Abstract
Background
Pregnancy induces physiological changes which affect biochemical and haematological parameters. As the significance of laboratory test results change throughout pregnancy, the reference interval (RI) or key result interpretive guide should be specific to pregnancy. This study sought to establish trimester-specific-RIs for routine biochemical and haematological tests in healthy white European women with singleton pregnancies with comparison to RIs for non-pregnant European adults.
Methods
A retrospective analysis of a prospective longitudinal single-centre study of healthy pregnant women conducted between November 2018 and December 2020 in a tertiary academic hospital with approximately 3000 births annually. Inclusion criteria: signed informed consent, age ≥18 years, white European, body mass index (BMI) <25 kg/m2, blood pressure <140/90mmHg, non-smoker, no previous pathology or gestational diabetes. Trimester defined as T1: up to 13 weeks + 6 days, T2: 14–27 weeks + 6 days and T3: ≥28–41 weeks + 6 days. Baseline demographics, anthropometric and laboratory measurements were recorded. In total, 31 biochemical and 10 haematological ISO15189:2012 accredited tests were measured using Roche Cobas® and Sysmex XN-9100™ analysers, respectively. RIs were established according to the International Federation of Clinical Chemistry (IFCC) recommended method.
Results
Apparently healthy pregnant women (n = 124) with bio-banked serum samples in each trimester were recruited. At the booking visit, 49.2% (n = 61) of participants were nulliparous, with median age of 34.4 (IQR: 31.3–37.3) years, gestational age of 89 (IQR: 84–93) days, BMI of 22.5 (IQR: 21.0–23.7) kg/m2 and systolic and diastolic blood pressure of 116 (110–125) mmHg and 67 (61–75) mmHg, respectively.
Conclusions
Normative trimester-specific biological intervals for routinely requested biochemical and haematological medical laboratory tests were established. These RIs will be invaluable to result interpretation and the management of pregnant women.
Introduction
Reference intervals (RIs) are a range of analyte concentrations usually encompassing the 95% central distribution of values observed in healthy individuals and are designed to aid result interpretation and clinical decision-making. 1 Knowledge of physiological changes is fundamental and essential to both understanding and developing reference intervals. 2 Healthy physiological changes occur throughout pregnancy to accommodate the needs of both mother and fetus. These pregnancy-induced physiological adaptations impact most organ systems and many biological markers.3,4 In laboratory medicine, this is of great importance because many biochemical and haematological test results have different significance depending on the gestational age of the woman at the time of testing.5,6 However, despite this, many laboratories do not provide trimester-specific-RIs for healthy pregnant women and often quote RIs from non-pregnant women or adults.4,7 The use of inappropriate RIs leads to an increased risk of misinterpreting normal pregnancy-induced changes in biomarkers as pathological or overlooking a truly abnormal result.8,9 The clinical risk of such practice is not insignificant with the potential to lead to patient misdiagnosis and/or mismanagement; therefore, ideally laboratories should establish/provide pregnancy-specific RIs for biochemical and haematological parameters across all three trimesters. 10
Establishing RIs per se is challenging 11 but endeavouring to do so for the different stages of pregnancy is even more demanding. 2 This is because to develop a RI a priori (direct) requires a minimum of 120 healthy people. 11 Moreover, the 9 months (40 weeks’ gestation) of human pregnancy have traditionally been divided into three 14-week periods, but often the partitioning of gestation is arbitrary. 12 Consequently, indirect or data mining methods are considered by many to be the practical way forward and perhaps the best approach to verify RIs.13,14 Notwithstanding these issues, development of trimester-specific-RIs necessitates careful consideration of the degree of change in analyte concentration throughout pregnancy, appropriateness of timing of partitioning of data, comparison of the magnitude of the change observed to that of the non-pregnant state and, finally, deciding whether the change identified is clinically significant. 5
There are few sources in the literature that provide RIs for white European pregnant women and those published rarely measure analytes longitudinally or tend to focus on selected analytes, particularly thyroid function tests. 4 In addition, many define the trimesters of pregnancy differently.
To address this unmet clinical need, this study sought to establish trimester-specific-RIs for routinely requested biochemical and haematological ISO15189:2012 15 accredited tests in healthy pregnant white European women.
Materials and methods
Ethical approval
Ethical approval for the study was granted by the research ethics committee at Galway University Hospitals (GUH) (Ref GUH: CA 2026). This study was conducted in accordance with the ethical principles as set out by the World Medical Association Declaration of Helsinki. Prior to study initiation, written/signed informed consent was obtained from each participant.
