Abstract
Creatinine-based estimated glomerular filtration rate equations (eGFRcreatinine) are used to measure excretory kidney function in clinical practice. Despite inter and intra-patient variability, eGFRcreatinine has excellent clinical utility and provides the basis for the classification system for chronic kidney disease (CKD), for kidney function monitoring, treatment interventions and referral pathways. The 4-variable modification of diet in renal disease (MDRD) eGFRcreatinine equation was introduced in 2000 and recommended by the National Institute for Health and Care Excellence (NICE) in 2008. Subsequently, the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) eGFRcreatinine equation was introduced in 2009 and is more accurate than MDRD in patients with mild and moderate CKD. In 2014, NICE recommended that CKD-EPI eGFRcreatinine replace MDRD eGFRcreatinine in routine clinical practice across England. Both equations originally incorporated adjustments for age, gender and ethnicity. However, the evidence for ethnicity adjustment has been increasingly questioned, and in 2021 NICE recommended that kidney function should be estimated by CKD-EPI eGFRcreatinine without using ethnicity adjustment. Recently, a CKD-EPI equation has been presented without ethnicity adjustment; however, this has not been validated outside of North America and NICE continues to recommend CKD-EPI 2009. We review the status of eGFRcreatinine in clinical practice, including the limitations of eGFRcreatinine and the rationale for removal of ethnicity adjustment and the potential impact of this change on clinical care for patients with kidney disease.
Introduction
Due to differences in creatinine synthesis and excretion, assessment and monitoring of individuals with stable kidney function, including those with chronic kidney disease (CKD), is usually undertaken using glomerular filtration rate estimating equations based on serum (or plasma) creatinine. The original estimating equation, the modification of diet in renal disease (MDRD) eGFRcreatinine, was introduced in 1999 and the 4-variable version of the equation requiring knowledge of serum creatinine, age, gender and ethnicity (Black ethnicity) was published in 2000.1,2 The Chronic Kidney Disease epidemiology collaboration (CKD-EPI) eGFRcreatinine equation was introduced in 2009 and provided improved accuracy at a higher level of eGFR compared to MDRD eGFRcreatinine. 3 The use of MDRD eGFRcreatinine was recommended by the National Institute for Health and Care Excellence (NICE) in 2008, and in 2014 this recommendation was changed to CKD-EPI eGFRcreatinine. 4
The use of an ethnicity adjustment in eGFRcreatinine has been increasingly questioned, and national professional groups including the National Kidney Foundation (NKF) and American Society of Nephrology (ASN) (through an NKF-ASN taskforce), and the UK Kidney Association have issued statements recommending this practice is discontinued.5–7 Subsequently, the 2021 NICE CKD guideline update recommended that the CKD-EPI 2009 equation is used without ethnicity adjustment, and this should be the current standard of care in the UK. 4 A new CKD-EPI eGFRcreatinine equation was introduced in 2021, which did not incorporate ethnicity and adjusted eGFR so that eGFR for individuals of Black ethnicity and all other ethnicities have the same bias when compared to measured GFR. 8 This equation has not yet been validated for use in the UK and NICE continue to recommend the CKD-EPI 2009 equation. Here, we review the current status of creatinine-based eGFR testing including the rationale for the removal of ethnicity adjustment. We will also explore the potential impact of removing the ethnicity correction from eGFR on clinical care for patients with kidney disease and how this may influence future patient outcomes and health inequalities.
