Abstract
Background
This study investigated the predictive potential of the albumin-to-alkaline phosphatase ratio (AAPR) for cerebrovascular accident (CVA) occurrence in patients with antineutrophil cytoplasmic antibody–associated vasculitis.
Methods
This study included 239 AAV patients and reviewed their medical records retrospectively. AAPR was calculated using the following formula: AAPR = serum albumin (g/dL)/serum alkaline phosphatase (IU/L). CVA was defined only as cerebral infarction after AAV diagnosis in this study. In patients with CVA and those without CVA, the follow-up duration based on CVA was defined as the period from AAV diagnosis to CVA occurrence and to the last visit day, respectively.
Results
The median age of 239 AAV patients (130 microscopic polyangiitis, 64 granulomatosis with polyangiitis, and 45 eosinophilic granulomatosis with polyangiitis) was 59.0 years and 32.6% were men. The median serum albumin and alkaline phosphatase levels, and AAPR were 3.7 g/dL, 70.5 IU/L and 0.051, respectively. Nineteen patients had CVA during the median follow-up duration of 34.8 months. Using the receiver operator characteristic curve analysis, the optimal cut-off of AAPR for CVA occurrence was obtained as ≤ 0.035. AAV patients with AAPR ≤ 0.035 showed a significantly higher risk of CVA occurrence after AAV diagnosis than those with AAPR >0.035 (relative risk 5.000, p < 0.001). In the multivariable Cox analysis, only AAPR ≤ 0.035 was independently associated with CVA occurrence among AAV patients (hazard ratio 3.195, 95% confidence interval 1.014, 10.062).
Conclusion
This study demonstrated the predictive potential of AAPR calculated at AAV diagnosis for CVA occurrence after AAV diagnosis among AAV patients.
Keywords
Introduction
Antineutrophil cytoplasmic antibody (ANCA)–associated vasculitis (AAV) is a small vessel vasculitis, which affects capillaries, arterioles, venules and occasionally medium-sized arteries. And according to the clinical characteristics, AAV is categorized into three subtypes; microscopic polyangiitis, granulomatosis with polyangiitis, and eosinophilic granulomatosis with polyangiitis.1,2 Central nervous system involvement of AAV is not a frequently observed systemic manifestation, and cerebrovascular accident (CVA) may occur in a minority of AAV patients. 3 As such, the low frequency of CVA involvement makes it difficult to predict and detect among AAV patients. However, given that CVA may lead to permanent brain dysfunction including cognitive impairment and sensorimotor loss, it is clinically important to discover a new index at the time of AAV diagnosis for predicting CVA occurrence in AAV patients.
The albumin-to-alkaline phosphatase ratio (AAPR) was first introduced to predict poor prognosis of cancers and was demonstrated to be inversely associated with mortality rates.4,5 Recently, in addition to the predictive potential for the poor prognosis of cancers, AAPR has been reported to be able to predict the poor prognosis of coronary artery disease by demonstrating that the alkaline phosphatase-to-albumin ratio, the inverse form of AAPR, was significantly associated with long-term adverse outcomes of coronary artery disease. 6 On the other hand, several studies have proposed an association between serum alkaline phosphatase levels and either cerebrovascular disease-related prognosis or small cerebral vascular diseases in healthy individuals.7–9 Although there is no direct evidence that AAPR could predict CVA occurrence, given that CVA occurrence in AAV patients might be closely related to small cerebral vascular diseases, and alkaline phosphatase is a parameter comprising the formula of AAPR, it could be reasonably assumed that AAPR calculated at AAV diagnosis could predict CVA occurrence after AAV diagnosis. However, to date, no studies have confirmed this assumption. Therefore, this study investigated the predictive potential of AAPR calculated at AAV diagnosis for CVA occurrence after AAV diagnosis.
Methods
Study subjects
The study subjects were selected from the Severance Hospital ANCA-associated VasculitidEs cohort, which is an observational cohort that includes patients diagnosed with AAV from 2000 to date. This study included AAV patients who fulfilled the inclusion criteria described in previous studies.10,11 Additionally, patients with the results of both serum albumin and alkaline phosphatase measured at AAV diagnosis were included. Finally, 239 patients with AAV were included in this study and their medical records were retrospectively reviewed.
