Abstract
Background
During the outbreak of coronavirus disease 2019 (COVID-19), many studies have investigated laboratory biomarkers in management and prognostication of COVID-19 patients, however to date, few have investigated arterial blood gas, acid–base and blood pressure patterns. The aim of the study is to assess the arterial blood gas and acid–base patterns, blood pressure findings and their association with the outcomes of COVID-19 patients admitted to an intensive care unit.
Methods
A single-centre retrospective, observational study in a dedicated COVID-19 intensive care unit in Cape Town, South Africa. Admission arterial blood gas, serum electrolytes, renal function and blood pressure readings performed on COVID-19 patients admitted between 26 March and 2 June 2020 were analysed and compared between survivors and non-survivors.
Results
A total of 56 intensive care unit patients had admission arterial blood gas performed at the time of intensive care unit admission. An alkalaemia (pH > 7.45) was observed in 36 (64.3%) patients. A higher arterial pH (median 7.48 [interquartile range: 7.45–7.51] versus 7.46 [interquartile range: 7.40–7.48], P = 0.049) and partial pressure of oxygen in arterial blood (median 7.9 kPa [interquartile range: 7.3–9.6] versus 6.5 kPa [interquartile range: 5.2–7.3], P = <0.001) were significantly associated with survival. Survivors also tended to have a higher systolic blood pressure (median: 144 mmHg [interquartile range: 134–152] versus 139 mmHg [interquartile range: 125–142], P = 0.078) and higher arterial HCO3 (median: 28.0 mmol/L [interquartile range: 25.7–28.8] versus 26.3 mmol/L [interquartile range: 24.3–27.9], P = 0.059).
Conclusions
The majority of the study population admitted to intensive care unit had an alkalaemia on arterial blood gas. A higher pH and lower partial pressure of oxygen in arterial blood on arterial blood gas analysis were significantly associated with survival.
Introduction
Since the novel coronavirus disease 2019 (COVID-19) was declared a pandemic, the impact globally has been unparalleled. At the time of writing this manuscript, there are more than 18.3 million confirmed cases with over 690 000 deaths worldwide, with more than 188 countries affected. 1 The first cases of atypical pneumonia were described in Wuhan, China, late 2019. 2 The World Health Organization (WHO) was notified January 2020 and the novel coronavirus 2 (CoV-2) was identified as the causative agent shortly thereafter. 3 , 4
As the angiotensin-converting enzyme 2 (ACE2) is the port of entry for SARS-CoV-2, 5 the renin angiotensin system (RAS) is affected in COVID-19. When SARS-CoV-2 enters the cell, there is downregulation of ACE2, with upregulation of the classic RAS pathway leading to angiotensin II and aldosterone production. Both angiotensin II and aldosterone affect the renal handling of hydrogen (H+) and bicarbonate (HCO3), which may cause acid–base disturbances.6–8
As the severity of COVID-19 progresses, many patients require intensive care unit (ICU) admission leading to frequent arterial blood gas (ABG) analysis. Various laboratory results have been identified as risk predictors that may assist in disease staging, monitoring and treatment and prognostication of COVID-19 patients. 2 ,8–17
However, the majority of these studies have focused on biochemical and haematological laboratory parameters with very little data available on ABG analysis.18–20 Also, there is a paucity of data on the effects of SARS-CoV-2 on ACE2 and aldosterone and the ensuing acid–base abnormalities. We therefore aimed to investigate ABG results, specifically acid–base findings, along with electrolytes, renal function tests and the possible effects of RAS upregulation on blood pressure (BP) and electrolytes of COVID-19 patients admitted to a dedicated ICU.
Materials and methods
Study setting
This single-centre, retrospective, observational study was performed at Tygerberg Academic Hospital, a referral hospital in Cape Town, South Africa. The institution provides a tertiary service for a large population and its bed capacity and critical care services were escalated in anticipation of the COVID-19 surge. 21
Patients
The study population consisted of the first 56 consecutive patients with laboratory diagnosed COVID-19 admitted to the ICU between 26 March 2020 and 2 June 2020. All the patients were over the age of 18 years and were included irrespective of possible underlying disorders. The ABG results were obtained from the testing at admission to ICU and performed on a point of care analyser.