Chronic kidney disease and kidney function measurement
Chronic kidney disease (CKD) affects one in seven of the adult population both in the UK and globally.9–11 Furthermore, a large proportion of individuals who do not have CKD are at risk of developing CKD because of the high prevalence of long-term conditions that cause CKD such as diabetes and hypertension. 12 The diagnosis of CKD is based on two laboratory measurements, glomerular filtration rate (GFR) and urinary albumin:creatinine ratio (ACR) and/or evidence of structural renal disease; for example, polycystic kidney disease. EGFR and urinary ACR form the basis of a Kidney Disease Improving Global Outcomes (KDIGO) classification system that informs risk stratification and clinical decision making.13,14 Lower GFR and high urinary ACR are independently associated with a major increased risk of cardiovascular disease and therefore early mortality.15,16 This relationship is directly related to the severity of kidney disease.17,18
Whilst cardiovascular disease is the major risk associated with CKD, progression to end-stage kidney disease (kidney failure usually treated by dialysis or kidney transplantation) is also related to low GFR and high ACR, when corrected for the competing risk of mortality. 19 There is also an increased risk between CKD and other adverse outcomes including some cancers and infectious diseases. 20 Therefore, kidney function measurement is essential for clinical decision making around treatments that improve long-term outcomes for patients with CKD, including dose adjustment for drugs that have renal excretion and appropriate timing of preparation for treatment for kidney failure. 4
An important recent development in clinical practice is using kidney function measurement for risk stratification through a risk prediction tool. The kidney failure risk equation (KFRE) incorporates CKD-EPI eGFRcreatinine, urine ACR, age and gender to produce a 2-year and 5-year risk of treated kidney failure (defined as a requirement for dialysis or transplantation) for individuals with an eGFR less than 60 ml/min/1.73 m2.21,22 KFRE has been validated internationally and in the UK, and has recently been incorporated into the NICE CKD guideline, which recommends referral from primary to secondary care for individuals with a 5% or more risk at 5 years for treated kidney failure.4,23
Measured glomerular filtration rate
Gold standard measurement of GFR is performed by calculating the clearance of an exogenous molecule that is eliminated exclusively by glomerular filtration. 24 Inulin clearance was the original reference measurement for GFR; however, inulin is not widely available, is difficult to measure and requires either collection of urine or constant infusion for best results, limiting its use in routine clinical practice.25,26 Measured GFR (mGFR) is now more commonly undertaken in clinical practice and research by calculating the plasma clearance of radio-isotopes such as technetium 99 m diethylenetriamine pentaacetic acid (99mTc-DTPA), chromium 51-ethylenediamine tetraacetic acid (51Cr-EDTA) or 125I-iothalamate following a single injection. 27 Measured GFR remains a time-consuming and expensive test however and may also involve exposure to radiation, so use is restricted to situations where optimal precision in kidney function measurement is required, such as assessment of potential living kidney donors, chemotherapy dosing and clinical trials of intervention. 24
Whilst mGFR is the most precise measurement of kidney function at a single time-point, there is substantial intra-test variability of between 4.5% and 9.8%. 28 This variability is due to multiple biological factors, including hydration status (extra-cellular volume) and protein loading. Furthermore, whilst mGFR provides a more accurate measure of kidney function than eGFR at a single time-point, clinical practice requires repeat testing of kidney function with time to assess disease trajectory and there is no evidence that mGFR provides greater precision for assessing change in kidney function with time than eGFRcreatinine.29,30
Estimated glomerular filtration rate
Kidney function testing by eGFR requires measurement of one or more endogenous filtration markers and adjustment for surrogate non-GFR determinants.
31
The ideal endogenous marker would be produced at a constant rate in the body, freely filtered by the glomerulus, and not absorbed, metabolised or secreted into the renal tract. Whilst no endogenous marker fulfils all these criteria, creatinine has acceptable accuracy, is cheap and is the established marker for use in routine clinical practice (Figure 1).