The present study was approved by the institutional review board (IRB) of Severance Hospital (
Clinical data
Regarding variables at AAV diagnosis, age, sex, body mass index and smoking history were collected as demographic data. The AAV-specific variables included AAV subtype, ANCA type and AAV-assessing indices. Comorbidities and laboratory results, including erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP), were also collected. Regarding variables after AAV diagnosis, the frequency and duration of CVA occurrence were collected. In general, CVA refers to both ischaemic stroke and haemorrhage; 12 however, CVA was defined only as cerebral infarction in this study, as two cases of subdural haemorrhage were closely related to the administration of warfarin rather than AAV complications. In addition, cases of previously occurred CVA were not included in this study. For patients with CVA, the follow-up duration based on CVA was defined as the period from AAV diagnosis to CVA occurrence. Whereas, for patients without CVA, the follow-up duration based on CVA was defined as that from AAV diagnosis to the last visit. There were no patients who were actively smoking from AAV diagnosis and during follow-up based on CVA. The number of patients who were administered glucocorticoids and immunosuppressive drugs was recorded.
The measurement of serum albumin level and alkaline phosphate activity
Serum albumin level and alkaline phosphatase activity were measured by using Atellica CH 930 Analyzer (Simens Health care Diagnostics, Marburg, Germany) in our hospital. Bromocresol green was used to measure serum albumin level. Inter-assay coefficients of variation for albumin were 2.4% at 2.1 g/dL, 2.0% at 3.4 g/dL and 1.6% at 5.1 g/dL. 2-amino-2-methyl-1-propanol buffer and para-nitrophenyl phosphate were used to estimate serum alkaline phosphatase activity. Inter-assay coefficients of variation for alkaline phosphatase activity were 1.3% at 87 IU/L, 0.7% at 277 IU/L and 1.5% at 841 IU/L.
