Abstract
Today, the world is affected by a COVID-19 epidemic. Early diagnosis can play an important role in the control and prevention of this disease, so the study and practice of biosafety among staff of diagnostic laboratories have a key role in the healthiness of health fighters against COVID-19. In this study, we will review laboratory biosafety considerations for the diagnosis process of SARS-CoV-2 in three phases: (1) the preanalytical phase, (2) the analytical phase, and (3) the postanalytical phase. It should be noted that employees with high-risk underlying health conditions, like lung or heart disease or diabetes, should be moved to lower-risk environments. We believe that this review will help to restrict the risk of developing COVID-19, while working in clinical laboratories.
Introduction
In the last days of 2019, a novel betacoronavirus, the agent of severe acute respiratory syndrome (SARS-CoV-2) has emerged. After some days, the resultant disease became the pandemic of coronavirus disease 2019 (COVID-19).1,2 The person-to-person transmission of this virus happens mainly via droplets and contaminated surfaces. 3 In this infection, the incubation period usually ranges from 2 to 14 days and after that, common symptoms including fever, muscle pain, fatigue, headache, sputum production, dry cough, shortness of breath, dyspnea, and sore throat will appear. 4 , 5
The World Health Organization (WHO) has implemented recommendations and regulations aimed at reducing the risk of infection transmission among laboratory staff. Based on the WHO Laboratory Biosafety Guide, the fundamental element of biosafety training is the process of risk assessment. 6 The primary determinant in the process of risk assessment involves the formulation of informed judgments by individuals with extensive knowledge regarding the characteristics of microorganisms, testing kits and tests, protocols, the containments, animal models, and other laboratory facilities. 7 The laboratory is considered a crucial component of diagnostic procedures within healthcare settings. The importance of accurate and reliable laboratory diagnosis cannot be overstated, as it plays a crucial role in facilitating early detection of diseases. This, in turn, significantly enhances the likelihood of successful treatment outcomes and effectively curbing disease transmission. 8
In order to mitigate the potential for laboratory-acquired infections with SARS-CoV-2, the implementation of biosafety procedures and adherence to established regulations are important. 9 The responsibility of laboratory supervisor entails ensuring that adequate and current risk assessments are conducted, consistently reviewed, and updated as needed. This process takes into account the incorporation of new information that aligns with the optimal risk management practices and other relevant data from scientific articles, as outlined in the WHO Laboratory Biosafety Guideline. 10 The objective of this study is to elucidate the prevailing difficulties encountered in laboratory settings during the SARS-CoV-2 pandemic and similar outbreaks.
Biosafety in clinical laboratories during the SARS-CoV-2 pandemic
The laboratory diagnostics process comprises three distinct phases, namely the preanalytical phase, the analytical phase, and the postanalytical phase. These phases are crucial in ensuring patient safety, as depicted in Figure 1. Therefore, mitigating the potential hazards associated with the various stages of the diagnostic process enhances the level of safety for healthcare workers.
11
Various specimens are received by laboratories in hospitals. Hence, the necessity for a comprehensive guideline is evident. When considering the development of a manual, an important initial consideration is the quantification of viral shedding in various types of samples.
12
A study is now being conducted to identify the presence of SARS-CoV-2 in several materials, including nasal and pharyngeal swabs, blood, sputum, bronchoalveolar lavage (BAL), and bronchoscope brush biopsy samples, feces, and urine. In this study, a total of 1,070 samples obtained from 205 individuals diagnosed with COVID-19. Among these samples, bronchoalveolar lavage (BAL) specimens had the greatest detection rates at 93%, followed by sputum samples at 72%, and nasal swabs at 63%. According to Chin et al. study, the specimens obtained using bronchoscope brush biopsy had a lower rate of detection at 46%. Pharyngeal swabs, feces, and blood, on the other hand, demonstrated detection rates of 32%, 29%, and 1% respectively.
13
Furthermore, none of the urine samples analyzed in the study conducted by Wang et al. yielded positive results.
14
In another research, it was shown that lower respiratory tract (LRT) samples, endotracheal aspirates and sputum exhibited significantly higher levels of SARS-CoV-2 RNA compared to nasopharyngeal swab (NPS) or oropharyngeal swab (OPS) samples.
