Abstract
Background
Levodopa (L-DOPA) provided to patients with Parkinson’s disease causes an increase in dopamine and methoxytyramine blood concentration which may lead to erroneous diagnosis of dopamine-producing tumours based on a plasma fractionated metanephrines and methoxytyramine assay. Considering that oral L-DOPA is mainly transformed in the gut wall into dopamine and methoxytyramine, we hypothesize that patients treated with L-DOPA produce predominantly sulphated methoxytyramine, whereas dopamine-producing tumours, devoid of sulfotransferase, will secrete free methoxytyramine. These metabolic differences may allow for discrimination between the two groups of patients through methoxytyramine plasma concentration.
Methods
We retrospectively investigated a cohort of 16 patients with a dopamine-secreting pheochromocytoma or paraganglioma and 22 patients treated for Parkinson’s disease to see whether the metabolic ratio of free and sulphated methoxytyramine differs.
Results
Receiver operating characteristic curve analysis indicates an absolute separation between the two groups when using a cut-off of free/total methoxytyramine (sum of free and sulphated methoxytyramine) ratio of 0.0059, corresponding to a free methoxytyramine fraction of 0.59% (
Conclusion
Dopamine secreted by tumours and exogenous dopamine (from Parkinson’s disease treatment) follow different metabolic pathways. We observed that free/total methoxytyramine ratio may be a useful tool in distinguishing between patients with a dopamine-secreting tumour from patients treated with L-DOPA when clinical information is incomplete or lacking.
Keywords
Introduction
Dopamine (DA) undergoes a complex metabolism that has been thoroughly studied over the last decades. However, differences in the fate of dopamine when it derives from endogenous pathologic secretion or when it is orally administrated in the form of levodopa (L-DOPA), are worth being investigated. While medication and diet are the main sources for exogenous L-DOPA, endogenous L-DOPA derives from tyrosine hydroxylation by tyrosine hydroxylase (TH, EC 1.14.16.2), the rate limiting enzyme for catecholamine (CAT) production in the cytoplasm of CAT-producing cells (e.g. nerve endings and chromaffin cells from the adrenal medulla). L-DOPA is known to increase the concentration of circulating DA and methoxytyramine (MT). L-DOPA treatment in Parkinson’s disease (PD) patients is a frequent preanalytical pitfall when diagnosing DA-secreting pheochromocytoma (PHEO) or paraganglioma (PGL) by measurement of plasma-fractioned metanephrines (MNs) and MT concentrations. 1
Several reports have highlighted the importance of a detailed drug history in order to prevent misdiagnosis of PHEO/PGL, especially for patients on PD medication.2–4 However, when the information on patient’s medication is limited, it can preclude the interpretation of high MT results.
A recent study has brought new insights to discriminate patients on L-DOPA therapy from the patients affected by a PHEO/PGL that produce normetanephrine (NMN) and metanephrine (MN) based on plasma and/or urine MNs tests. 5 However, as highlighted by the authors, this study did not include tumours exclusively secreting DA. These tumours are rare and difficult to diagnose because affected individuals are often asymptomatic or suffer from unspecific symptoms such as nausea, weight loss, or hypotension, but not hypertension.6–8
Considering the current knowledge of L-DOPA metabolism, our hypothesis is that the free/total MT ratio in plasma may differ in patients treated with L-DOPA co-prescribed with aromatic-amino acid-decarboxylase (AADC) and/or catechol-O-methyltransferase (COMT) inhibitors as compared with patients affected by PHEO/PGL which exclusively secrets DA and MT.
Materials and methods
We performed a retrospective, anonymized, systematic search of our lab results since 2008 for patients suspected of having PHEO/PGL, exhibiting isolated elevations in MT: high plasma concentrations of MT (free MT > 0.06 nmol/L and total MT > 4.19 nmol/L) and NMN and MN concentrations below our upper reference limit. 9 This search identified 208 records corresponding to 146 patients fulfilling the selection criteria. Among these, clinical information was available for 38 subjects, allowing for their inclusion in our investigation. Twenty-two (12 females) were PD patients treated with L-DOPA and 16 patients (7 females) were diagnosed with a DA-only secreting tumour based on biochemical tests (MNs, MT and CAT quantification) and confirmed by imaging techniques, or by histology in case of tumour resection. For all patients included in the L-DOPA group, no PGL or PHEO were diagnosed for at least several months after the biochemical measurements. Data from these two patient groups were further compared with the results of 75 control subjects (no PHEO/PGL or L-DOPA treatment) known for hyperaldosteronism and treated for hypertension recruited for another study (unpublished results). Free and total MT values are expressed in nanomoles of metabolite per litre of plasma.