Analytical methods
Admission ABG analyses were performed on a Gem Premier 3500™ (Instrumentation Laboratory, Werfen Group, USA) a dedicated point of care ABG analyser in the ICU. A potentiometric method is used to calculate partial pressure of carbon dioxide (pCO2) and pH, while partial pressure of oxygen (pO2) and lactate is determined amperometrically. HCO3 and base excess (BE) are calculated parameters. The instrument is subject to regular internal quality control with the on-board Intelligent Quality Management® system.
Admission serum electrolyte and renal function tests were performed on the Roche Cobas® 6000 (Roche Diagnostics, Germany) platform in an accredited laboratory. Sodium and potassium are determined by indirect ion-selective electrode potentiometry while urea and creatinine are based on kinetic and enzymatic assays, respectively.
Clinical and laboratory data collected
Laboratory and ABG data were collected retrospectively, along with demographic data, including age, sex, comorbidities and patient outcome. Admission vital signs, including BP, heart rate, respiratory rate and pulse oximeter oxygen saturation, were also collected. Survivor was defined as a patient discharged from ICU and hospital alive. Estimated glomerular filtration rate was calculated using the Chronic Kidney Disease Epidemiology Collaboration equation. Metabolic alkalosis was defined as high pH > 7.45 and HCO3 > 27 mmol/L.
Statistical analysis
Data were analysed with the statistical programming language R, version 3.6.3. An upset plot was created with the ComplexHeatmap package in R. 22 Medians and interquartile ranges (IQRs) were calculated for continuous variables. The Wilcoxon rank-sum test for differences in continuous variables, between groups, was used. The Fisher exact test was used for differences between proportions of categorical variables. P values below 0.05 were considered statistically significant.
Results
General characteristics of the study population
The study population included 56 patients of which 31 (55.4%) were survivors, while 25 (44.6%) died from COVID-19 complications. The median and IQR for age of both survivors and non-survivors were 51 years with IQR of 46–57.5 and 43–58, respectively (P = 0.967). There were 35 (62.5%) males and 21 (37.5%) females of which 37 (66.1%) had diabetes and 27 (48.2%) hypertension. The median length of hospital stay was 12 days (IQR: 7–16) with the median ICU stay being six days (IQR: 3–9). Table 1 summarizes the demographic findings, comorbidities and major risk factors for COVID-19 complications of the study population.
Study population demographics and comorbidities.

HIV, human immunodeficiency virus.
All variables are expressed as n (%), except for age which is median (IQR).
Vital signs, ABG and biochemical parameter findings
Table 2 is a comparative summary indicating the measured admission observations, ABG and biochemical parameters of survivors and non-survivors. When comparing vital signs between survivors and non-survivors, no statistically significant findings were observed. Of the 56 patients, seven (12.5%) had an acidaemia (pH < 7.35). This was observed in two of the 31 survivors (6.5%) and five of the 25 non-survivors (20.0%) (P = 0.223). An alkalaemia (pH > 7.45) was observed in 36 (64.3%) of the 56 patients. This included 23 of 31 survivors (74.2%) and 13 of 25 non-survivors (52.0%) (P = 0.101). Of those with an alkalaemia, no patients had hypokalaemia (K < 3.4 mmol/L), while only one had hypernatraemia. Fisher’s exact tests for differences in the proportion of survivors and non-survivors with low, normal and high pH were not statistically significant, but a higher pH was found to be a predictor of survival in our study population.
Summary of admission observations, arterial blood gas and biochemical parameters comparing measured parameters in survivors and non-survivors.
