Abstract
Background
Low faecal elastase-1 (FE-1) results are suggestive of pancreatic insufficiency, but watery diarrhoea may lead to falsely low results.
Methods
FE-1 results reported on watery samples over a three-year period were reviewed. Results in watery samples were compared to those from a formed sample. The follow-up of patients in whom an FE-1 result ≤199 ug/g stool (Schebo ELISA) was reported on a watery sample was also reviewed.
Results
In total, 288 watery samples were identified. All results (19/19) ≥200 ug/g in watery samples were also ≥200 ug/g when measured in a formed sample from the same patient. There were 41 results ≤199 ug/g in watery samples, of which 29 (71%) were ≥200 ug/g when measured in a formed sample. Thirty-seven patients with a single FE-1 value ≤199 ug/g from a watery sample were followed up. Pancreatic Enzyme Replacement Therapy (PERT) was commenced in 15 patients. This was inappropriate in at least one patient. Reporting practice was subsequently changed to not report FE-1 values ≤199 ug/g in watery samples. This change was assessed after 12 months. Repeat samples were received from 15/56 (27%) of patients. Overall, 10/15 (67%) of samples were ≥200 ug/g on repeat. PERT was not commenced inappropriately in any of these patients.
Conclusions
There is value in measuring FE-1 in watery samples, as 144/288 (50%) of watery samples analysed were ≥200 ug/g, enabling a diagnosis of exocrine pancreatic insufficiency to be excluded. Not reporting FE-1 values ≤199 ug/g in a first-time watery stool samples appears clinically safe and has potentially reduced inappropriate diagnoses and prescribing.
Background
Faecal elastase-1 (FE-1) is a commonly requested test to investigate gastrointestinal symptoms such as diarrhoea. Low FE-1 results are suggestive of pancreatic insufficiency, but watery diarrhoea, a common presenting complaint in pancreatic insufficiency, can lead to falsely low FE-1 due to dilution.1–3 Laboratories should adjust and standardize water content in stool samples prior to analysis 2 ; where this is not possible, a repeat test on a formed stool sample should be performed to ensure correct diagnosis. 3
This study aimed to evaluate the usefulness of FE-1 measured in watery samples unadjusted for water content by examining the diagnostic accuracy of both high and low FE-1 results and by reviewing the follow-up of patients with a low FE-1 result reported on a single watery stool sample.
Methods
FE-1 results reported on watery samples over a three-year period, 2018–2020, were obtained from the Laboratory Information Management System (LIMS).
FE-1 was measured using the Schebo ELISA assay. A cut-off value of ≤199 ug/g stool was used to define pancreatic insufficiency. 4 FE-1 was reported on all watery samples, but an additional comment stating: “Watery faecal sample received. Interpret result with caution and consider repeat on a formed stool sample” was added when FE-1 was ≤199 ug/g. Watery samples were defined as those which contained no solid material and could only be sampled using a pipette rather than the standard sampling device.
The diagnostic accuracy of FE-1 measured in watery stool samples was assessed by comparison of results to those obtained on another formed sample from the same patient obtained within 12 months (before or after).
The follow-up of patients in whom an FE-1 result ≤199 ug/g stool was reported on a watery sample was also reviewed by examination of clinical notes. In particular, other evidence of pancreatitis (surgical, imaging, histological or clinical) and/or a positive clinical response to Pancreatic Enzyme Replacement Therapy (PERT) was noted.