32
Determinants of serum level of endogenous filtration markers.1,32 Plasma level (P) of endogenous filtration marker is determined by (i) generation (G) from cells and diet, (ii) extrarenal elimination (E) by gut and liver and (iii) urinary excretion (UV) by the kidney. Urinary excretion is the sum of filtered load (GFR x P), tubular secretion (TS) minus tubular reabsorption (TR). By substitution and rearrangement, eGFR can be expressed as the ratio of the non-GFR determinants (G, TS, TR and E) to the plasma level.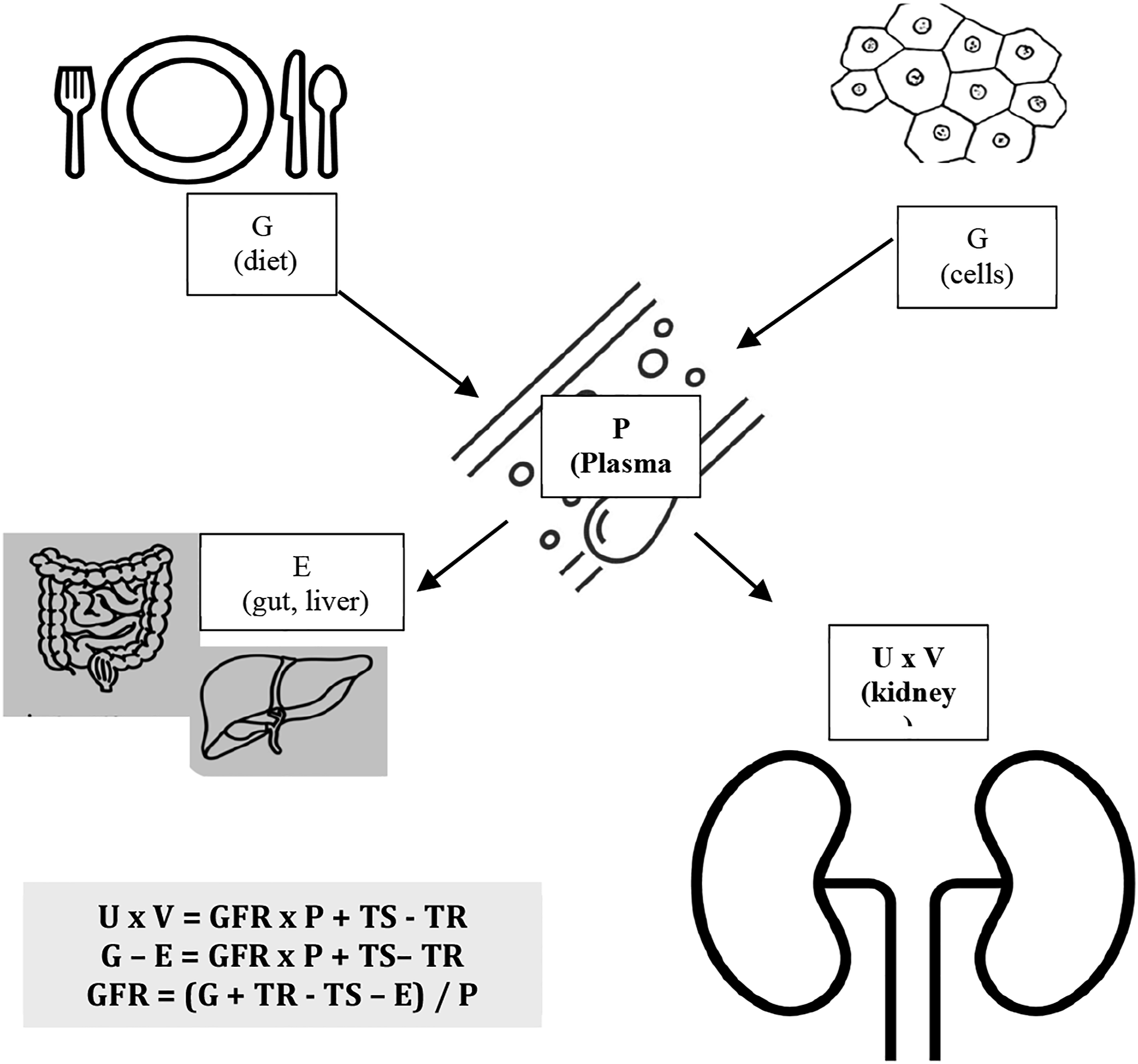
Creatinine-based eGFR equations
Creatinine is a breakdown product of muscle creatine. It is influenced by non-GFR factors including muscle mass, dietary protein intake, tubular secretion of creatinine and drug therapy. 33 Whilst it is impossible to adjust for all non-GFR determinants of serum creatinine in eGFRcreatinine equations, age and sex can provide surrogate information on muscle mass. 34 Until recently, ethnicity (Black or other ethnicity) was considered to provide an additional surrogate of muscle mass; the rationale for both inclusion and removal of ethnicity adjustment in eGFR equations is discussed in more detail below. Additionally, NICE guidelines include recommendations that patients should not eat meat and limit dietary protein intake in the 12 hours preceding a blood test for eGFRcreatinine; however, this can be difficult to implement in clinical practice. 