Abstract
Background
Effective prognostic markers are needed for antineutrophil cytoplasmic antibody-associated vasculitis (AAV). This study evaluated the clinical associations of serum vascular endothelial growth factor-A (sVEGF-A) and sVEGF-A165b (an antiangiogenic isoform of VEGF-A) concentrations with time to remission of AAV in a nationwide Japanese prospective follow-up cohort.
Methods
We collected samples from patients with AAV who were enrolled in the nationwide Japanese cohort study (RemIT-JAV-RPGN). We measured sVEGF-A and sVEGF-A165b concentrations using enzyme-linked immunosorbent assays in 57 serum samples collected 6 months before and after initiation of AAV treatment. Patients were classified based on AAV disease subtypes: microscopic polyangiitis, granulomatosis with polyangiitis and eosinophilic granulomatosis with polyangiitis (EGPA).
Results
Results revealed significant reductions in sVEGF-A and sVEGF-A165b concentrations in patients with microscopic polyangiitis and EGPA, respectively. However, despite the comparable concentrations of sVEGF-A and sVEGF-A165b during the 6 months of treatment in granulomatosis with polyangiitis patients, correlation analysis revealed that the differences in log2-transformed concentrations of sVEGF-A and sVEGF-A165b were inversely correlated with time to remission in granulomatosis with polyangiitis patients.
Conclusion
These results suggest that sVEGF-A and -A165b can serve as potential markers of time to remission in patients with granulomatosis with polyangiitis.
Keywords
Introduction
Antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis (AAV) comprises three distinct diseases: microscopic polyangiitis (MPA), granulomatosis with polyangiitis (GPA) and eosinophilic granulomatosis with polyangiitis (EGPA).1,2 AAV is characterized by pauci-immune necrotizing inflammation of small-to-medium-sized vessels and affects various organs. ANCA-associated activation of leukocytes and capillary endothelium has been implicated in the pathogenesis of vasculitis. 3 If remission induction therapy can be predicted early in the therapy, meticulous follow-up or application of additional regimens to the treatment could improve the patient outcome. Traditional acute-phase indicators, including C-reactive protein (CRP), have failed to predict the time to remission of AAV. 4 Researchers have reported candidate genes and serum IL-6 (sIL-6) as prognostic markers of AAV remission.5,6 However, few prognostic markers of AAV remission are known. Therefore, additional markers are needed to guide clinical management.
Angiogenesis refers to the formation and maintenance of blood vessels. A variety of signalling molecules are involved in the regulation of angiogenesis, including vascular endothelial growth factor-A (VEGF-A), an essential factor in both physiological and pathological angiogenesis. 7 VEGF-A binds to VEGF receptor-2 (VEGFR-2), thereby inducing receptor phosphorylation and the propagation of signalling cascades that regulate endothelial survival, proliferation and motility. 8 In contrast, alternative splicing of exon 8 in VEGF-A mRNA, at a site known as the distal splice site, has been identified; this results in a new family of VEGF-A isoforms, termed VEGF-Axxxb. 9 VEGF-A165b interacts with VEGFR-2 and VEGF-A165 with the same affinity. However, the altered C-terminal sequence causes insufficient activation of VEGFR-2, owing to its inability to bind to the co-receptor neuropilin-1, 10 resulting in an antiangiogenic activity. Recently, several studies have demonstrated the association of VEGF-A165b with some diseases, including obesity, 11 peripheral artery disease,12–14 coronary artery disease15–17 and chronic kidney disease. 18 Correlations of VEGF-A concentrations with serum CRP (sCRP) and anti-myeloperoxidase (MPO) titres have been demonstrated; these suggest that VEGF-A is a potential marker for monitoring disease activity in anti-MPO-associated vasculitis. 19 However, the relationship between VEGF-A and VEGF-A165b concentrations and patient prognosis in longitudinal samples is unknown.
The purpose of this study was to elucidate whether the concentrations of serum VEGF-A (sVEGF-A) and sVEGF-A165b were correlated with time to remission of AAV in a nationwide Japanese prospective cohort study.
