Abstract
Objective
Sex hormone-binding globulin (SHBG) is a glycoprotein which binds hormones such as testosterone. Around 97% of circulating testosterone is bound to SHBG or albumin and is therefore biologically unavailable; 2–3% of testosterone is free. Free testosterone is very technically challenging to quantify; in order to circumvent this problem, equations using testosterone and SHBG are used to estimate free testosterone. We decided to determine the effect of using different SHBG immunoassays on calculated free testosterone results.
Design
Anonymized surplus serum samples were analysed for SHBG on four different immunoassay platforms (Abbott Architect, Roche, Beckman and Siemens). The SHBG results were used to generate a Vermeulen calculated free testosterone.
Results
Beckman Access and Siemens Centaur both gave results close to the overall mean. Roche gave the highest SHBG concentrations with Abbott Architect producing the lowest results. Abbott Architect gave the highest calculated free testosterone results, followed by Beckman. Roche gave the lowest results. Sixty-five per cent of male samples had low calculated free testosterone and 27.5% of the females had high calculated free testosterone using the SHBG from the Abbott assay compared with 69% low male calculated free testosterone and 20% high female calculated free testosterone with the Roche assay.
Conclusion
Our results have shown significant differences in SHBG results produced by different analysers and subsequently the calculated free testosterone, which may affect result interpretation if method-specific reference ranges for calculated free testosterone are not used. Care should be taken to ensure reference ranges are appropriate for the analyser used to avoid misdiagnosis of hypo or hyperandrogenism, and ensure patients get the most appropriate treatment.
Keywords
Introduction
Sex hormone-binding globulin (SHBG) is an 87.4 kDa glycoprotein whose primary function is to bind steroid hormones such as testosterone, oestradiol and dihydrotestosterone. Hormones bound to SHBG or albumin are unable to enter the cell to bind and activate their receptors; therefore, only free hormones are biologically active. The use of total testosterone in the diagnosis of male hypogonadism and female hyperandrogenism can be misleading, especially when SHBG concentrations are altered due to pregnancy, obesity or use of oral contraceptives. 1 Measurement of free or bioavailable testosterone is technically challenging and not widely available; therefore, the free androgen index (FAI) and calculated free testosterone (CFT) using equations such as the Vermeulen, Sodergard, Nanjee-Wheeler and Ly-Handelsman equations2–5 are commonly used to estimate free testosterone in clinical practice. Recent clinical practice guidelines on the use of testosterone therapy in hypogonadal men also advocate the use of CFT in the diagnosis of hypogonadism. 6
The effects of using different testosterone assays on the estimated CFT have been studied in detail, 7 and reference measurement procedures and standard reference materials are available for testosterone to enable traceability of routine laboratory methods to higher order reference methods and materials.9–12 This has had some success in standardizing testosterone measurement across laboratories, although not all manufacturers of commercial assays have been involved and further improvement is still required. 8 However, only limited attempts have been made to standardize other components of the CFT equations, such as SHBG. The aim of this study was to compare SHBG results produced using four different immunoassay analysers commonly used in Clinical Biochemistry laboratories in the UK (Abbott Architect, Roche, Beckman and Siemens) and assess how differences in results would affect free testosterone calculated using the Vermeulen equation for CFT.
Methods
Surplus serum samples (40 male, 40 female) collected into Becton Dickinson SST II Advance serum separator tubes were obtained from the Clinical Biochemistry laboratory after routine analysis, anonymized and split into four different tubes. The samples were chosen randomly from discarded samples, then were frozen at −80°C and shipped frozen to participating laboratories where they were stored at −20°C prior to analysis.
