Abstract
Objectives
Up to 3% of patients with monoclonal gammopathies have multiple serum paraproteins. This article investigates whether multiple isotype-matched paraproteins, as seen on capillary zone electrophoresis, are truly biclonal.
Methods
Serum samples containing multiple isotype-matched paraproteins were treated with the reducing agent dithiothreitol, and capillary zone electrophoresis was performed pre- and post-treatment. Band resolution and effect of resolution on quantitation of paraprotein burden were assessed. The Hevylite
Results
Among patients with biclonal isotype-matched paraproteins, 23/24 (96%) IgA paraproteins resolved into a single band following treatment with dithiothreitol compared with only 1/12 (8%) IgG paraproteins. Daratumumab therapy accounted for the second band in 5/9 non-resolving IgGκ paraproteins. Where initially quantified as a single IgA ‘complex’ (multiple bands in close proximity), the single postdithiothreitol band averaged 2.8 g/L less (
Conclusions
These results suggest that disulphide-mediated polymerisation of IgA paraproteins is more common than true biclonal gammopathy and support dithiothreitol treatment of samples with isotype-matched IgA bands before quantifying by capillary zone electrophoresis. The Hevylite
Background
Detection and monitoring of patients with myeloma or other paraprotein-related diseases most commonly uses serum protein electrophoresis (with immunofixation or immunosubtraction methods for identifying the paraprotein type).
The majority of patients with a paraprotein have just one band detected by serum electrophoresis; however, two or more bands have been reported to occur in 2–3% of all patients with a gammopathy.1–5 This is sometimes referred to as ‘double gammopathy’ or ‘biclonal gammopathy’, and can occur in multiple myeloma, other lymphoproliferative disorders or in Monoclonal Gammopathy of Uncertain Significance (MGUS). 6
When these bands are different heavy–light chain combinations, it is likely that these paraproteins originate from separate clones of cells (although double-staining of plasma cells has demonstrated incidences where one cell can produce two different paraproteins). 3 This could occur from the proliferation of two distinct plasma cell clones or as a consequence of class switching whereby the type of paraprotein synthesized by a subset of cells changes. The most commonly arising combinations of paraproteins are IgG–IgA, IgG–IgM and IgG–IgG. 6
Where typing of multiple bands reveals an identical heavy–light chain combination (isotype matched), whether these are truly from separate clones of plasma cells is less certain.
One possible explanation for a single paraprotein resulting in multiple bands on electrophoresis is the formation of polymers. It has been reported that treatment of the serum samples of patients with two isotype-matched IgA paraproteins with β-mercaptoethanol can resolve multiple bands that are a result of paraprotein polymerisation, leaving one band on electrophoresis. 7 Indeed, Kyle et al. suggested in 1981 that ‘in IgA gammopathy, the two bands on the cellulose acetate tracing are most often monomers and polymers of IgA rather than components of biclonal gammopathy’. The authors also suggested that it is ‘essential to exclude aggregates of IgG monoclonal protein when making a diagnosis of a biclonal IgG gammopathy’, although how this is achieved is not discussed. 3
The presence of multiple bands of the same heavy–light chain type complicates how these paraproteins should be quantified and reported – assuming they are representative of two smaller clones of cells may give a different impression of tumour cell burden versus interpreting them as one paraprotein load from one clone. However, studies have demonstrated no clinical difference between monoclonal and biclonal gammopathy in terms of patient survival, response to therapy and rate of myeloma-associated anaemia, renal dysfunction or lytic bone lesions.3,8
The International Myeloma Working Group 2015 criteria for the diagnosis of multiple myeloma suggest the use of serum monoclonal protein concentration in the classification of monoclonal gammopathies but do not address the effect of biclonal isotype-matched (BIM) gammopathy. 9 This study aims to improve understanding of potential biclonal gammopathies of the same heavy and light chain (HLC) combination and to help with consistency of paraprotein reporting in patients being monitored. IgA and IgG BIM paraproteins were investigated here, both with capillary zone electrophoresis (CZE) and the Hevylite® assays that quantitate individual HLC combinations due to the potential benefits compared with traditional electrophoresis-based methods.
Materials and methods
All samples were collected and analyses performed within Barts Health NHS Trust biochemistry department.
