Abstract
Objective
A series of physiological changes in thyroid function occur during pregnancy and differ from those non-pregnant women. This study aimed to establish the pregnancy-specific reference intervals of TSH and FT4 using an indirect method based on the healthy pregnant women from southwest China population.
Methods
Thyroid function test results which available on the Laboratory Information System (LIS) were collected from the pregnancies who visited the Obstetric Clinic or the Department of Gynecology between 1 January 2015, and 30 December 2020. We grouped the data by trimesters to establish the reference intervals (RIs) based on the clinical consensus of different levels of TSH and FT4 at different weeks of gestation. All arrangements were referenced to the document CLSI EP28-A3C.
Results
A total of 33,040 thyroid function test results of pregnant women, aged 31 (28,33) years were statistical analyzed. Estimated RIs for TSH and FT4 in the first, second and third trimesters corresponding to the 2.5th and 97.5th percentiles in TPOAb negative were 0.02–5.23, 0.03–5.24, 0.37–5.68 mIU/L, 11.66–20.69, 10.1–18.59, 9.85–16.86pmol/L, respectively.
Conclusion
This study provides trimester-specific RIs for TSH and FT4 among healthy pregnant women in southwest China which guides clinicians to diagnosis and screen for thyroid disorders in this region.
Introduction
It is well known that a series of physiological changes in thyroid function occur during pregnancy, including a remarkable increase in the concentrations of thyroid hormone transport proteins due to the estrogen stimulation, and a decrease in thyroid stimulating hormone (TSH) levels due to rapidly elevated human chorionic gonadotropin levels.1-3 Maternal thyroid dysfunction is associated with an increased risk of various adverse maternal and child outcomes, including miscarriage, intrauterine growth retardation, hypertensive disorders, preterm delivery, and fetal mortality.4-9 Therefore, it is critical to evaluate thyroid function accurately during pregnancy.
The reference intervals (RIs) of hormones related to thyroid function are one of the critical basis for clinicians to judge whether the test result is normal or not and to make clinical decisions. Serum TSH and FT4 vary with different regions, ethnic, genders, and gestational age.4, 10,11 To date, several studies and guidelines12-15 encourage every laboratory to establish its own RI and recommend using pregnancy specific RIs for thyroid hormones during pregnancy. Meanwhile, guidelines 13 issued by the American Thyroid Association (ATA) for the diagnosis and management of thyroid diseases during pregnancy and the postpartum period recommend “Accurate estimation of the FT4 concentrations can also be done by calculating a FT4 index.” Caution is advised about this view. 16 The FT4 index should not be used as a substitute for trimester-specific reference intervals which is a calculated value based on the total T4 measurement and thyroid-binding capacity. It was valuable in the era when reliable commercial FT4 assays were not readily available. However, in view of pregnant women have higher concentrations of thyroxine-binding globulin and nonesterified fatty acid that could, in theory, alter the properties of immunoassays developed and validated with use of non-pregnant serum, the immunoassays could potentially be inaccurate during pregnancy. So, there is no doubt that trimester-specific reference intervals should be established for each commercial immunoassay. Nowadays, commercial immunoassays of TSH and FT4 are the mainstreams, and it is essential to establish assays and population-specific reference intervals.
Although some researches17-20 have reported the RIs of TSH and FT4 in China, there have not been reported the pregnant women from southwest China population so far, and these platform-specific reference intervals cannot be found in a related recommendation list of Chinese authoritative guidelines. 21 As a consequence, maternal thyroid hormones assessment should be performed using pregnancy-specific reference intervals ideally based on locally pregnant women data from southwest China population.
The document CLSI EP28-A3C published by the Clinical and Laboratory Standards Institute expound that the direct method for establishing RIs strictly requires the inclusion of a large number of individuals with normal thyroid function, whereas the indirect method is based on data and easier to perform. 12
Accordingly, this study aimed to establish the pregnancy-specific RIs of TSH and FT4 using the indirect method based on the healthy pregnant women from southwest China population Which guides clinicians to diagnosis and screen for thyroid disorders in this region.