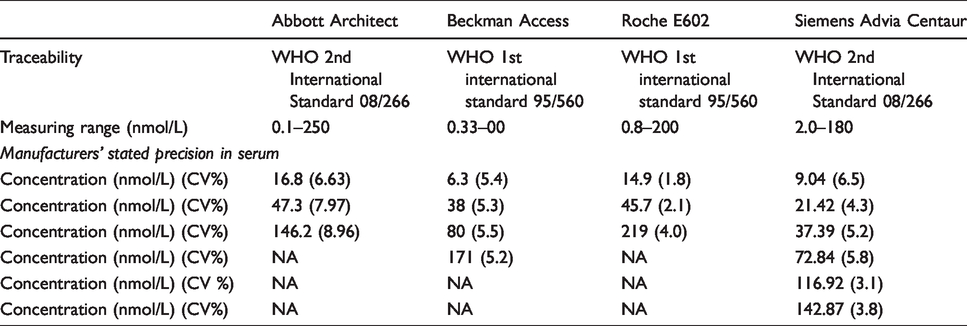
Samples were analysed using four different immunoassay platforms: Abbott Architect, Beckman Access, Roche E602 and Siemens Advia Centaur. All analysers use a two-step (sandwich) chemiluminescent immunoassay. An overview of the performance characteristics of each assay is provided in Table 1.
Characteristics of each assay used in the study.
Free testosterone was calculated for each sample using the Vermeulen equation (www.issam.ch/freetesto.htm), with 10 nmol/L testosterone used for male samples and 1.5 nmol/L testosterone used for female samples. This equation was chosen instead of the FAI due to the inaccuracy of FAI at extremes of SHBG concentrations 13 and because this is reported to be the most robust method for CFT calculation. 14 These concentrations for testosterone were chosen, as they are close to the lower limit of the reference range for male patients and the upper limit of the reference range for female patients used in the laboratory at the Wythenshawe Hospital.15,16 Albumin was set at 40 g/L for all samples.
Results
Comparison of SHBG concentrations
As there is no higher order reference method available for SHBG, results produced from each analyser were compared with the SHBG result obtained from each of the other analysers and also to the mean result obtained from all four analysers. The details