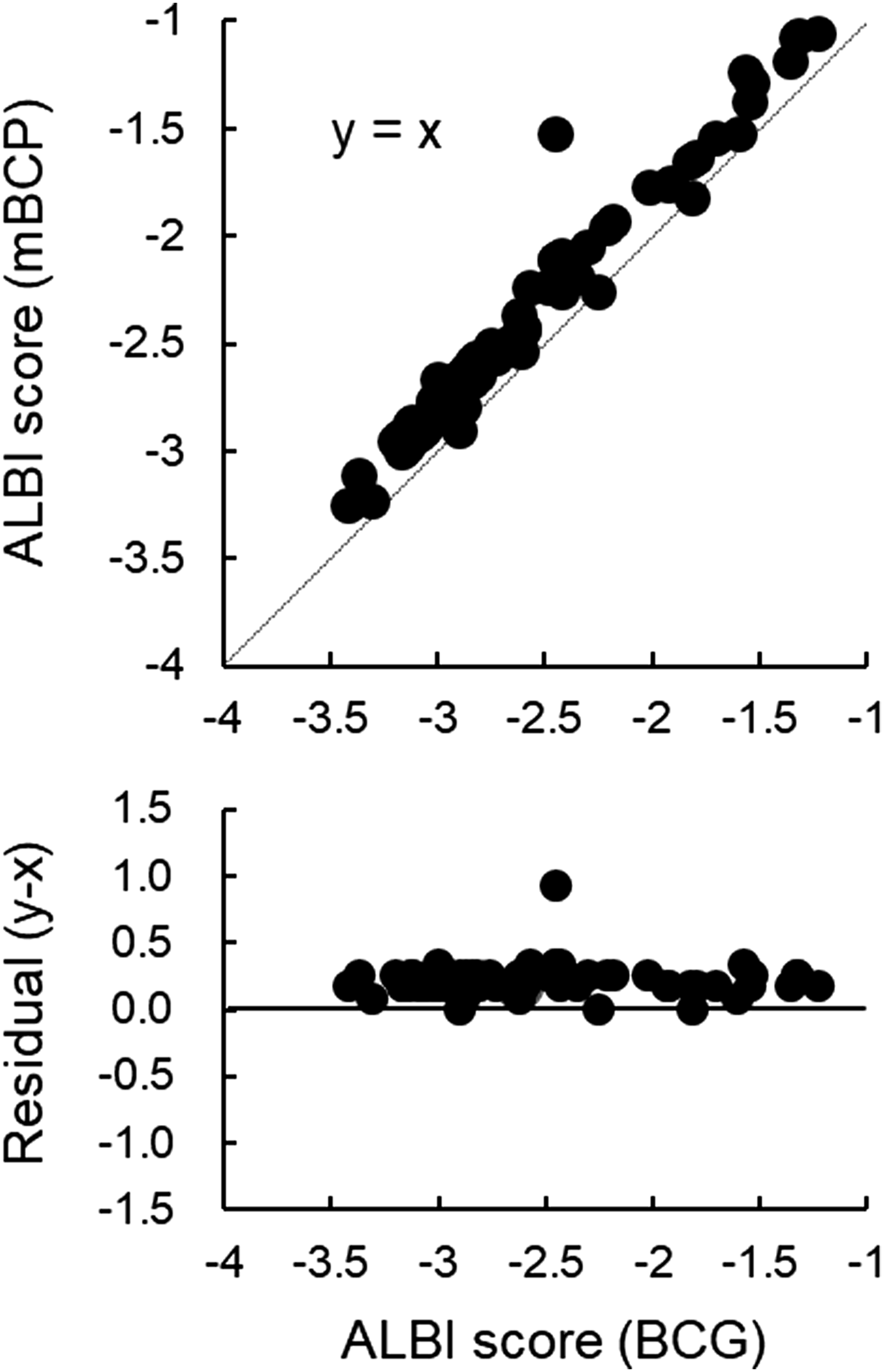
Rate of change in modified ALBI grade between different methods of albumin measurement
Rate of change of modified albumin–bilirubin (ALBI) grade between the BCG method and the modified BCP method.

aALBI scores were classified into three ALBI grades (1, 2, and 3). ALBI grades 1, 2, and 3 = ≤ −2.60, > −2.60 to −1.39, and > −1.39, respectively. ALBI grade 2 was further classified into two subgroups using an ALBI score of −2.270 as the cutoff value (modified ALBI grade).
Clinical characteristics of patients for whom the modified albumin–bilirubin (ALBI) grade differed between albumin measurement methods.