Formula for calculating AAPR
Albumin-to-alkaline phosphatase ratio was calculated using the following formula: AAPR = serum albumin (g/dL)/serum alkaline phosphatase (IU/L). 4
Statistical analyses
All statistical analyses were performed using IBM SPSS Statistics for Windows, version 26 (IBM Corp., Armonk, NY, USA). Continuous variables are expressed as medians with interquartile ranges, whereas categorical variables are expressed as numbers (percentages). The optimal cut-off was extrapolated by performing the receiver operator characteristic (ROC) curve analysis and one value having the maximized sum of sensitivity and specificity was selected. The relative risk (RR) of the cut-off for the high activity of AAV was analysed using contingency tables and the chi-square test. The multivariable Cox hazard model using variables with statistical significance in the univariable Cox hazard model was conducted to appropriately obtain the hazard ratios (HRs) during the considerable follow-up duration. Serum albumin and alkaline phosphatase were not included in the multivariable Cox analysis because they are the parameters comprising the formula of AAPR. Statistical significance was set at p < 0.05.
Results
Characteristics
Characteristics of AAV patients (N = 239).
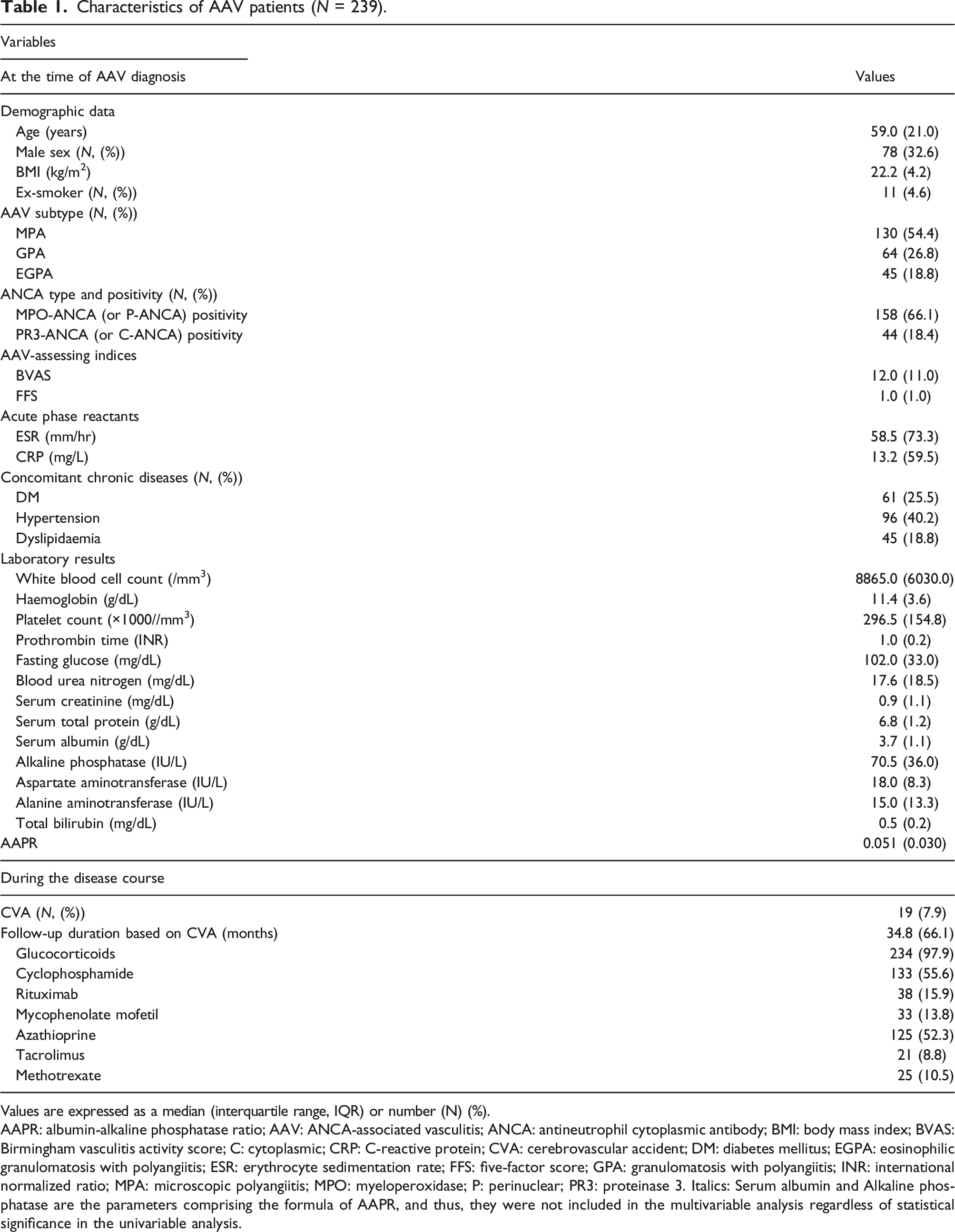
Values are expressed as a median (interquartile range, IQR) or number (N) (%).
AAPR: albumin-alkaline phosphatase ratio; AAV: ANCA-associated vasculitis; ANCA: antineutrophil cytoplasmic antibody; BMI: body mass index; BVAS: Birmingham vasculitis activity score; C: cytoplasmic; CRP: C-reactive protein; CVA: cerebrovascular accident; DM: diabetes mellitus; EGPA: eosinophilic granulomatosis with polyangiitis; ESR: erythrocyte sedimentation rate; FFS: five-factor score; GPA: granulomatosis with polyangiitis; INR: international normalized