Study design
This was a retrospective sub-analysis of a larger prospective longitudinal study which aimed to evaluate novel biochemical tests for the diagnosis and follow-up of gestational diabetes. 16 This single-centre study of healthy pregnant women was conducted between November 2018 and December 2020 in a tertiary academic hospital with approximately 3000 births annually. Participants were recruited consecutively at the first antenatal visit at Galway University Hospitals (GUH). Each participant had an early dating scan for an agreed estimated due date (EDD). This sonographically determined EDD was subsequently used to determine the gestational age of the women at each study sampling timepoint. The gestational period was divided into three 14-week periods or trimesters (T) as follows: T1: up to 13 weeks + 6 days (97 days), T2: 14–27 weeks + 6 days (98–195 days) and T3: ≥28–41 weeks + 6 days (196–293 days). 12
Reference population
Healthy women, aged 18–47 years with singleton pregnancy, were recruited to this study. The inclusion criteria were: signed informed consent, age ≥18 years, white European, body mass index (BMI) <25 kg/m2, blood pressure (BP) <140/90mmHg, non-smoker and no previous pathology or gestational diabetes.
Data collection
Following informed written consent, at the booking visit, baseline clinical data including age, ethnicity, BMI, BP, gravida, parity, anthropometric and HbA1c measurements were recorded. Weight was measured in kilogrammes using the Seca® scale and height in metres using a Seca® wall-mounted stadiometer, according to departmental standard operating procedures. Systolic blood pressure (SBP) and diastolic blood pressure (DBP) were measured in mmHg using an automated oscillometric device (Omron®), after participants had been seated quietly for 5 minutes in accordance with the 2013 European Society of Hypertension/Cardiology guidelines. 17
Sample collection
Eligible participants were required to undergo phlebotomy at the first antenatal visit and again in trimesters 2 and 3 of pregnancy. The participants were not required to fast or make any special preparation before having blood collected at each study timepoint. Venous whole blood was collected into appropriate specimen tubes, one 7 mL (Greiner Vacuette®) plain plastic for biochemical tests and one 3 mL (Greiner Vacuette®) potassium ethylenediaminetetraacetic acid (K3EDTA) tube for measurement of haematological parameters. Samples for biochemical tests were maintained at room temperature (RT) for 30 min to allow clot formation, and all specimens were transported to the laboratory within 1 h of blood draw. On receipt, samples were centrifuged at 2000g for 15 min at RT, serum separated, aliquoted and stored at −80°C pending batch analyses. EDTA blood samples were analysed for haematological (full blood count (FBC)) parameters within 3h of phlebotomy.
Parameters evaluated and analytical methods
All biochemical and haematological parameters were measured by standard laboratory methods and in accordance with the respective manufacturer’s instructions for use.
Biochemical testing included:
The haematology
Statistical analyses
Statistical analyses were carried out using MedCalc® Statistical Software (Version 18.2.1) and Analyse-it® (Version 17). Continuous parametric data were represented as mean (±standard deviation (SD)) and non-Gaussian data, as median (interquartile range (IQR)). Categorical data are summarised with frequencies (in percentage). For non-parametric data, the Mann–Whitney U test and the Kruskal–Wallis multiple comparison test with Dunn’s post hoc multiple comparison test were used. A p-value of < 0.05 was deemed statistically significant.
The RIs were defined according to the Clinical Laboratory Standards Institute (CLSI)/International Federation for Clinical Chemistry and Laboratory Medicine (IFCC) non-parametric method (EP28-A3c). 11 This requires a minimum of 120 individual reference values to establish the central 95% distribution of results and the 90% confidence interval (CI) of the lower reference limit (LRL) and upper reference limit (URL), respectively. Where reference values were less in number than those required by the non-parametric method, the Robust method 19 was employed.
