Abstract
Background
Pancreatic elastase-1 (PE1) can be measured to assess exocrine activity of the pancreas. A semi-automated particle-enhanced, open-channel turbidimetric immunoassay has been introduced by Bühlmann (fPELA turbo, Bühlmann Laboratories AG, Schoenenbuch, Switzerland). Published evaluation data is lacking. We therefore verified performance of the assay on the Binding Site Optilite benchtop analyser and undertook a sample comparison with the DiaSorin PE1 assay on the Liaison.
Methods
Accuracy, imprecision, lower limit of quantitation (LLoQ) and linearity of the Bühlmann fPELA turbo assay on the Binding Site Optilite analyser was ascertained. Comparison with the DiaSorin Liaison PE1 assay was also undertaken. Difference between assays was evaluated using the Wilcoxon signed-rank test and method comparison was undertaken using Spearman’s rank correlation (rs), Bland–Altman and Passing–Bablok regression analyses.
Results
The fPELA turbo assay was linear between 5 and 2500 μg/g. The LLoQ was 5 µg/g. Intra- and inter-assay imprecision was <6%. There was a good agreement (rs = 0.92) and no significant bias (5.8 µg/g, P = 0.29) present between the Bühlmann fPELA turbo and DiaSorin PE1 assays.
Conclusion
The Bühlmann fPELA turbo assay performs well on the Binding Site Optilite analyser. Faecal elastase results are commutable between with Bühlmann fPELA turbo and DiaSorin Liaison PE1 assays.
Introduction
Pancreatic elastase-1 (PE1), a protease enzyme excreted in the faeces, can be measured to screen patients for pancreatic insufficiency. Faecal PE1 concentrations <100 µg/g indicate severe pancreatic insufficiency, 100–199 µg/g mild to moderate insufficiency and ≥200 µg/g normal pancreatic sufficiency. 1 A semi-automated, particle-enhanced, open-channel turbidimetric immunoassay has been introduced by Bühlmann (fPELA turbo, Bühlmann Laboratories AG, Schoenenbuch, Switzerland). 2 The Optilite is a benchtop analyser available from the Binding Site (Edgbaston, Birmingham, UK) which is optimized for protein analysis and has greater than 50 serum protein assays available. Furthermore, several third party assays are available for use and is therefore well suited for small to medium throughput laboratories. Due to a lack of published evaluation data for the Bühlmann fPELA turbo assay, we verified performance of the assay on the Binding Site Optilite benchtop analyser (Edgbaston, Birmingham, UK) and undertook a sample comparison with the DiaSorin PE1 assay on the Liaison.
Materials and methods
Samples and analysis
Excess stool submitted to the laboratory for routine analysis of faecal PE1 was used. Samples were from individuals attending general medicine and gastroenterology clinics for the investigation of diarrhoea and pancreatic insufficiency. Samples were collected and transported to the laboratory on the day of collection. Stool consistency was assessed against the Bristol Stool Form Scale. Liquid samples (Bristol stool type 7) were not suitable for analysis. Elastase was extracted using the Bühlmann Calex Cap stool extraction device and analysed on the same day of collection using the Bühlmann fPELA turbo assay on the Optilite analyser following manufacturer’s instructions. A portion of faeces was sent frozen to an external laboratory for analysis using the DiaSorin PE1 assay on the Liaison (DiaSorin S.p.A., Saluggia, Italy). Time between collection and analysis was within 14 days, for which faecal elastase is stable. 3
Method verification
Intra- and inter-assay imprecision and accuracy and comparability of methods were in accordance with Clinical Laboratory and Standards Institute guidelines.4,5 Internal quality control (IQC) material provided with the fPELA turbo reagent kit at low and high concentrations, and two samples containing elastase from the INSTAND (Düsseldorf, Germany) external quality assurance scheme for faecal markers were analysed in quintuplicate on the same day and on five different days.
Lower limit of quantitation (LLoQ) was assessed by dilutions of an extracted patient sample (PE1 concentration approximately 200 µg/g) with Calex Cap stool extraction buffer, using an acceptance criterion of <10% inaccuracy and imprecision (mean of triplicates).
Linearity was assessed by dilutions of an extracted patient sample (PE1 concentration 2500 µg/g) with Calex Cap stool extraction buffer. Differences were calculated between the nominal and measured concentrations, with acceptance criteria being within 10% (mean of triplicates).
Carry-over was determined by the analysis of samples containing PE1 at low (L; 50 µg/g) and high (H; 2000 µg/g) concentrations followed by a blank (B; Bühlmann Calex Cap stool extraction buffer) in the sequence: L, L, L, B, B, B, and H, H, H, B, B, B.