Identification of samples
Protein electrophoresis and paraprotein quantitation was routinely carried out on a Sebia Capillarys CZE. Band quantitation was via the perpendicular drop method (including baseline) that is the quantitation method used in routine analyses.
Patients with BIM IgA or IgG gammopathy were identified from twice weekly searches of the laboratory routine protein electrophoresis results and 70 residual samples were retrieved (58 IgA and 12 IgG). Samples were collected over a period of five months (February 2018–June 2018), stored at –20°C and thawed on the day of analysis. For DTT treatment, BIM samples were stored at 4°C for no more than one week before analysis (hence, lower numbers of samples were treated with DTT, see below). Samples from patients with single IgA (
DTT treatment of samples with isotype-matched paraproteins
A total of 24 IgA BIM paraproteins and 12 IgG BIM paraproteins were treated with DTT to evaluate possible polymerisation.
Prior to CZE, 360
Hevylite® assay verification
The performance of IgA kappa, IgA lambda, IgG kappa and IgG lambda Hevylite® assays (The Binding Site™, Birmingham, UK) on the SPAPlus analyser was verified as described by Khatami et al. 10 ACB spread sheets A (Kallner, version 6.84), B (Kallner, version 3.7) and D (Kallner, version 1.7) were used to statistically assess imprecision and bias. 11
All Hevylite® assays achieved acceptable repeatability and intralaboratory imprecision (CV <3% for both IgA and IgG assays).
EQA material was obtained from The Binding Site™ IMMPROVE quality assurance scheme – distributions 138–143. Bias was assessed using Z-score, desirable bias, desirable total error and desirable minimum error as defined in Ricos et al. 12 using non-paraprotein biological variation data for immunoglobulins.13,14 No significant bias was noted.
Using high concentration paraproteins (46 g/L IgGκ, 53 g/L IgGλ, 60 g/L IgAκ, 43 g/L IgAλ), linearity was deemed acceptable since the slope of the line of best fit for measured concentration versus expected concentration was close to 1 and the intercept close to zero and the R2 value was equal to or greater than 0.99.
Assessment of Sebia Capillarys linearity
A high concentration IgGκ paraprotein (reported paraprotein concentration of 94.7 g/L) was diluted in HPLC grade water for linearity assessment of Sebia Capillarys. Linearity was deemed acceptable as explained above.
Statistical analysis
The two-tailed paired
Results
IgA BIM paraproteins are multimers of monoclones rather than biclonal paraproteins
DTT was used as a reducing agent to investigate the possibility that BIM paraproteins are in fact polymers of a monoclonal paraprotein.
In total, 36 samples were treated with DTT: 24 IgA BIM samples (from 21 patients) and 12 IgG BIM samples (from 11 patients).
Conclusive results were obtained with IgA paraproteins – 96% of these resolved to one band after treatment with DTT (14/15 IgAκ and 9/9 IgAλ). Conversely, 92% of IgG samples did not resolve upon treatment with DTT (8/9 IgGκ and 3/3 IgGλ). Four patients had single repeat samples included in this data-set which responded consistently to DTT treatment. Example electropherograms for different heavy–light chain combinations are shown in Figure 1.