4 NICE also recommend that eGFRcreatinine should be interpreted with caution in extremes of muscle mass, for example, in bodybuilders, people who have had an amputation or people with muscle wasting disorders. 4
Substantial variability in accuracy, both within and between laboratories, of the assays currently used to measure serum creatinine is a recognised concern worldwide.35,36 There have been global efforts to try and standardise creatinine measurements to ensure that all assays are directly traceable to isotope dilution mass spectrometry (IDMS) standards. 37 In the UK, the kinetic Jaffe and enzymatic creatinine assays are the most widely used. Although the Jaffe assay is cheaper, NICE have recommended enzymatic creatinine assays as the preferred method of creatinine measurement, due to their improved accuracy and reduced risk of interference by endogenous and external compounds, including bilirubin, ketones and cephalosporins.4,38 Laboratories in the United Kingdom participate in external quality assurance to assess analytical variation. 39
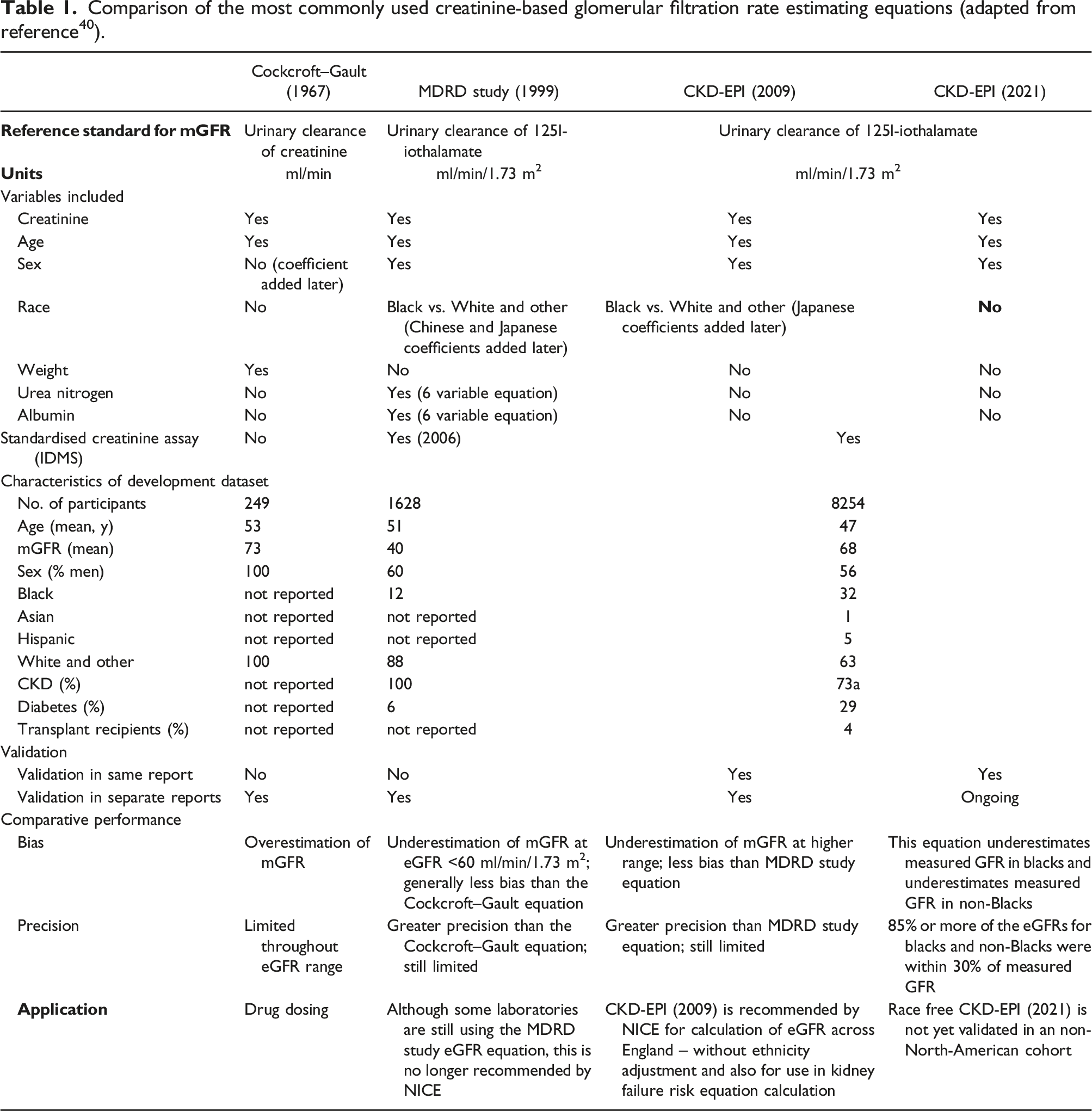
Comparison of the most commonly used creatinine-based glomerular filtration rate estimating equations (adapted from reference 40 ).