of all the results are shown in supplementary data Table 1. One sample was mis-sampled on the Roche E602 and was excluded from data analysis as there was insufficient sample for repeat analysis. As the data showed a non-parametric distribution, the Wilcoxon signed-rank test was used to compare the results obtained from each analyser. The results for all combinations of analysers were significantly different, with P < 0.0001 in all cases. The Beckman Access and the Siemens Advia Centaur gave results closest to the overall mean, with Passing-Bablock regression equations of Beckman [SHBG (nmol/L) = 1.02 × mean SHBG (nmol/L)−0.01] and Siemens Advia Centaur [SHBG (nmol/L) = 1.00 × mean SHBG (nmol/L)−0.57] (Figure 1). The Abbott Architect gave the lowest SHBG results (Abbott Architect SHBG = 0.9 × mean SHBG (nmol/L) +0.85), whereas the Roche E602 gave the highest results (Roche E602 SHBG (nmol/L) = 1.09 × mean SHBG (nmol/L)−0.66). Bland-Altman analysis showed the same trend; the Abbott Architect had a mean bias of −5.015 nmol/L from the overall mean, Siemens Advia Centaur had a mean bias of −0.457 nmol/L, the mean bias for Beckman was 1.156 nmol/L and Roche showed a mean bias of 4.414 nmol/L (Figure 2).
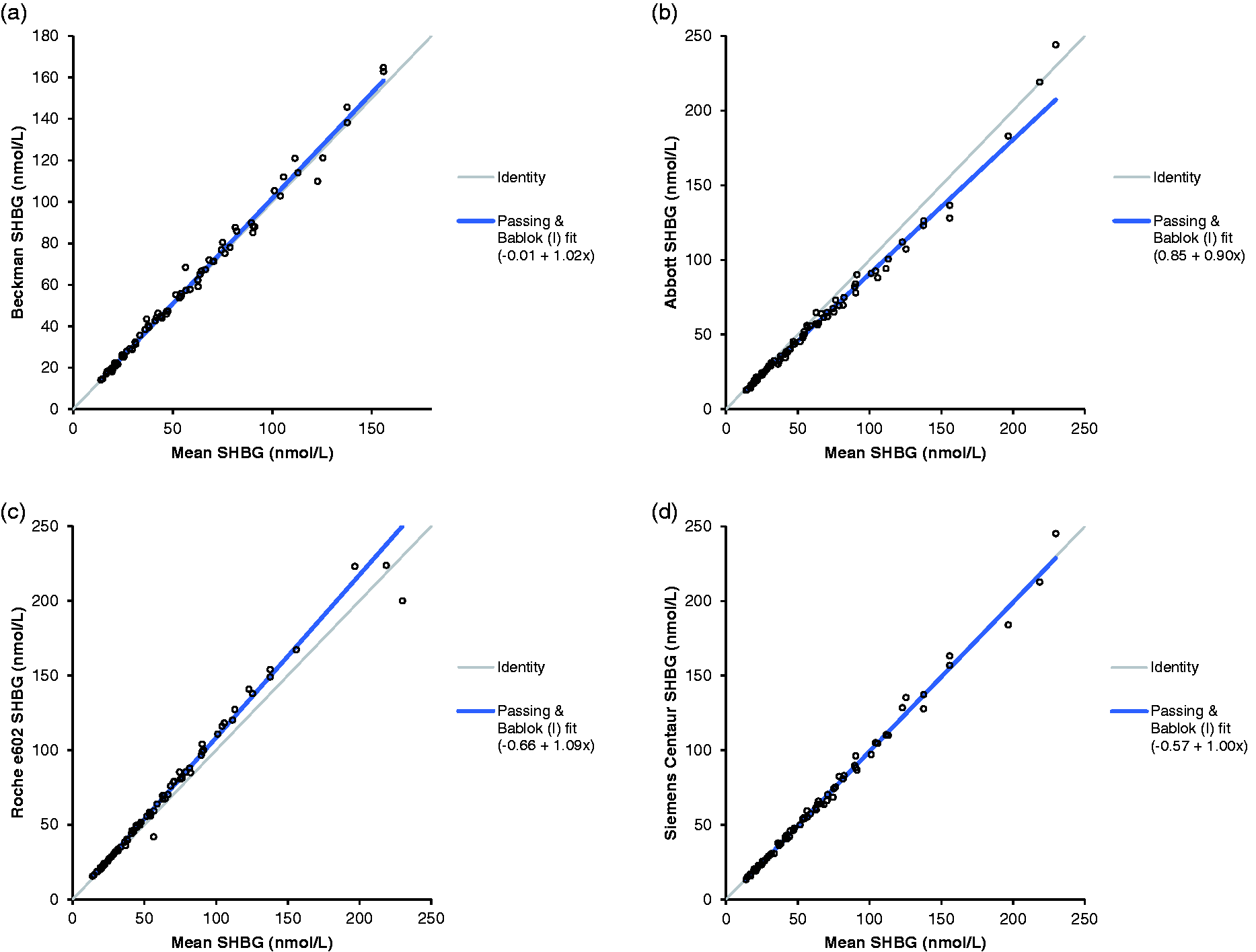