15
Therefore, it may be inferred that the necessary biosafety methods will vary depending on the specific samples being handled. Following an analysis of viral titer in various specimens, this paper will proceed to examine the variations in diagnostic protocols across different regions. Chart demonstrating laboratory diagnostics process of SARS-CoV-2.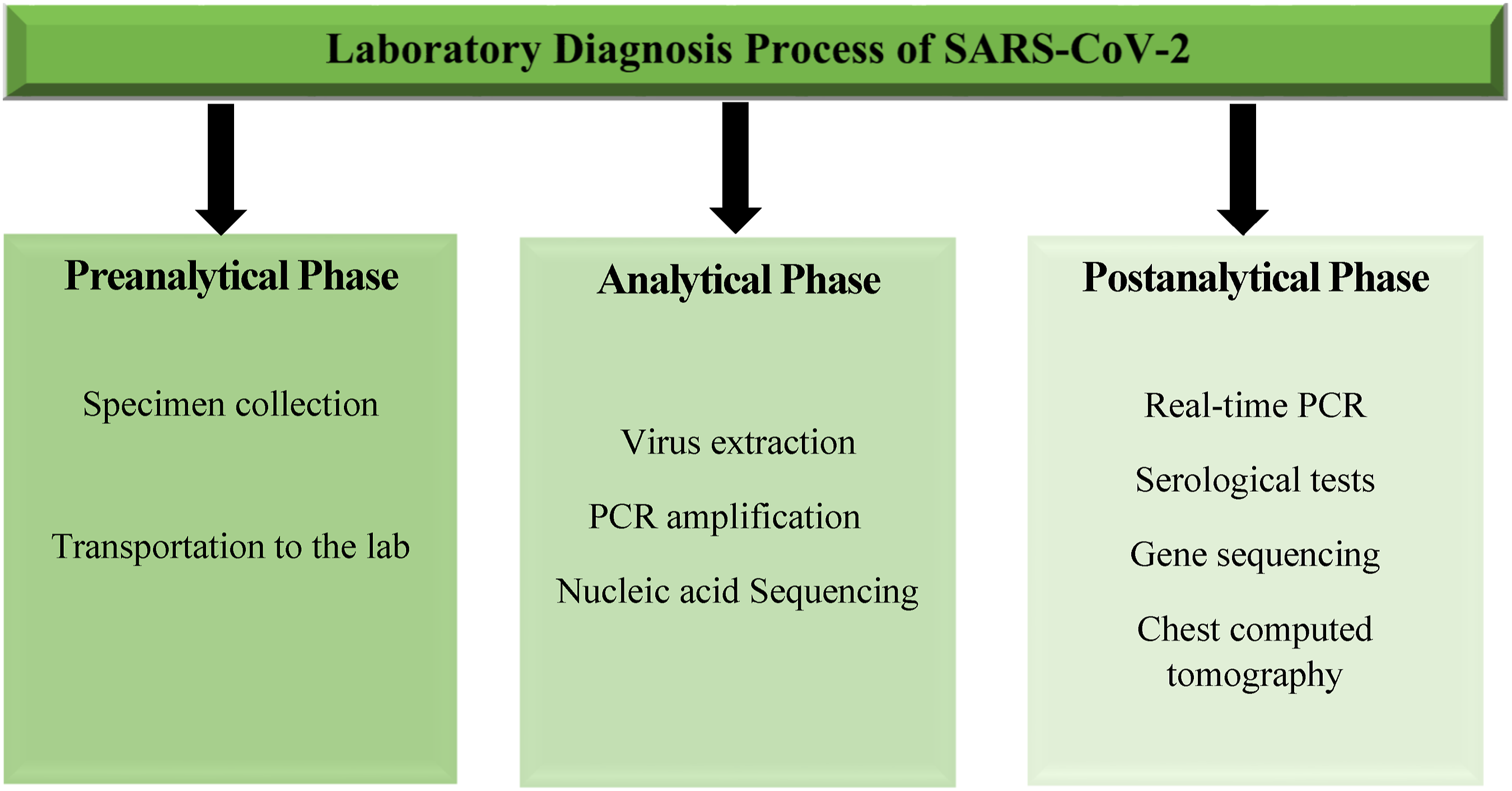
Preanalytical phase
The first and most significant stage for laboratory diagnosis is safe and correct specimen collection. Viral RNA could be detected in the upper respiratory tract (URT), lower respiratory tract (LRT), blood, feces, and urine of SARS-CoV-2-infected persons. Among these, the samples obtained from URT and LRT exhibit the most significant shedding of virus.16,17 After collection, swabs should be placed in transport medium for fast transportation to the clinical microbiology laboratory, ideally under refrigerated conditions.18,19 In regard to specimen transportation, the WHO recommendation is that all clinical samples (blood, respiratory, urine, feces, and others) be firmly covered, and transportation occurs in safe zip-keeps within a sealed cool box with a noticeable biohazard label. 20 In contrast to previous advisories, the Centers for Disease Control and Prevention (CDC) currently authorizes the transportation of all laboratory specimens through pneumatic tube systems, except respiratory specimens, which must be manually transported. In addition, other specimens may be collected after the hospitalization of patients with severe infections. 21 Consequently, it is imperative to ensure that behaviours are implemented throughout several tiers of biosafety.