Reference values within each assays’ reportable range were used to establish the RIs. If results were below an assay’s limit of detection (LoD), for statistical purposes, the LoD was assigned. Firstly, data was illustrated (histogram and box and whiskers plot) together with the Shapiro–Wilk normality test to assess normality using Analyse-it® (Version 17). The frequency distribution of the individual analytes in each trimester of pregnancy was examined visually for results that were out-with the majority of reference values, that is, potential outliers. All apparent outliers were then statistically evaluated based on the Dixon 20 and Reed, 21 criteria, an approach supported by the IFCC working group. 10 Data was subdivided according to trimester and the appropriateness of partitioning evaluated using the Harris and Boyd’s standard deviate test. 22
Results
In total, 1522 pregnant women with singleton pregnancies were recruited to the original study, 117 withdrew or failed to complete the oral glucose tolerance test (OGTT). Of the remaining 1405 eligible participants, 279 comprised the reference population, 124 of whom had bio-banked serum samples in each trimester (Figure 1). At the booking visit, 49.2% (n = 61) of participants were nulliparous, with a median age of 34.4 (IQR: 31.3–37.3) years, gestational age of 89 (IQR: 84–93) days, BMI of 22.5 (IQR: 21.0–23.7) kg/m2 and SBP and DBP of 116 (110–125) mmHg and 67 (61–75) mmHg, respectively (Table 1). Recruitment schematic. Baseline characteristics of the reference population. BMI: body mass index; SBP: systolic blood pressure; DBP: diastolic blood pressure. kg: kilogramme; m; metre; data presented as †median (interquartile range) as appropriate.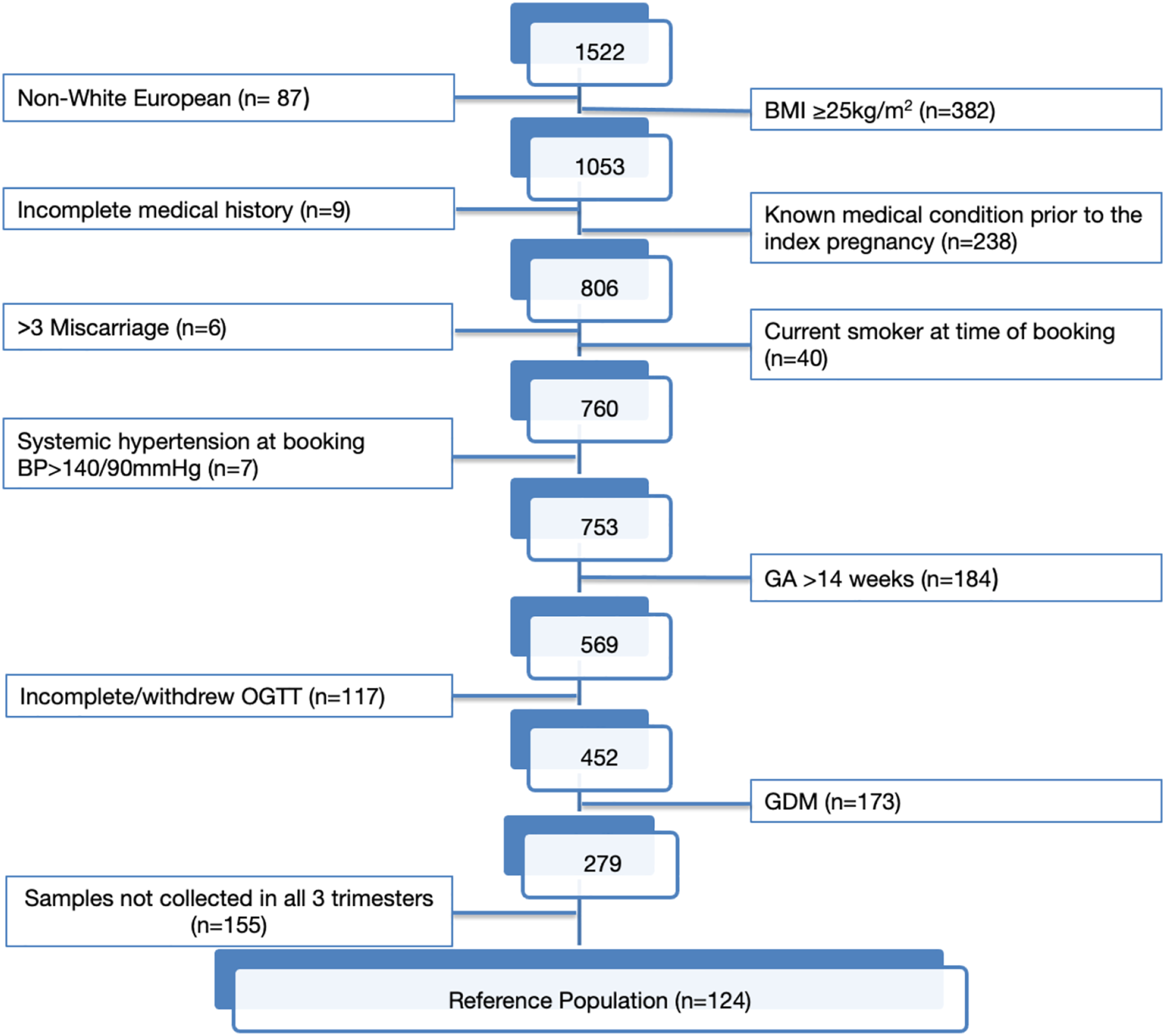

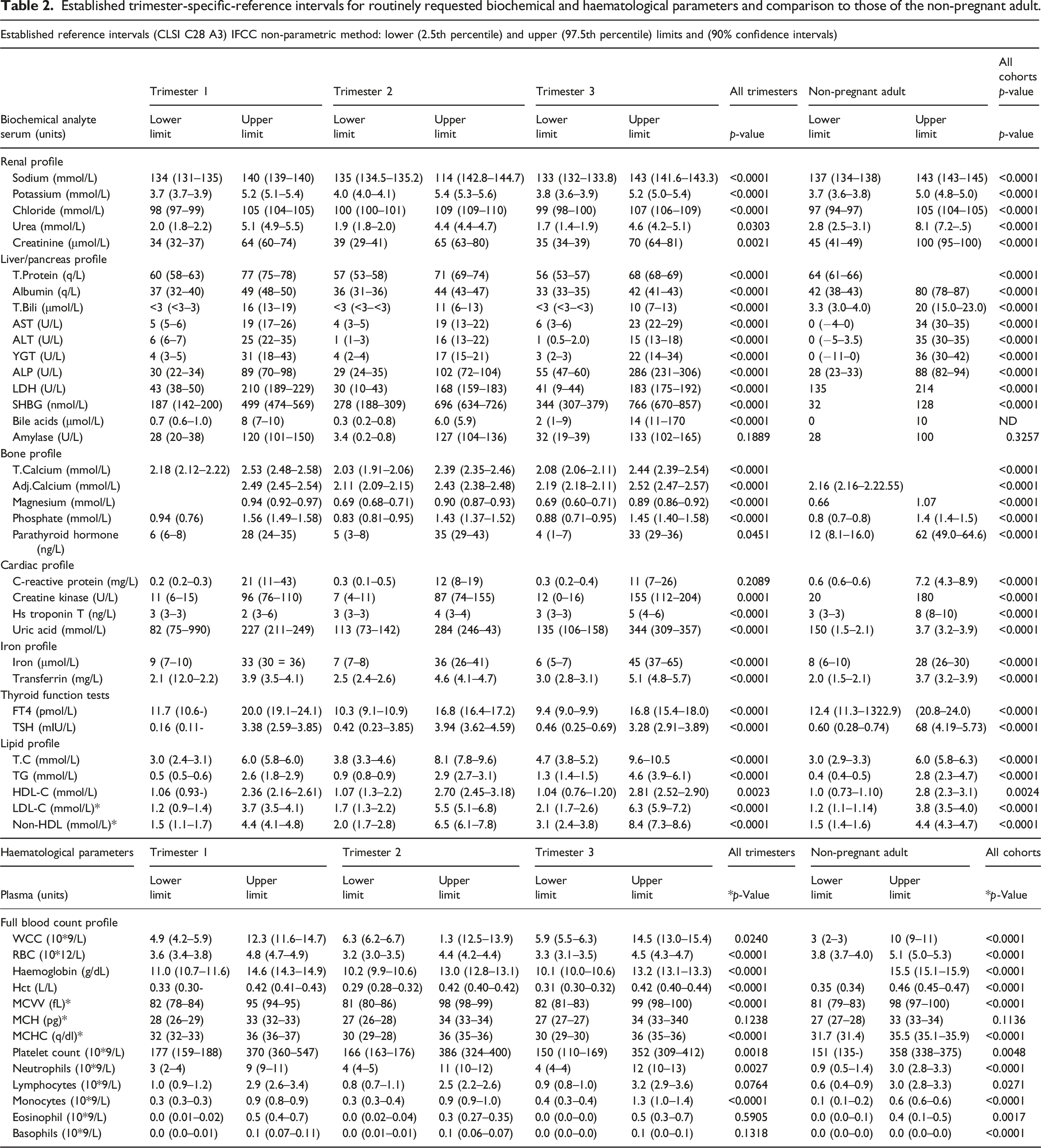
Established trimester-specific-reference intervals for routinely requested biochemical and haematological parameters and comparison to those of the non-pregnant adult.
Comparison of established trimester-specific-reference intervals to those of the non-pregnant adult.