Precision and bias in eGFR equations
The differences between eGFR and mGFR can be expressed through the P30 value, referring to the percentage of individuals with an eGFR within 30% of mGFR. The P30 value is recommended as an assessment standard by the NKF.
42
The P30 shows the bias (difference between mGFR and eGFR), precision (variance around the bias) and accuracy of eGFR (Figure 2). For MDRD eGFRcreatinine, P30 values range between 73% and 93%.
43
Illustration of the concepts of test bias and precision when comparing estimated GFR values (circles) to measured GFR (black solid line).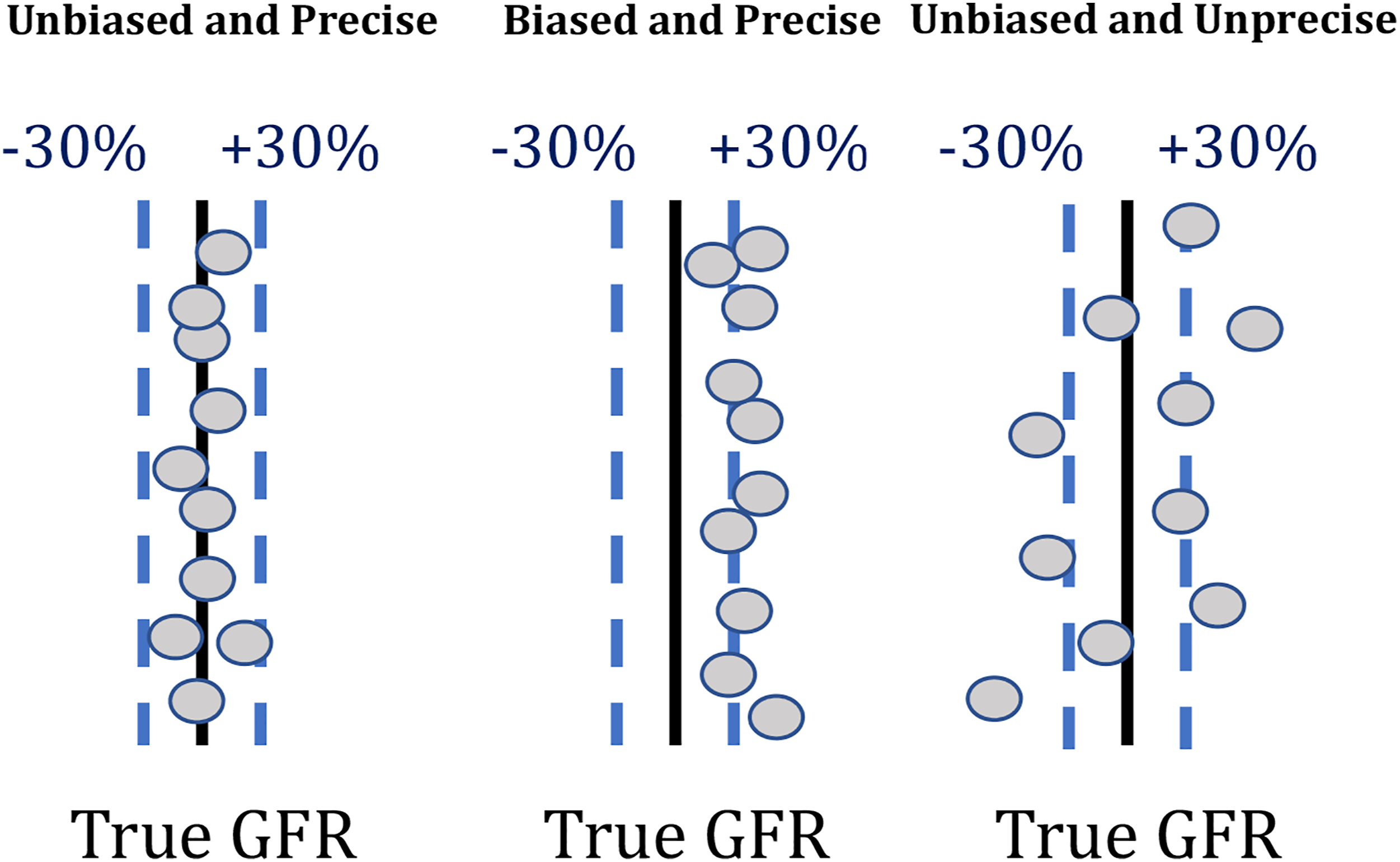
Many laboratories in the UK are still reporting MDRD eGFRcreatinine despite NICE recommending the 2009 CKD-EPI eGFRcreatinine in 2014. 4 The MDRD eGFRcreatinine underestimates GFR (particularly in individuals with a GFR of 60 mL/min/1.73 m2 or more) and has poor precision.1,2,44 The CKD-EPI eGFRcreatinine has a better P30 than MDRD eGFR at higher levels of kidney function, although this is not the case at lower levels of GFR, and direct comparisons between CKD-EPI and MDRD across all ranges of GFR show only a small benefit of CKD-EPI. 45 Using CKD-EPI eGFRcreatinine rather than MDRD eGFRcreatinine leads to reclassification of a proportion of patients with an eGFR less than 60 mL/min/1.73 m2 to 60 mL/min/1.73 m2 or more. 44 This has important implications as it decreases the number of individuals inadvertently classified as having CKD by MDRD eGFRcreatinine and ensures more accuracy for treatment and referral pathways. 3 The 2009 CKD-EPI equation is also the equation that is used in the kidney failure risk equation (KFRE). 21
Cystatin-based eGFR equations
Cystatin C is a low-molecular weight protein that is produced by all nucleated cells. It is freely filtered at the glomerulus and not reabsorbed. Measurement of plasma cystatin C is now increasingly used as an endogenous filtration marker.46,47 An international standard for measuring cystatin C was introduced in 2010 and the CKD-EPI consortium have published both a CKD-EPIcystatin and CKD-EPIcreatinine-cystatin equation.8,47 These equations were updated in 2021 following recommendations for removal of ethnicity adjustments. 