Analytical phase
Diagnostic tests typically be conducted as part of regular practice within a BSL-2 laboratory utilizing standard safety measures. Because a blood sample is one of the general specimens for analysis in the laboratory, recent research has compared measure of serumic SARS-CoV-2 RNA of 48 patients with various severities. Out of the individuals under consideration, only five had positive serum results, all of whom were diagnosed with serious illnesses. Notably, two of these patients unfortunately succumbed to their conditions during the course of treatment. 22 Hence, the risk of handling blood samples from COVID-19 patients is accepted not to be high and this implies that BSL-2 laboratory is enough to handle blood isolated from SARS-CoV-2 infected individuals. 23 Nevertheless, there is still a need for more research to determine the level of infectivity present in blood and its derivatives obtained from individuals infected with COVID-19. It is crucial to minimize any potential risk factors to healthcare personnel during this time. Also, Wang et al. assessed the titer of SARS-CoV-2 after exposing the specimens to various temperatures for a variety of periods. 24 They showed that storing the samples at 56°C for 30 min completely can inactivate any infectious SARS-CoV-2; however, viral RNA was detectable. Virus isolation for cell culture of SARS-CoV-2 specimens is recommended only at the biosafety level 3 (BSL-3) facilities. Multiple assays are available for the diagnosis of COVID-19 with different sensitivity and specificity. 25
The virus detection process includes three steps: (i) virus extraction, (ii) PCR amplification, and (iii) confirmation by nucleic acid sequencing when necessary. 26 The isolation process and culture should be done at the biosafety level 3 laboratory (BSL-3), and laboratory staff should use personal protective equipment, 27 containing wrap-around gowns, disposable gloves, coveralls with sleeves, head coverings, shoe covers, respiratory protection, and eye protection. 28
To compare all features of 4 levels of biosafety in work with viral infectious samples.

Postanalytical phase
Detection methods
Significantly 4 methods for SARS-CoV-2 detection have been recommended by the CDC 30 : real-time PCR, serological tests, 30 gene sequencing, 30 and (4) chest computed tomography (CT). 31
The CDC recommendation for laboratory diagnosis of COVID-19 is the real-time reverse transcription-PCR (RT-PCR) test. 32 Detection by real-time RT-PCR assays has great advantages, that is capability to amplification and analysis done simultaneously in a closed system to reduce false-positive results associated with the amplification stage of PCR.33,34
Discharged patients
Also, screening individuals recovered from COVID-19 may be essential once they need to be released from isolation and discharged. Discharging individuals with continued shedding of viable coronavirus may increase the prevalence of infection.32,35 According to the WHO, it is recommended that certain individuals observe a period of self-quarantine lasting at least one month. In certain cases, the use of NP and OP swabs alone may not be sufficient for the detection of infection. However, additional evaluations are necessary to address this limitation. 14 There is an approach to testing for recovery by demonstrating two repeated negative real-time PCR tests on rectal swabs; this recommendation is based on the fact that SARS-CoV was cultured from stool during the SARS epidemic and SARS-CoV-2 was cultured from stool during the COVID-19 outbreak. 36 Hence, the detection of a positive real-time PCR result from a rectal swab suggests the presence of SARS-CoV-2 shedding in the patient’s fecal matter, indicating the potential for infectiousness.