Materials and methods
Study subjects
Serum samples were obtained from patients with AAV who were enrolled in the Remission Induction Therapy in Japanese Patients with AAV and rapidly progressive glomerulonephritis (RemIT-JAV-RPGN) cohort study (n = 57). 20 The diagnosis of AAV was based on the criteria for primary systemic vasculitis proposed by the European Medicines Agency (EMEA) algorithm. 21 Out of 321 patients who had been initially enrolled in RemIT-JAV-RPGN, serum samples were collected from 172 patients at diagnosis, and 57 patients after 6 months of treatment. AAV disease subtype of each patient was classified based on the EMEA algorithm as MPA (100 patients), GPA (25 patients), EGPA (21 patients) and unclassifiable disease (26 patients). The clinical and laboratory data for the RemIT-JAV-RPGN study population were extracted from the database. 20 We analysed paired serum samples obtained at 0 and 6 months from 57 patients (30 MPA, 9 GPA, 8 EGPA and 10 unclassifiable diseases). Samples were frozen at –80°C until use. The research complied with the Declaration of Helsinki and the study protocol was approved by the ethics committee of the Nagoya University School of Medicine (No. 2017-0304). All patients in the RemIT-JAV-RPGN study provided written informed consent.
Outcome measures
Details of the RemIT-JAV-RPGN study protocol have been reported previously. 20 Organ involvement was evaluated in accordance with the Birmingham Vasculitis Activity Score version 3 (BVAS) system. Remission was defined as a BVAS score of 0 on two consecutive occasions at least 1 month apart, according to the European League Against Rheumatism recommendations. 22
sVEGF-A and sVEGF-A165b measurements
sVEGF-A concentrations were measured using the Human VEGF Quantikine ELISA Kit (DVE00, R&D Systems, Minneapolis, MN, USA) according to the manufacturer’s instructions. The limit of detection was 9 pg/mL. Intra- and interassay coefficients of variation were 4.5% and 7.0%, respectively. VEGF-A ELISA Kit does not discriminate between pro- and antiangiogenic isoforms of VEGF-A. sVEGF-A165b concentrations were measured using the Human Vascular Endothelial Growth Factor-165b ELISA Kit (MBS720132, MyBioSource, San Diego, CA, USA) according to the manufacturer’s instructions. The limit of detection was 1 pg/mL. Intra- and interassay coefficients of variation were both <10%.
Statistical analyses
Data are expressed as medians and interquartile ranges, or n (%). We compared continuous variables between 0 and 6 months using the Wilcoxon signed-rank test. We assessed correlations between time to remission in AAV patients and differences in log2-transformed clinical parameters before and after remission induction therapy. Two AAV patients were excluded from the correlation analysis of the differences in the clinical parameters and time to remission (one patient who died of infectious disease and another one who did not ingratiate the remission definition). The correlations were assessed by Pearson correlation analysis. All P values were two-tailed and P values < 0.05 were considered to indicate statistical significance. All statistical analyses were performed using SAS software, version 9.4 (SAS Institute Inc.) and Prism 6 software (GraphPad Software, San Diego, CA, USA).
Results
Clinical characteristics of patients with AAV at diagnosis and following treatment
The clinical characteristics of the study population are summarized in Table 1. Baseline sVEGF-A and sVEGF-A165b concentrations showed no significant difference between AAV subtypes (data not shown). We observed significant decreases in white blood cell (WBC) counts (P = 0.016) and sCRP (P < 0.001), sVEGF-A (P < 0.001) and sVEGF-A165b (P = 0.002) concentrations after 6 months of treatment in AAV patients (Table 1 and Figure 1). For patients with MPA, concentrations of sCRP and serum creatinine were significantly reduced (P < 0.001), and neutrophil counts were significantly increased after 6 months of treatment (P = 0.030). For patients with GPA or unclassifiable disease, concentrations of sCRP were significantly reduced (P = 0.004 and P = 0.004, respectively). For patients with EGPA, WBC counts and sCRP concentrations were significantly reduced (P = 0.008 and P = 0.008, respectively), while neutrophil counts were significantly increased (P = 0.008). For patients with MPA, sVEGF-A concentrations were significantly reduced (P < 0.001), whereas sVEGF-A165b concentrations were significantly reduced in patients with EPGA (P = 0.023) (Figure 1(c) and (h)).
Patient characteristics and circulatory parameters at 0 and 6 months in identical AAV patients from the RemIT-JAV-RPGN cohort.