8 Cystatin is less influenced by factors such as age and extremes of muscle mass in comparison to creatinine. 5 There is also emerging data to suggest that cystatin-based eGFR measurements may be better at predicting long-term cardiovascular disease and mortality risk. 48 Lack of availability and standardisation of cystatin C assays have limited their widespread use in clinical practice and these concerns have led to the withdrawal of the recommendation of use of cystatin C by NICE for CKD in clinical practice. 4
Ethnicity and eGFR equations
Precise assessment of kidney function testing is of great importance to avoid exacerbation of health inequalities; all individuals with or at risk of CKD should have access to appropriate and accurate monitoring and management to provide the opportunity for best outcomes. Although the reported prevalence of CKD is similar between different ethnic groups, people of non-White ethnic groups are at increased risk of rapid progression of CKD and kidney failure treated with dialysis or kidney transplantation.49–51 For example, people of Black ethnicity represent 7.8% of UK patients requiring renal replacement therapy (RRT), whereas only 3% of people living in the UK are of Black ethnicity.52,53
Ethnicity adjustment originated from the Modified Diet in Renal Disease (MDRD) Study, which reported higher measured GFR by iothalamate clearance in African Americans compared to Caucasians with the same serum creatinine concentration. 1 This difference was largely attributed to greater muscle mass in individuals of Black ethnicity, although the supporting evidence for this was unconvincing. The explanation for increased muscle mass was derived from a study describing increased total body potassium (as a surrogate for lean body mass) in individuals of Black ethnicity (n = 47) compared to individuals of White ethnicity (n = 67). 54 In a separate study, Worrall et al. found higher levels of serum creatine kinase, without significant differences in lean body mass, in individuals of Black ethnicity compared to individuals of White ethnicity. 55 Neither of these studies demonstrated a causal association between ethnicity, muscle mass and serum creatinine.