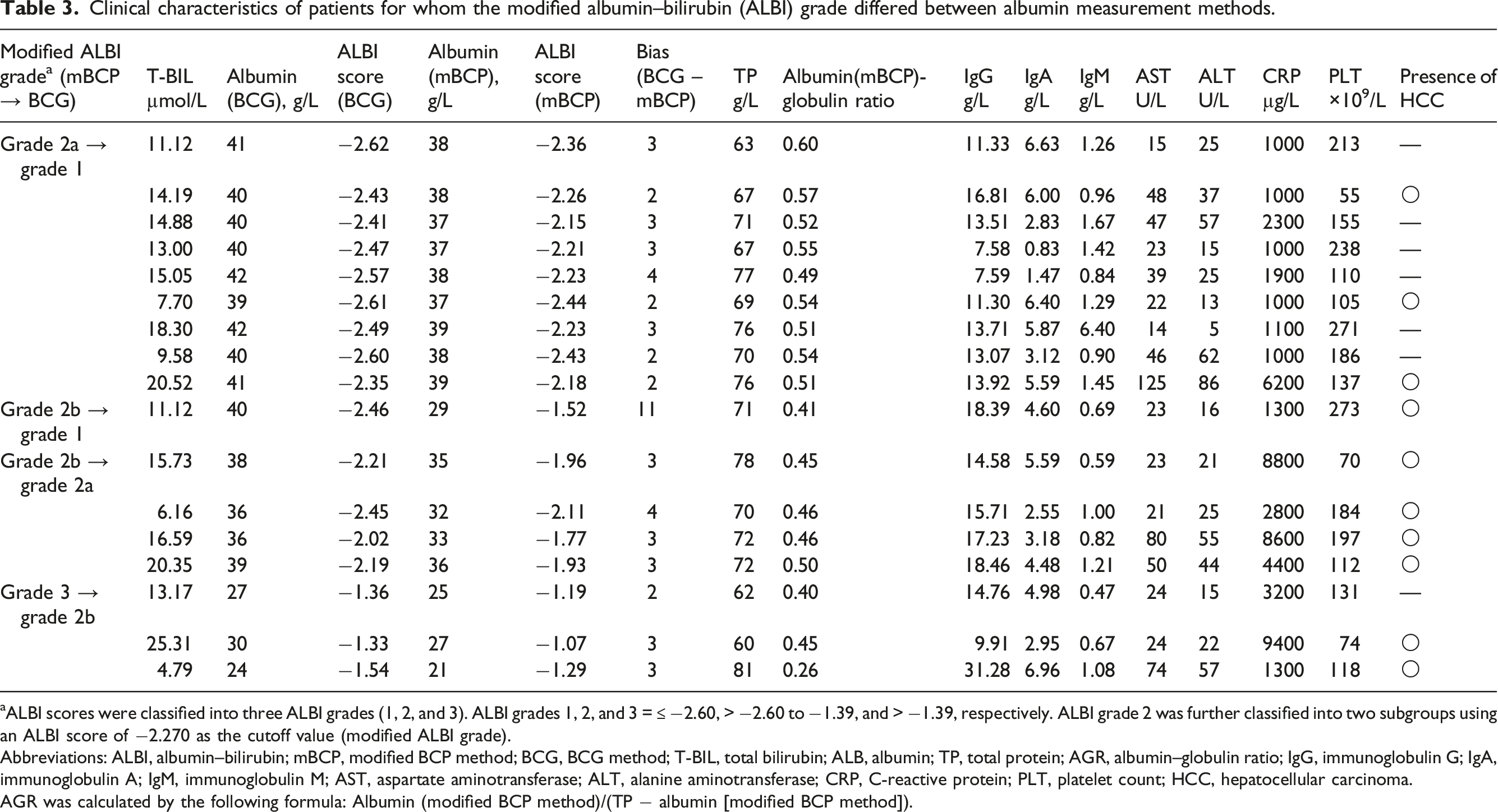
aALBI scores were classified into three ALBI grades (1, 2, and 3). ALBI grades 1, 2, and 3 = ≤ −2.60, > −2.60 to −1.39, and > −1.39, respectively. ALBI grade 2 was further classified into two subgroups using an ALBI score of −2.270 as the cutoff value (modified ALBI grade).
Abbreviations: ALBI, albumin–bilirubin; mBCP, modified BCP method; BCG, BCG method; T-BIL, total bilirubin; ALB, albumin; TP, total protein; AGR, albumin–globulin ratio; IgG, immunoglobulin G; IgA, immunoglobulin A; IgM, immunoglobulin M; AST, aspartate aminotransferase; ALT, alanine aminotransferase; CRP, C-reactive protein; PLT, platelet count; HCC, hepatocellular carcinoma.
AGR was calculated by the following formula: Albumin (modified BCP method)/(TP − albumin [modified BCP method]).
