Abstract
Background
There is limited published data on the stability of calcitonin, chromogranin A, thyroglobulin and anti-thyroglobulin antibodies in serum. The aim of this study was to determine stability at three temperature conditions over 7 days, reflecting current laboratory practices.
Methods
Surplus serum was stored at room temperature, refrigerated and in the freezer; for 1, 3, 5 and 7 days. Samples were analysed in batch and analyte concentrations compared to that of a baseline sample. Measurement Uncertainty of the assay was used to determine the Maximal Permissible Difference and thus the stability of the analyte.
Results
Calcitonin was found to be stable for at least 7 days in the freezer but only 24 h refrigerated. Chromogranin A was stable for 3 days when refrigerated and only 24 h at room temperature. Thyroglobulin and anti-thyroglobulin antibodies were stable under all conditions for 7 days.
Conclusion
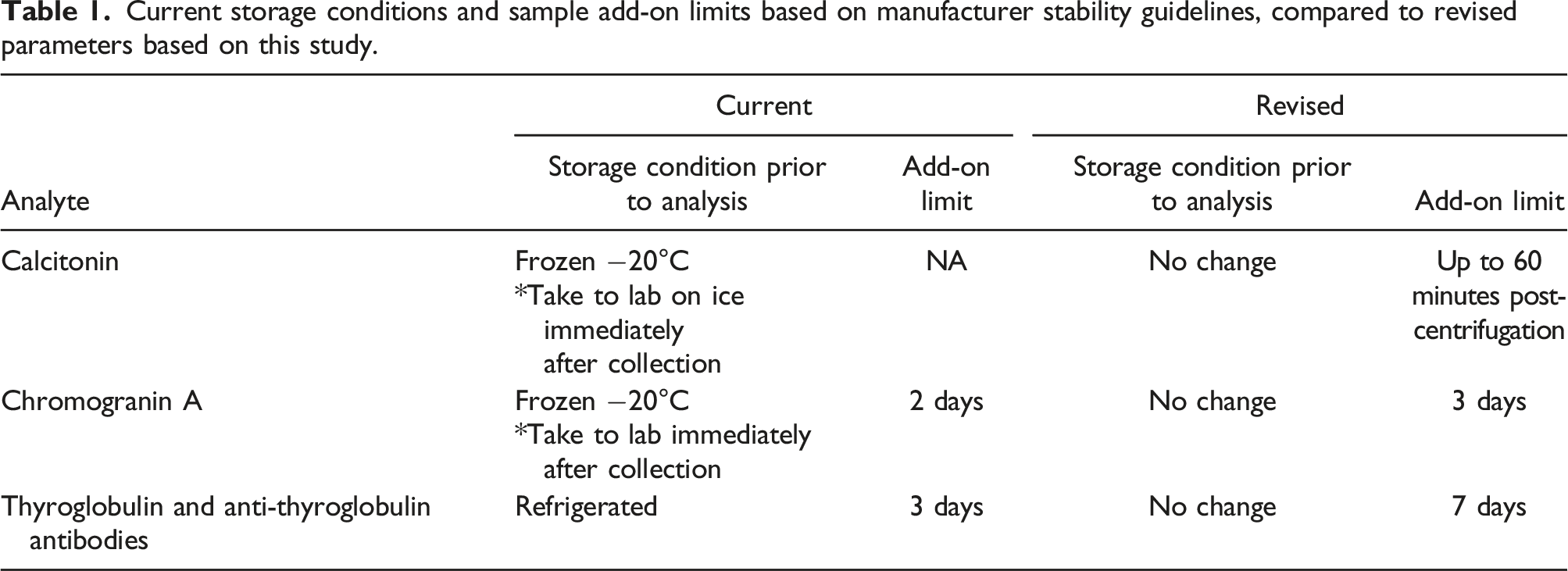
This study has enabled the laboratory to increase the add-on time limit of Chromogranin A to 3 days, and up to 60 min for calcitonin and inform optimal storage and transportation conditions for referring specimens.
Introduction
Thyroglobulin and calcitonin are tumour markers used in the monitoring of follicular and medullary thyroid cancer, respectively, and chromogranin A in the monitoring of neuroendocrine malignancies. Anti-thyroglobulin antibodies are always measured alongside thyroglobulin due to the high levels of interference exhibited in thyroglobulin immunoassay in the presence of these auto-antibodies.