All blood samples were collected using a forearm venous cannula, with the patient kept supine for at least 15 min before sampling. Patients were instructed to fast and to abstain from caffeinated and alcohol beverages and tobacco for 24 h before blood collection. All samples were collected onto ice and centrifuged within 30 min after puncture. Plasma was kept at −80 °C until analysis. Total MNs and MT were desulphated using perchloric acid method or treated with sulphatase as described in a previous study. 10 Total and free MNs and MT were extracted on µElution plate and quantified by ultraperformance liquid chromatography-tandem mass spectrometry (UPLC-MS/MS).11,12 The validation of the method to quantify free MNs and MT was described by Peaston and colleagues and calculated CV inter-assay from three different concentrations and 20 different assays was reported to be lower than 13% for MT. 12 In no case would the inter-assay imprecision interfere with the high significance of the free/total MT ratio. The method used for free and total MT quantification is identical after the deconjugation treatment applied to the sulphated MNs and MT. 10
Calculations were performed on a Microsoft Excel® sheet. The Stata® software was used for all further statistical calculations (version 14.0 StataCorp, College Station, TX, USA). Graphs were prepared using the Prism software (Version 7.03, GraphPad Software, La Jolla, CA, USA).
This investigation was conducted according to the Swiss law for clinical research, the international good clinical practice (GCP) and ICH requirements. The regional ethics committee for the Canton de Vaud reviewed and approved this study.
Results
Thirty-eight subjects were included in our investigation: 22 in the L-DOPA treatment group and 16 in the DA-tumour group. Patients in the L-DOPA group are older than those in the DA-secreting tumour group: geometric means (CI 95%) are 75.1 (71.7–78.7) and 47.9 years (39.4–58.2), respectively (
Free MT, total MT and free/total MT concentration in plasma in nmol/L, age (years) and GFR values (ml/min) for the three groups.
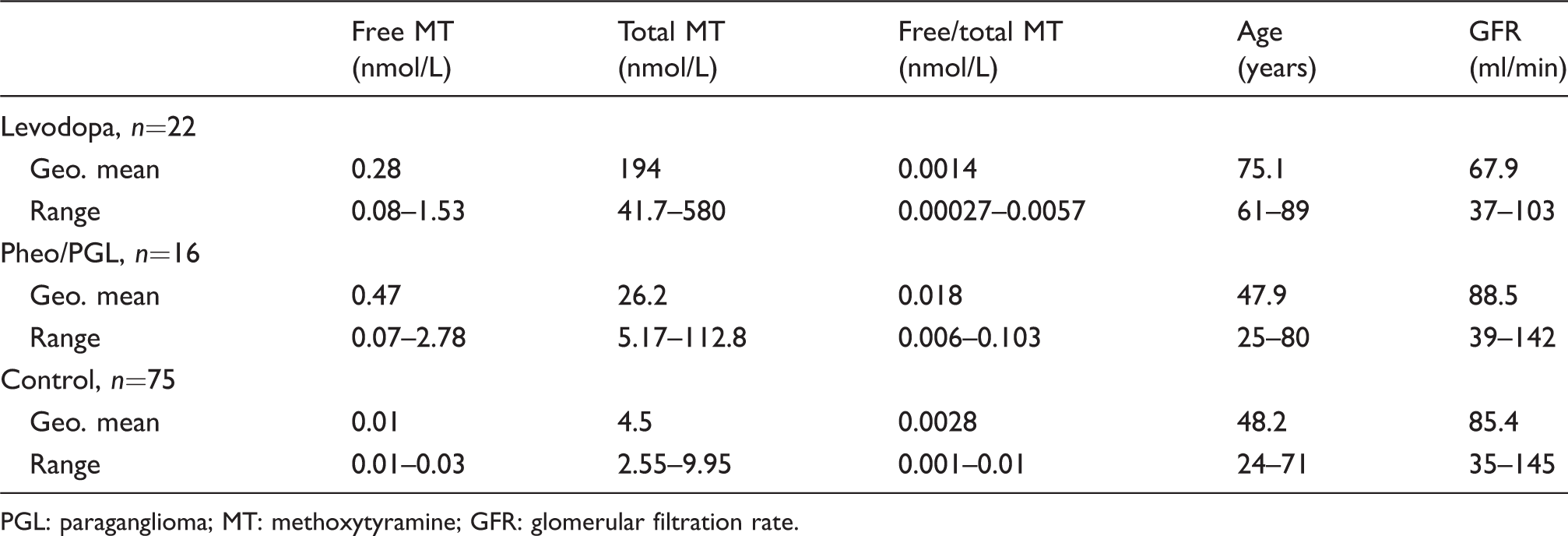
PGL: paraganglioma; MT: methoxytyramine; GFR: glomerular filtration rate.
