Abstract
Background
Quantitative faecal immunochemical tests (FIT) for haemoglobin are being used for colorectal cancer (CRC) screening for asymptomatic populations and are being indicated as a suitable test to rule out CRC in symptomatic populations. Faecal samples are typically collected by patients using a probe attached to the cap of a device which is inserted into a collection device into the preservative buffer, passing through a collar to remove excess sample: this process has potential for pre-analytical error. This study investigates whether faecal haemoglobin concentration (f-Hb) results are affected by the mass and method of sample collection.
Methods
Faecal samples with detectable f-Hb were loaded into collection devices from four manufacturers using increasing masses of sample. The f-Hb in the device buffer was measured using the relevant analyser. The results from the minimum recommended load were compared with results of ‘sample overloading’.
Results
The variation in the faecal mass added to the probe (overall CVs: EXTEL HEMO AUTO-MC Collection Picker 300%, OC-Auto Sampling Bottle 3 237%, SENTiFIT pierceTube 264%, Specimen Collection Container A 250%), was more than the variation in f-Hb (respective overall CVs: 62%, 35%, 47%, 39%). The mass of faeces added to the probes increased significantly (
Conclusion
The mass of sample loaded onto the probe did not impact the f-Hb significantly using all four tested devices.
Introduction
The faecal immunochemical test (FIT) for f-Hb is used in many bowel cancer screening programmes worldwide 1 and is being indicated as a suitable test for ruling out CRC in patients with low-risk symptoms. 2
Qualitative and quantitative systems are available for f-Hb, and this study focuses on the latter. For quantitative FITs, faecal samples are typically collected at home by the participant/patient into a FIT sampling device. A small mass of faeces (2 mg or 10 mg depending on the manufacturer) is loaded onto a grooved or dimpled probe and then inserted into a device containing a preservative buffer. FIT devices have an internal ‘collar’ that is claimed to remove excess faeces leaving a standard mass in the buffer. Manufacturers provide simple instructions to guide the sample collection; however, there is likely to be variability in the mass of sample provided. An evaluation of four systems in 20123 found that all the devices delivered more sample into the buffer than the manufacturers quoted. A recently published paper 4 has demonstrated that the sample collection devices from different manufacturers collect a mass of sample that is not consistent with the quoted sample mass.
Once loaded, the devices are sent to a laboratory for analysis. While in transit, the buffer washes the faeces from the probe and if f-Hb is present it is eluted in the buffer. The buffer acts as a preservative to slow deterioration of the globin moiety.
Laboratory analysers specific to the sampling devices are used to measure the f-Hb. F-Hb results expressed as ng Hb/mL buffer are converted to μg Hb/g faeces using the manufacturers’ defined mass of faeces and buffer volume as recommended by Fraser et al. 5
In this study, we investigated the effect of varying sample quantities loaded on to the probe on f-Hb.
Method
Portions of faecal samples, originally for calprotectin analysis, were used for this study. Each sample was collected by the patient into a faecal collection pot, then sent to the laboratory and refrigerated before a portion was taken, anonymised and mixed to distribute any f-Hb present. The initial f-Hb of each sample was measured using one of four FIT sampling devices/analysers: EXTEL HEMO AUTO-MC Collection Picker/HM-JACKarc (Kyowa Medex Co. Ltd), OC-Auto Sampling Bottle 3/OC-SENSOR DIANA (Eiken Chemical Co. Ltd), SENTiFIT pierceTube/SENTiFIT 270 (Sentinel Diagnostics), or Specimen Collection Container A/NS-Prime (Alfresa Pharma Corp). Nine faecal samples with detectable f-Hb were selected to include a range of f-Hb, within and above the analytical range of the four FIT systems. The consistency for all the samples was similar to type 4 on the Bristol Stool Chart. 6
The four different FIT devices were each loaded with the samples using the following techniques: (a) sample loaded according to the manufacturers’ instructions (minimum sample on the grooved/dimpled part of the probe only, with none on the tip of the probe), this method was used as the comparator; (b) minimum sample on the grooved/dimpled part of the probe only, with faeces also on the tip of the probe; (c) slight excess of faeces around the grooved/dimpled part of the probe, and higher up the stick to where it protrudes though the collar into the buffer, with none on the tip.