Differences in ALBI scores between albumin assays and their relationship to laboratory values
The effect of globulin or AGR on the difference in ALBI scores calculated by the BCG and modified BCP methods was evaluated. The difference in ALBI scores between the BCG and modified BCP methods did not correlate with IgG, IgA, IgM, their combined globulin concentration, or AGR (IgG: r = −0.049, IgA: r = −0.057, IgM: r = 0.063, globulin: r = −0.064, and AGR: r = −0.269). However, consistent differences in ALBI scores were observed independent of their values (Figure 4). Relationship between various measurements (x) and difference in albumin-bilirubin (ALBI) score calculated by the BCG and modified BCP methods (y) in patients with liver disease. (a) IgG; r = −0.049. (b) IgA; r = −0.057. (c) IgM; r = 0.063. (d) Globulin; r = −0.064. Globulin was defined as the sum of IgG, IgA, and IgM. (e) Albumin–globulin ratio (AGR); r = −0.269. AGR was calculated by the following formula: Albumin (modified BCP method)/(TP − albumin [modified BCP method]).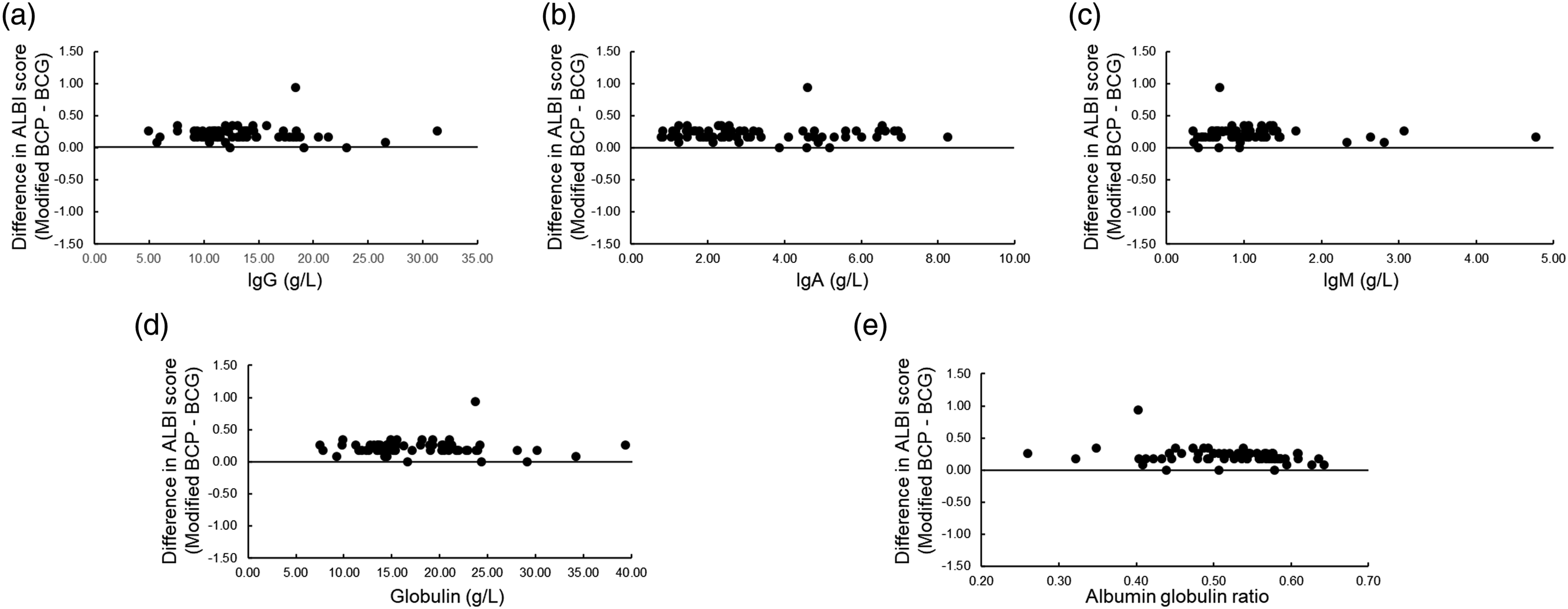
Discussion
During clinical examinations, it is important to note that differences in measurement methods can alter measured values and affect clinical assessment. In this study, we first examined the effect of the methods used in measuring the T-BIL, which is one of the ALBI grade components; however, no change was observed in the measured values (Figure 1(a)). Currently, two methods of measuring T-BIL are mainly used in Japanese laboratories: the vanadate oxidation and enzymatic methods. However, few studies have compared these two methods using a large number of samples. A recent study using artificially prepared bilirubin materials from the Japan Society of Clinical Chemistry reported no difference between the two methods, 22 which is consistent with the results of the present study. These results suggest that differences in measurement methods may not affect ALBI grade, at least in cases with low T-BIL concentrations as in this study.