Materials and methods
Data collection
Thyroid function test results were collected from 33,040 pregnant women who visited the Obstetric Clinic or the Department of Gynecology at the West China Second Hospital of Sichuan University between 1 January 2015, and 30 December 2020. According to clinical and demographic information available on the Laboratory Information System (LIS), pregnant women with personal history of thyroid disease, receiving treatment with drugs able to influence thyroid function, i.e. amiodarone, lithium, levothyroxine, or anti-thyroid treatment were excluded. Women with either multiple gestation pregnancies or relevant abnormalities detected by echography were excluded to establish TSH and FT4 reference values in each trimester. For clinical reasons, we also excluded those women with TSH values<0.01mIU/L and FT4 level above the general population reference value (0.55–4.78 mIU/L for TSH and 11.5–22.7pmol/L for FT4) in trimesters, since these women were considered to suffer from undiagnosed thyroid disease. The first trimester (T1) was defined as 4–12 weeks gestation, second trimester (T2) was defined as 13–28 weeks gestation, and third trimester (T3) was defined as > 28 weeks gestation. Gestational ages were determined using the calculations based on the last menstrual period (LMP), or early obstetric scan if available. Owing to the retrospective and anonymous nature of this study, informed consent was not required. This study protocol was approved by the Ethics Committee of West China Second Hospital of Sichuan University and was performed in accordance with the Declaration of Helsinki.
Instruments and reagents
All thyroid hormones, including TSH, FT4 and TPOAb were measured with an ADVIA Centaur XPT automatic chemiluminescence immunoassay analyzer (Siemens Healthineers, Erlangen, Germany) using supplied reagents and calibrators. Our laboratory uses Westgard multi-rules (12s,13s,22s, R4s) for internal quality control. Results were determined via a calibration curve, which is instrument specifically generated, and a master curve provided via the reagent barcode. The analytical measuring range for TSH was 0.005–150 mIU/L and that of FT4 was 1.3–155 pmol/L22. A value of less than 60 kIU/L for TPO antibodies was considered negative as per the manufacturer quoted reference range. The cumulative coefficient of variation (CV) at both high and low levels were less than 8%. Over the past years, we also participated in External Quality Assessments by the National Center for Clinical Laboratories and the College of American Pathologists to guarantee the accuracy and reliability of our results.
Reference intervals establishment methods
First, we used the Skewness–Kurtosis test to analyze the normality of the data. When the skewness value and the kurtosis value are less than 1.96 times, the standard deviation of the corresponding skewness and kurtosis, respectively, it can be judged as a normal distribution. The non-normally distributed data is transformed into approximately normal distribution by the BOX-COX transformation. Second, Tukey method was used to eliminate outliers. The upper limit is P75 plus 1.5 times the interquartile range (IQR), and the lower limit is P25 minus 1.5 times the IQR. All values outside this range should be considered outliers. The standard normal deviate test (z-test) suggested by Harris and Boyd was used to assess whether or not to partition RIs by the subclass. Finally, the non-parametric method was used to estimate the 95% distribution RI of TSH and FT4. All arrangements were referenced to the document CLSI EP28-A3C published by The Clinical and Laboratory Standards Institute.
Statistical analysis
All statistical analyses were performed using the Excel 2010 (Microsoft Inc., Redmond, WA, USA) and SPSS 21.0 software (IBM Inc., Armonk, NY, USA). Kolmogorov-Smirnov tests were used to evaluate the distribution of data; normally distributed data are presented as mean ± SD, whereas non-normally distributed data are presented as the medians (quartiles). Using Stata 15.0 statistical software performed Box-Cox normality transformation of data (undetermined parameters λ are obtained by maximum likelihood method). Interquartile ranges were also calculated, and non-parametric analyses were applied to calculate the median, as well as the 2.5th and 97.5th percentiles (P2.5–P97.5) using SPSS. p-values <0.05 were considered statistically significant.