There is a paucity of literature surrounding the stability of these tumour markers with the laboratory currently relying on manufacturer-supplied and historical sample acceptance and storage guidance. International databases such as Norwegian database Noklus exist, but this does not provide data for analytes within this study. These criteria may be more stringent than is required, leading to unnecessary rejection of samples and repeat testing.
As a tertiary oncology centre, the laboratory service offers testing for these specialist tumour markers to the North West region of the United Kingdom and beyond, where referral sites are also using their own criteria for sample acceptance/rejection and storage. The aim of this study was to assess the stability of these analytes in serum stored at ambient laboratory temperature, refrigerated and in the freezer (−20°C) over 7 days, in order to provide an evidence-base for these practices. Time points were selected to reflect current sample storage, reflective add-on order time limits, and to allow for assessment throughout the length of time samples are stored in the laboratory.
Methods
Pools of surplus serum collected from Sarstedt gel separated tubes, across the analytical range were prepared for each of calcitonin, chromogranin A, thyroglobulin and thyroglobulin antibodies. A baseline aliquot was frozen at −80°C and further aliquots stored at laboratory ambient temperature (AT), refrigerator (4°C) and freezer (−20°C). Aliquots were transferred to the −80°C freezer after 24 h, 3, 5 and 7 days, reflecting transit times from referral laboratories, laboratory storage times, and the potential window for reflective testing. Temperature was assessed by Traceable thermometers (VWR). Prior to pooling, blood samples were centrifuged (4 min, 3000xg) on receipt to the laboratory and were stored as follows: calcitonin and chromogranin A were separated and frozen at −20°C on receipt, thyroglobulin and thyroglobulin antibodies were stored at 4°C.
To assess calcitonin stability over a short time period a further three surplus serum samples for calcitonin were stored at AT for 2 h, with aliquots frozen (−80°C) at t = 0, and then every 15 min for 2 h.
To minimise between-batch variation, and to reflect our standard laboratory processes for these analytes, samples were analysed once in one batch; Siemens Immulite 2000 (thyroglobulin, thyroglobulin antibodies and calcitonin) and Thermo Fisher Scientific Kryptor Compact (chromogranin A, Gen II assay). The data presented here was from one experiment, with the exception of thyroglobulin antibodies where results of two experiments were combined. Internal Quality Control (IQC) was analysed with each batch and passed acceptance criteria for all analyses. Samples were analysed once, and the results obtained from the stored samples were compared to the baseline and expressed as a percentage of the baseline sample value. The mean and standard deviation was calculated using Microsoft Excel 2010.