While we find comparable free MT plasma concentrations between both patient groups (
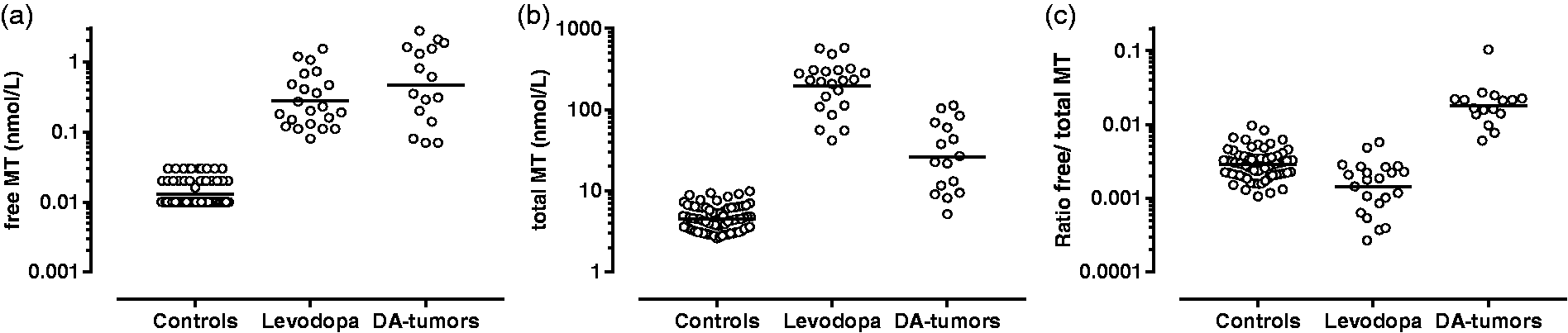
Plasma concentration of free MT (a), total MT (b) and ratio free/total MT (c) for control patients, levodopa-treated patients and patients affected by a DA-only secreting tumours.
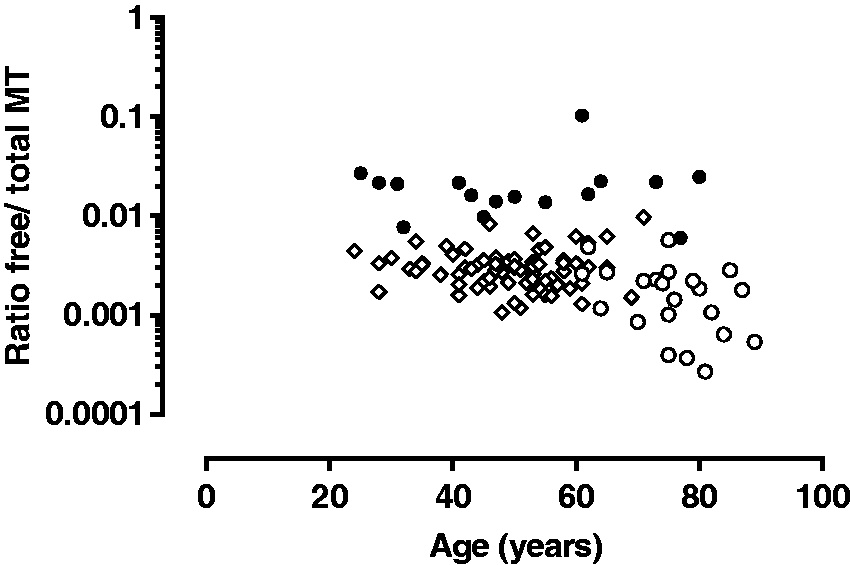
Free/total MT ratio according to age in the L-DOPA (○), DA-secreting tumour (●) and control subjects (◊) group.
