Abstract
Objective
The aim of this study was to identify arterial blood gas (ABG) abnormalities, with a focus on a high anion gap (AG) metabolic acidosis and evaluate outcomes in coronavirus disease 2019 (COVID-19) patients admitted to the ICU.
Methods
A retrospective, observational study was conducted in a tertiary hospital in Cape Town during the first and second COVID-19 waves. Age, gender, sodium (Na), potassium (K), chloride (Cl), bicarbonate (HCO3std), pH, partial pressure of carbon dioxide (pCO2), creatinine, estimated glomerular filtration rate (eGFR), lactate levels and ABG results were obtained. The Pearson χ2 test or Fisher exact test and the Wilcoxon rank-sum test were used to compare mortality and survival. To identify factors associated with non-survival, a multivariable model was developed.
Results
This study included 465 patients, 226 (48%) of whom were female. The sample population’s median (IQR) age was 54.2 (46.1–61.3) years, and 63% of the patients died. ABG analyses found that 283 (61%) of the 465 patients had alkalosis (pH ≥ 7.45), 65 (14%) had acidosis (pH ≤ 7.35) and 117 (25%) had normal pH (7.35–7.45). In the group with alkalosis, 199 (70.3%) had a metabolic alkalosis and in the group with acidosis, 42 (64%) had a metabolic acidosis with an increased AG of more than 17. Non-survivors were older than survivors (56.4 years versus 50.3 years, p < .001).
Conclusion
Most of the COVID-19 patients admitted to the ICU had an alkalosis, and those with acidosis had a much worse prognosis. Higher AG metabolic acidosis was not associated with patients’ characteristics.
Introduction
The coronavirus disease 2019 (COVID-19) pandemic has caused over 537,880,995 infections and 6,315,850 deaths globally as of 17 June 2022. 1 South Africa has been the most affected country in Africa with 3,983,675 infections and 101,584 deaths to date. 1 As COVID-19 progressed, many patients required intensive care unit (ICU) admission leading to frequent arterial blood gas (ABG) analysis. The most common ABG abnormality found in ICU admissions is usually a metabolic acidosis. 2 We have previously described that severely ill COVID-19 patients in our setting often presented with a metabolic alkalosis.2,3 Tropism of the virus for the lungs and kidneys may theoretically lead to frequent acid–base alterations due to pneumonia and acute kidney injury (AKI), respectively.4–6 We postulated that this may be due to activation of the renin–angiotensin–aldosterone system (RAAS) due to the virus binding to the angiotensin-converting enzyme-2 (ACE2) receptor. 7 Although there are few articles on ABG abnormalities in COVID-19, some studies have corroborated this finding of a metabolic alkalosis in ICU admissions.6,8,9 In fact, metabolic acidosis is associated with an increased mortality in these patients.6,10–12 The anion gap (AG) is described as the difference between measured anions and cations and is determined in metabolic acidosis to try to determine the underlying cause. 13 An increased AG metabolic acidosis is due to an accumulation of unmeasured anions such as lactate, ketones and those found in renal failure or poisoning. 14 There is a paucity of studies on ABG abnormalities in COVID-19 ICU admissions, and to our knowledge, Bezuidenhout et al. is the only study in Africa looking at ABG abnormalities in these patients. 2 No previous study has specifically examined AG in COVID-19. The aim of this study was to determine the ABG abnormalities and specifically investigate high AG metabolic acidosis and outcomes in COVID-19 ICU admissions during the first and second waves in a tertiary hospital in Cape Town, South Africa.
Materials and methods
Study setting
This retrospective cohort study took place at Tygerberg Hospital, a 1380-bed tertiary hospital in Cape Town, South Africa. The hospital provides tertiary services to approximately 3.5 million people from the Western Cape Province.
Study population
The study population consisted of all patients with a laboratory diagnosis of COVID-19 (admitted to the ICU from 27 March 2020 to 29 October 2020 and 4 November 2020 to 10 February 2021). This coincided with the first and second waves of COVID-19 experienced in South Africa. All the patients were over the age of 18 years and were included irrespective of possible underlying disorders.