An analyte was said to be stable if the percentage deviation from the baseline sample was within the Measurement Uncertainty (MU) of each assay at the time of analysis. MU was calculated based on laboratory internal quality control data collected over a 6-month period and thus incorporated inter-batch precision. Ethical approval was not required as all serum was surplus from standard of care bloods for these assays, and all pools were anonymised and could not be traced back to the primary sample.
Results
Calcitonin
Three pooled samples were used with baseline calcitonin concentrations of 101, 138 and 12,267 ng/L. The assay had a mean MU of 8.2%. Figure 1(a) shows that stability of calcitonin in serum declines rapidly in samples stored at AT, but as shown in Figure 1(b) (calcitonin concentrations in samples of 4.58, 14.6 and 1455 ng/L) calcitonin is stable for up to 60 min, which would allow ample time for sample pre-processing and storage and permits the laboratory to increase the add-on time limit up to 60 min for calcitonin requests. Calcitonin is only stable for 24 h refrigerated, but stable frozen for at least 7 days (−1.5%, SD 2.2% from baseline). Stability of Calcitonin, Chromogranin A, thyroglobulin and anti-thyroglobulin antibodies in serum over 7 days. The dotted lines show ± Measurement Uncertainty for each analyte and error bar show standard deviation.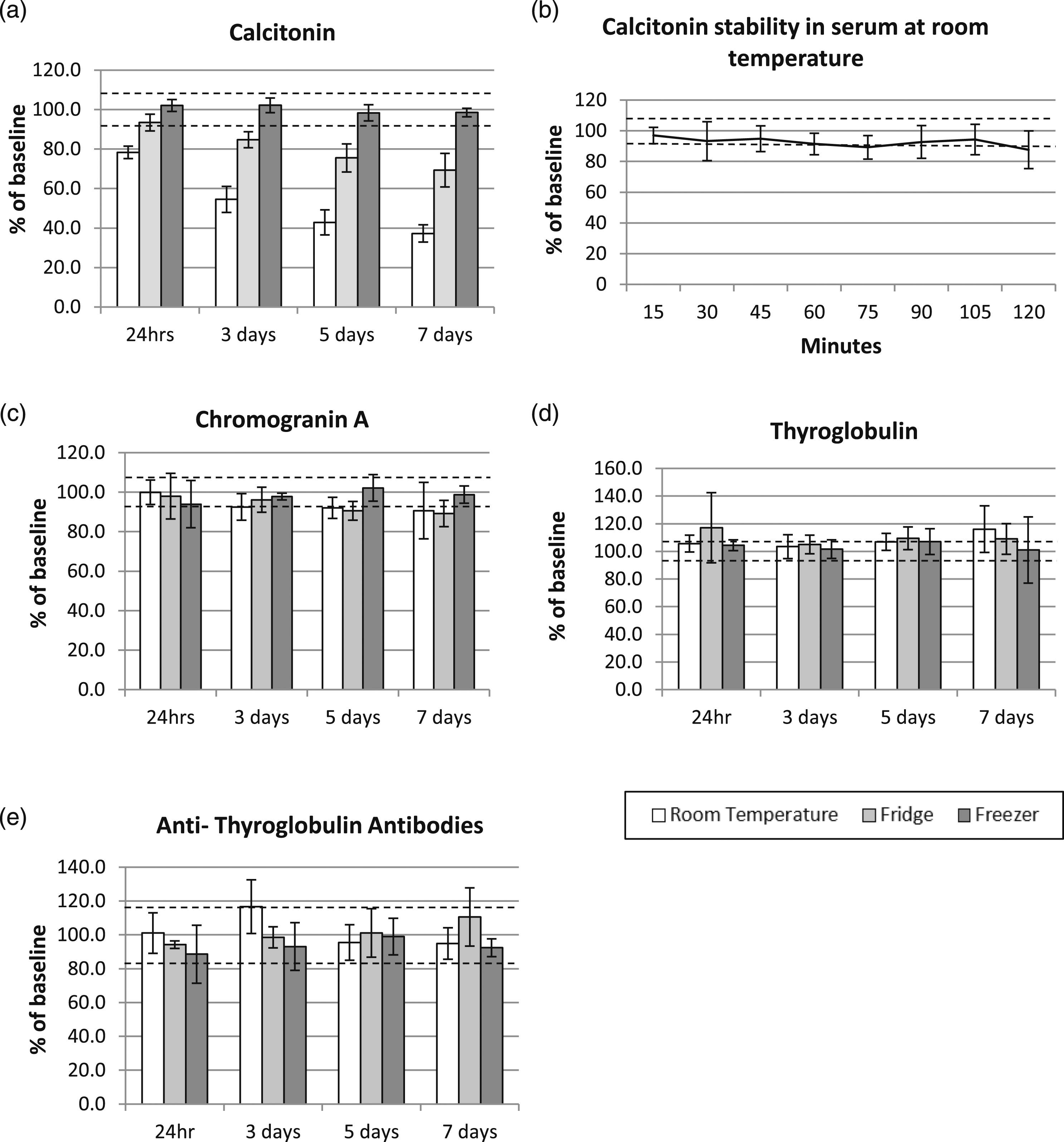
Chromogranin A
Four pooled samples were used with baseline chromogranin A concentrations of 25, 75, 160 and 26,200 ng/mL. The assay had a mean MU of 6.9%. Figure 1(c) shows that chromogranin A is stable for 24 h at AT (0% deviation from baseline, SD 6.2%) however, by 3 days concentrations decreased 7.5%, SD 6.2% from baseline which is greater than the MU of the assay. Chromogranin A is stable for 3 days refrigerated (−3.8% SD 6.3% from baseline) and for at least 7 days frozen (−1.2% SD 4.3% from baseline).
Thyroglobulin
Three pooled samples were used with baseline thyroglobulin concentrations of 21, 135 and 27,314 ng/mL. The assay had a mean MU of 6.9%. Figure 1(d) shows that thyroglobulin is stable for at least 7 days under all storage conditions (+16% SD 16.8%, +9.1% SD 11.8%, and +1.0 SD 24.0%) from baseline at 7 days with storage at AT, fridge and freezer, respectively).