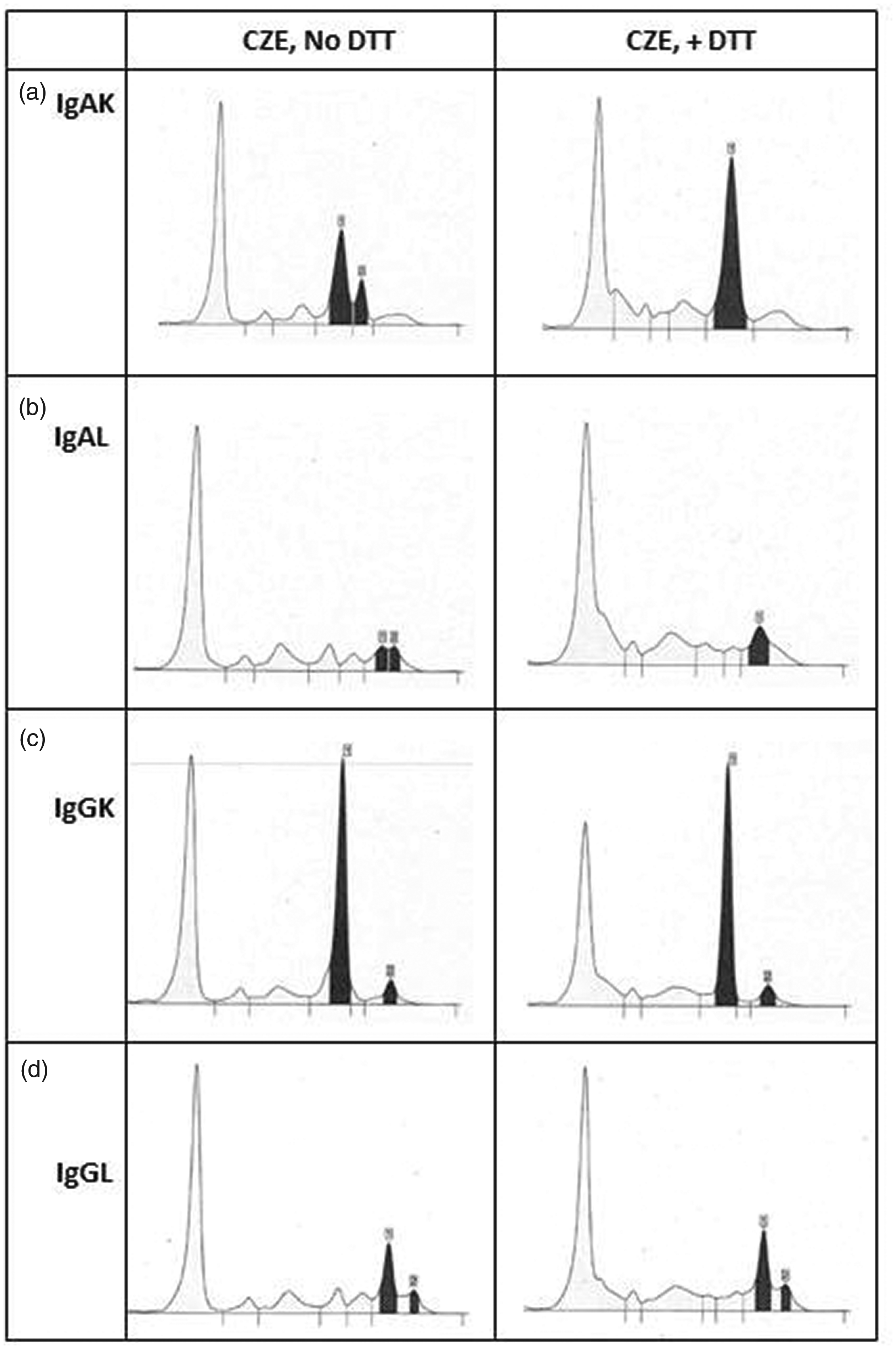
Example electropherograms for samples with BIM gammopathy. Examples pre-DTT are shown in the left panel, and post-DTT in the right panel. (a) IgAκ, (b) IgAλ, (c) IgGκ and (d) IgGλ.
Five of the nine samples with IgGκ BIM paraproteins were from four patients retrospectively identified to be on daratumumab treatment. This is a monoclonal IgGκ antibody and hence resolution into a single band could not be expected.