Results
Distribution of data and elimination of outliers
According to clinical and demographic information available on the Laboratory Information System (LIS), a total of 33,040 thyroid function test results of pregnant women, aged 31 (28,33) years were statistical analyzed (Figure 1). 476 test results of pregnant women showed TSH and FT4 values out-of-range and 807 test results of pregnant women who had multiple gestations. We further excluded 1434 individuals who received treatment with drugs or abnormalities detected by echography. Ultimately, 30,323 thyroid function test results of pregnant women who did not show significant abnormalities in results of the physical examination were included. Positive TPOAb were detected in 10,358 out of the 30,323 participants (34.16%). The data shows a non-normal distribution by the Skewness–Kurtosis test (Supplement Table S1), and it turns into an approximately normal distribution after Box-Cox conversion. Tukey method was used to eliminate 916 cases of outliers (TPOAb negative: 210 cases; TPOAb positive: 706 cases) (Supplement Table S2). Flow chart of sample sources and exclusions.
Levels of TSH and FT4 in different subgroups across pregnancy
TSH and FT4 levels showed differences between gestational weeks. As Figure 2 shows, TSH levels increased across the pregnancy period, and were significantly higher in the second and third trimesters with respect to the first one. On the other hand, FT4 levels decreased significantly and continuously across the pregnancy period. There were significant differences in TSH levels between TPOAb negative and positive pregnant women among the same gestational weeks. In contrast, the difference of FT4 levels in early pregnancy was not statistically significant between TPOAb negative and positive pregnant women. And differences of FT4 levels between mid- and late-pregnancy in TPOAb negative and positive pregnant women were consistent with the performance of TSH levels. The distribution of TSH and FT4 in different subgroups across pregnancy.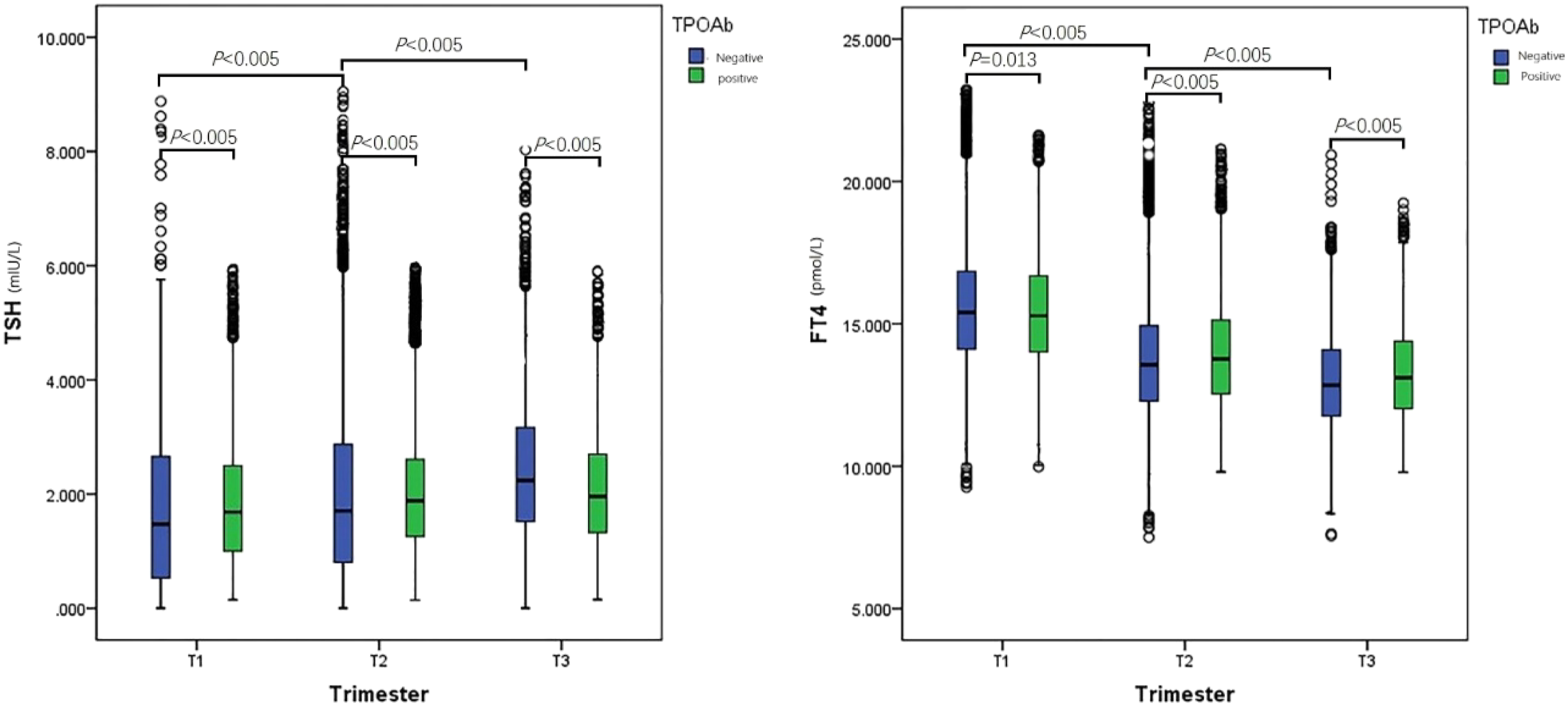
Establishment of the reference interval
Numbers of pregnant women in different gestational weeks.