The devices were weighed both before and after loading the faeces using a Sartorius BP210S balance (Goettingen, Germany), with weighing reproducibility (standard deviation) ⩽ ± 0.0001 g. The devices were incubated overnight in the laboratory at room temperature to ensure that the devices were exposed to the same conditions, although the samples would normally be returned by post and exposed to a variety of temperatures and transit times. The following day, f-Hb was measured using the corresponding analyser.
The mean, SD and CV of the faecal masses and f-Hb were calculated for each sample and loading technique for each of the four FIT devices. The overall CVs of the faecal masses and f-Hbs were calculated (√(sum of CV2/number of CV)) for each FIT device type. The Kruskal Wallis test was used to test the significance of the difference between the sample weights and also between the f-Hb of the samples loaded with techniques (a) to (c).
Results
Table 1 shows the mass of faeces added to the nine samples for each device and the f-Hb for the loading techniques (a) to (c), and the results of the statistical analyses.
Faecal sample masses added to FIT sampling devices using three loading techniques, and corresponding f-Hb.
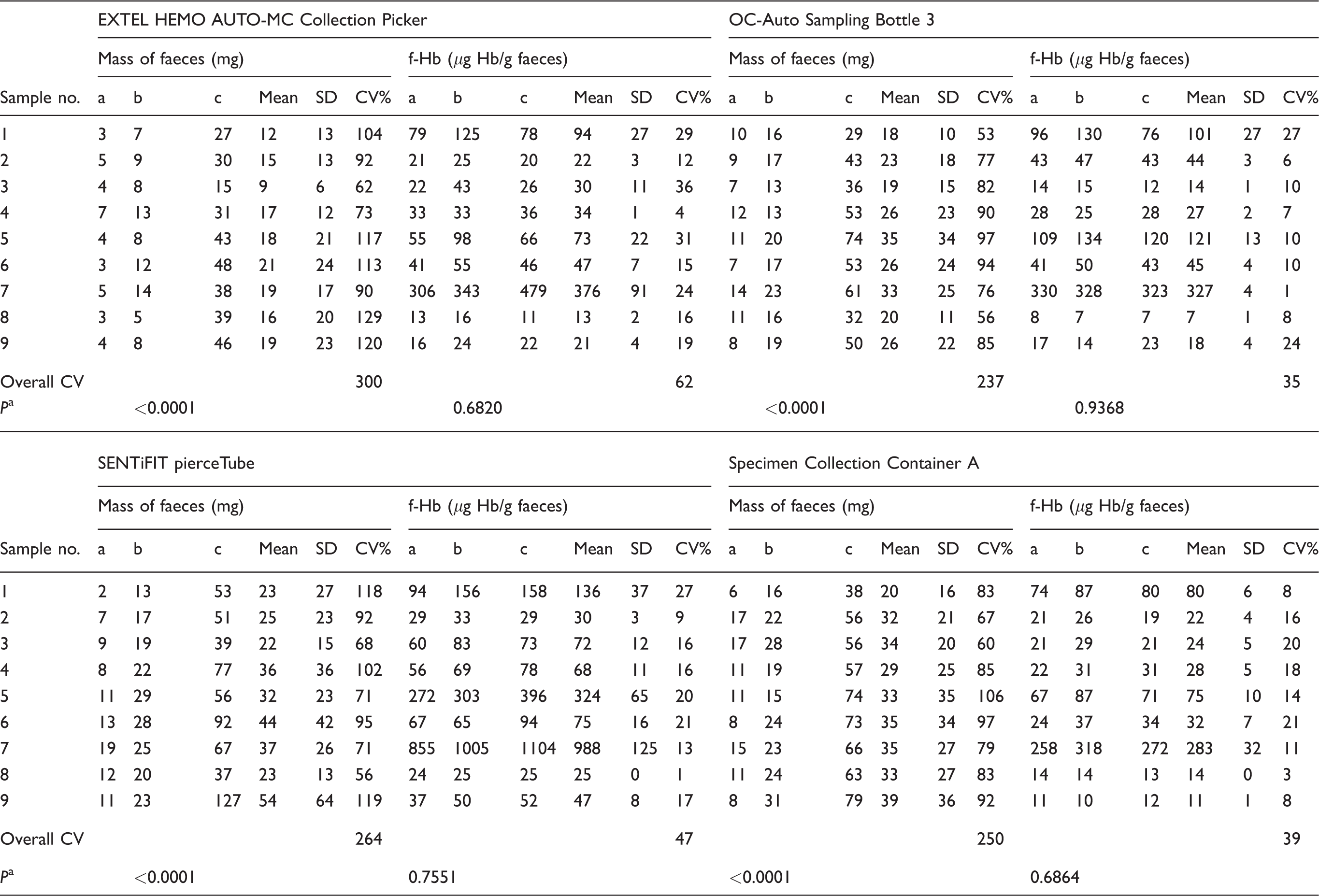
aThe significance of the difference between the masses or f-Hb for values for sampling techniques (a), (b) and (c).