The effect of DTT on quantitation of BIM paraproteins
For IgA paraproteins which were originally quantified as two discrete separate bands (that were added together) and resolved into one band on CZE, excellent correlation was obtained between quantitation pre- and post-DTT (R2=0.995, y = 1.01x–0.28,
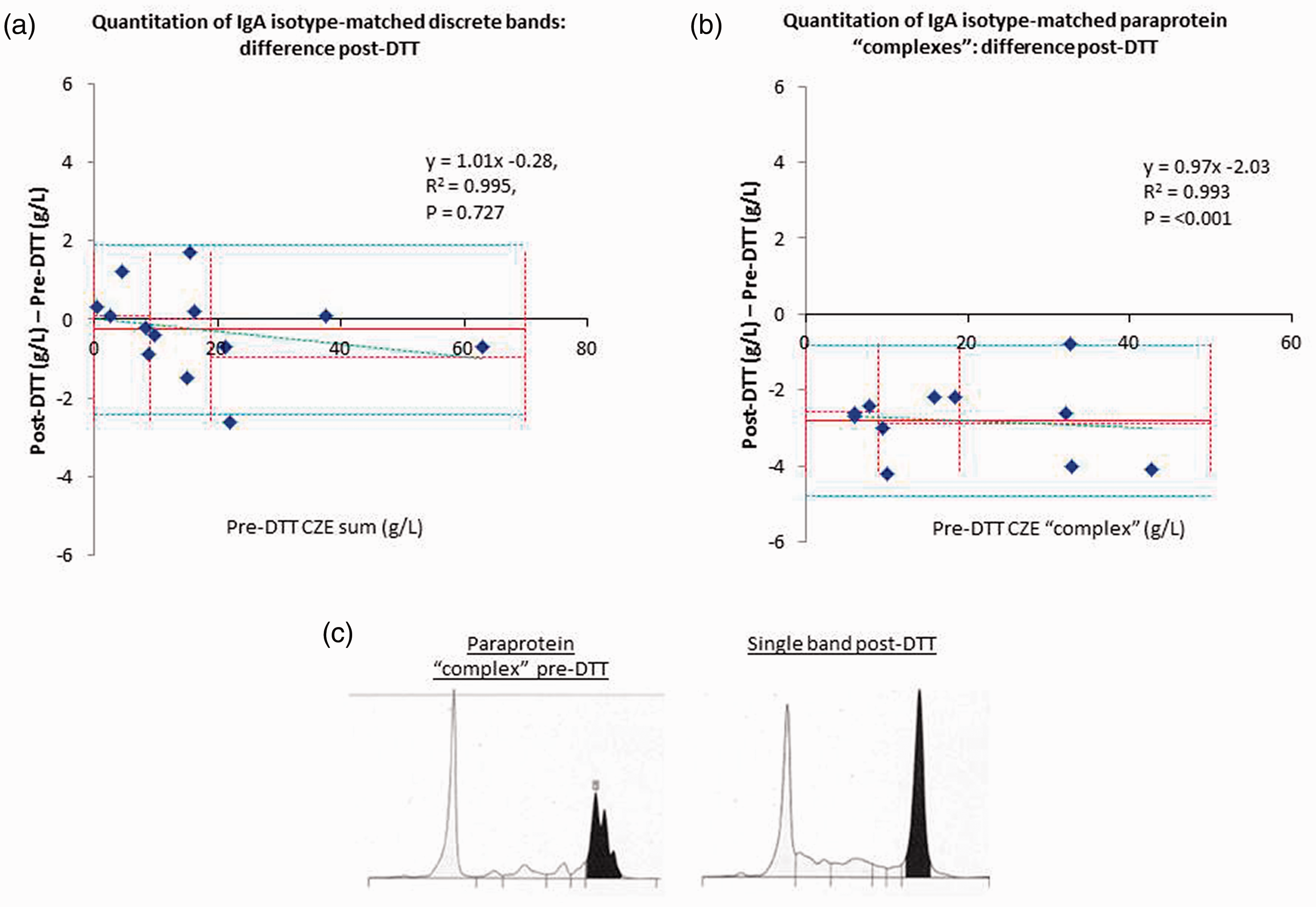
Comparison of IgA discrete BIM paraproteins and ‘complex’ paraprotein quantitation pre- and post-DTT. (a) IgA BIM discrete bands
Where multiple isotype-matched bands migrate closely on CZE, these are sometimes quantified as one ‘complex’, inclusive of any background proteins.
Quantitation of paraproteins originally quantified as one ‘complex’ were consistently lower after DTT and resolution to one band (Figure 2(b) mean –2.8 g/L, range –0.8 to –4.2 g/L,
Note that in both groups (discrete bands and complex paraproteins) the IgA paraproteins migrated in both the beta and gamma regions. There did not appear to be a preference for migration in a particular zone based on whether the bands were initially discrete or complex.
