Abstract
Aims
Physiological changes that occur during pregnancy can influence biochemical parameters. Therefore, using reference intervals based on specimens from non-pregnant women to interpret laboratory results during pregnancy may be inappropriate. This study aimed to establish the essential reference intervals for a range of analytes during pregnancy.
Methods
A cross-sectional study was performed in 13,656 healthy pregnant and 2634 non-pregnant women. Fifteen biochemical measurands relating to renal and hepatic function were analysed using an Olympus AU5400 analyzer (Olympus, Tokyo, Japan). All the laboratory results were checked for outliers using Dixon’s test. Reference intervals were established using a non-parametric method.
Results
Alanine aminotransferase, aspartate aminotransferase, albumin, cholinesterase, creatinine, direct bilirubin, gamma-glutamyl transpeptidase, total bilirubin, total bile acid and total protein showed a decrease during the whole gestational period, while alkaline phosphatase and uric acid increased. Urea nitrogen, β2-microglobulin and cystatin-C fell significantly during the first trimester and then remained relatively stable until third trimester. Reference intervals of all the measurands during normal pregnancy have been established.
Conclusions
The reference intervals established here can be adopted in other clinical laboratories after appropriate validation. We verified the importance, for some measurands, of partitioning by gestational age when establishing reference intervals during pregnancy.
Introduction
During pregnancy, serum progesterone and oestrogen concentrations increase progressively and reach maxima during late pregnancy. 1 Those sex steroids have effects on synthetic, metabolic and excretory hepatic functions. 2 Some authors have claimed that there is a subclinical and physiological cholestasis during pregnancy. 3 An increase in plasma volume that occurs during pregnancy decreases the concentration of serum protein.4,5 Serum alkaline phosphatase (ALP) concentrations increase during the third trimester of pregnancy because of both an increase in the bone isoenzyme and a production of the placental isoenzyme.6–8 Renal plasma flow and glomerular filtration rate (GFR) increase by 50 to 85% and 40 to 65%, respectively, during normal pregnancy. 9 The net effect is an early decrease in serum creatinine, urea and uric acid (UA) concentration.10,11 Not surprisingly, therefore, changes in liver and kidney function tests occur during pregnancy.
For many commonly used laboratory assays, reference intervals based on a population of healthy pregnant women at different stages of gestation are of importance for proper interpretation of laboratory results during pregnancy. Therefore, it is advisable to establish reference intervals for several biochemical substances relating to renal and hepatic function during normal pregnancy.
Although some reference intervals for clinical chemistry assays during normal pregnancy have been previously reported,12–15 there are several problems with the analyses: some do not fulfil the recommended minimum sample size of 120 individuals 16 ; some do not remove the outliers before analysis, as recommended 17 and some do not establish reference intervals by a non-parametric method. 18
In this study, we report gestational age-specific reference intervals for several biochemical substances relating to hepatic and renal function during normal pregnancy in China.
Subjects and methods
Study population
A total of 24,212 healthy Chinese Han women aged 18–39 years with a singleton pregnancy were recruited from the outpatient department of the Third Affiliated Hospital of Zhengzhou University from January 2012 to May 2016, with nearly equal numbers at each gestational week. Normal pregnancy was defined as singleton pregnancy with no proteinuria and with diastolic blood pressure (DBP) <90 mmHg. Exclusion criteria were the following: (a) taking drugs other than folic acid or vitamins; (b) nutritional deficiency diseases, documented liver, kidney or other diseases that might affect the values of selected measurands; (c) pregnancy-related complications; (d) gestational pruritus and cholecystectomy; (e) excessive alcohol consumption (≥30 g/day) and smoking (≥12 cigarettes/day); (f) exposure to hepatitis or human immunodeficiency virus (HIV). Blood pressure and other clinical data were obtained from the medical record. Participants who met the study criteria completed a lifestyle questionnaire designed for this study and underwent blood tests and physical examination. Information regarding age, gestational age, weight, height, smoking or alcohol intake, environment, ethnic background, prescription medications, physical activity, dietary habits, as well as disease history was obtained for each subject. Pregnant women were divided into five different gestational groups: <12 weeks (n = 2748); 13–≤20+6 weeks (n = 2727); 21–≤27+6 weeks (n = 2755); 28–≤35+6 weeks (n = 2754); and 36–≤42+6 weeks (n = 2672). A total of 2634 healthy non-pregnant women aged 18–40 years served as controls and subject to the same exclusion criteria as the healthy pregnant women.