Levels of biosafety in a laboratory and considerations
Expansion BSL levels include further training for workers, raising the level of personal protective equipment (PPE), and other severe controls. For instance, at BSL-2, during working and special processes are performed in biosafety cabinets or other containment equipment, accessibility to laboratories and other controlled work areas is restricted.37,38 At BSL-3, surplus to controlling the availability of laboratories and work areas, every task, including the handling of infectious materials, is performed in biosafety cabinets or other containment equipment. To further protect employees, the work must be done in a correctly maintained and certified biosafety cabinet (BSC). Class I BSCs, by using negative pressure and high-efficiency particulate arrestance (HEPA) filters, protect staff and the environment. 39 Higher levels of containment and filtration that also protect samples or other products in the BSC from contamination were provided by class II and III BSCs. 40 To make sure that the whole process, including manipulation of untreated specimens or those that have the potential to generate aerosols (like vortexing or sonication of specimens in an open tube, etc.), is performed in a BSC, and to also use proper physical containment devices (like sealed centrifuge rotors or safety carriers with gaskets) for centrifugation.41,42
Throughout the entirety of the work process, it is imperative that all personnel possess knowledge regarding the possible infectivity of each biological sample. This novel coronavirus may pose a serious risk to healthcare personnel that is an important issue that has been emphasized by many worldwide health organizations43,44 and still there is low awareness about SARS-CoV-2 and using disinfectants, particularly those capable of effectively combating enveloped viruses. The biocidal agents that can be used are: 62%–71% ethanol, 0.5% accelerated hydrogen peroxide, quaternary ammonium compounds, and phenolic compounds (following the manufacturer’s recommendations). 45 Other solutions, like 0.02% chlorhexidine digluconate or 0.05%–0.2% benzalkonium chloride, may be less effective. Moreover, sodium hypochlorite at a concentration of 0.1% is recommended for general surface, whereas a concentration of 1% is recommended for the blood spills. 46