Hevylite® overestimates IgA BIM paraprotein concentration
Single IgA paraprotein results (
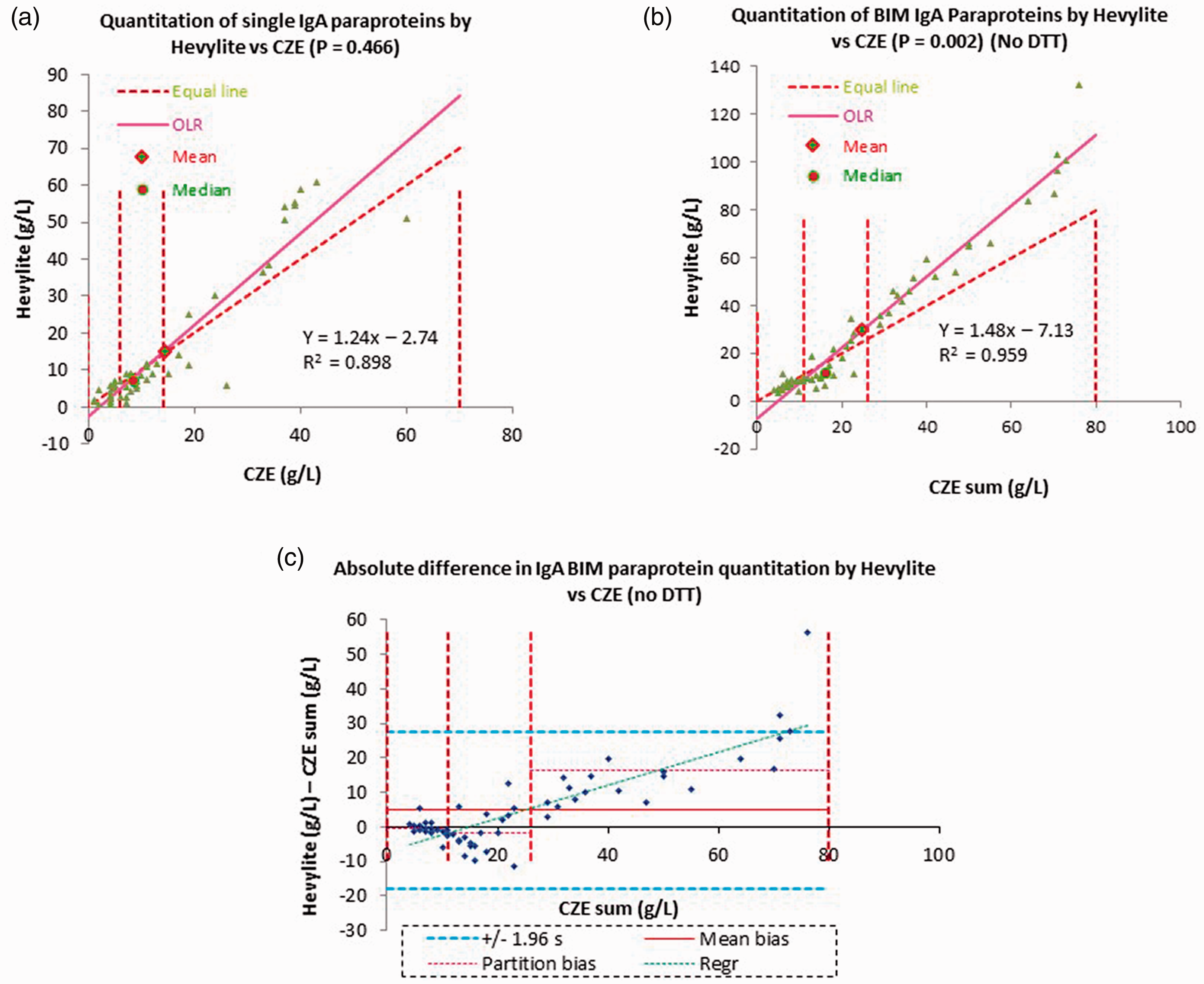
Comparison of quantitation of multiple paraproteins by Hevylite® assay and CZE vs. quantitation of single paraproteins. (a) Single IgA paraproteins
For IgG paraproteins, quantitation by Hevylite
Underestimation of paraprotein concentration due to signal saturation on CZE was ruled out by establishing assay linearity to at least a paraprotein concentration of 94 g/L (data not shown).
Discussion
Up to 3% of patients with monoclonal gammopathies have two or more serum paraproteins, and some of these are isotype matched. It has been reported that treating samples with multiple isotype-matched bands on electrophoresis with the reducing agent β-mercaptoethanol can resolve these bands into one, suggesting that these are polymers of IgA. 7
In this study, 96% of IgA BIM paraproteins resolved into one band after treatment with DTT. This strongly suggests that these patients have a single monoclonal IgA gammopathy. Whereas DTT treatment of samples with discrete isotype-matched IgA paraproteins did not affect quantitation, for samples containing IgA paraprotein complexes that resolved into one band, the quantity of paraprotein was significantly lower indicating overestimation of the original CZE result, likely due to inclusion of underlying globulins in the wider baseline pre-DTT.