Statistical analysis
Statistical analysis was performed using Analyse-it (Analyse-it Software Ltd, Leeds, UK) for Microsoft Excel. Normality distribution was assessed by the Shapiro–Wilk test. Data were non-parametric and expressed as medians and 95% confidence intervals (95% CI). Difference between assays was evaluated using the Wilcoxon signed-rank test and method comparison undertaken using Spearman’s rank correlation (rs), Bland–Altman and Passing–Bablok regression analyses.
Results
Summary accuracy and imprecision data for faecal elastase measured using the Bühlmann fPELA turbo assay on the Binding Site Optilite analyser.
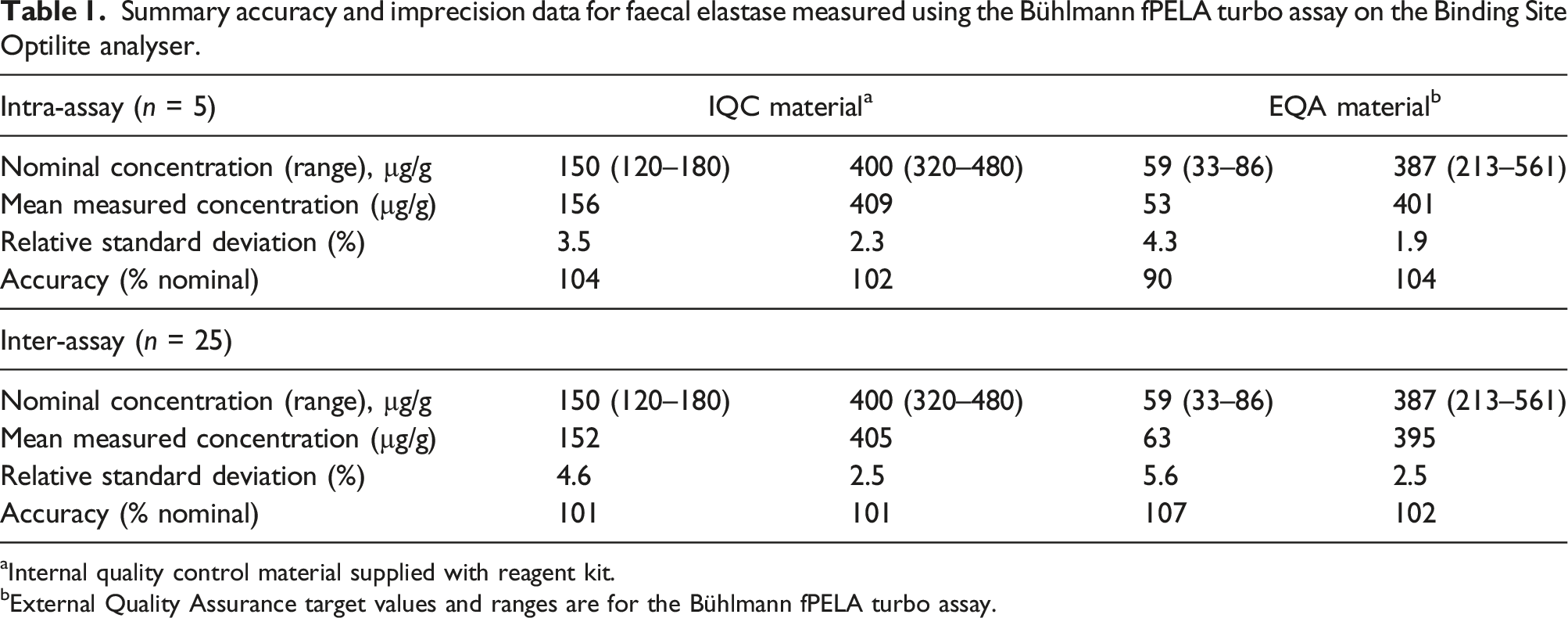
aInternal quality control material supplied with reagent kit.
bExternal Quality Assurance target values and ranges are for the Bühlmann fPELA turbo assay.
Thirty-five samples were analysed (14 male, median age [range] 65 [39–80] y; 21 female 57 [11–85] y). All samples were Bristol stool forms type 2–6 (i.e. firm to loose consistency).
Between assays, there was a very good correlation (rs = 0.92 [95% CI = 0.85–0.96], P = <0.0001). Passing–Bablok regression also indicated good agreement (slope = 1.06 [95% CI = 0.92–1.13], intercept = −0.28 [95% CI = −21.1–14.1] µg/g). The Bland–Altman plot indicated that median elastase (323 µg/g) measured using the Bühlmann fPELA assay was 5.8 (95% CI = −9.0–46.5) µg/g (2.9, 95% CI = −3.1–13.1%) higher compared to the DiaSorin Liaison PE1 assay (P = 0.29).