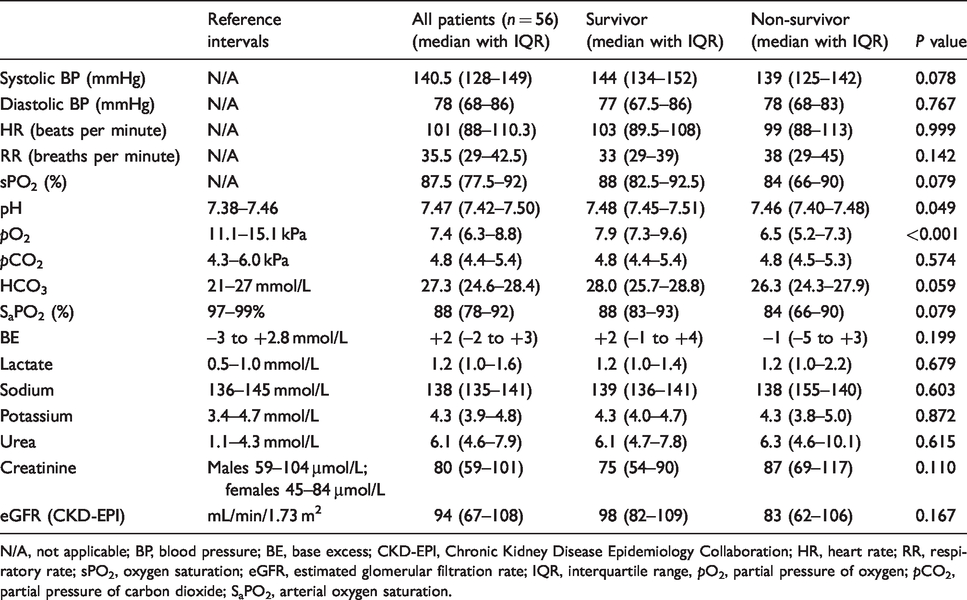
N/A, not applicable; BP, blood pressure; BE, base excess; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; HR, heart rate; RR, respiratory rate; sPO2, oxygen saturation; eGFR, estimated glomerular filtration rate; IQR, interquartile range, pO2, partial pressure of oxygen; pCO2, partial pressure of carbon dioxide; SaPO2, arterial oxygen saturation.
Figure 1 shows the overlap in pH group membership and comorbidities (diabetes mellitus and hypertension), for survivors (Figure 1(a)) and non-survivors (Figure 1(b)). Figure 1(a) shows that the largest grouping (n = 10) for survivors are those with a combination of alkalaemia, diabetes and hypertension, compared with five in Figure 1(b) with the same variables. We noted that 16 of 25 non-survivors (64%) had diabetes, compared with 21 of 31 survivors (67.7%) and this difference was not statistically significant (Fisher’s exact test, P value 0.784).
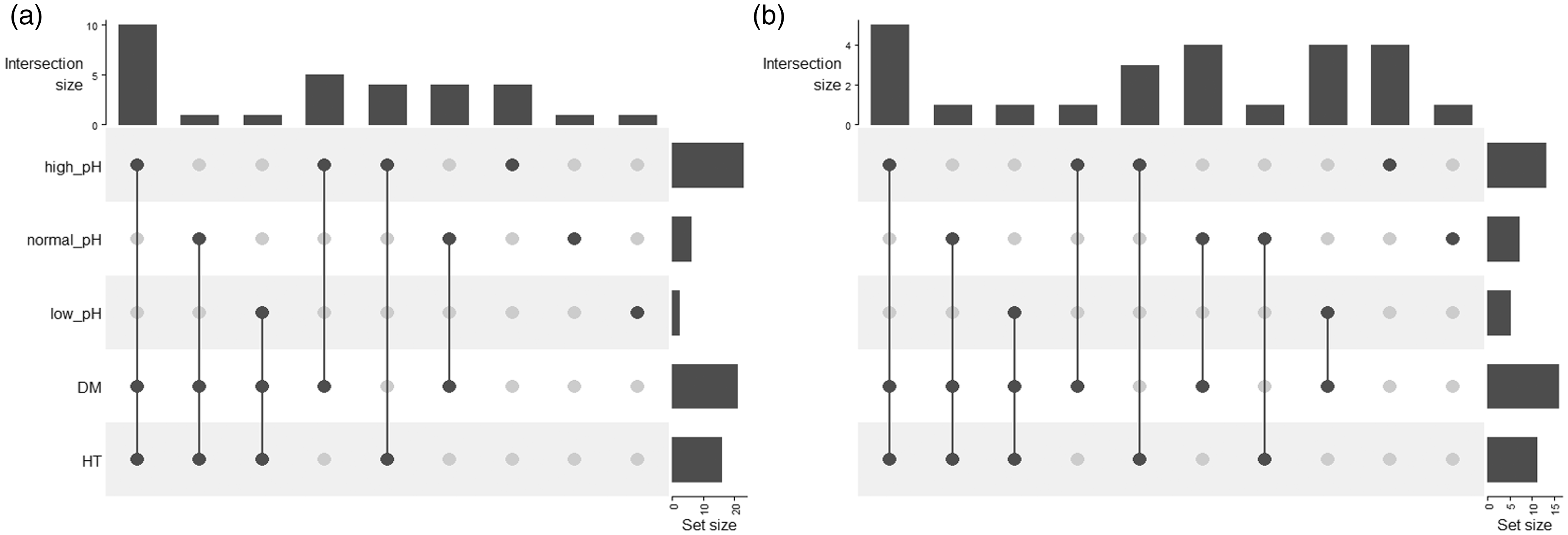
Overlap in pH group membership and comorbidities. (a) Survivors and (b) non-survivors. DM, diabetes mellitus; HT, hypertension.