The majority of samples with BIM IgG peaks on CZE did not resolve after DTT treatment, suggesting that these are more likely to be paraproteins from separate clones rather than multimers, except where a patient is known to be treated with a therapeutic monoclonal antibody such as daratumumab, which accounts for a large proportion of such cases (5/9 non-resolving IgGκ BIM paraproteins). The failure of non-daratumumab IgG BIM paraproteins to resolve is in agreement with the fact that IgG is monomeric, whereas IgA is known to form dimers and higher polymers. 15 The cysteine residue in the secretory tailpiece of the heavy chain of IgA and the joining chain (J-chain) are involved in the formation of dimers and higher polymers of IgA through disulphide linkage.15–18 IgG lacks these features, explaining the ability of DTT to resolve multiple bands on CZE for IgA but not IgG paraproteins through reduction of disulphide bridges.
Glycans account for up to 15% of the molecular weight of IgG, and altered glycosylation of both IgG and IgA has been linked to diseases such as IgA nephropathy and rheumatoid arthritis. 19 Therefore, while it appears that most IgA BIM paraproteins are due to multimerization, treatment of IgG BIM paraproteins in particular with de-glycosylating enzymes may demonstrate whether glycosylation is responsible for multiple IgG bands on CZE when therapeutic monoclonal antibodies are not the cause.
This study also evaluated the Hevylite® immunoturbidimetric assay for the quantitation of single and BIM paraproteins. Proposed benefits of the Hevylite® assay include greater sensitivity over electrophoretic methods due to the additional information given by heavy–light chain pair suppression, 20 improved quantitation of paraproteins migrating in the beta region on electrophoresis 21 and improved quantitation of broadly migrating paraproteins. 22
Here, Hevylite® demonstrated a significant concentration-dependent positive bias for IgA but not IgG BIM paraproteins. This positive bias was worse than that seen for single IgA paraproteins and is likely due to polymerisation leading to an enhanced signal in the turbidimetric assay, in a similar manner to that seen for polymerized serum-free light chains.
While the Hevylite® assay does have limitations with high concentration IgA BIM paraproteins, it may have added value in samples with hidden BIM gammopathy where a second smaller band is hidden on CZE. Kyle et al. demonstrated that only 18/57 patients with two paraproteins had multiple bands on cellulose acetate electrophoresis. More patients had one band by this technique and a second band only seen with immunofixation. They postulated that this was due to masking of the second band in the beta or gamma region by other serum proteins. 3 The use of Hevylite® may allow for true quantitation of the overall paraprotein burden without missing smaller monoclonal proteins, although the clinical significance of such small additional paraproteins is questionable.
In conclusion, it is demonstrated that the majority of apparent IgA BIM paraproteins are in fact multimers of single IgA clones, and that quantitation and reporting of these paraproteins can be improved by utilizing reducing agents such as DTT to resolve such paraproteins into one quantifiable band on CZE. DTT treatment allows these paraproteins to be quantified as one entity and may lead to more consistent and clear reporting of these results from the laboratory. DTT treatment of IgA ‘complex’ paraproteins would also reduce overestimation of paraprotein burden that is due to inclusion of background serum proteins. It is recognized, however, that the most important aspect of laboratory paraprotein reporting is consistency in reporting to enable changes over time to be monitored effectively.
Caution should be exercised if Hevylite® is used to quantitate IgA apparent BIM paraproteins due to their propensity to polymerize. This likely extends to IgM BIM paraproteins but has not been evaluated here. Where IgGκ BIM paraproteins are observed on CZE, the possibility of therapeutic monoclonal therapy should be considered before reporting the presence of a second paraprotein.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Hevylite® kits used in this project were funded by The Binding Site™; however, the Binding Site™ have had no input to this paper, or the poster and MSc dissertation associated with it.
Ethical approval
Not applicable.
Guarantor
HV.
Contributorship
HV and AD researched literature and conceived the study. HV performed laboratory experiments and data analysis. HV wrote the draft paper with review by AD.