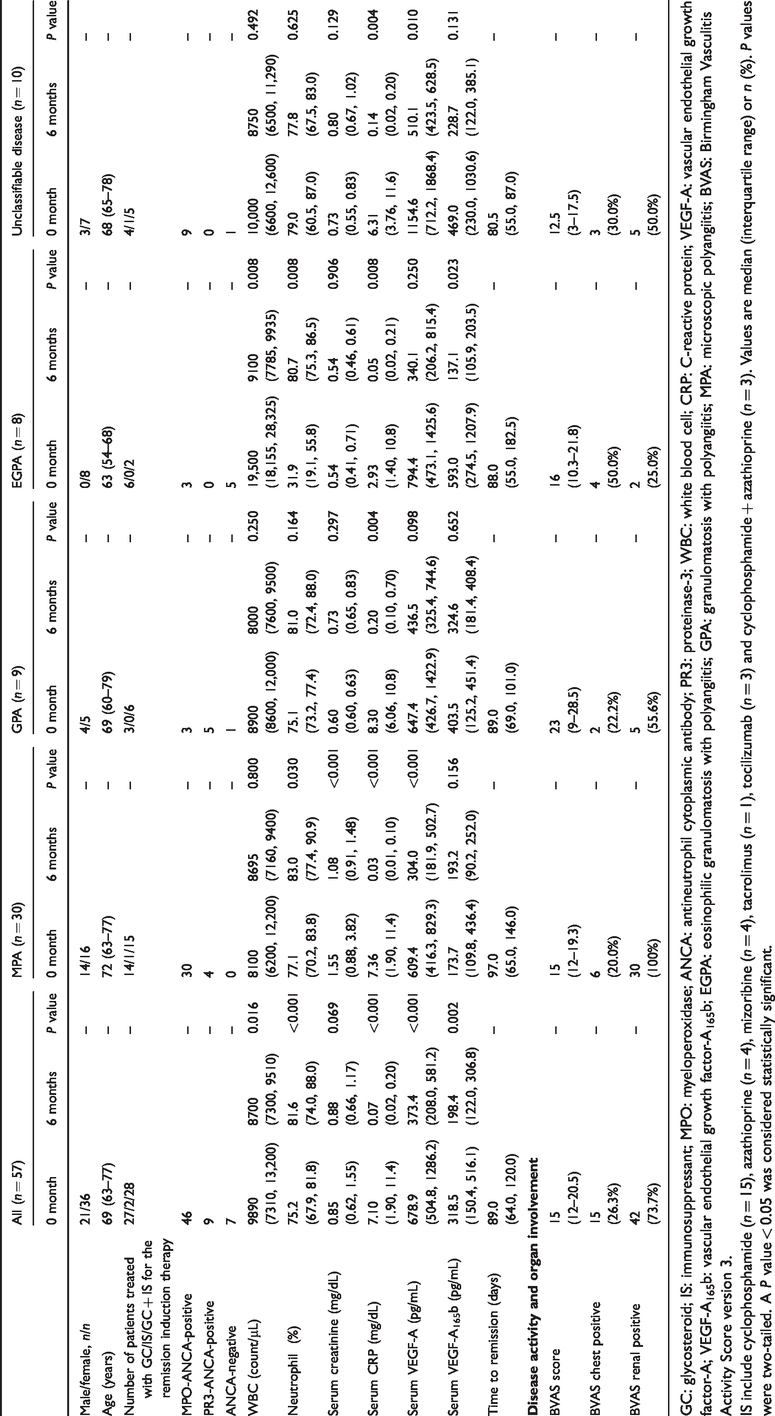
GC: glycosteroid; IS: immunosuppressant; MPO: myeloperoxidase; ANCA: antineutrophil cytoplasmic antibody; PR3: proteinase-3; WBC: white blood cell; CRP: C-reactive protein; VEGF-A: vascular endothelial growth factor-A; VEGF-A165b: vascular endothelial growth factor-A165b; EGPA: eosinophilic granulomatosis with polyangiitis; GPA: granulomatosis with polyangiitis; MPA: microscopic polyangiitis; BVAS: Birmingham Vasculitis Activity Score version 3.