It has long been known that the results of the modified BCP method have lower albumin levels than the BCG method because of its high specificity for albumin. 11 The difference is particularly large when albumin levels are low, and the effect is significant when assessing diseases in which low albumin levels are present.13,15 In terms of liver function, a report on C-P classification 12 showed that of 14 patients with an albumin score of 2 (28–35 g/L) using the BCG method, two patients were re-scored as 3 (<28 g/L) using the modified BCP method, and of 66 patients with an albumin score of 1 (>35 g/L) using the BCG method, nine patients were re-scored as 2 using the modified BCP method. In the present study, an alteration in the ALBI grade was observed in 25.7% of the patients (Table 2), suggesting that the albumin measurement method influenced the ALBI grade.
The ALBI grade is reportedly useful not only for HCC but also for other diseases, including intrahepatic cholangiocarcinoma, 23 lung cancer, 24 and acute heart failure. 25 In a patient with HCC, its utility has been reported regardless of the treatment method, region where the treatment was performed, or era, and it has become a universal prognostic formula.5,8 Indeed, jaundice is less likely to be associated with HCC because of advances in antiviral therapy and early detection of HCC. 6 The ALBI score is calculated using albumin and T-BIL values; therefore, the ALBI grade is greatly influenced by albumin levels. As noted above, although the method of measurement affects the resulting albumin levels, which are important in determining the ALBI grade, few clinical trials using the ALBI grade have described the method of albumin measurement.
The influence of albumin measurement methods on ALBI grade has been discussed previously. Kumada et al. 6 reported that the most recent patients with HCC have good liver function (modified ALBI grade 1 or 2a) and that the cutoff value of albumin to distinguish between modified ALBI grades 2a and 2b is 35 g/L. Furthermore, since the greatest discrepancy between the BCG and modified BCP methods occurred when albumin was less than 35 g/L, they argued that the method of albumin measurement did not affect the determination of grades 1 and 2a.
However, in this study, of the 12 patients that were determined as grade 2a using the modified BCP method, nine (75%) changed to grade 1 using the BCG method, and of the 16 patients determined as grade 2b using the modified BCP method, four (25%) changed to grade 2a and one (6%) changed to grade 1 using the BCG method (Tables 2 and 3). These results showed that the patients with ALBI 1 and 2a were also affected by the methods of albumin.
The use of anti-angiogenic antibodies in the treatment of unresectable HCC has become increasingly important. 26 Ramucirmab is an anti-angiogenic antibodies that was found to be more effective in the Japanese population than the global population.27,28 Kudo suggested that this could be due to differences in albumin measurement methods between Japan and other countries, which suggested that caution was necessary in global clinical trials because the C-P score determined by the modified BCP method in Japan represents better liver function than the C-P score measured by the BCG method in most other countries. 14
The fact that the BCG method cross-reacts with globulin means that differences of albumin value are more likely to occur with the higher globulin. In addition, γ-globulin is known to be elevated in liver disease. 29 Therefore, IgG, IgA, and IgM were measured to examine the effect of difference in ALBI scores between albumin assays. Our results showed that the ALBI score by the modified BCP method was consistently higher regardless of the values of IgG, IgA, IgM, globulin, and AGR (Figure 4). The small effect of γ-globulin on the difference between albumin assays has been reported in basic 30 and clinical 31 studies and is consistent with the results of this study. The lack of association between globulin or AGR and differences in ALBI scores may be due to the fact that only cases with low CRP were included in this study, and therefore there were no differences in α1- and α2-globulins, which are strongly associated with divergence between albumin measures. 13
The utility of the ALBI grade has been widely demonstrated, and its value may not be diminished due to differences in albumin measurement methods; however, a problem arises when comparisons are made between facilities that use different albumin measurement methods. It is expected that the modified BCP method will become widespread in the future.
In conclusion, caution should be exercised when evaluating the ALBI grade in international clinical trials because the method of albumin measurement affects the ALBI grade. Standardization of albumin measurement is needed worldwide.
Footnotes
Acknowledgements
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Satoshi Takahashi received speaker honoraria from MSD K.K. and Fujirebio Inc., and research grants from Shino-Test Corporation, Roche Diagnostic K. K., Fujirebio Inc., and Abbott Japan Co., Ltd. The other authors declare that they have no conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by the institutional review board of Sapporo Medical University Hospital (ID:322-116) and was conducted in accordance with the principles of the Declaration of Helsinki.
Guarantor
S Takahashi.
Contributorship
All the authors contributed substantially to the conception and design of the study. MT acquired the data. MT and S Tanaka analysed and interpreted the data. S Takahashi was the chief investigator who analysed the data. ES and RK provided logistical support and discussed the data. S Takahashi directed this project. All the authors contributed to the writing of the final manuscript and approved the submitted version.