Correlation with survival
Median arterial pH, pO2, HCO3 and BE values were higher in survivors, although only the differences in partial pressure of oxygen in arterial blood (PaO2) and pH were statistically significant. In the survivor group, only one patient (3.2%) had acidaemia (pH < 7.35), low PaO2 (<8 kPa) and high lactate (>1.0 mmol/L), while two patients (8.0%) in the non-survivor group matched this pattern. We found that 36 patients had alkalaemia, of whom 23 (64%) survived and 13 (36%) died (odds ratio [OR] 2.61 [95% CI, 0.75–9.54], P = 0.101).
Discussion
Main findings
In a general ICU population, one would anticipate hypoxaemia with accompanying acidaemia on ABG analysis. However, in our study population, we observed that the majority of our patients were alkalaemic. Even more unexpected, was the fact that observed high pH was significantly associated with survival.
Acid–base disturbances in ICU patients are common, with the majority of patients having a metabolic acidosis with lactic acidosis being the most common cause. 23 However, previous studies in this regard were performed in general ICU patients and did not include COVID-19 patients. To our knowledge, this is the first study to discuss ABG results and risk of non-survival in COVID-19 patients.
Risk in COVID-19
The first study published on COVID-19 patients reported 41 cases with a median age of 49 years, with 73% of the study population being male. Of this cohort, 32% of patients were admitted to ICU. 2 In another study describing the clinical features of COVID-19 in 710 patients, 52 patients were found to be critically ill. The average age of the critically ill patients was 59.7 years, 67% were male and 40% had a comorbid condition. The fatality rate was 61.5% in this population. 24 Similarly, our study population had an average age of 50 years and 62.5% were male. The high incidence of comorbid conditions and their association with non-survival in our study population is also notable.
Other ABG studies in COVID-19
Limited data on ABG findings in critically ill COVID-19 patients are available. In a single, small study conducted in Wuhan No. 1 Hospital in China at the start of the pandemic, the authors analysed the ABG results of ICU COVID-19 patients requiring intubation and ventilation. However, only 20 patients were included in this study. The main ABG findings of the study and their correlation in survivors and non-survivors were assessed. The study does not state the time of ABG collection but compares ABG readings prior and post intubation. The authors reported that despite their observation that intubation improved the patients’ oxygen saturation and reduced the pCO2 and blood lactate, seven out of the 20 patients included in the study still died. The non-survivors had significantly lower pO2 and higher pCO2 and lactate concentrations, compared with the survivors. The authors concluded that the results from the non-survivors were due to ensuing hypoxaemia, lactic acidosis and respiratory acidosis, indicating respiratory failure and possibly multiorgan failure. 18 However, they did not assess outcomes of alkalaemia for survival. In our study, we found that most patients had a higher pH and normal lactate concentrations.
Similar to our study, an Italian study 20 compare admission ABG results with the extent of pulmonary inflammation, determined by computer tomography, and the prognosis of 79 emergency department patients with SARS-CoV-2 infection. They concluded that admission ABG findings could be indicative of the extent of pulmonary involvement and poorer prognosis of patients with COVID-19. 20 They also found higher pH values in agreement with our findings but did not compare these with outcome. The survivor subgroup of our study also had higher pH and HCO3 values on admission ABG analysis when compared with non-survivors. These findings also correlated with the study done in Wuhan No. 1 Hospital. 18
In another study 25 evaluating admission ABGs in 91 patients of which 60 were still admitted at the time of write-up, an increased pH was noted in 29.4%. However, this study focused more on epidemiological and clinical characteristics and did not correlate ABG findings to patient outcomes.