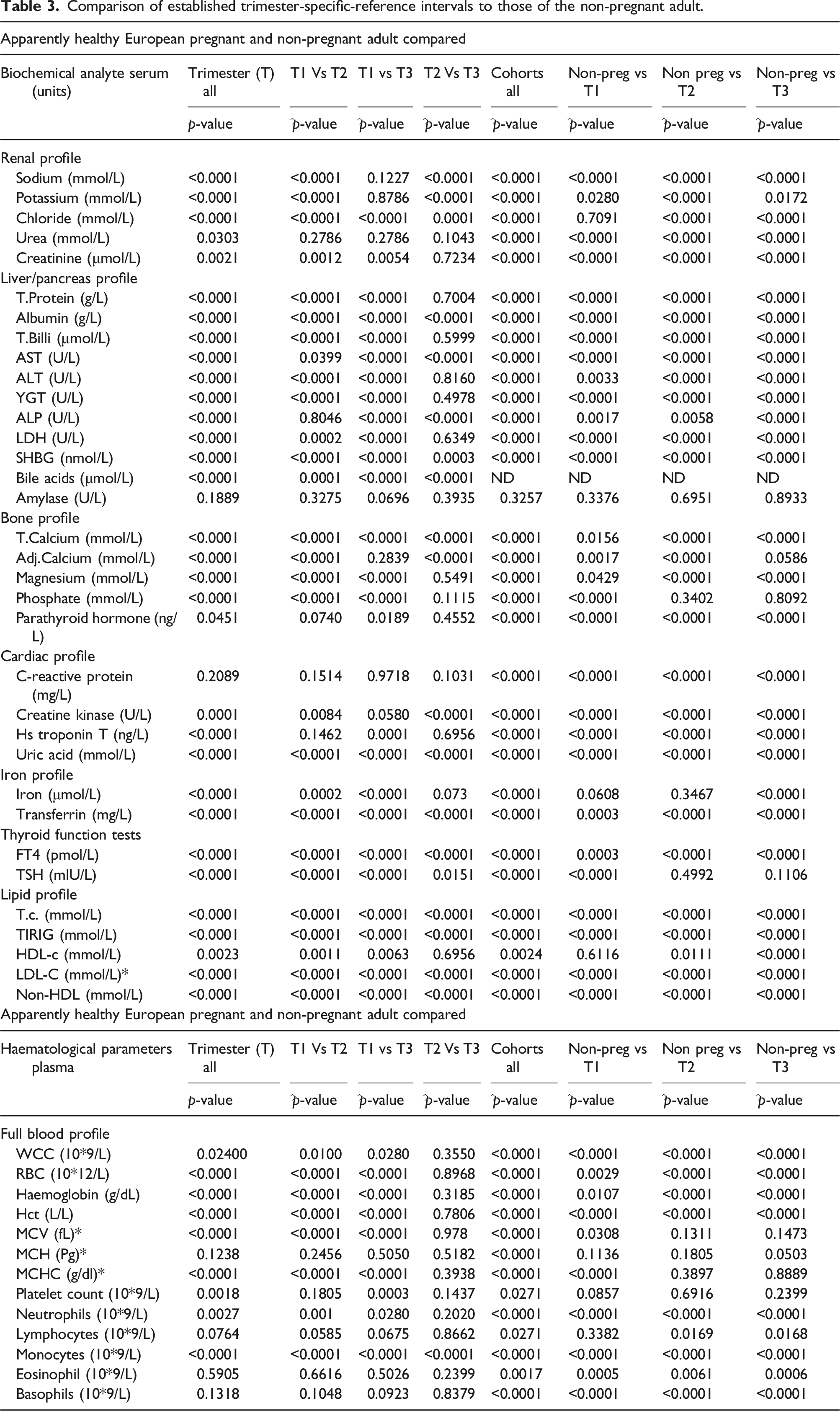
Of the FBC parameters evaluated, no differences were observed between trimester-specific-RIs for MCH, lymphocytes, eosinophils and basophils. Comparison with those for non-pregnant women determined that there was no statistical difference in MCH RIs for any trimester. While the differences observed for eosinophils and basophils were statistically different and for lymphocytes for trimesters 2 and 3 only, all were clinically insignificant. For all remaining trimester-specific biochemical and haematological RIs, the observed differences were statistically different, as was the overall comparison with the respective RIs established in the non-pregnant cohort. Levels of RBC, Hb and Hct were lower, and levels of WCC, platelets, neutrophils and monocytes were higher across trimesters and in comparison to non-pregnant healthy women. These differences were both clinically and statistically significant.
Discussion
Main findings
Normative trimester-specific-RIs for 41 routine biochemical and haematological tests in white European pregnant women with singleton pregnancies were established.
Strengths and limitations
This study comprised a comprehensive retrospective analysis of a longitudinal study establishing trimester-specific-RIs for routinely requested biochemical and haematological tests in healthy white European women with singleton pregnancies. This study design permitted the identification of pregnant women in good health (the reference population), 24 reducing the potential for confounders to influence the established trimester-specific-RIs.