Analytical methods
Admission ABG analyses were performed on a Gem Premier 3500™ (Instrumentation Laboratory, Werfen Group, USA) point of care ABG analyser used in the ICU. The pCO2 and pH were measured using a potentiometric method, and pO2 and lactate were measured amperometrically. The bicarbonate (HCO3) and base excess (BE) are calculated parameters. Regular internal quality control using the on-board Intelligent Quality Management® system was performed. Admission serum electrolyte and renal function tests were performed on the Roche Cobas® 6000 (Roche Diagnostics, Mannheim, Germany) platform. Sodium (Na) and potassium (K) were determined using indirect ion-selective electrode potentiometry, while urea and creatinine determination was based on kinetic and enzymatic assays, respectively. The laboratory is ISO15189 accredited by the South African National Accreditation Services (SANAS), result quality was validated with internal quality control and the laboratory participates in external quality control schemes. The estimated glomerular filtration rate (eGFR) was calculated using the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation without correction for race, a calculation recommended by the Kidney Disease Improving Global Outcomes (KDIGO) report. 15 AG was determined using the formula Na – (Cl + HCO3). Lactate levels, eGFR and diabetes status were assessed to find a possible cause for increased AG.
Clinical and laboratory data collection
Due to infection control risk, data were captured prospectively using images of written clinical notes at the bedside, which were securely stored electronically, and clinical data (demographic data, including age, sex, comorbidities and patient outcome) were entered remotely by a data-capturer into a Redcap® database. Admission vital signs, including blood pressure (BP), heart rate (HR), respiratory rate (RR) and pulse oximeter oxygen saturation, were also collected. Survivor was defined as a patient discharged