Ensuring the safe handling of samples obtained from patients exhibiting symptoms or having a confirmed diagnosis of COVID-19 necessitates the imperative task of providing comprehensive training to all laboratory professionals regarding any supplementary protocols or procedures (Figure 2). This contains education on the communication processes that affect the clinical and laboratory workers. Moreover, training should be provided throughout the scheduled work hours, and there is no cost for workers.41,47 Adhering to the guidelines recommended by the WHO and the European and US CDC, has significant importance in mitigating the risk of contracting COVID-19. Standard precautions and measures to prevent the transmission of SARS-CoV-2.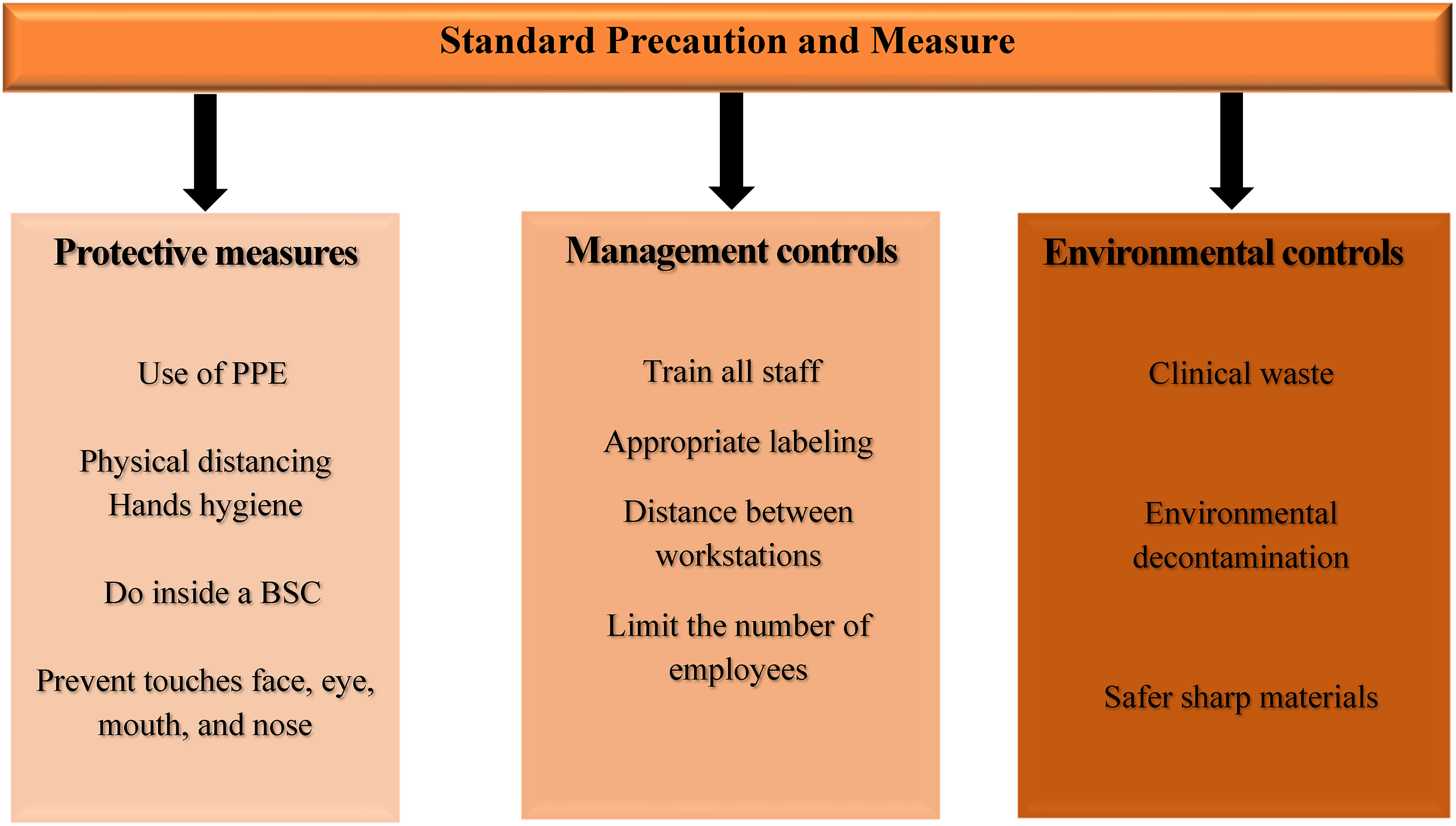
In summary, a number of safe work practices encompass the following measures: • To make ensure proper announcement and to properly label the suspected or confirmed SARS-CoV-2 contaminated specimens. • For reduce the formation of aerosols and droplets, use technical processes and conduct any necessary aerosol-generating procedures in containment (e.g., inside a BSC) and/or while using proper precautions, including worker PPE. • Be careful since handling needles or other sharps, and eliminate contaminated sharps in puncture-proof, labeled, closable sharps containers. • To inactivate infectious material waste before disposal, use an autoclave. • Employees should prevent touching their faces involving their eyes, mouths, and noses, especially until after they have carefully washed their hands as soon as their work is completed and removing PPE. • Hands should be often washed with soap and water for at least 20 s and if they are not available, use an alcohol-based hand sanitizer with a minimum of 60% ethanol or 70% isopropanol. • Tie lab coat tightly and use double gloves that extend over the sleeve of the gown or lab coat. • Before exiting the BSC, dispose of the outer pair of gloves, and cover a new pair until re-entering the BSC. • Personal eyeglasses do not have enough eye protection. They should apply surgical masks and either goggles or face shields. • To prevent worker exposure, cloth face coverings are unacceptable for PPE. • Limit the number of employees per shift and make sure workstations have distance a minimum of 6 feet apart.