We acknowledge that an increased sample size would likely increase the reliability of results. Restricting the study to white European women may mean the findings are not generalizable to other ethnic groups. We accept that the study cohort of healthy pregnant women precludes comments on the performance of any of the analytes evaluated to detect disease.
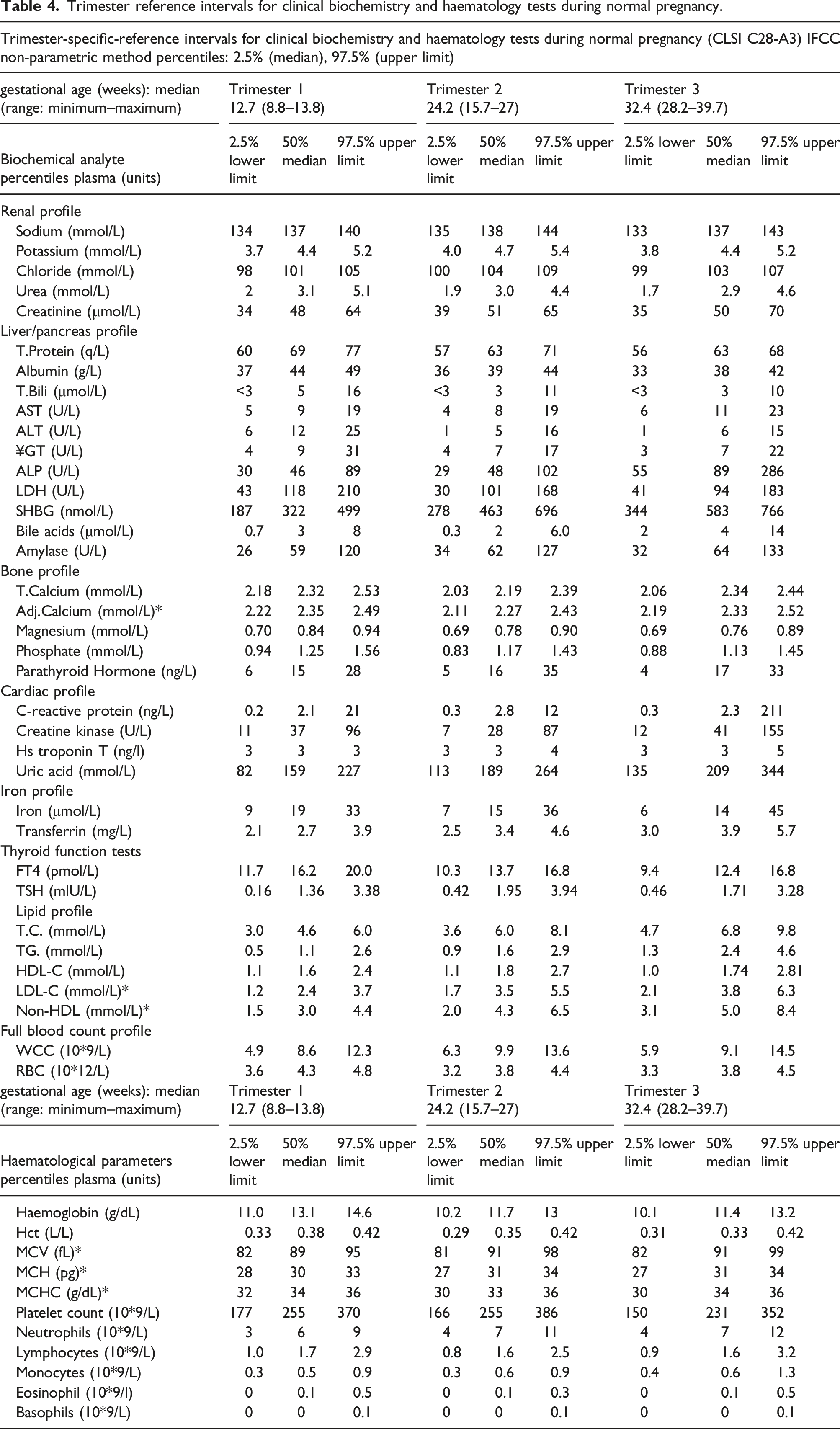
Trimester reference intervals for clinical biochemistry and haematology tests during normal pregnancy.