Data from the US-based National Health and Nutrition Examination Survey (NHANES) described that Black participants had on average 11% increased muscle mass compared to White participants. Furthermore, creatinine concentrations for Black patients on haemodialysis were found to be 15–25% higher compared to patients of other ethnic groups.56,57 However, there was no significant association between difference in serum creatinine and body mass, assessed by body impedance analysis. 57 Although muscle mass does affect creatinine generation, differences in serum concentration between different ethnic groups, independent of GFR, may be more related to nutrition, physical activity and creatinine excretion, via tubular handing and extrarenal metabolism.58,59
A major weakness of using a single Black ethnicity correction factor is that it covers over one billion people with broad and diverse backgrounds. In continental African populations, there is substantial evidence that an adjustment factor is not required.60–68 For example, in a cohort of 93 healthy Congolese participants, both MDRD and CKD-EPI (creatinine only and creatinine-cystatin C) equations demonstrated significantly reduced bias compared to GFR, measured using plasma clearance of iohexol, by removing the ethnicity adjustment factor.31,60 Cystatin C containing CKD-EPI equations also performed better than creatinine equations alone, suggesting than serum creatinine concentration is more dependent on non-GFR factors. 31
Differences in BMI and diet may also contribute to the difference between African Americans and Africans, as BMI effects the accuracy of eGFR. The Chronic Renal Insufficiency Cohort (CRIC) study reported reduced bias from 20.3% to 3.3% in African Americans when body composition variables were included. 69 A cohort of 944 individuals recruited in rural Ghana and with a mean BMI of 21.1 kg/m2 were compared to US individuals recruited in the AASK and CRIC cohort studies, with mean eGFRs of 29.0 kg/m2 and 31.3 kg/m2, respectively. Conventional eGFR equations overestimated mGFR, and CKD-EPI without the ethnicity adjustment factor was the most accurate assessment of mGFR assessed by 24-hour urinary creatinine clearance.60,61,69,70
Dietary protein consumption increases serum creatinine levels through protein catabolism rather than decreased clearance.58,59 Therefore, lower dietary protein intake, as described in Black South Africans compared to African Americans, combined with reduced body surface area (likely less than the standardised 1.73 m2 in eGFR equations), may account for differences in accuracy in eGFR between African and African-American populations and the use of accurate anthropometric characteristics in place of ethnicity correction may mitigate this. 71
In Europe, where dietary content may be more similar to USA, overestimation of GFR in Black populations has been reported in France and the United Kingdom.72,73 Flamant et al. found eGFR was overestimated, with a mean bias of 7.2 ml/min/1.73 m2 compared to mGFR in African Europeans and calculated that a reduced correction factor of 1.08 was required. 72 In the UK, a recent retrospective study, consisting of 2333 patients (314 Black; 2019 White self-reported ethnicity), compared GFR measured by 51Cr-EDTA to MDRD and CKD-EPI equations and found that both significantly overestimated mGFR when ethnicity adjustment was incorporated. Removal of the adjustment factor reduced percentage bias by 22.3% and 17.7% for MDRD and CKD-EPI respectively, and resulted in reclassification of CKD stage in 24% of patients of Black ethnicity. 74
Ethnicity is a social construct based on cultural, religious and linguistic factors.75,76 Increasing global awareness of ethnicity-related health disparities has highlighted the negative impact of using ethnicity adjustment factors to estimate GFR. While genetics and environmental factors may contribute to GFR, adjusting with a singular correction factor is unlikely to accurately represent the broad heterogeneity that exists within all ethnic groups. As highlighted previously, in 2021, the NKF-ASN taskforce, the UKKA, and NICE recommended discontinuing adjustment for ethnicity when calculating eGFR in people of Black ethnicity.4,6 These recommendations were based on both the unacceptable practice of conducting ethnicity-based medicine and the wide range of P30 values from studies assessed, which included different ethnicities and variation in contributing factors including muscle mass across ethnic groups. 4
Recommendations for clinical practice
Management of patients with CKD is often dependent on specific eGFR thresholds and the removal of the ethnicity correction factor is a welcome change to clinical practice, which may address some current health inequalities by enabling people of Black ethnicity to benefit from earlier recognition of CKD and timely planning for renal replacement therapies, including kidney transplantation.