Persistence of the SARS-CoV-2 on the surfaces and specimens
The persistence of SARS-CoV-2 on different surfaces and materials since the emergence of SARS-CoV-2 at the end of 2019, has raised concerns about the potential transmission of the virus through inanimate objects. 48 One material of particular interest is Ethylene Vinyl Acetate (EVA), a flexible, stable, and inexpensive material often used in toys, gym mats, and other surfaces that come into contact with both children and adults. These settings have been frequently linked to COVID-19 outbreaks, making the persistence of the virus on EVA a significant concern. 49
In addition to EVA, other materials commonly used in the food supply chain, such as aluminum, Tetrapak, Polyethylene terephthalate (PET) plastic, and glass, have also been examined for their potential to harbor the virus. 48 A previous study reported the detection of SARS-CoV-2 RNA on the surface of a salmon chopping board, though the viability of the virus was not assessed. 50 Other research has shown that SARS-CoV-2 can remain viable on PET surfaces for up to 96 h 51 Researchers have explored whether the material properties of various surfaces and objects may influence the viability of SARS-CoV-2. While some studies have observed lower survival rates of the virus on porous surfaces, 51 other research has found no clear correlation between material features and virus viability. 52
The COVID-19 pandemic has also led to a significant increase in the demand for protective equipment, such as N95 and surgical masks, protective gloves, and Tyvek suits. Studies have shown that SARS-CoV-2 can have a biological half-life of 10–32 h and a survival time of 2–21 days on the surfaces of these protective materials at 22°C. The virus has been found to remain infectious for up to 9 h on N95 and N100 masks, 4 h on Tyvek, and 10 h on chemical gloves. 53 Additionally, SARS-CoV-2 has been shown to survive longer on the outer layer of masks (>7 days) compared to the inner layer (4–7 days). 54
The virucidal activity of disinfectants against SARS-CoV-2.

In the term of persistence of SARS-CoV-2 RNA in various organs of the body and different laboratory specimens, previous studies demonstrated while the respiratory tract was the most common site of persistent SARS-CoV-2 RNA, a significant proportion of late-stage COVID-19 cases showed RNA persistence in a variety of non-respiratory tissues. 55 A recent study found that over 50% of late cases had detectable SARS-CoV-2 RNA in the myocardium, lymph nodes in the head, neck and thorax, the sciatic nerve, ocular tissues, and throughout the central nervous system, excluding the dura mater.55,56
Notably, the researchers observed that the stark difference in viral RNA levels between respiratory and non-respiratory tissues, which was over 100-fold in early cases, greatly diminished over time. 56 The researchers observed that the virus was able to proliferate in various non-respiratory parts of the body during the initial two weeks after the onset of symptoms. Moreover, they identified the presence of subgenomic RNA, an indicator of ongoing viral replication, in at least one tissue sample from 14 out of the 27 cases examined, even beyond the 14-day mark. This finding implies that the virus may have the capacity to persist and continue replicating in non-respiratory tissues for an extended period, potentially lasting several months. This suggests that SARS-CoV-2 may have a more difficult time being cleared from certain non-respiratory sites compared to the respiratory tract.
The authors hypothesize that tissue-specific differences in the virus’ ability to evade cellular detection of viral mRNA, interfere with interferon signalling, or disrupt antigen processing and presentation could contribute to less efficient viral clearance in non-respiratory compartments. Further research is needed to fully elucidate the mechanisms underlying the persistence of SARS-CoV-2 RNA in the body over the course of infection.
Conclusion
In summary, our review on the critical importance of robust biosafety practices in laboratory settings particularly when handling SARS-CoV-2, provides valuable insights that are highly relevant to the scientific community. By emphasizing the importance of increased understanding and adherence to laboratory biosafety measures, this work provides a practical roadmap for researchers, clinicians and laboratory personnel working with this highly infectious virus. Our study underscores the significance of prioritizing the health and well-being of laboratory personnel, especially those with underlying health conditions that place them at higher risk. We advocate for the relocation of high-risk individuals to environments with reduced risk levels, thereby minimizing potential exposure to SARS-CoV-2 and similar pathogens. Moreover, our findings stress the critical need to implement and rigorously adhere to standard preventive measures within laboratory settings. By following the strategies outlined in our study, we propose that the risk of contracting COVID-19 and related illnesses can be effectively mitigated. Overall, our research not only addresses the immediate biosafety concerns associated with SARS-CoV-2 but also offers a foundation for future research endeavours aimed at enhancing safety protocols and protective measures when working with infectious agents. We believe that our work can serve as a valuable resource for guiding research practices and fostering a culture of safety within the scientific community.
Footnotes
Acknowledgments
The authors thank Research Consultation Center for improving the article’s writing.
Author Contributions
Study concept: E.B; Study design: E.B; Bench work: M.M; Manuscript drafting: M.M, N.A, M.S; Critical revision of the manuscript: H.M, E.B. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