Anti-Thyroglobulin antibodies
Three pooled samples were used with baseline anti-thyroglobulin antibody concentrations of 196, 607 and 2147 IU/mL. The assay had a mean MU of 16.4%. Figure 1(e) shows that anti-thyroglobulin antibodies are stable for at least 7 days under all storage conditions (−5.2% SD 9.3%, +10.5% SD 17.2%, and −7.6% SD 5.8% from baseline at 7 days with storage at AT, fridge and freezer, respectively). Raw data for all analytes can be found in the supplementary file.
Discussion
The stability of calcitonin, chromogranin A, thyroglobulin (and anti-thyroglobulin antibodies) was assessed over 7 days to determine if current storage practices and add-on limits are appropriate and to provide analyte-specific advice to our service users. Assessment of sample stability is important in ensuring correct storage and add-on advice so that we are not reporting results on poor quality samples stored in inappropriate/suboptimal conditions and that we are not rejecting add-on requests due to over-cautious, historical add-on limits.
Our data shows that calcitonin is very unstable, and requires freezing as soon as possible after receipt, however, samples can be stored for up to 60 min once separated if required, and the test can be added on within this very short time-frame. Baylin et al. (1977) 1 found a reduction in measured calcitonin (measured by radioimmunoassay) of 54% in 5 h in patients with hypercalcaemia and 22% in plasma from healthy volunteers, suggesting there would be rapid deterioration in calcitonin concentrations on taking this study beyond 2 h. In addition, there is a knowledge gap concerning the stability of calcitonin in whole blood prior to sample separation.
Chromogranin A was shown to be stable when refrigerated for 3 days, allowing the laboratory to increase the reflective test request add-on time-window from 2 days to 3 days. Pedersen et al. (2014) 2 showed loss of chromogranin A of 15.6% after 24 h and 44% after 48 h in samples stored at 4°C in the serum of 10 healthy subjects. This difference is explained by use of the CgA Generation II assay used in this study which has been shown to exhibit increased stability at ambient temperature compared to the Generation I assay, 3 highlighting that stability is assay-specific. Several reasons for this observation have been hypothesised including proteolysis, protein folding, CgA epitope stability or exposure.
Current storage conditions and sample add-on limits based on manufacturer stability guidelines, compared to revised parameters based on this study.
A major challenge faced by this study was selecting samples with detectable concentrations of calcitonin and thyroglobulin, which is likely due to use of these markers in tumours that are treated with curative intent and where these markers are more commonly used for early detection of relapse, meaning there was only sufficient sample volume for single sample analysis. In addition, stability at some time points was increased beyond MU for both thyroglobulin and anti-thyroglobulin antibodies, resulting in wide standard deviations. Whilst the cause of this is not known this could be due to artefacts of the freeze-thaw, or of the age of samples used in the patient pools, which had likely been stored some time due to limited sample availability at higher concentrations Due to this, further patient sample pools are required for a more comprehensive long-term stability assessment; however, this study has provided an evidence-base to guide sample storage conditions and test request add-on limits, specific to the analytical platforms within this report, which can be used to better inform laboratory service users and reduce rejection of samples for analysis.
Since this study, European Federation of Clinical Chemistry and Laboratory Medicine (EFLM) Working Group for the Preanalytical Phase (WG-PRE) has released a checklist for the reporting of stability studies, 6 including clinical details of all samples included in any pooled sample, method of phlebotomy and preanalytical sample handling, which would be utilised in any extension of this work.
Supplemental Material
Supplemental Material - Stability assessment of serum tumour markers: Calcitonin, chromogranin A, thyroglobulin and anti-thyroglobulin antibodies
Supplemental Material for Stability assessment of serum tumour markers: Calcitonin, chromogranin A, thyroglobulin and anti-thyroglobulin antibodies by Sally Thirkettle, Patrycja Blaszczyk, Rachel Evans, Michael Wheatley, Madihah Abbas, Joanne Russell and Phillip J Monaghan in Annals of Clinical Biochemistry
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
N/A.
Guarantor
PM.
Contributorship
TS – draft manuscript, design, data analysis; PB, RE, MW, MA, JR – study set up and sample analysis; PM – study design, manuscript review.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