ratio; MPA: microscopic polyangiitis; MPO: myeloperoxidase; P: perinuclear; PR3: proteinase 3. Italics: Serum albumin and Alkaline phosphatase are the parameters comprising the formula of AAPR, and thus, they were not included in the multivariable analysis regardless of statistical significance in the univariable analysis.
Optimal cut-off of AAPR for CVA
Because AAPR calculated at AAV diagnosis was negatively associated with CVA occurrence after AAV diagnosis, we set the dependent variable in the ROC curve analysis as the absence of CVA to obtain an area under the curve (AUC) of more than 0.5. Using the ROC curve analysis, the optimal cut-off of AAPR for CVA occurrence was obtained as ≤ 0.035 (AUC 0.683, 95% confidence interval [CI] 0.540, 0.826), and the sensitivity and specificity were 82.5% and 52.6%, respectively (Figure 1(a)). Cut-off of AAPR and its relative risk for CVA occurrence. (a) Using the receiver operator characteristic curve analysis, the optimal cut-off of AAPR for CVA occurrence was obtained as ≤ 0.035. (b) AAV patients with AAPR ≤ 0.035 showed a significantly higher risk of CVA occurrence after AAV diagnosis than those with AAPR >0.035. AAPR: albumin-to-alkaline phosphatase ratio; CVA: cerebrovascular accident; AAV: antineutrophil cytoplasmic antibody–associated vasculitis.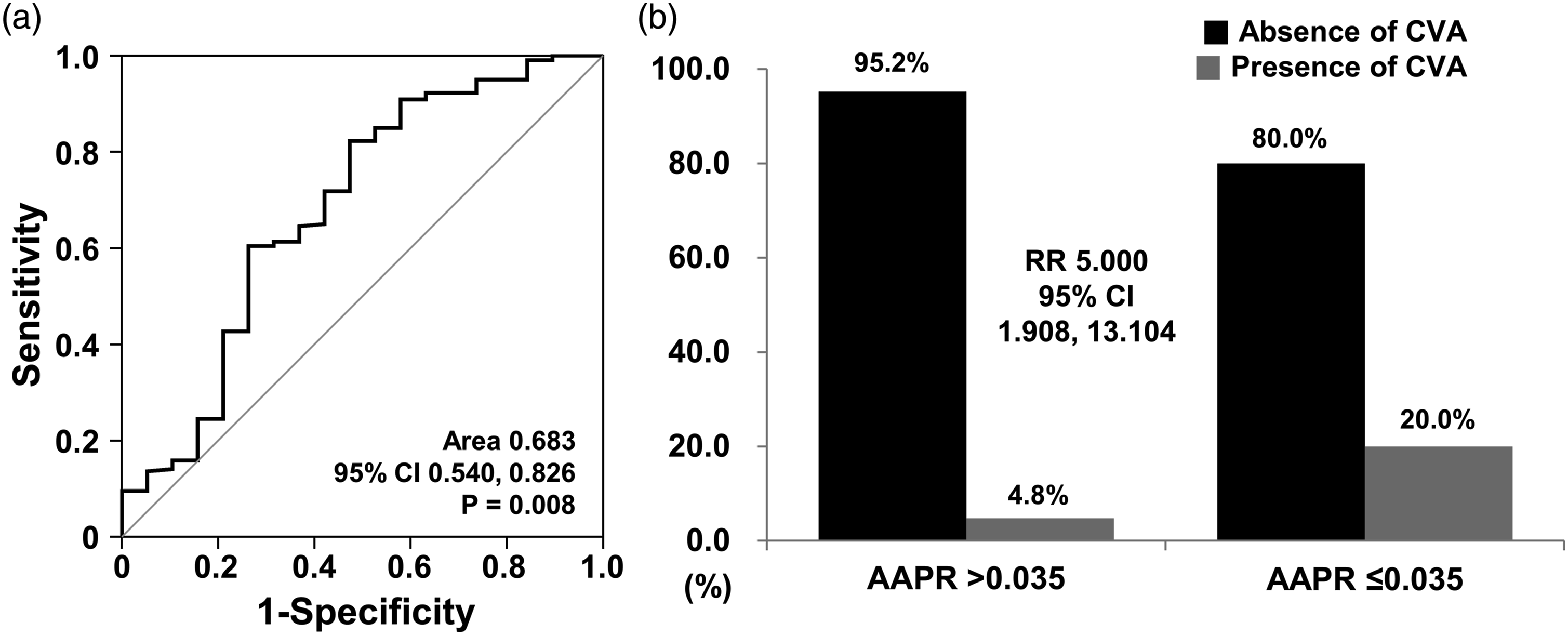
RR analysis
When AAV patients were divided into two groups according to AAPR ≤ 0.035, CVA occurrence after AAV diagnosis was identified more common in AAV patients with AAPR ≤ 0.035 than in those with AAPR >0.035 (20.0% vs. 4.8%, p < 0.001). Furthermore, AAV patients with AAPR ≤ 0.035 showed a significantly higher risk of CVA occurrence after AAV diagnosis than those with AAPR >0.035 (RR 5.000, 95% CI 1.908, 13.104) (Figure 1(b)).
Cox hazards model analyses
Cox hazards model analysis of variables at the time of AAV diagnosis for CVA during the disease course in AAV patients.