Interpretation
The quality of the RI is critically dependent on the selection of the reference population. 1 Stringent inclusion criteria ensured that the health of the reference population was well-characterized, representative, and closely resembles the population where the tests will be used; 1 while the numbers failing to meet the inclusion criteria are significant, a similar study recruited 801 healthy Caucasian pregnant women amongst 2147 attending first trimester screening with 391 participants comprising the reference population. 4
Few biochemical analytes measured remained unchanged during pregnancy (Table 2). The reference values for sodium were lower than those for non-pregnant adults’ findings supported by others4,8,10 and consequent to the increased plasma volume (30%–50%), extracellular volume and cardiac output. 25 Potassium values were slightly increased, with 97.5th percentile values in each trimester > 5.0 mmol/L and in agreement with Larsson et al. 8 In contrast, Klajnbard et al. reported lower potassium levels in all trimesters, while Friis Petersen et al. reported lower 2.5th percentile values in the first trimester than in non-pregnant adults.4,10 This may be related to pre-analytical issues, for example, the use of lithium heparin as compared to serum in the current study.
The observed lower RIs for creatinine concur with the well-recognized increase in glomerular filtration rate (GFR), approximately 50% in early pregnancy. 26 It is readily apparent that the use of non-pregnant RIs could potentially result in a missed diagnosis of pregnancy associated acute kidney injury and suboptimal patient care and mismanagement.
The haemodilution effect which occurs because of increased extracellular volume results in a decrease in protein concentrations in each trimester, findings that are consistent with others.4,8 ALT and AST values remained stable across all trimesters and correlate with previous studies.4,27,28 However, others observed lower levels of ALT and AST during pregnancy 8 which may relate to assays that do not include pyridoxal phosphate. 4 Other liver profile analytes (Table 2) increased with each trimester.
Intrahepatic cholestasis of pregnancy (ICP) and haemolysis, elevated liver enzymes and low platelets (HELLP) syndrome are fatal life-threatening conditions that can occur during pregnancy. 6 ICP, characterized by pruritus and elevated BA, is associated with an increased risk of adverse pregnancy outcomes. In our study, total BA levels ranged from 0.3 to 14 μmol/L and rose minimally in the third trimester. A similar study performed in a predominantly white European population (94%) gave comparable results. 29
Preeclampsia and eclampsia are hypertensive disorders of pregnancy which cause significant amounts of cellular damage. While pregnancy itself does not affect LDH levels, an increase in LDH reflects the occurrence of pre-eclamptic complications and the severity of pre-eclamptic disease.6,30 Our study shows lower levels of LDH in pregnant women than in non-pregnant women. Friis Petersen et al. reported decreasing levels of LDH with increasing weeks’ gestation in the first trimester, illustrating how gestational changes can already be seen in early pregnancy. 10
Calcium homeostasis is altered during pregnancy to ensure the growing fetus receives sufficient minerals for bone mineralization. 31 Calcium in serum is either present in an ionised (or free) state or bound to proteins, principally albumin. Commonly, calcium is measured as total calcium (i.e. the sum of protein-bound, complexed and free (ionised) calcium) in plasma or serum. Consequently, variations in albumin concentration will alter total calcium levels independently of the physiologically active ionised (free) calcium concentration. In patients with hypoalbuminaemia, formulae are used to adjust patients’ total calcium result to that expected had the albumin concentration been normal. Albumin levels are lower in healthy pregnant women and the use of these formulae is essential to prevent result misinterpretation. Our results show a slight decrease in calcium and magnesium, while phosphate levels decreased after the first trimester; these findings are in accord with Larsson et al. 8
PTH RIs were significantly lower in each trimester compared to the non-pregnant adult interval. A lack of awareness of these changes in pregnancy may mask primary hyperparathyroidism or incorrectly diagnosis hypoparathyroidism. While previous studies have reported lower PTH levels31–33 during pregnancy which is consistent with our findings, others have reported unchanged levels in comparison to the non-pregnant state. 34
Sepsis has been reported as the third most common cause of maternal mortality worldwide. 35 Pregnancies complicated by sepsis are associated with increased risk of adverse outcomes. 36 CRP is widely used as a marker of inflammation in the diagnosis of sepsis. 37 In our study, CRP concentrations increased throughout pregnancy which is broadly in accord with a recent study reporting a range of 0.5–34 mg/L in a healthy pregnant cohort. 38
Cardiac troponin tests are essential for diagnosing myocardial injury; however few studies have investigated higher-sensitivity assays in pregnancy. In this study, hs-TnT values remained unchanged across the trimesters of pregnancy and were not clinically different from the non-pregnant RI established in healthy women. 39 hs-TnT has rarely been compared in pregnant and non-pregnant women. However, Adamcova et al. reported normal hs-TnT levels during the third trimester of pregnancy although the demographics of the reference population and 99th percentile URL are unknown. 40
RIs for TC, TG, LDL-C and non-HDL were significantly and clinically different from the non-pregnant RIs. The changes observed are dramatic; TC, TG and LDL-C concentrations increase throughout normal pregnancy due to reduced activity of lipoprotein lipase, insulin resistance and oestrogen stimulation.6,41 Lipid results in pregnancy are routinely assessed using RIs specific to normal non-pregnant adults. During pregnancy, normal lipid results may be considered as highly abnormal if interpretated using these inappropriate RIs.