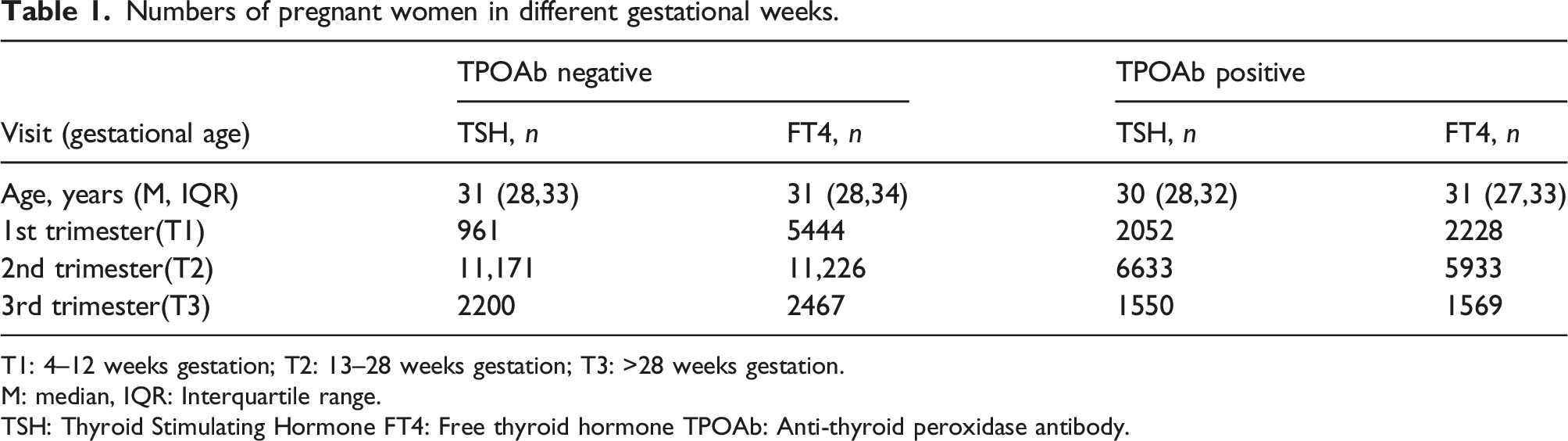
T1: 4–12 weeks gestation; T2: 13–28 weeks gestation; T3: >28 weeks gestation.
M: median, IQR: Interquartile range.
TSH: Thyroid Stimulating Hormone FT4: Free thyroid hormone TPOAb: Anti-thyroid peroxidase antibody.
Reference intervals of TSH and FT4 in pregnant women with negative and positive thyroid autoantibodies (P2.5, 95% CI, P 97.5,95% CI).