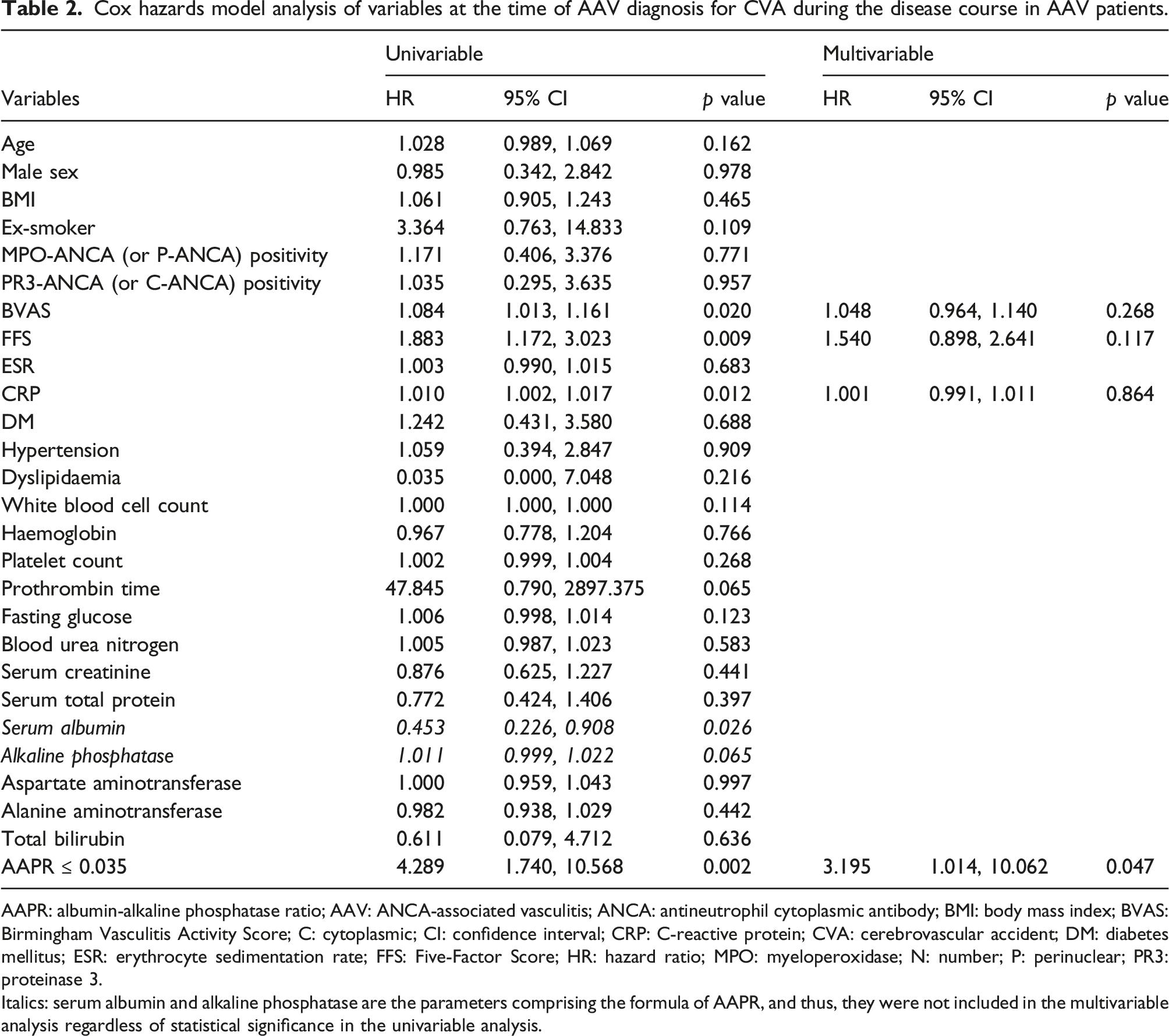
AAPR: albumin-alkaline phosphatase ratio; AAV: ANCA-associated vasculitis; ANCA: antineutrophil cytoplasmic antibody; BMI: body mass index; BVAS: Birmingham Vasculitis Activity Score; C: cytoplasmic; CI: confidence interval; CRP: C-reactive protein; CVA: cerebrovascular accident; DM: diabetes mellitus; ESR: erythrocyte sedimentation rate; FFS: Five-Factor Score; HR: hazard ratio; MPO: myeloperoxidase; N: number; P: perinuclear; PR3: proteinase 3.
Italics: serum albumin and alkaline phosphatase are the parameters comprising the formula of AAPR, and thus, they were not included in the multivariable analysis regardless of statistical significance in the univariable analysis.
Discussion
This study investigated the predictive potential of AAPR calculated at AAV diagnosis for CVA occurrence after AAV diagnosis among AAV patients. Among the 239 patients, 19 had CVA during the median follow-up duration of 34.8 months. When the cut-off of AAPR calculated at AAV diagnosis was set at ≤ 0.035, AAV patients with AAPR ≤ 0.035 showed a significantly higher risk of CVA occurrence than those with AAPR >0.035 (RR 5.000). In the multivariable Cox analysis with BVAS, FFS, CRP and AAPR ≤ 0.035, only AAPR ≤ 0.035 was independently associated with CVA occurrence after AAV diagnosis among AAV patients. Therefore, we concluded that this study demonstrated the predictive potential of AAPR calculated at AAV diagnosis for CVA occurrence after AAV diagnosis among AAV patients.