This study was approved by the Ethics Committee of the Third Affiliated Hospital of Zhengzhou University, and all the subjects gave informed written consent.
Specimen collection and handing
Performed tests, abbreviations, methods and analytical details.

Note: The total analytical imprecision for the experimental method used to calculate the reference intervals is given for each assay as an average variation coefficient (CV%) of two concentrations of internal controls through one year. Samples with preanalytic handling time longer than the stability in the blood (in days) at room temperature were removed from the calculations.
IFCC: International Federation of Clinical Chemistry.
Statistical methods
The outliers were removed from this study when D/R ratio was over 1/3, 19 where D is the absolute difference between an extreme value (small or large) and the next value (smallest or largest), and R is the range of all observations. Gestational age-specific reference intervals (2.5th and 97.5th percentiles with confidence intervals) were established for each measurand using the non-parametric method performed with RefVal 4.11 program (RefVal, Rykkinn, Norway) according to the recommendations of the International Federation of Clinical Chemistry (IFCC).16,20 Statistical analyses were performed using SPSS 17.0 (SPSS Inc., Chicago, IL, USA) for windows. Normality of the data was assessed by applying the Kolmorov-Smirnov test. All the data were found to be non-normally distributed. The Kruskal-Wallis test followed by the Wilcoxon test was used to assess the differences in each analyte concentration/enzyme activity across pregnancy. Differences between pregnant and non-pregnant groups were evaluated by the Mann-Whitney U test. Spearman’s rank correlation test was used to assess the relationship between concentrations of the measurands and weeks of gestation. Statistical significance was defined as P < 0.01.
Results
There were 10,204 exclusions: 8094 for taking drugs other than folic acid or vitamin, 1173 for pregnancy-related complications, 554 for diseases that could affect the values of selected measurands and 383 for excessive drinking or smoking. A total of 14,008 pregnancies (57.86%) passed all exclusion criteria, and of these 352 pregnant women dropped out during the study (Figure 1). Participant numbers, age and body mass index (BMI) are shown for each gestational age group and for non-pregnant control subjects along with numbers of participants in each group removed from further analysis because of results that were classified as outliers (Table 2). Numbers of detected outliers are also listed in Table 2. There were no significant differences between the ages or BMI between the pregnant and non-pregnant groups. There were no differences in maternal age between the gestational age groups (Table 2).
Participant flow diagram. Median, interquartile range and body mass index for each gestational age group. IQR: interquartile range.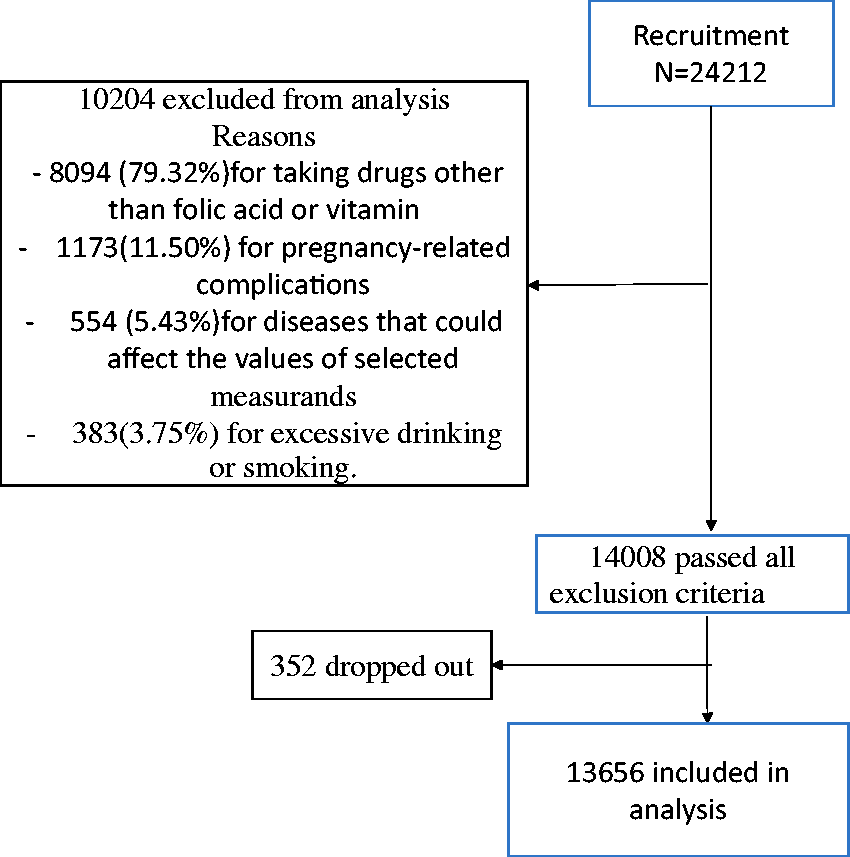
Median and 95% reference intervals for liver and kidney function tests during the normal pregnancy.