Receiver operating characteristic (ROC) curve analyses (Figure 3) are used to discriminate between patients treated with L-DOPA from those having a DA-secreting tumour. ROC curves indicate no significant discriminating potential for free MT (
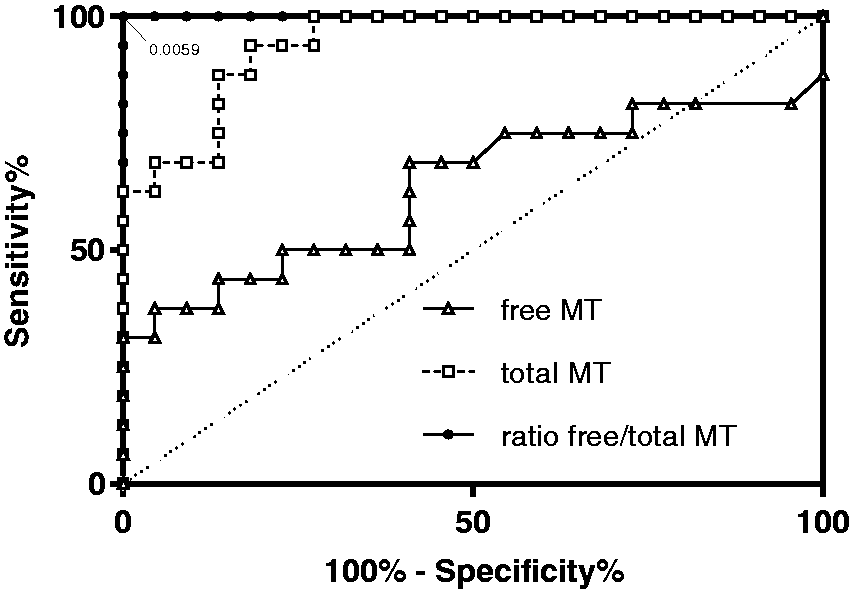
ROC curves for free MT, total MT and ratio free/total MT to discriminate between L-DOPA medication and DA-secreting tumours.
Discussion
L-DOPA is a prodrug. After oral administration, its bioavailability is approximatively 70–100% when co-prescribed with carbidopa or benserazide. These two inhibitors prevent the peripheral transformation of L-DOPA into DA, enabling a larger proportion of L-DOPA to cross the blood–brain barrier. 13 Its absorption occurs in the duodenum and proximal jejunum. 14 L-DOPA then undergoes a complex metabolism. It is transformed by the action of AADC (EC 4.1.1.28) into DA, which is further metabolized into MT within cells containing the enzyme COMT (EC 2.1.1.6). Sulfotransferase 1A3 (SULT1A3, EC 2.8.2) is the main enzyme involved in L-DOPA, DA and MT sulphation. 15 Only a small fraction of L-DOPA in plasma is detected in its sulphated form, while a large proportion of DA (>90%) and MT circulate in their sulphated form. 16 According to the literature, DA sulphate production takes place mainly in the gastrointestinal tract16,17; therefore, mesenteric organs receiving exogenous Tyrosine, L-DOPA, and DA harbour strong expression of SULT1A3 and are considered as the major production site of DA- and MT-sulphate conjugates. 17 This contrasts with DA-secreting tumours devoid of SULT1A3 which exclusively produce free DA from tyrosine, and subsequently MT through the action of local COMT. 15
Our data found significantly higher total MT concentrations in the L-DOPA group compared with the DA-secreting tumour group, whereas comparable free MT plasma concentrations were observed in both groups. We observed that L-DOPA, depending on its origin (medication or tumoural), follows different metabolic pathways. After oral administration, L-DOPA is transformed into sulphated DA and MT, causing a massive amount of sulphated MT to enter the portal circulation, decreasing the free/total ratio of MT in L-DOPA-treated patients compared with the DA-secreting tumour group. Nutriments rich in tyrosine, the precursor of DA, may also give rise to an increase of both free and sulphated MT, which could ultimately influence our results. 18 However, in our study, the free and total MT values above our upper reference limits were due to either a tumour or to levodopa medication.