As a result, the potential risk for misdiagnosis (e.g. familial hypercholesterolaemia) or mismanagement is increased. Moreso, evolving evidence suggests that lipids can be used for identifying maternal and foetal complications, as well as individuals at risk of developing GDM, highlighting the importance of providing RI specific to pregnancy.42–45 Women with underlying hypertriglyceridaemia are at risk of a severe rise in triglycerides during pregnancy and potentially pancreatitis. 46
Thyroid disease is common in pregnancy and when untreated can lead to a plethora of adverse outcomes. In 2014, the European Thyroid Association recommended the use of trimester-specific-RIs to avoid misclassification of thyroid dysfunction in pregnant women. 47 Our results show a decrease in both FT4 and TSH across all three trimesters in comparison to the non-pregnant RI, findings which are in accord with numerous studies reported on this topic. 48
The haematological trimester-specific-RIs for RBC, WCC, Hb, Hct, platelet count and neutrophils were strongly in agreement with those reported by Markus et al. 49 The decrease in Hb which is often referred to as ‘physiologic anaemia’ of pregnancy and decrease in Hct are common phenomena observed in pregnant women and have been previously reported.4,50 The elevation of WCC and neutrophils in all trimesters is consistent with Dockree et al. 51 The WCC is frequently measured when there is suspicion of infection; thus, it is imperative that WCC results are interpreted in the context of normal pregnancy. Furthermore, it is critical to appreciate the influence of ethnicity on haematological parameters. Compared with whites, Africans show significantly lower levels of Hb, Hct, WBC, neutrophils and platelet counts.52,53 This highlights the importance of knowing the characteristics of the reference population from which the RIs are established to ensure accurate diagnosis and patient management.
Conclusion
Education of clinicians and healthcare professionals to the physiological changes that occur in normal pregnancy together with the provision of appropriate trimester-specific-RIs on laboratory reports is essential to mitigate the clinical risk to pregnant women of missed diagnoses, misdiagnoses or mismanagement. Normative biological intervals defined in a healthy pregnant white European population for routinely requested laboratory parameters will be invaluable to result interpretation and management.
Supplemental Material
Supplemental Material - Reference intervals for clinical biochemistry and haematology tests during normal pregnancy
Supplemental Material for Reference intervals for clinical biochemistry and haematology tests during normal pregnancy by Wendy Groenendijk, Delia Bogdanet, Louise Dervan, Oisin Finn, Md Nahidul Islam, Helen Doheny, Tomas P Griffin, Liam Blake, Mark Lyons, Mary Kilcooley, Janusz Krawczyk, Ruth Gilmore, Damian G Griffin, Geraldine Gaffney, Fidelma P Dunne and Paula M O’Shea in Annals of Clinical Biochemistry
Footnotes
Author Contributions
PMOS: study concept and design, responsibility for laboratory analysis, data integrity, statistical analysis and interpretation. PMOS/DB/DGG/RG/JK/GC/FD: study conduct and protocol development. WG/DB/LD: patient recruitment, clinical data acquisition and collation. WG/LD/OF/MNI/HD/TPG/LB: laboratory analysis. HD/ML/MK: laboratory analysis, laboratory data integrity, acquisition and collation. WG and PMOS: first draft of this manuscript. All authors critically reviewed, edited and approved the final version of the manuscript.
Acknowledgements
We wish to express our gratitude to all volunteers who made this study possible. Special thanks to the scientific, nursing and medical staff at the Centre for Endocrinology, Diabetes and Metabolism and the Departments of Obstetrics and Gynaecology, Clinical Biochemistry and Haematology at Saolta University Health Care Group (SUHCG), University Hospital Galway, Ireland.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for the study was granted by the research ethics committee at Galway University Hospitals (GUH) (Ref: GUH: CA 2026 on 10.10. 2018).
Guarantor
PMOS.
Contributorship
PMOS: study concept and design, responsibility for laboratory analysis, data integrity, statistical analysis and interpretation. PMOS/DB/DGG/RG/JK/GC/FD: study conduct and protocol development. WG/DB/LD: patient recruitment, clinical data acquisition and collation. WG/LD/OF/MNI/HD/TPG/LB: laboratory analysis. HD/ML/MK: laboratory analysis, laboratory data integrity, acquisition and collation. WG and PMOS: first draft of this manuscript. All authors critically reviewed, edited and approved the final version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