Results
Between 2018 and 2020, 5046 samples were received for elastase measurement, of which 288 samples (5.7%) were identified as watery. A summary of the results is shown in Figure 1. Watery samples received between 2018 and 2020. FE-1 = Faecal Elastase-1. PERT = Pancreatic Enzyme Replacement Therapy.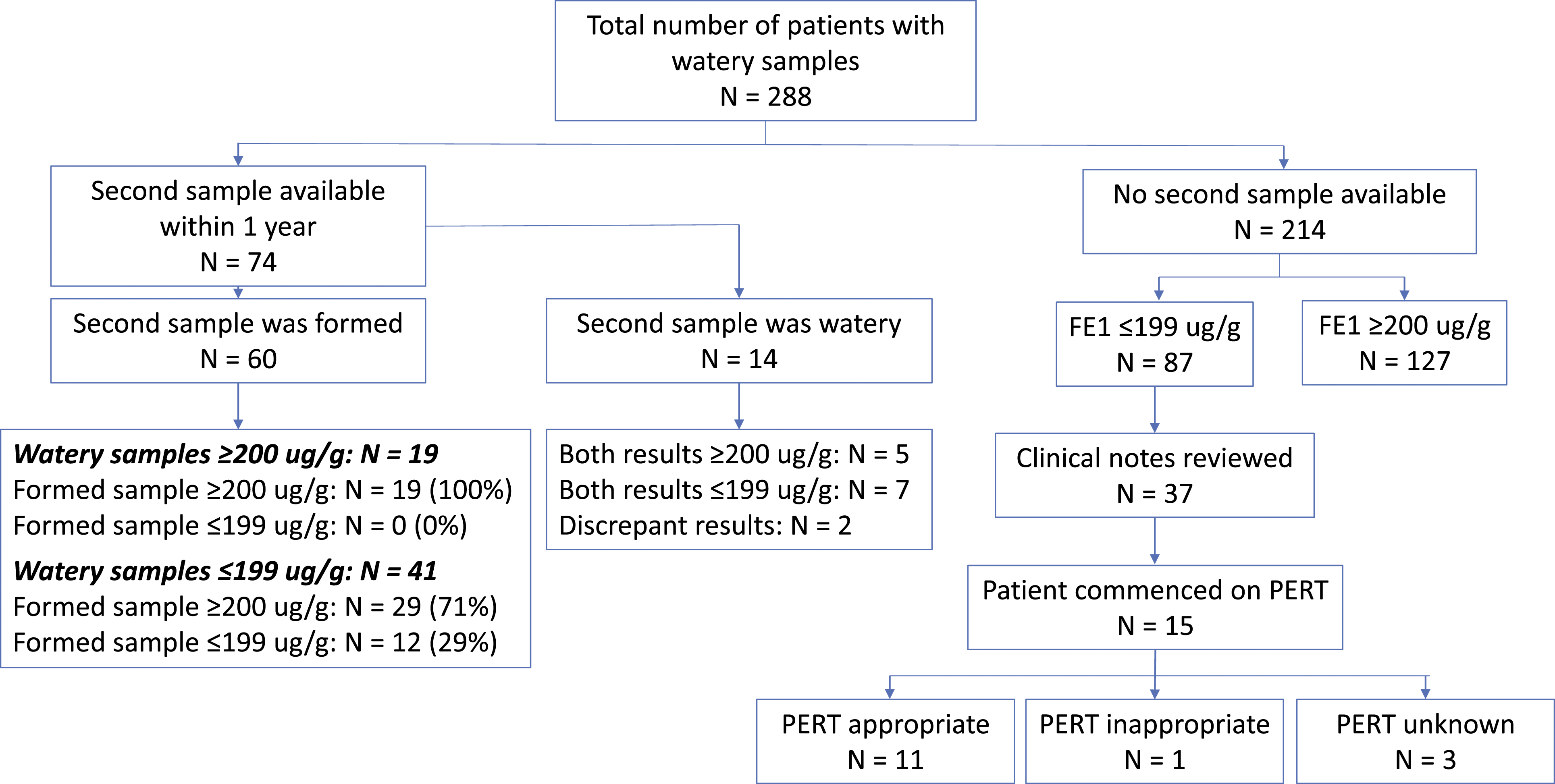
Comparison of results between watery samples and formed stool samples (figure 1 – left-hand side)
Sixty patients sent both a watery and a formed stool sample within 12 months. All (19/19) results ≥200 ug/g from watery samples were also ≥200 ug/g when measured in a formed sample. Pancreatic insufficiency was suggested by the FE-1 result in 41 watery samples (≤199 ug/g), but in 29/41 (71%), the result from the formed sample was normal (≥200 ug/g).