Passing-Bablock analysis of SHBG concentrations from each analyser compared with the mean results from all four analysers. (a) The results from the Beckman Access, (b) Abbott Architect and (c) Roche E602. (d) The results from Siemens Advia Centaur.
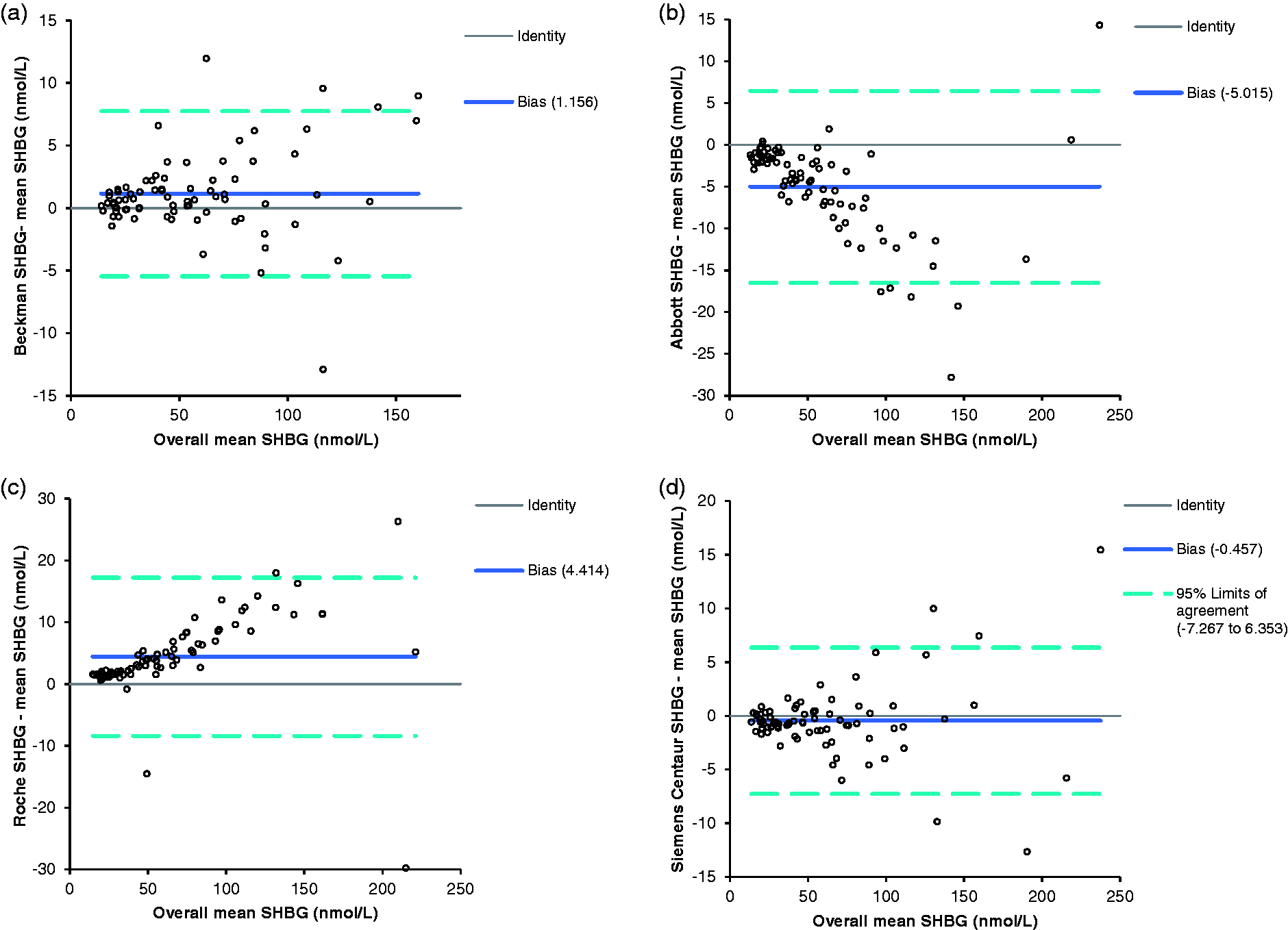
Bland–Altman plot of SHBG concentrations from each analyser compared with the mean results from all analysers. (a) The results from the Beckman Access, (b) Abbott Architect and (c) Roche E602. (d) The results from Siemens Advia Centaur. The solid line shows the mean difference between results, the broken lines show ±1.96 SD from the mean.
Comparison of CFT
The Vermeulen equation was used to calculate the free testosterone for all 80 samples. The Wilcoxon signed-rank test was used to compare the free testosterone calculated using the SHBG results from each of the analysers, and the results were significantly different for all combinations of analysers (P < 0.0001). As the males and females had significantly different CFTs, Bland-Altman analysis was carried out separately for the two groups (Figures 3 and 4). The Abbott results were the furthest away from the mean for both sets of data, with results above the overall mean, whereas the Roche gave the lowest results. Beckman Access also gave results below the mean, whereas the Siemens Centaur results were above the overall mean, but both analysers gave results much closer to the mean than the Roche or the Abbott (Figures 3 and 4).