IS include cyclophosphamide (n = 15), azathioprine (n = 4), mizoribine (n = 4), tacrolimus (n = 1), tocilizumab (n = 3) and cyclophosphamide + azathioprine (n = 3). Values are median (interquartile range) or n (%). P values were two-tailed. A P value < 0.05 was considered statistically significant.
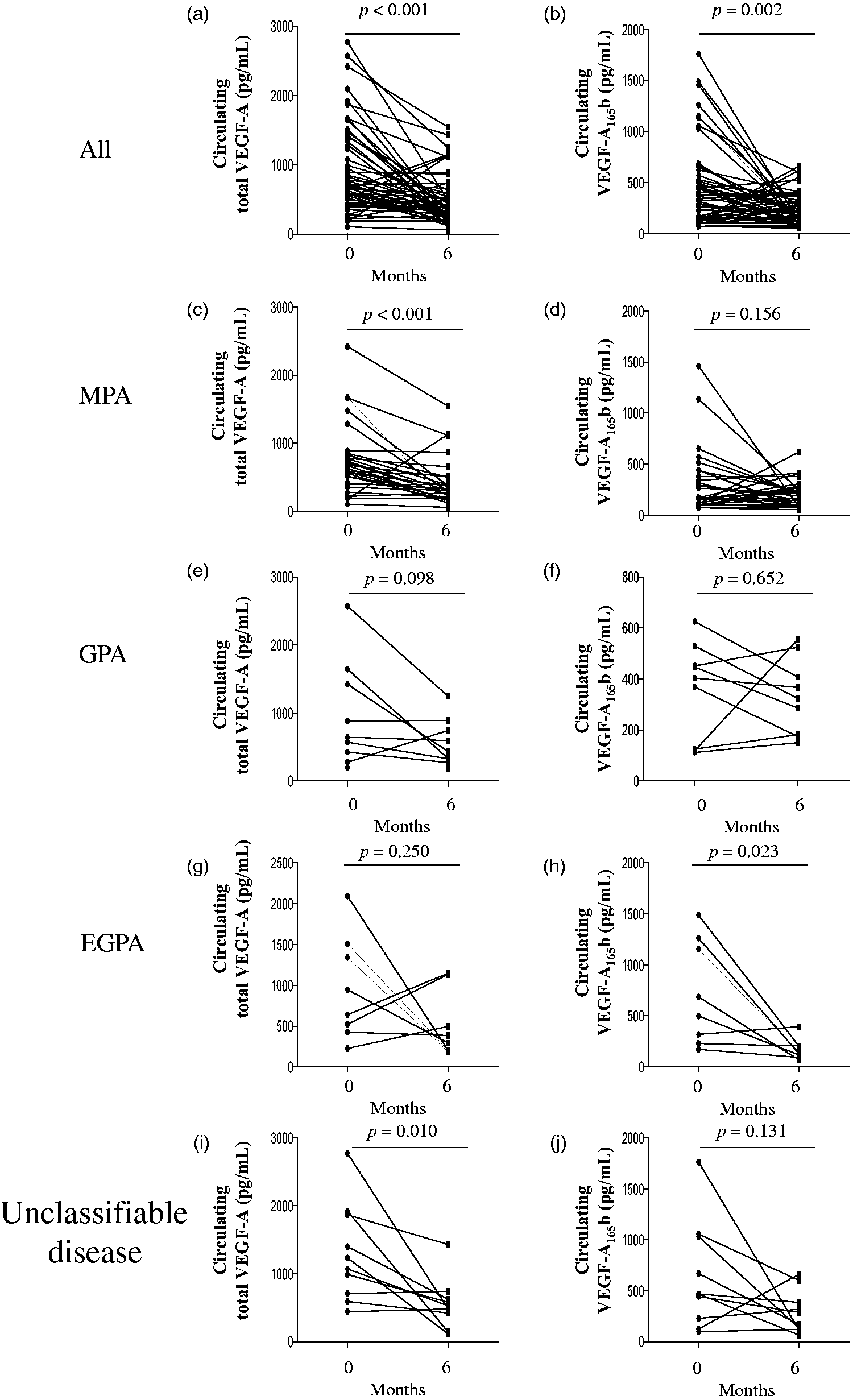
sVEGF-A and sVEGF-A165b concentrations at diagnosis and 6 months after treatment. sVEGF-A and sVEGF-A165b concentrations were measured in paired serum samples collected at diagnosis (0 month) and 6 months after the start of treatment, from 57 patients with AAV (all; a and b). Thirty patients were diagnosed with MPA (c and d), nine with GPA (e and f), eight with EGPA (g and h) and 10 with unclassifiable disease (i and j). Lines connect data obtained from each patient. AAV: ANCA-associated vasculitis; EGPA: eosinophilic granulomatosis with polyangiitis; VEGF-A: vascular endothelial growth factor-A; VEGF-A165b: vascular endothelial growth factor-A165b; sVEGF-A: serum concentration of total VEGF-A; sVEGF-A165b: serum concentration of VEGF-A165b.