The mass of the faeces added to each group (a) device were less than group (b), and group (b) sample masses were less than group (c). The CVs for the masses of sample loaded with the three techniques for each sample was greater than the CVs for the corresponding f-Hb results, for all four device types. The overall CVs for the masses of faeces added using the three techniques were: EXTEL HEMO AUTO-MC Collection Picker 300%, OC-Auto Sampling Bottle 3 237%, SENTiFIT pierceTube 264%, Specimen Collection Container A 250%. The overall CVs for the corresponding f-Hb were 62%, 35%, 47%, 39%.
The weights of samples were significantly different for sample loading techniques (a), (b) and (c) for all the four device types (
Discussion
The results show that the internal collar of the devices removed some of the excess faeces because the f-Hb showed less variation than the mass of faeces added. Despite having a lower sample mass, devices loaded using technique (b) often gave higher f-Hb results than (c). We suggest this is because the sampling probe tips for technique (b) were covered in faeces in addition to that on the grooves/dimples and the additional faeces could remain on the tip when the probe passed through the collar, or, if inserted at an angle, could be wiped off inside the device before the collar.
However, the degree of consistency of the results varied between the samples and the devices, with samples loaded into the OC-Auto Sampling Bottle 3 showing the least variation in f-Hb (overall CV 35%) and samples loaded into the EXTEL HEMO AUTO-MC Collection Picker showing the most variation (overall CV 62%). The most likely explanation for this is the EXTEL HEMO AUTO-MC Collection Picker collects 2 mg of sample whereas the other devices collect 10 mg of sample.
The manufacturers’ instructions typically state to collect a portion of faeces directly and without delay into the FIT sample collection device. The samples used in this study were first collected into pots, and there was a delay of several days before the sample was loaded into the collection device, during which time there is likely to be degradation of any f-Hb present. While the use of this type of sample is not ideal, it provided a range of sample material that contained a natural source of f-Hb, which is otherwise difficult to obtain and which we consider is an acceptable surrogate marker for addressing the study question which is whether excess sample on the probe impacts the f-Hb result.
In conclusion, this small study supports the manufacturers’ claims that the presence of a ‘collar’ means that excess loading of faeces on to the probe does not significantly affect f-Hb. In this study, the faecal sampling devices were easy to load because the sample consistency was soft; further work is required to look at the effect of more liquid or firmer faeces. Further work is also required to look at the effect of loading higher masses of faeces.
Footnotes
Acknowledgements
We thank FIT system UK suppliers (Alpha Laboratories, Eastleigh, Hants; Mast Diagnostics Division, Bootle, Merseyside; Sysmex UK Ltd, Milton Keynes, Bucks; Alere Ltd, Stockport, Cheshire) for supplying the analysers and consumables, and Berkshire & Surrey Pathology Services for providing the faecal samples.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
CP.
Contributorship
CP conceived the study with input from HB and CJ. CP and CJ processed the samples. CP analysed the data and prepared the manuscript with input from CJ, HB and SB.