from ICU and hospital alive. Laboratory and ABG data were collected retrospectively. The laboratory results were exported from the National Health Laboratory Service (NHLS) Laboratory Information System (TrakCare® Lab Enterprise) into the database. Data were checked to ensure quality and reliability.
Statistical analysis
Statistical analyses were performed using Stata (V.16, Stata Corp, College Station, Texas, USA) and R (V, 4.1.0, R Core Team) with R Studio (V.1.3, R Studio Team) statistical software. Continuous variables were expressed as mean and SD for normally distributed data and median with inter-quartile range for skewed data. We expressed categorical variables as frequencies and percentages and compared their parameters among those who were discharged and died. To compare between mortality and survival, we used the Pearson χ2 test or Fisher exact test where appropriate for categorical variables and the Wilcoxon’s rank-sum test for continuous variables. A multivariable model was developed for demographics, comorbidities, clinical symptoms and laboratory parameters to identify factors independently associated with non-survival. Factors that were associated with non-survival at p < .15 in the unadjusted model were included in a multivariable model. Adjusted risk ratios at p < .05 and their 95% CIs were used to identify independent measures associated with non-survival.
Results
Study population
Socio-demographic and clinical characteristics associated with mortality among COVID-19 patients admitted to ICU.
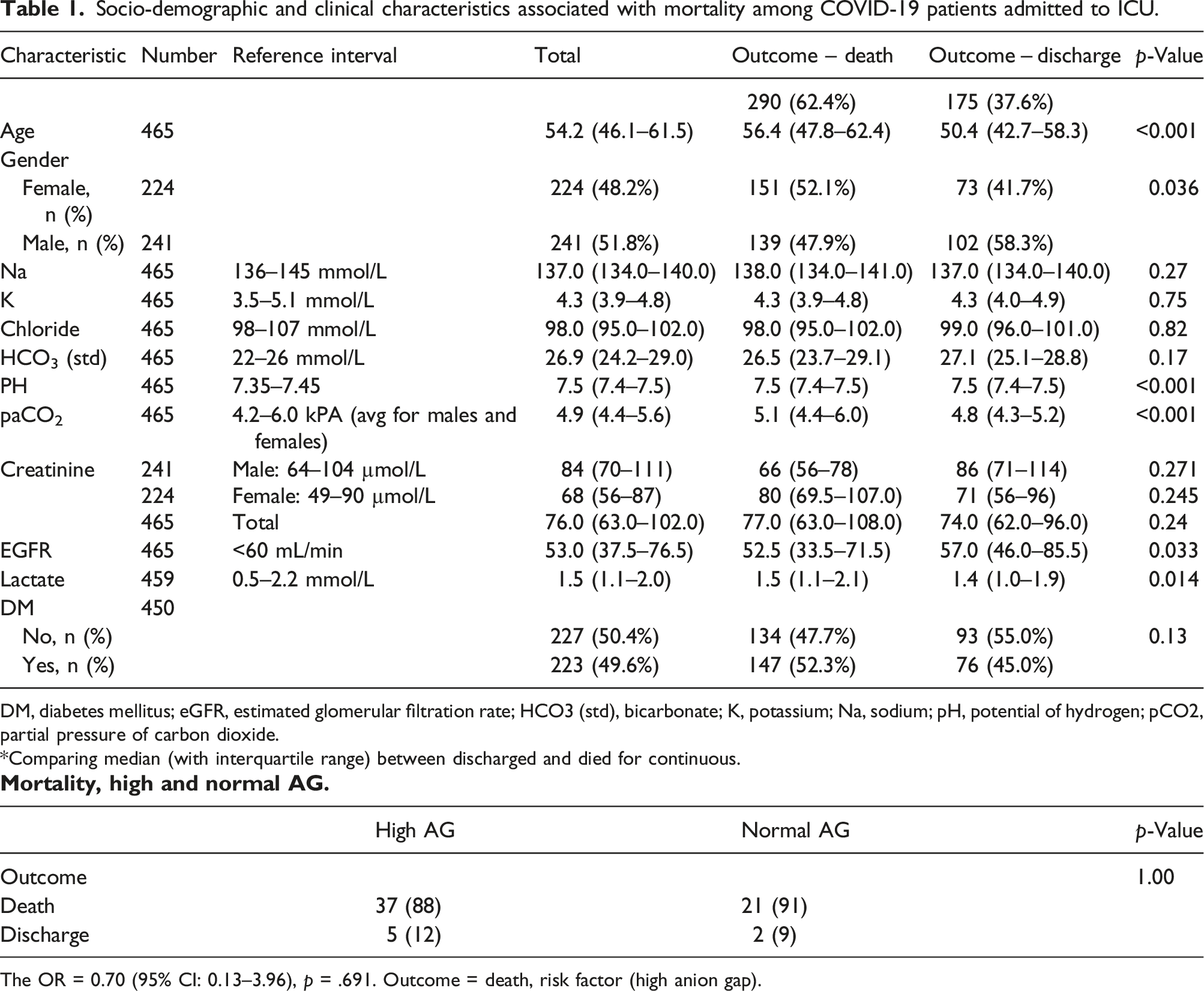
DM, diabetes mellitus; eGFR, estimated glomerular filtration rate; HCO3 (std), bicarbonate; K, potassium; Na, sodium; pH, potential of hydrogen; pCO2, partial pressure of carbon dioxide.
*Comparing median (with interquartile range) between discharged and died for continuous.
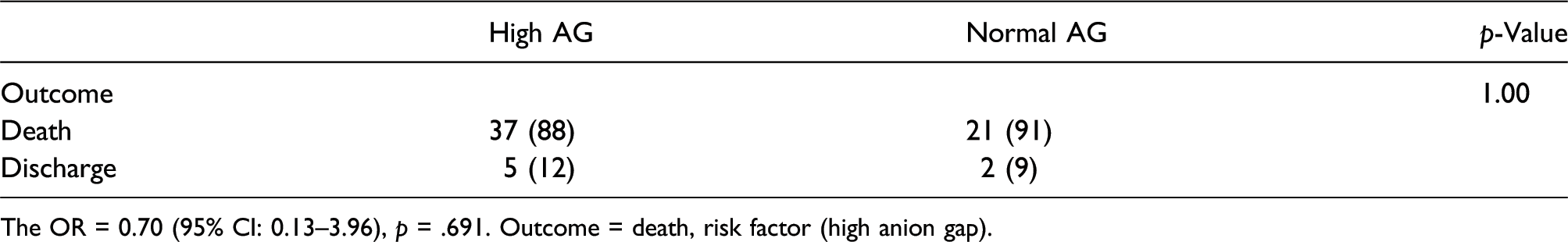
The OR = 0.70 (95% CI: 0.13–3.96), p = .691. Outcome = death, risk factor (high anion gap).