Excess total MT concentration is found in plasma of patients taking L-DOPA, causing a lower free/total MT ratio which enables discrimination between patients from both groups, as supported by a ROC curve analysis. According to our ROC curve analyses, no significant discriminating potential was identified for free MT, while total MT allowed for distinguishing both groups. However, an absolute separation of both groups (100% sensitivity and specificity) was observed with the free/total MT ratio with a cut-off corresponding to a free MT fraction of 0.59%.
Alterations in the metabolic reaction sequence in the L-DOPA pathway may also account for some of the observed differences in MT ratio between both groups. Enzymatic inhibitions (AADC and COMT) may provide more L-DOPA available to SULT1A3, possibly resulting in an increased conversion of both exogenous and endogenous free L-DOPA to sulphated L-DOPA in peripheral tissue. AADC and COMT inhibition may be incomplete, allowing transformation into DA and MT. Moreover, a recent study reported that DA was able to induce SULT1A3 expression.
19
The administration of large amounts of L-DOPA may therefore increase the production of sulphated DA and MT. In favour of this hypothesis, the free/total MT ratio shows an increased sulphation of MT in the L-DOPA group compared with the DA tumour group (0.1% vs. 1.8% of free forms,
The observed age difference between our two groups is inherent to the nature of the studied diseases, and in addition, older subjects are more prone to have a lower GFR. A statistically significant but modest influence of GFR was observed on total MT concentrations and on the free/total MT ratio. However, this impact should be balanced, as it may only reflect the confounding influence of age on both total MT concentrations and GFR. Indeed, testing together age and GFR revealed a significant (but clinically modest) impact only for age.
A limitation of this study relies on factors not considered in our analysis that may also account for the large variability of free and total plasma MT concentration such as L-DOPA administration route, the amounts of drug or the presence of AADC and/or COMT inhibitors. Although pharmacokinetic considerations have not been accounted for in this study, we are able to consistently observe the high discriminating potential of the free/total MT ratio.
Conclusion
In this study, we aim to decipher how L-DOPA metabolism, specifically its transformation into DA and MT and their sulphation by SULT1A3, may differ depending on the metabolic path and production site. Oral administration of high amounts of L-DOPA, which undergoes hepatic first path transformation, associated to AADC and/or COMT inhibitors lead to a metabolite ratio which differs from DA produced and secreted by tumour cells. We indeed observed that quantification of free and total MT in plasma and calculating the free MT fraction allows differentiation between patients taking L-DOPA from patients affected by a DA-only secreting tumour.
Supplemental Material
Supplemental material for High concentration of plasma methoxytyramine: dopamine-producing tumour or Parkinson’s disease therapy?
Supplemental Material for High concentration of plasma methoxytyramine: dopamine-producing tumour or Parkinson’s disease therapy? by Haithem Chtioui, Samira M Sadowski, Bettina Winzeler, Oliver Tschopp, Eric Grouzmann and Karim Abid in Annals of Clinical Biochemistry
Footnotes
Acknowledgements
We are grateful to Dr G. Wuerzner (Service of Nephrology and Hypertension, Department of Medicine, Lausanne University Hospital, Lausanne, Switzerland) for providing data on patients included in the control group. We also thank Mrs C. Centeno and C. Seghezzi for excellent technical help for metabolites quantification.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by the regional ethics committee for the Canton de Vaud (Reference number:2017–01865).
Guarantor
EG.
Contributorship
KA, HC and EG conceived the study. Laboratory technicians performed the practical work. SMS, BW, OT, KA, HC and EG interpreted the data and wrote the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