Follow-up of patients with a single FE-1 result ≤199 ug/g reported on a watery sample (figure 1 – right-hand side)
There was no formed sample received from 87 patients with an FE-1 value ≤199 ug/g on a watery sample. A large proportion of samples were received from external hospitals, so it was only possible to review clinical notes in 37 patients.
Pancreatic Enzyme Replacement Therapy (PERT) was commenced in 15 patients. Of these, seven patients had surgical, imaging, histological or clinical evidence of pancreatitis. Of the remaining eight patients, a positive response to PERT was observed in four; one patient remained symptomatic on PERT and there was no reference in the clinical notes on the effect of PERT in the remaining three patients. PERT therefore appears to have been an appropriate treatment in at least 11/15 patients, and inappropriate in at least one.
The potentially unreliable FE-1 result and/or need for repeat sample was only recorded in 5 of the 37 patient notes examined.
Based on these findings, reporting practice was changed in April 2021. FE-1 results ≤199 ug/g in watery samples are not reported. Instead, text suggesting the FE-1 result may be unreliable is provided and a repeat formed sample is requested. If a repeat sample is received and is also watery, the measured value is reported within the text of a cautionary comment.
The impact of this change was reviewed after 12 months (May 2021–April 2022): Fifty-six results with an FE-1 value ≤199 ug/g in a watery sample were identified. Repeat samples were received from 15/56 patients (27%), of which 14 were formed stools. Overall, 10/15 (67%) of samples were found to be normal on repeat.
Clinical notes from 20 out of the 41 patients with no repeat sample were reviewed. No reference to the FE-1 report was found in any of the patients’ notes. Two patients had a previous diagnosis of pancreatic insufficiency and were already on PERT. Two patients were commenced on PERT based on other evidence of pancreatic insufficiency. No reference was found to PERT or pancreatic insufficiency in the remaining 16 patients.
Conclusions
The study demonstrates clear value in measuring FE-1 in watery samples, as 144 out of 288 (50%) of watery samples reviewed over the three-year period produced a result ≥200 ug/g. This enabled a diagnosis of pancreatic insufficiency to be excluded without requiring a repeat, formed sample to be collected, saving time and money and reducing inconvenience for patients.
However, consistent with previous reports,1-3 false positive (≤199 ug/g) FE-1 results may be caused by sample dilution due to watery diarrhoea. This was found to be the case in approximately seven out of ten patients tested in this study.
In the initial data review, 87 FE-1 results ≤199 ug/g on watery samples were not repeated despite a comment advising this. PERT may have been commenced inappropriately in some patients, suggested by a review of a sample of available patient notes. This demonstrates the limitation of relying on interpretative comments to highlight potentially unreliable results to clinical users. There is an increasing volume of publications discussing the added value of interpretive comments on reports.5,6 However, there is little, if any, assessment of the proportion of such comments that are actually viewed by clinicians.
Since stopping reporting FE-1 results in first-time watery samples, no patients have been started on PERT based on a single, potentially falsely low FE-1 result, and no cases of pancreatic insufficiency appear to have been missed. The practice appears clinically safe and has potentially reduced inappropriate diagnoses and prescribing.
However, reporting results ≥200 ug/g from watery samples enables a diagnosis of exocrine pancreatic insufficiency to be excluded without requiring a repeat, formed sample to be collected.
Footnotes
Acknowledgements
Thanks to Dr Daniel Turnock for advice on structure and assistance in redrafting.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The audit was registered with the Clinical Governance department at York and Scarborough NHS Foundation Trust.
Guarantor
Alison M Jones.
Contributorship
The study was conceived, designed and carried out by AMJ. The manuscript was written solely by AMJ.