Arterial blood gas (ABG) findings
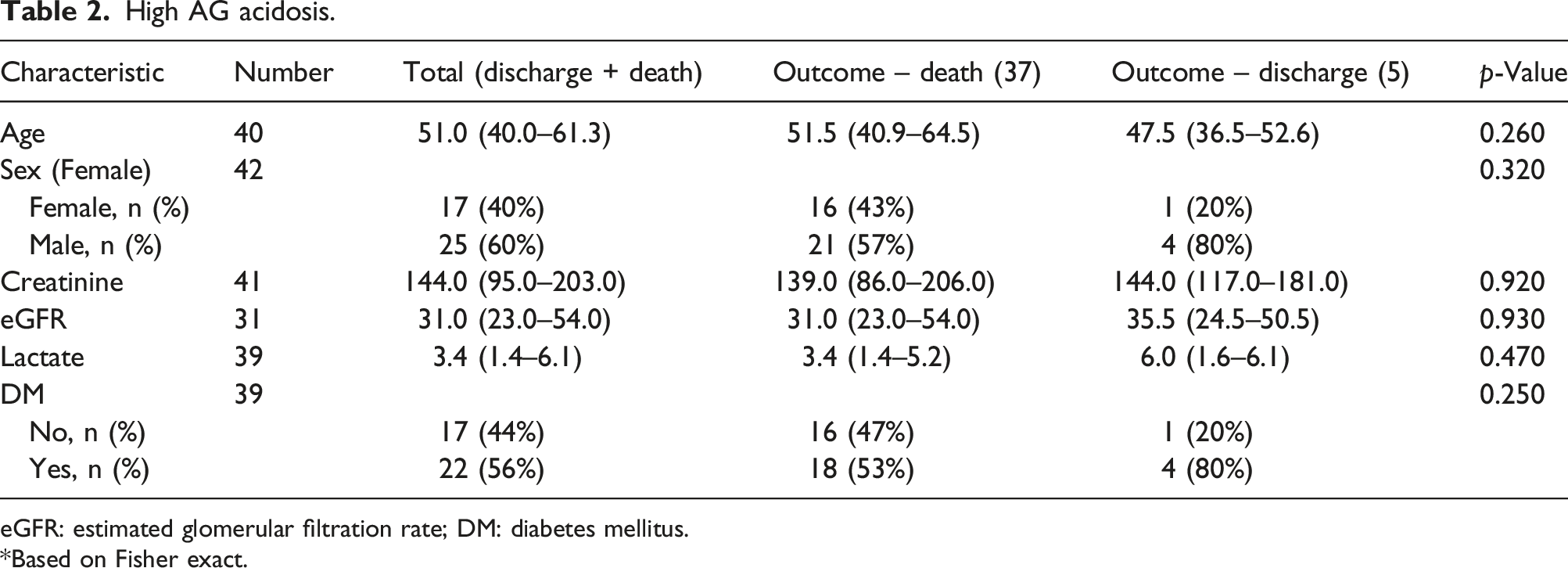
The ABG findings for this cohort are shown in Figure 1. Of the 465 patients in this cohort, 283 (61%) had an alkalosis (pH ≥ 7.45), 65 (14%) had an acidosis (pH ≤ 7.35) and 117 (25%) had a normal pH (pH 7.35–7.45). In the group with alkalosis, 199 (70.3%) had a metabolic alkalosis. In the metabolic acidosis group, 42 (64%) had an increased AG of more than 17. Table 2 shows the characteristics of those patients with acidosis and increased AG. Of these 42 patients, 37 (88%) did not survive. To attempt to determine the cause of the raised AG, lactate levels, diabetes status (as a proxy for ketones) and eGFR were assessed. Lactate levels were increased in the total group (median 3.4 mmol/L) with no significant difference between survivors and non-survivors (median 6 mmol/L versus 3.4 mmol/L, p = .455). The eGFR was decreased in the total group (median 31 mL/min) with no significant difference between survivors and non-survivors (median 35.5 mL/min versus 31 mL/min, p = .903). Similarly, although 57% of this group was diabetic, there was no significant difference between survivors and non-survivors. Acid–base status of COVID-19 patients admitted to ICU. High AG acidosis. eGFR: estimated glomerular filtration rate; DM: diabetes mellitus. *Based on Fisher exact.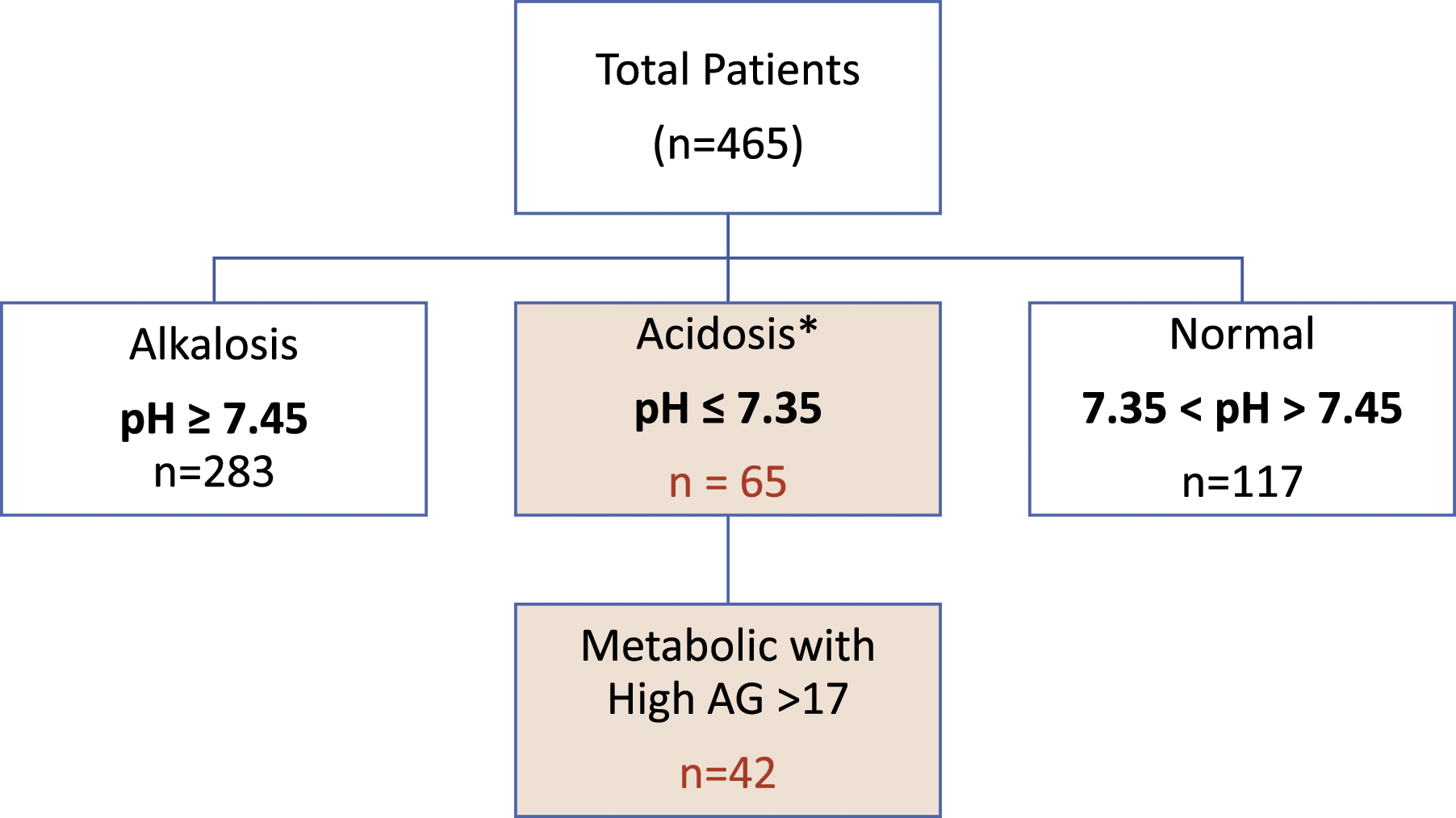
Discussion
ABG disturbances in ICU patients are common. In keeping with previous literature, most patients (61%) in our cohort presented with an alkalosis, of which the majority was of a metabolic origin, and a minority had metabolic acidosis. According to our findings, those who survived ICU admission had a higher pH, while those who died were acidotic. This is a common finding as shown in previous studies.2,3 Alkalosis is uncommon in ICU patients who usually present with a metabolic acidosis. The risk exposure and clinical outcome are multifactorial with lactic acidosis, sepsis and multiorgan failure contributing. When closely examining acidosis and an increased AG in our study cohort, our findings demonstrate what has been previously shown before, namely, that there was a worse prognosis with 88% non-survival with acidosis. When examining the possible cause for a raised AG, data show that the lactate levels were raised and eGFR was decreased. However, this was not significantly different between the groups although the eGFR was slightly lower in non-survivors than in survivors.