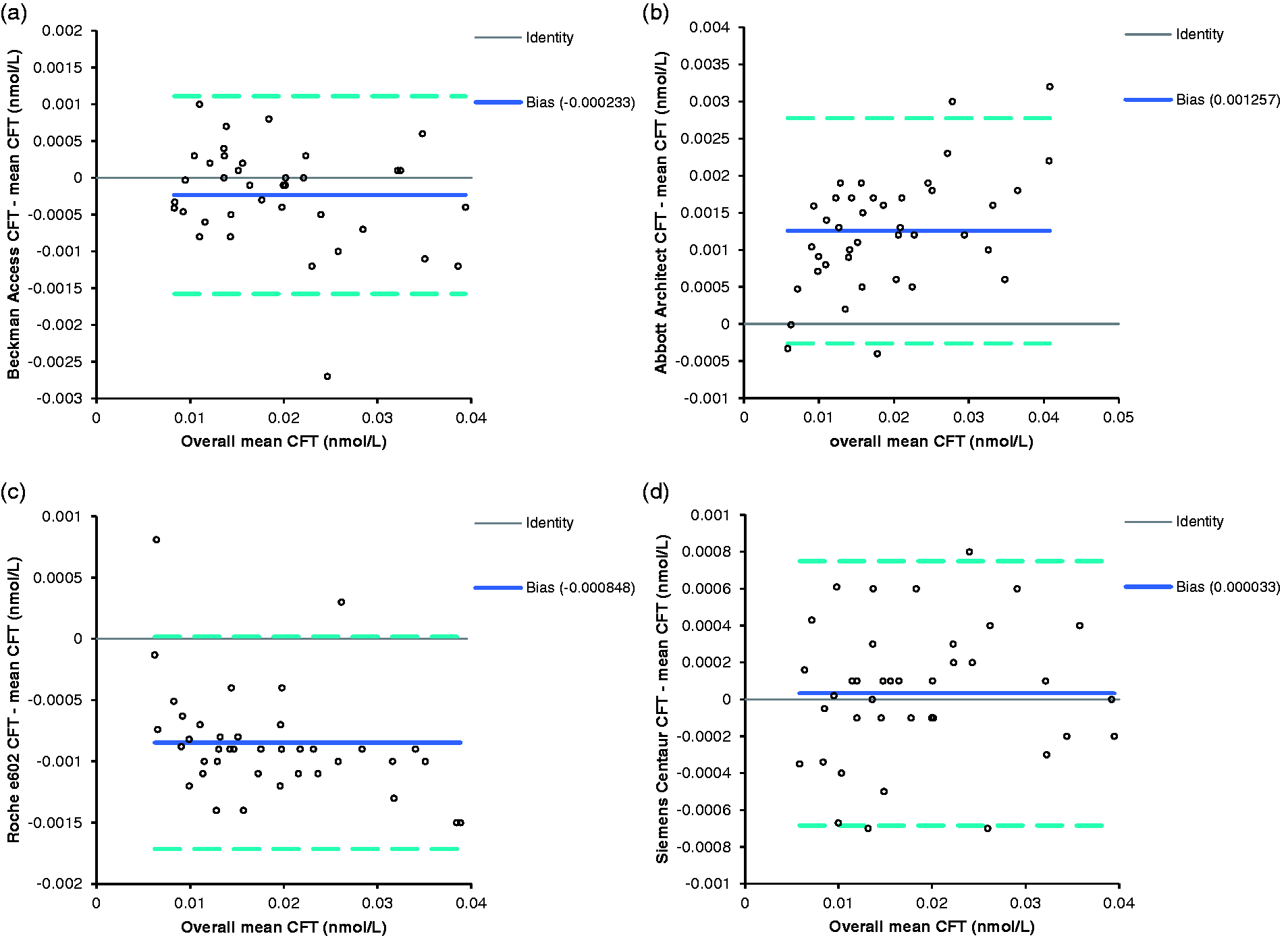
Bland–Altman plot of CFT in female samples calculated using the SHBG from each analyser compared with the CFT calculated using the mean results from all analysers. (a) The results from the Beckman Access, (b) Abbott Architect and (c) Roche E602. (d) The results from Siemens Advia Centaur. The solid line shows the mean difference between results, the broken lines show ±1.96 SD from the mean.