Correlation between differences in log2-transformed clinical parameters before and after remission induction therapy and time to remission in AAV patients
Correlation analysis of the differences in log2-transformed clinical parameters from 0 to 6 months and time to remission of AAV is summarized in Table 2. Neither WBC counts nor sCRP concentrations were correlated with time to remission in any AAV subtype. Neutrophil counts were inversely correlated with time to remission in patients with unclassifiable diseases (r = −0.72, P = 0.030), but no correlation was observed for the other AAV subtypes. Serum creatinine concentrations were positively correlated with time to remission in patients with EGPA (r = 0.82, P = 0.013), but no correlations with other subtypes were observed. Interestingly, sVEGF-A and sVEGF-A165b were significantly and inversely correlated with time to remission only in patients with GPA (r = −0.67, P = 0.046, and r = −0.78, P = 0.014, respectively), but not in patients with other AAV subtypes.
Correlation analysis of the differences in log2-transformed clinical parameters from 0 to 6 months, and time to remission of AAV patients from the RemIT-JAV-RPGN cohort.
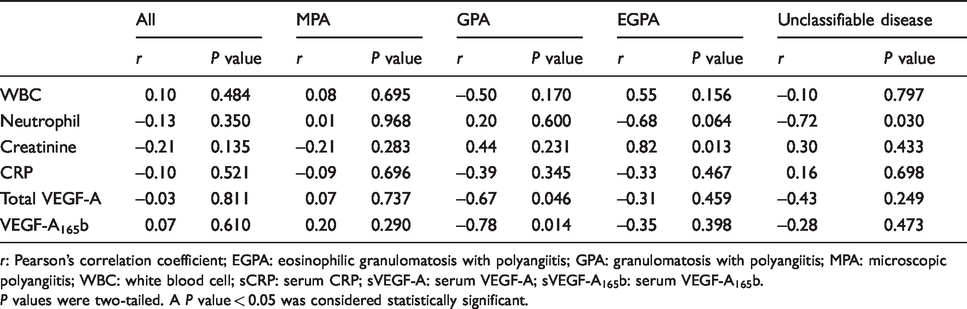
r: Pearson’s correlation coefficient; EGPA: eosinophilic granulomatosis with polyangiitis; GPA: granulomatosis with polyangiitis; MPA: microscopic polyangiitis; WBC: white blood cell; sCRP: serum CRP; sVEGF-A: serum VEGF-A; sVEGF-A165b: serum VEGF-A165b.
P values were two-tailed. A P value < 0.05 was considered statistically significant.
Discussion
In the current study, we demonstrated for the first time that concentrations of sVEGF-A and sVEGF-A165b decreased significantly in response to 6 months of treatment in patients with AAV in the RemIT-JAV-RPGN cohort. Significant reductions in sVEGF-A and sVEGF-A165b concentrations in patients with MPA and EGPA following the treatment were observed. At 6 months, sVEGF-A and sVEGF-A165b concentrations did not significantly differ compared with concentrations observed at 0 month in patients with GPA, although changes in sVEGF-A and sVEGF-A165b concentrations inversely correlated with time to remission.