In terms of serum albumin, no study clearly demonstrating the direct link between serum albumin level and CVA occurrence has been reported to date; however, it can be inferred that serum albumin level might be associated with CVA by three facts: first, serum albumin, which was known to modulate inflammation and reduce oxidative stress, is a well-known index inversely reflecting the inflammatory burden in critical illness;13,14 second, Inflammation is a risk factor for stroke and small vessel diseases 15 ; and third, serum albumin level in patients with acute cerebral infarction (ischaemic stroke) is inversely associated with the risk of poor outcomes.16,17
In terms of serum alkaline phosphatase, high serum alkaline phosphatase levels have been reported to be a significant biomarker for stroke and significantly increase the risk of stroke.18,19 Based on the results of the previous studies, two conclusions can be drawn. First, low serum albumin levels can predict CVA occurrence by reflecting a high burden of inflammation which may subsequently be associated with CVA occurrence. Second, high serum alkaline phosphatase levels play a role as a biomarker for CVA occurrence. Since AAPR is calculated using the formula where serum alkaline phosphatase is the denominator and serum albumin is the numerator, AAPR may be inversely associated with CVA occurrence and can independently predict CVA occurrence in AAV patients.
When the AUCs of the three variables (AAPR, serum albumin and alkaline phosphatase levels) were compared using the ROC curve analysis, the AUC of serum alkaline phosphatase levels was not statistically significant. In contrast, the AUC of serum albumin (0.694, p = 0.005) was larger than that of AAPR (0.683, p = 0.008). If so, what is the advantage of AAPR compared to serum albumin in predicting CVA occurrence in AAV patients? AAPR is considered more clinically meaningful than serum albumin alone. One reason is that AAPR is more stable than the single variable of serum albumin because it is composed of two variables. Second is that AAPR can further amplify its predictive potential because it places serum albumin and alkaline phosphatase as the denominator and numerator, respectively.
The advantage of this study is that it is the first to demonstrate that AAPR calculated at AAV diagnosis could predict CVA occurrence after AAV diagnosis among AAV patients. Given that the biomarkers or predictive indices for CVA occurrence in AAV patients are not widely known, the results of this study might have clinical implications in that AAPR can be helpful in actual clinical practice. However, this study had several limitations. The critical limitations were the retrospective and single-centre study design. First, due to the retrospective study, it was difficult to control the confounding variables at AAV diagnosis. In particular, although the association between CVA and smoking doses (pack-years) is more important clinically than that between CVA and smoking history (yes or not), since the clinical research forms included only an item of smoking history but not smoking doses, we could not retrospectively collect and calculate smoking doses from the study subjects. Second, due to the small number of patients regardless of CVA occurrence, it might be uneasy to generalize the results of this study to all AAV patients. Also, the 95% CI for the RR of the cut-off of AAPR for the presence of CVA was not statistically powerful. If the sample size is increased in the future and significance is maintained, the lowest value of the 95% CI may be expected to increase up to 2. Third, there was a previous study result that when albumin is measured using bromocresol green assay, it is measured higher in inflammatory conditions. 20 However, it was found that AAPR calculated with albumin measured by bromocresol green assay has an association with CVA occurrence, so it is thought that it can be used clinically. When measuring albumin by a method other than the bromocresol green assay, it may be necessary to reset the AAPR cut-off value associated with CVA occurrence. To overcome these limitations, a prospective nationwide multicentre study should be conducted.
Conclusion
This study demonstrated the predictive potential of AAPR calculated at AAV diagnosis for CVA occurrence after AAV diagnosis among AAV patients. In hospitals that measure albumin with the bromocresol green assay, AAPR can be calculated at the time of AAV diagnosis to predict the possibility of CVA.
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Korea Health Technology R&D Project through the Korea Health Industry Development Institute, funded by the Ministry of Health and Welfare (
Ethical approval
The present study was approved by the institutional review board (IRB) of Severance Hospital. Given the retrospective design of the study and the use of anonymised patient data, the requirement for written informed consent was waived by the IRB.
Guarantor
S-WL.
Contributorship
JWH, JYP, SSA and S-WL designed data collection tools, monitored data collection, wrote the statistical analysis plan, cleaned and analysed the data, and drafted and revised the paper. JWH, JJS, Y-BP and S-WL drafted and revised the paper.