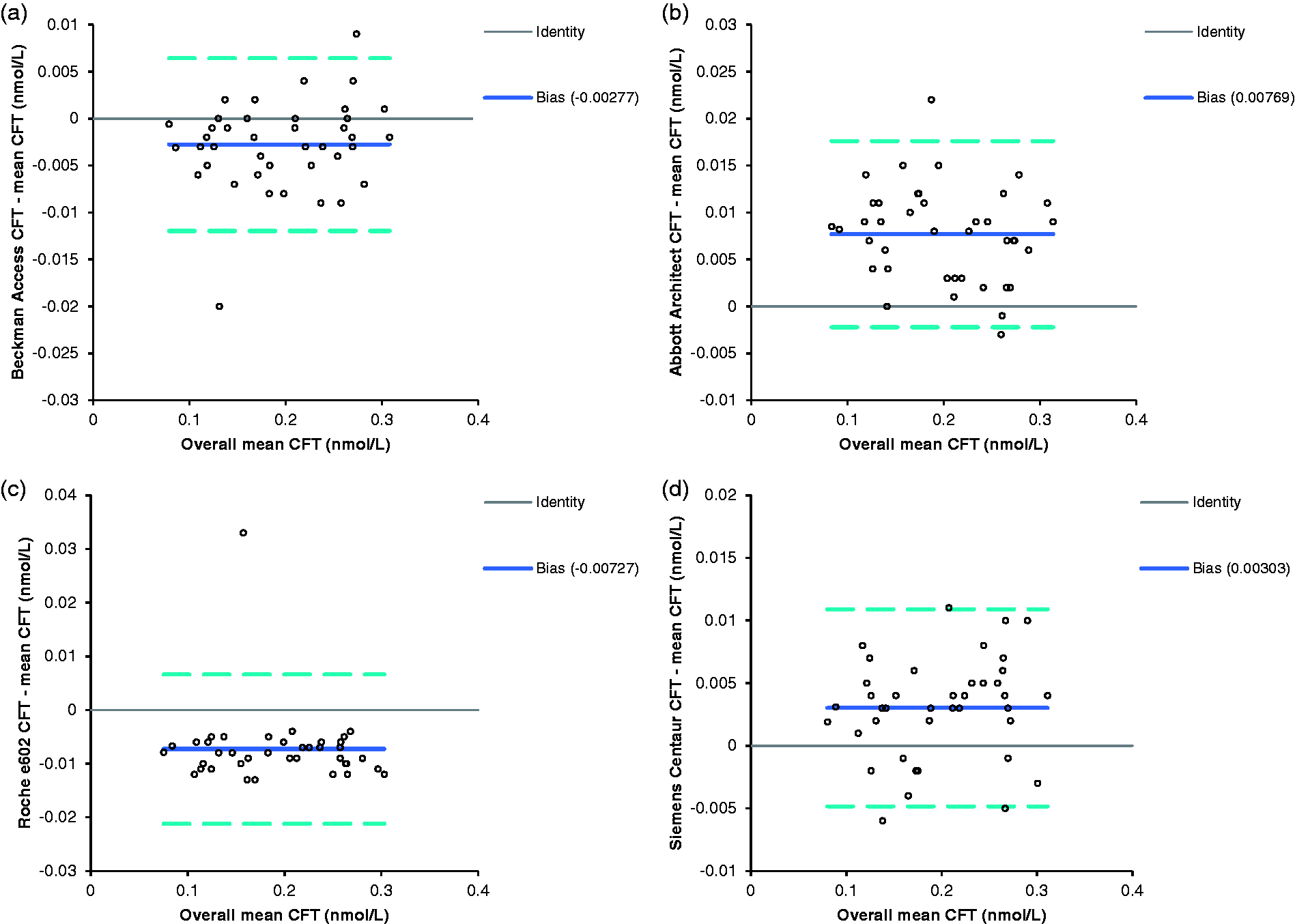
Bland–Altman plot of CFT in male samples calculated using the SHBG from each analyser compared to the CFT calculated using the mean results from all analysers. a) The results from the Beckman Access, (b) Abbott Architect and (c) Roche E602. (d) The results from Siemens Advia Centaur The solid line shows the mean difference between results, the broken lines show ±1.96 SD from the mean.