A higher likelihood of alkalosis and a worse prognosis in acidosis has been described in COVID-19 patients. 6 An explanation for the metabolic alkalosis in these patients may be due to the SARS-CoV-2 virus binding to the ACE2 receptor and affecting the RAAS. 7 Lakhani et al. studied 80 COVID-19 patients admitted to the ICU in India. 10 They performed serial ABG on admission, day five and day 10. 10 Most of their cohort had mixed ABG disorders and mixed respiratory, and metabolic acidosis was associated with non-survival. 10 Furthermore, Pal et al. found that acidosis predicted mortality in COVID-19 patients with diabetic ketoacidosis. 11 However, Alfano et al. studied 211 baseline ABG of COVID-19 patients admitted to ICU in Italy and found that metabolic alkalosis was the main abnormality among this group of patients. They postulated that this was due to stimulation of the RAAS. 6 Our study showed similar findings, with metabolic acidosis being uncommon and associated with a worse prognosis. A further study by Alfano et al. found that 41% of patients admitted to hospital with COVID-19 had hypokalaemia, and again this was postulated to be due to activation of the RAAS. 16 In our cohort, irrespective of the pH, potassium levels were not significantly abnormal.
An increased AG has been shown to be associated with all-cause mortality in critically ill patients. Cheng et al. investigated the effect of AG on the prognosis of critically ill COVID-19 patients with acute kidney injury and found that high AG was associated with increased risk of mortality at 30, 90 and 365 days. 17 Similar findings have been described in patients with disseminated intravascular coagulation (DIC) 18 and pancreatitis. 19 Dafal et al. described an increased length of hospital stay and higher mortality among ICU patients with increased AG. 20
There is a paucity of studies on AG in patients with COVID-19. A systematic review conducted by Papadopoulos et al. among diabetes-related COVID-19 patients found sufficient evidence demonstrating the association between raised AG and mortality. 21
Our study conducted an analysis of all consecutive patients admitted to a single ICU with the use of baseline laboratory data prior to ICU interventions. Our study has several limitations. The study originated from a single centre that analysed baseline ABG and laboratory results on ICU admission. Secondly, administration of oxygen therapy (O2) due to hypoxaemic state and or assisted ventilation might have affected ABG results and therefore introduced bias into the study findings. Additionally, when looking for causes of a raised AG, we used diabetic status as a proxy for ketone levels. However, our study has numerous strengths. We had a large sample size and examined all consecutive patients admitted to the ICU prior to medical interventions over two COVID-19 waves. To our knowledge, this is the first study on raised AG in severely ill COVID-19 patients in Africa.
Conclusion
Our study found that most of the seriously ill COVID-19 patients admitted to the ICU had an alkalotic state and that those with acidosis had a worse prognosis. We postulate that patients with COVID-19 admitted to ICU with a high AG metabolic acidosis are at increased risk of poor outcome and therefore need a more aggressive clinical approach to improve outcome.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was carried out with support of The COVID-19 Africa Rapid Grant Fund supported under the auspices of the Science Granting Councils Initiative in Sub-Saharan Africa (SGCI) and administered by South Africa’s National Research Foundation (NRF) in collaboration with Canada’s International Development Research Centre (IDRC), the Swedish International Development Cooperation Agency (Sida), South Africa’s Department of Science and Innovation (DSI), the Fonds de Recherche du Québec (FRQ), the United Kingdom’s Department of International Development (DFID), United Kingdom Research and Innovation (UKRI) through the Newton Fund and the SGCI participating councils across 15 countries in sub-Saharan Africa.
Ethical approval
The investigators obtained ethical approval and waiver of consent from the Health Research Ethics Committee of the Faculty of Medicine and Health Sciences, Stellenbosch University and Research Committee of the Tygerberg Hospital, Ethics approval number N20/04/002_COVID-19.
Guarantor
PSN.
Contributorship
Conceptualization: AEZ and LNS; study design: AEZ and PSN. data collection: BWA, VN, AEZ and LNS; statistical analyses: LNS and AY; draft manuscript: AEZ, LNS, OW and PSN; final drafting of the manuscript: AEZ, PSN, BWA, CK, EMI, UL, RE, TM, LNS, AY, JLT, OW and AZ. All authors approved the last version of the manuscript.