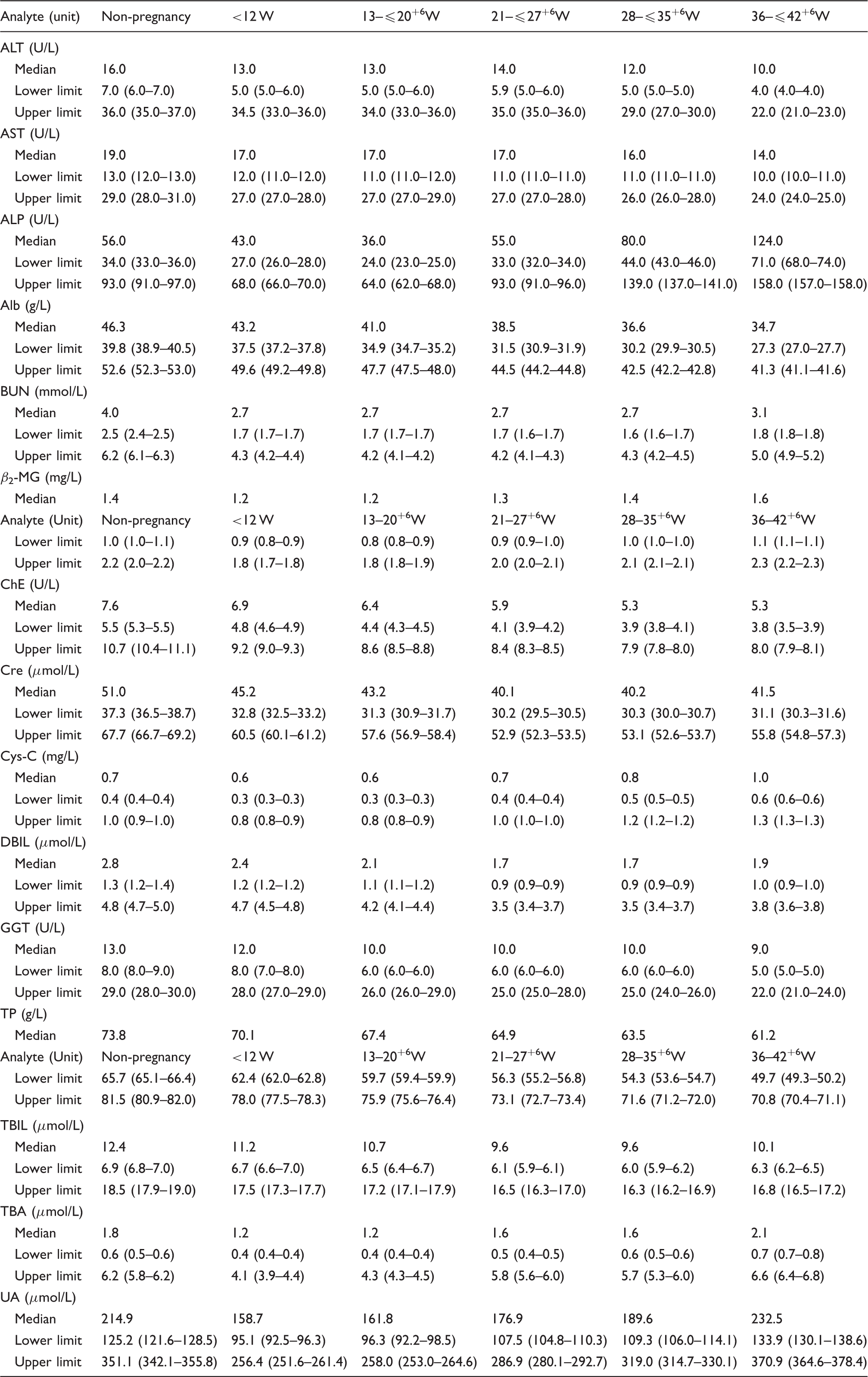
Note: The 90% confidence intervals for the lower and upper limits are given in parentheses.