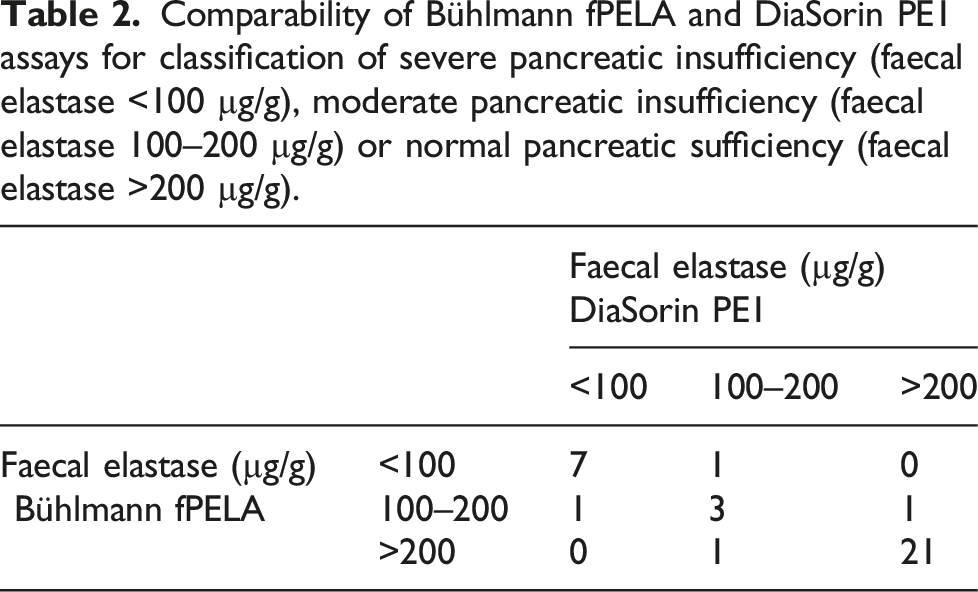
Comparability of Bühlmann fPELA and DiaSorin PE1 assays for classification of severe pancreatic insufficiency (faecal elastase <100 µg/g), moderate pancreatic insufficiency (faecal elastase 100–200 µg/g) or normal pancreatic sufficiency (faecal elastase >200 µg/g).
Total analysis time for a batch of 30 patient’s samples after sample extraction was approximately 15 min, with an additional 10 min required for sample preparation. The fPELA turbo assay was calibrated once at the beginning of the verification and was not required again as ascertained by acceptable IQC results. This indicated an on-board calibration curve stability of up to 4 weeks; further testing could not be undertaken as the reagent cassette was empty after this period.
Discussion
To our knowledge, verification of the Bühlmann fPELA turbo assay has not been published, although Bühlmann do provide analytical specifications for some clinical chemistry analysers (e.g. Roche Cobas 6000 c501). We found the Bühlmann fPELA turbo assay on the Optilite provided acceptable performance and compared well with the DiaSorin Liaison assay, with no statistically or clinically significant bias present – a similar finding has been reported when reviewing proficiency testing data. 6 A faecal elastase linear range of 3–5024 µg/g has been reported by Bühlmann for the fPELA assay on the Roche Cobas 6000 c501 chemistry analyser. Although we were only able to confirm a faecal elastase linearity of up to 2500 µg/g, this upper limit is more than 10 times the concentration associated with normal pancreatic sufficiency and concentrations above this offer limited clinical value. Likewise, the LLoQ identified in this study of 5 µg/g was well below the faecal elastase concentration associated with severe pancreatic insufficiency and is well within the requirements for clinical utility.
Batch analysis time was relatively short, and the added benefit of using the Bühlmann assay is that faecal calprotectin is also available. In our institution, approximately 50% of faecal elastase requests also require calprotectin measurement. As both assays use the Bühlmann Calex Cap stool extraction device, sampling from the same extract could be undertaken for both assays, thereby reducing the need for separate sample aliquots.
We found the calibration curve to be stable for up to 4 weeks, which allows efficient use of reagents and operator time. A limitation of the assay is that liquid samples are not suitable for analysis. This is inherent to most faecal elastase assays, and should a liquid sample be submitted, re-collection on another day is recommended. Regarding study limitations, we did not investigate the effect of potentially interfering substances (e.g. drugs and haemoglobin), undertake recovery studies or assess imprecision using patient stool samples.
Conclusions
In conclusion, the fPELA turbo assay on the Optilite is well suited for rapid analysis of faecal elastase providing short turn-around times. Elastase results are commutable between Bühlmann fPELA turbo and DiaSorin Liaison PE1 assays.
Footnotes
Acknowledgements
We thank INSTAND for permission to quote EQA data and Dr DS Handley for constructive criticism of the draft manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethics approval was not required as work undertaken does not include studies on humans.
Guarantor
LP.
Contributorship
SH and ND researched literature and conceived the study. ND and TW were involved in protocol development and data analysis. SH and LP drafted the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