VEGF-A reportedly participates in the inflammatory process and is associated with disease severity in rheumatoid arthritis (RA). 23 In patients with RA and osteoarthritis who experience clinical remission, wingless-related integration site 5a (Wnt5a) is increased in the synovium. 24 Wnt5a can promote inflammation-driven VEGF-A165b expression, which has been shown to impair vascular collateralization in murine hindlimb ischaemic models. 12 Little is known about the role of VEGF-A165b in human disease, although it appears to be expressed ubiquitously in different tissues, including the eyes, muscle, vascular endothelium and skin.25–27 Therapeutic modulation of VEGF-A165b is currently being investigated in cancer and angiogenic eye disorders and is being considered to be developed for treating the autoimmune disorder, systemic sclerosis.28,29 Besides, several reports have described the involvement of VEGF-A in systemic vasculitis, such as GPA (previously known as Wegener’s granulomatosis), 30 giant cell arteritis 31 and Kawasaki disease. 32 Studies have shown the clinical value of VEGF-A in monitoring disease activity in vasculitis, including MPA.19,33 Shulman et al. 34 reported that VEGF-A expression is altered in many glomerular diseases. Nitta et al. 35 reported that VEGF-A concentrations increased in human RPGN, which is a clinical manifestation of AAV-related renal involvement.
sVEGF-A and sVEGF-A165b may be important for the diagnosis and monitoring of AAV and/or AAV-related renal involvement. Corticosteroids and cyclophosphamide can inhibit VEGF-A production by pulmonary cells36,37 and leukocytes, 38 respectively. In the current study, significant reductions in sVEGF-A and sVEGF-A165b concentrations in patients with MPA and EGPA, respectively, were observed. This result may be due to immunosuppressive therapy, as cyclophosphamide or rituximab were incorporated into glucocorticoid therapy, improving the survival of patients with AAV.39,40 Literature indicates that VEGF-A in the bloodstream is transported by blood cells, including leukocytes and platelets, and that platelet count and sIL-6 correlate with sVEGF-A. 41 Therefore, changes in VEGF-A or VEGF-A165b of MPA and EGPA during immunosuppressive therapy might be related to immunosuppressant-induced leukopenia, neutropenia and eosinopenia. Human eosinophils constitutively express messenger RNA encoding VEGF-A and also store VEGF-A, and eosinophils release VEGF-A following stimulation with granulocyte-macrophage colony-stimulating factor or interleukin-5. 42 VEGF-A released from eosinophils might contribute to the recruitment of inflammatory cells, including T-cells and eosinophils, by increasing vascular permeability, during the development of EGPA. Interestingly, sVEGF-A concentrations did not change beyond the 6-month treatment period in EGPA. Glucocorticoid is known to promote apoptosis of eosinophils and inhibit apoptosis of neutrophils. 43 Therefore, we assume that the massive release of VEGF-A from the apoptotic eosinophils might explain the high VEGF-A concentrations 6 months after the start of therapy in EGPA. In contrast, sVEGF-A165b was significantly reduced after 6 months of treatment in EGPA. The source of circulating VEGF-A165b in EGPA is not apparent from the current study. However, our data may provide evidence against the notion that immunosuppressants are the major inhibitors of sVEGF-A165b in EGPA.
We found that sVEGF-A concentrations did not significantly differ in patients with GPA before and after treatment, although the change in sVEGF-A from 0 to 6 months was inversely correlated with time to remission of GPA. We observed similar results for sVEGF-A165b concentrations in GPA. GPA is a rare systemic autoimmune disease of unknown aetiology that is characterized by granulomatous inflammation, tissue necrosis and vasculitis in small- and medium-sized vessels. 44 A recent study demonstrated that a subpopulation of granuloma macrophages produces VEGF-A, which recruits immune cells to the granuloma by a non-angiogenic pathway. 45 Moreover, selective blockade of VEGF-A in myeloid cells showed that granuloma macrophage-derived VEGF-A regulates granulomatous inflammation. Hence, it was suggested that the severity of granuloma-related inflammation can be ameliorated by the inhibition of VEGF-A. Our current results from patients with GPA revealed disassociations between changes in sVEGF-A and sVEGF-A165b during treatment and inverse associations between sVEGF-A and sVEGF-A165b concentrations and time to remission of GPA. In other words, the less change in sVEGF-A165b before and after treatment hastened the time to remission. The sVEGF-A165b, which is an antiangiogenic isoform of VEGF-A, may improve the granuloma-related inflammation of GPA and may result in a shorter time to remission. Therefore, it is assumed that endogenous VEGF-A165b is cytoprotective for endothelial, epithelial and neuronal cells in pathological conditions, explaining the unpredicted anti-VEGF-A side effects. These results may provide insights into the differential roles of two VEGF-A-related molecules into GPA pathogenesis. However, this result should be considered preliminary and further validation is warranted.