In primary care settings, diagnosis and management of CKD (including testing of urine ACR, regular monitoring of renal function, lowered blood pressure targets, statin prescription and lifestyle/dietary advice) are dependent on patients reaching CKD G3 (eGFR less than 60 ml/min/1.73 m2). 4 Until 2021, the most prominent referral thresholds in the UK for specialist nephrology care has been an eGFR less than 30 ml/min/1.73 m 2 and is now a kidney failure risk (which includes eGFR) of more than 5% at 5 years. 4 When patients are under specialist nephrology care, an eGFR threshold, such as less than 20 ml/min/1.73 m2, can trigger transfer of patients into multidisciplinary advanced CKD care clinics. For patients who are fit for a kidney transplant, activation on the transplant waiting list will usually not happen until the eGFR is less than 15 ml/min/1.73 m2. With these specific eGFR thresholds, inaccurate correction of eGFR for patients of Black ethnicity, which may potentially overestimate GFR, raises important concerns about delays in this patient group for receiving optimum CKD care and may contribute to inequalities in health outcomes. However, it is also important to recognise that for some self-identifying Black individuals, removal of the correction factor will result in underestimation of GFR, which may be anxiety provoking and also affect access to medications where prescribing is based on GFR thresholds. 77
Creatinine remains an imperfect measure of glomerular function with considerable intra and inter-patient variability. Interpretation of serum creatinine and derived eGFR for an individual should be assessed as a trajectory on multiple testing and considered in the context of other factors which influence creatinine synthesis and excretion, such as muscle mass and catabolism, sex, age, body size and renal tubular handling of creatinine.
The use of other filtration markers, such as cystatin C, may address some of creatinine’s limitations as it is made by all nucleated cells and is therefore less dependent on muscle mass. Furthermore, Inker et al., modified the CKD-EPI equation in 2021 omitting ethnicity to produce an equation with smaller differences between Black and non-Black patients, when using creatinine and cystatin C. This has contributed to the NKF-ASN taskforce recommending increased routine use of cystatin C, especially to confirm eGFR in adults who have or are at risk of having CKD.8,78 In the UK, the NICE guideline removed recommendations for use of cystatin C, based on the possible increased risk of false positive results in confirmatory tests, but have encouraged further research in this area. 4 The eGFR-C study will report soon and will provide evidence on eGFRCystatin C and eGFRCreatinine-Cystatin C. 79
Summary
Kidney function measurement by eGFRCreatinine is the current standard of care for measurement of excretory kidney function in clinical practice, although there is substantial intra and inter-patient variability. The removal of ethnicity adjustment is an important development and has focused the laboratory and clinical community on the importance of standardisation of assay measurement and eGFR reporting. Further iterations of the CKD-EPI eGFR equations have been published and these require validation in non-North-American populations, and work to further refine eGFRCreatinine equations should continue. Interpretation of kidney function measurement includes consideration of non-ethnic variables that affect inter and intra-patient variability, including supporting the patient to ensure that they can make fully informed choices about their clinical care.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
PC.
Contributorship
RG – Conceptualization, Literature Review, Writing – original draft, Writing – review & editing JP – Conceptualization, Literature Review, Writing – original draft, Writing – review & editing KB - Conceptualization, Literature Review, Writing – original draft, Writing – review & editing PC - Conceptualization, Literature Review, Writing – original draft, Writing – review & editing.