ALT: alanine aminotransferase; AST: aspartate aminotransferase; ALP: alkaline phosphatase; Alb: albumin; BUN: urea nitrogen; β2-MG: β2-microglobulin; ChE: cholinesterase; Cre: creatinine; Cys-C: cystatin-C; DBIL: direct bilirubin; GGT: gamma-glutamyl transpeptidase; TP: total protein; TBIL: total bilirubin; TBA: total bile acid; UA: uric acid.
Correlation relationships between biochemical analyte concentrations and gestational weeks.
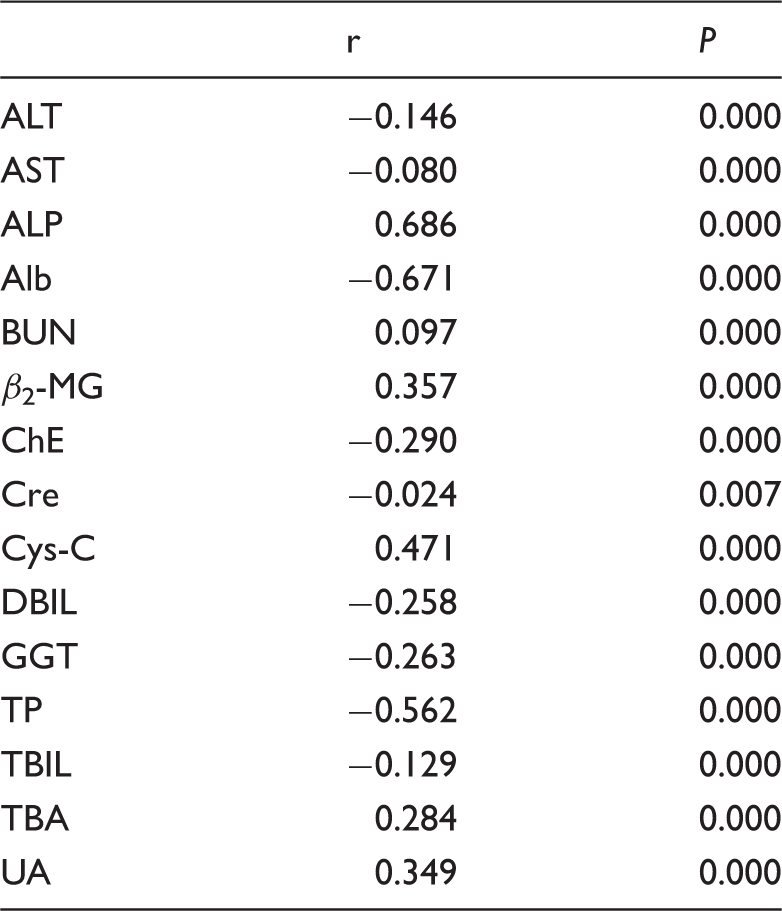
ALT: alanine aminotransferase; AST: aspartate aminotransferase; ALP: alkaline phosphatase; Alb: albumin; BUN: urea nitrogen; β2-MG: β2-microglobulin; ChE: cholinesterase; Cre: creatinine; Cys-C: cystatin-C; DBIL: direct bilirubin; GGT: gamma-glutamyl transpeptidase; TP: total protein; TBIL: total bilirubin; TBA: total bile acid; UA: uric acid.
Discussion
Blood tests are commonly requested during pregnancy to identify pregnancy-associated complications or to investigate intercurrent illness. Appropriate reference intervals for clinical laboratory tests are therefore important in supporting clinical decision-making.21–23
Although previous studies have addressed trimester-specific reference intervals for selected biochemical measurands, considerable variation can be observed in the population because serum biochemical measurand concentrations of pregnant women can be influenced by many factors such as dietary habits, nutritional status, lifestyle, ethnicity, which can differ significantly among pregnant women in different geographical areas.