It is unclear why such a large proportion of COVID-19 ICU patients exhibit alkalaemia, which is traditionally considered uncommon in critical care. 26 Certainly, the most likely causes would appear to be alkalaemia generated at the level of the kidney, with excessive mineralocorticoid activation (either endogenous or exogenous) as a possibility. COVID-19 has been postulated to upregulate the classic pathway of RAS and may cause metabolic alkalaemia. The RAS is primarily involved in the regulation of BP, water balance, electrolyte concentrations and the acid–base status of the body. It comprises two well described arms, the classic vasoconstrictive and the protective pathway. The classic pathway leads to vasoconstriction, cell proliferation, fibrosis, oxidative stress and aldosterone release which, in turn, leads to metabolic alkalosis. However, under these circumstances, one would expect hypernatraemia and hypokalaemia. All our patients had normokalaemia. Additionally, urinary electrolytes would have been helpful, however, these were not available.
The protective pathway leads to vasodilation, natriuresis, anti-inflammatory and antiproliferation effects ensue. In COVID-19, the protective pathway is downregulated while the classic pathway is upregulated. 27
Alternatively, corticosteroid administration could be a contributing factor due to its effect on the mineralocorticoid system. The use of dexamethasone attracted significant attention for use in critically ill patients on ventilatory support with better survival outcomes observed. 28 Mineralocorticoid activation will result in hypertension, alkalaemia and hypokalaemia. However, our patients had ABG analysed on admission before any treatment that could affect these results had been initiated. Although these ABGs were performed at admission, 43 (77%) of our cohort were on non-invasive high flow nasal prong oxygen.
In our study, 66% of the patients were known type 2 diabetics and 48% suffered from hypertension. Diabetes is associated with a worse outcome in COVID-19 with a higher proportion of ICU admission, ARDS and mechanical ventilation being observed. 29 In a multicentre observational study, it was found that patient suffering from hypertension also had a worse outcome. 30 We however found that alkalaemia in these patients was protective and associated with survival and postulate that this may be due to activation of the protective arm of the RAS system. However, our numbers were too small to correct for possible confounders.
We postulate that because of the high proportion of patients with alkalaemia, the presence of an acidaemia which has been shown to be a poor prognostic factor, may reflect a secondary insult in the form of ensuing sepsis, renal failure or hyperlactataemia.
Limitations and strengths
Our study has some limitations. First, this was a relatively small, single-centre study. Also, the study only focused on admission ABG and biochemistry results and no further results or urine electrolytes were analysed during the patients’ ICU stay to monitor trends. Another limitation is the lack of chronic and acute medication history in these patients.
However, our study has several strengths. To our knowledge, this is the first study that focuses on ABG, acid–base and BP findings in severely ill COVID-19 patients in Africa. The study population included the first 56 consecutive patients that required ICU admission and evaluated their data prior to any intervention or corticosteroid administration in the ICU.
Conclusion
In conclusion, we found the majority of patients in our study population to have an alkalaemia on admission ABG. A higher pH was associated with survival. This is an unusual finding in that, generally, patients admitted to ICU have acidaemia which is in contrast with what we observed. We postulate that this may be due to activation of the classical arm of RAS and the subsequent increase in aldosterone action. Larger studies on arterial ABG analysis and acid–base status of COVID-19 patients are needed to substantiate our findings. ABG analysis may be useful for predicting outcomes and risk stratification in critically patients with COVID-19.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by Stellenbosch Health Research Ethics Committee (reference number N20/05/023_COVID-19). As all patient data were anonymized with no direct treatment intervention, a waiver of consent was approved.
Guarantor
AEZ.
Contributorship
MCB and OJW: data analysis and interpretation, literature review, original and final draft writing, reviewing and editing. AAK-W: data analysis and interpretation, original and final draft reviewing and editing. AEZ and BWA: conceptualization and final draft reviewing and editing. EM: data analysis and interpretation. UL and CFNK: final draft reviewing and editing. MRD: data analysis, final draft reviewing and editing. DM: data collection.