Discussion
CFT is commonly used in clinical practice to diagnose hypogonadism in men and hyperandrogenism in women. Testosterone, SHBG and albumin are used in the equations to derive the free testosterone, and treatment decisions are based on the results; however, there is little appreciation of the effect of differences in SHBG assays on the calculation. This study has shown that commonly used assays can give different results for SHBG, even when traceable to the same international standard. The Abbott Architect and Siemens Advia Centaur assays are both traceable to the WHO 2nd International Standard 08/266, but there is a mean difference of 4.558 nmol/L between the results. The Roche E602 and Beckman Access are both traceable to the WHO 1st International Standard 95/560, but there is a mean difference of 4.2% between their results. In contrast, although the Beckman Access and Siemens Advia Centaur assays are traceable to difference in international standards, the mean difference between their results is only 3.5%. The difference between the Abbott Architect and the Roche E602 results are the greatest, with the Roche E602 results being a mean 16.7% higher than the Abbott Architect results.
The results of this study reflect a snapshot in time of the relative performance of four routine methods, run on individual analysers in different departments. A drawback of this study is the fact that we looked at data from individual analysers, and these may not necessarily be representative of the performance of that manufacturers’ method across a range of laboratories. Data from external quality assessment schemes can give a better representation of overall method performance. RIQAS data from the period of study (nine distributions) shows Roche have a positive bias of 0.20% compared with the overall mean, whereas Abbott, Siemens and Beckman had negative biases of 0.75%, 1.26% and 6.02%, respectively (with kind permission of RIQAS). UKNEQAS data show that there is a 10–15% spread of bias within individual method groups (Personal communication, from UKNEQAS). This may account for the closer agreement between the Beckman, Siemens and Abbott assays seen in the EQA scheme than we found in our study. Nevertheless, EQA data confirm our finding that Roche has a positive bias compared with the other methods.
This difference is also reflected in the CFT. There is a mean difference of 10.4% in males and 11.2% in females between CFT calculated using the SHBG from the Roche and Abbott, with Roche SHBG giving lower CFT than Abbott. Figures 3 and 4 show the Bland-Altman plots of the CFT calculated using the SHBG results from each analyser compared with the CFT calculated using the mean SHBG. Moving between analysers, for instance if a patient was being followed up in Primary and Secondary care and the care providers using different laboratories, or if a laboratory changes immunoassay platform could cause diagnostic confusion if the same reference ranges for CFT were used.
Although these differences may seem small, due to the difficulty in establishing reference ranges for CFT, laboratories may quote ranges determined using SHBG and testosterone concentrations obtained using assays different to those used in their own laboratory. Because of the differences in SHBG between assays and the subsequent difference in CFT, it would be expected that the use of a generic reference range for CFT would result in an altered clinical interpretation between assays. While the difference in the proportion of patients diagnosed with hypogonadism or hyperandrogenism based on CFT was consistent with the bias of the various SHBG assays, the difference was not found to be clinically significant. This may have been due to low total sample numbers and using randomly selected samples in which the proportion of samples that would have an SHBG concentration resulting in a CFT near the limits of the reference intervals and so more likely to yield a different clinical interpretation in the various assays when using a generic reference range is small. Further and larger studies are required.
The specific SHBG assay used may cause the difference between male patients receiving or being denied testosterone replacement, or results supporting or being inconsistent with the diagnosis of PCOS in a female patient. Untreated male hypogonadism causes symptoms such as erectile dysfunction, infertility, osteoporosis, fatigue and gynaecomastia, and untreated PCOS can cause oligo or amenorrhoea, infertility, hirsutism and weight gain. Misclassification of individual patients can therefore have a big impact on the quality of life of these patients. In conclusion, we recommend the use of analyser-specific reference ranges for CFT until such a time that standardization of SHBG measurement is improved to avoid inequality of access to treatment and diagnostic confusion solely due to the analyser used to measure SHBG.
Supplemental Material
ACB888549 Supplemetal Material - Supplemental material for Ramifications of variability in sex hormone-binding globulin measurement by different immunoassays on the calculation of free testosterone
Supplemental material, ACB888549 Supplemetal Material for Ramifications of variability in sex hormone-binding globulin measurement by different immunoassays on the calculation of free testosterone by Joanne Adaway, Brian Keevil, Annmarie Miller, Phillip J Monaghan, Nicola Merrett and Laura Owen in Annals of Clinical Biochemistry
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
JA.
Contributorship
BK, LO and JA conceived the project and sourced the samples. AM, NM, JA and PJM analysed the samples. JA wrote the first draft of the article. All authors reviewed and edited the article and approved the final version of the article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