Compared with the non-pregnant women, the values of most measurands were significantly different. This may reflect the biochemical and physiological changes that occur during pregnancy. In agreement with previous studies, ALT and AST showed a decrease during the whole gestational period. 13 GGT activity significantly decreased at 13– ≤ 20+6 gestational weeks, remained relatively unchanged at 21– ≤ 27+6 and 28 ≤ 35+6 gestational weeks, and significantly decreased at 36– ≤ 42+6 gestational weeks. ChE activity decreased in first trimester and continued to fall throughout pregnancy being significantly lower during the third trimester compared with the first and second trimesters. TBIL and DBIL – again similar to ChE – decreased in early pregnancy (compared with non-pregnant) and then continued to fall. Highest pregnancy levels were noted in the first trimester. This was associated with the slight but non-significant decrease observed during early pregnancy related to haemodilution. β2-MG and Cys-C concentrations were significantly higher during the third trimester than during the first and second trimesters. Although statistically significant changes were noted in ALT, AST, GGT, ChE, TBIL, DBIL, β2-MG and Cys-C, the quantitative changes were small and unlikely to be of clinical importance. These changes may reflect the scatter of results, as the median values did not change. However, there was a general trend of minor reduction in ALT, AST, GGT, ChE, TBIL, DBIL, β2-MG and Cys-C in pregnancy compared with non-pregnant women, and the values of these measurands continued to decrease slightly over pregnancy.
Cre decreased in early pregnancy and fell further during pregnancy. In contrast, BUN fell significantly in early pregnancy and then remained relatively stable until late gestation. Although these changes are not large, it may be appropriate to use pregnancy-specific reference intervals for these measurands to allow the early detection of renal disease during pregnancy.
Although reproductive hormones have been implicated in the pathogenesis of cholestasis, and obstetric cholestasis often presents during late pregnancy, studies regarding TBA concentration during pregnancy have reported conflicting results. Some studies showed that TBA concentration was significantly higher during later pregnancy than during early pregnancy,13,24 whereas another study showed no difference. 25 In addition, a longitudinal study of 56 pregnant women showed a rise in TBA concentration with increasing gestation. 26 In the present study, TBA concentration increased progressively with gestation, which was in accordance with the report by Carter et al. 27 Serum TP and Alb values decreased progressively with gestation, which may be explained by the haemodilution that occurs during pregnancy. As expected, serum ALP activity was significantly higher during late pregnancy (a consequence of the production of placental ALP). There was also a slight but significant decrease observed during early pregnancy related to haemodilution. Serum UA concentration increased progressively with gestational age. The low concentration of UA in early pregnancy may be due to the raised GFR. Increased serum UA concentration in late pregnancy may be related to an altered renal handling concentration of urate. We suggest that it may be important to have gestational age-related reference intervals for TP, Alb, ALP, TBA and UA, all of which differ from those in non-pregnant women and change with gestational age.
We also analysed the correlation relationships between gestational weeks and the concentrations of the measurands. Our results showed that all the measurands concentrations were significantly correlated with weeks of gestation (all P < 0.05).
The strengths of our study are the large sample size and use of strict exclusion criteria. One limitation in the study design is the cross-sectional analysis of several hepatic and renal function parameters. A longitudinal study design, where the same pregnant women are followed up at different time points of gestational weeks, would have been more appropriate. However, to undertake such a longitudinal study in a large number of participants would have been challenging. A further limitation is that for those measurands which demonstrate a high degree of between subject variability, population-based reference intervals are of limited value, i.e. an individual patient result may fall within the reference interval but be highly abnormal for that patient.
Reference intervals of several renal and hepatic function parameters during normal pregnancy have been established. Any clinical laboratory can evaluate those reference ranges with 20 specimens, if no more than two results are outside the proposed reference range, it is statistically valid for the clinical laboratory to adopt the reference range as its own, as recommended in the Clinical and Laboratory Standards Institution document. 16
We verified that when establishing reference intervals during pregnancy, partitioning by gestational age is frequently necessary. We hope that those reference intervals may assist the clinicians in distinguishing between pathological and physiological changes states during pregnancy.
Footnotes
Acknowledgements
We would like to express our sincere thanks to all the pregnant women who participated in this study. And we also thank our co-workers at the Department of Clinical Laboratory.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by the Ethics Committee of the Third Affiliated Hospital of Zhengzhou University on 1 January 2012, number 2012/011.
Guarantor
EY.
Contributorship
Study concept and design: YD, JL
Acquisition of data: EY
Interpretation of data/results: LY, WQ
Data analysis: LW, YS
Drafting of the manuscript: YD, JL
Critical revision of the manuscript: YD, JL
Polish language: JL, EY
All authors approved the final version of the manuscript.