Our study revealed the specificities of sVEGF-A and sVEGF-A165b for AAV disease subtypes. The action of VEGF-A depends not only on its total amount but also on the local environment. Thus, different responses to induction therapy among AAV types may result in different responses to VEGF-A, which is involved in time to remission.
The main strength of this study is the analysis of the RemIT-JAV-RPGN cohort of patients, a nationwide Japanese prospective follow-up cohort study of AAV. Patients in the RemIT-JAV-RPGN cohort received remission induction therapy according to the Japanese MULWA Guidelines of Treatment for AAV reflected in the JMAAV protocol.20,46 Nonetheless, a limitation of this study is that changes in marker concentrations during mild relapses could not be assessed. Further, the study was not designed to predict future relapses or distinguish AAV from other diseases that may influence marker concentrations. Moreover, blood samples were collected at 0 and 6 months in the RemIT-JAV-RPGN study cohort. Therefore, we could not show the data at prior to 6 months.
Conclusion
These results suggest that sVEGF-A and VEGF-A165b can serve as potential markers of time to remission in patients with granulomatosis with polyangiitis.
Footnotes
Data statement
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Acknowledgements
We express our sincere gratitude to Keiko Hongo, Tomomi Maruyama, Eri Katsuyama, Takayuki Katsuyama, Mariko Narazaki, Noriko Toyota-Tatebe and Kouichi Sugiyama for their valuable assistance in data management and to the patients and physicians who participated in RemIT-JAV and RemIT-JAV-RPGN (see ![]() ).
).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Grant-in-Aid for Young Scientists (Grant Number 18K15386), Japan Foundation for Applied Enzymology and Kurozumi Medical Foundation, to R. Kikuchi. The funding organization played no role in the study design; in the collection, analysis and interpretation of data; in the writing of the report; or in the decision to submit the report for publication.
Ethical approval
The ethics committee of the Nagoya University School of Medicine approved this study (No. 2017-0304).
Guarantor
RK.
Contributorship
RK, NT, K-ES, YY, SM, ToM, TaM, and Research Committee of Intractable Vasculitis Syndrome and Research Committee of Intractable Renal Disease of the Ministry of Health (KM, TA, YT, SI, HH, HD, TI, HM, SM), Labour and Welfare of Japan, designed the study. RK and AS performed the laboratory work. RK and MN performed the statistical analysis. RK and MN prepared the figures. RK and NT wrote the manuscript. All the authors have accepted responsibility for the entire content of this submitted manuscript and have approved submission.
Appendix 1
The Japan Research Committee of Intractable Vasculitis (JPVAS), and Research Committee of Intractable Renal Disease of the Ministry of Health, Labour, and Welfare, Japan. In addition to the authors, the following investigators and institutions participated in this study: Department of Respiratory Medicine, Toho University Omori Medical Center (Sakae Homma); Hamamatsu University School of Medicine (Noriyoshi Ogawa); Department of Nephrology, Faculty of Medicine, University of Tsukuba (Kunihiro Yamagata); Department of Medicine, Kidney Center, Tokyo Women’s Medical University (Kosaku Nitta); Tokyo Medical University Ibaraki Medical Center (Masaki Kobayashi); Nephrology and Rheumatology, First Department of Internal Medicine, Kyorin University School of Medicine and Kichijoji Asahi Hospital (Yoshihiro Arimura); Department of Pharmacovigilance and Department of Medicine and Rheumatology, Graduate School of Medical and Dental Sciences, Tokyo Medical and Dental University (Masayoshi Harigai); Department of Rheumatology, Shimane University Faculty of Medicine (Yohko Murakawa); Division of Nephrology, Tokyo Medical University Hachioji Medical Center (Masaharu Yoshida); Department of Nephrology, School of Medicine, Fujita Health University (Yukio Yuzawa); Kurobe City Hospital (Shinichi Takeda); and Division of Rheumatology, Department of Internal Medicine, Keio University School of Medicine (Tsutomu Takeuchi).