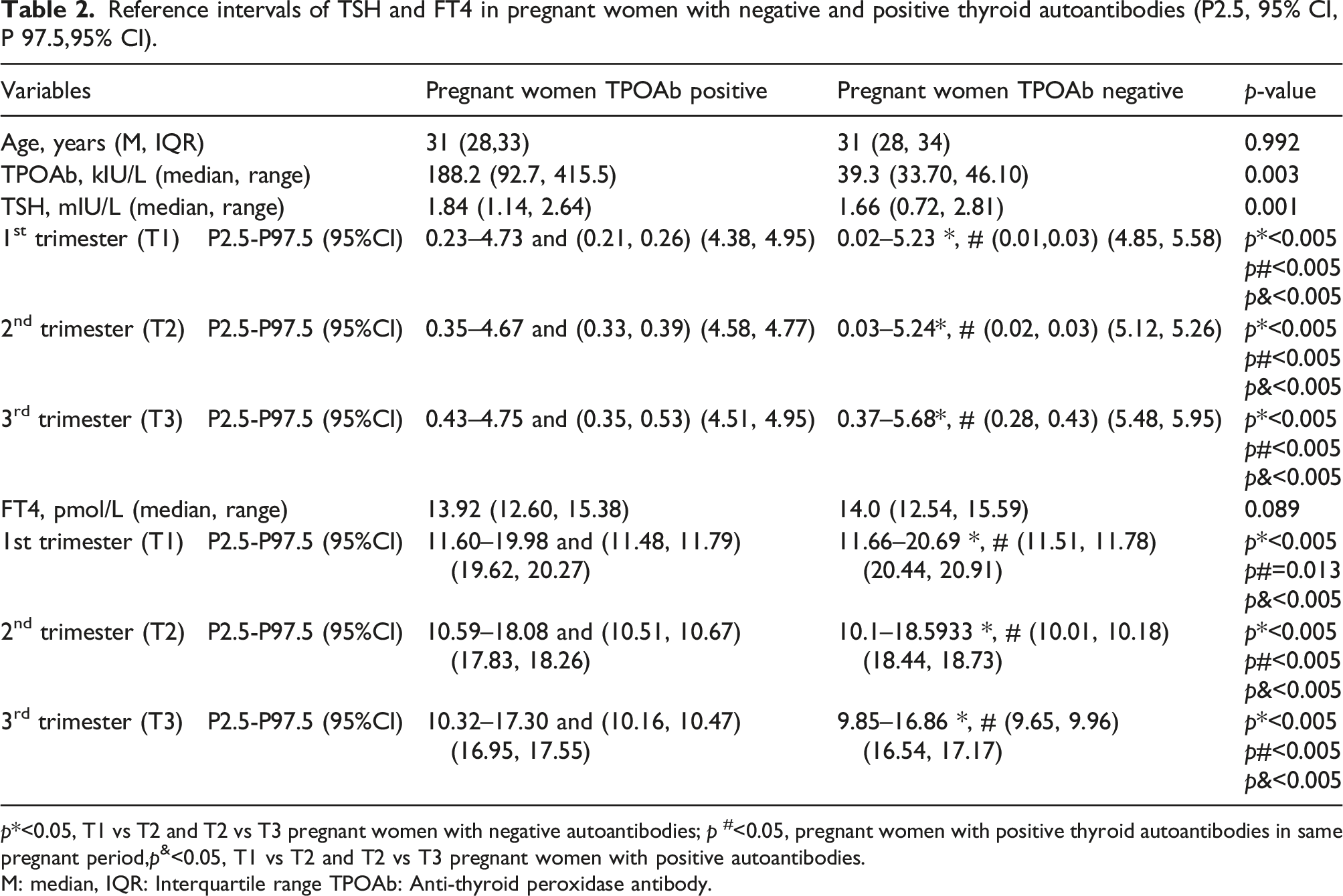
p*<0.05, T1 vs T2 and T2 vs T3 pregnant women with negative autoantibodies; p #<0.05, pregnant women with positive thyroid autoantibodies in same pregnant period,p&<0.05, T1 vs T2 and T2 vs T3 pregnant women with positive autoantibodies.
M: median, IQR: Interquartile range TPOAb: Anti-thyroid peroxidase antibody.
Comparison of TSH and FT4 RIs in pregnant women across previous studies
Reference intervals of TSH and FT4 among TPOAb negative pregnancy established by other studies using indirect methods in recent years.
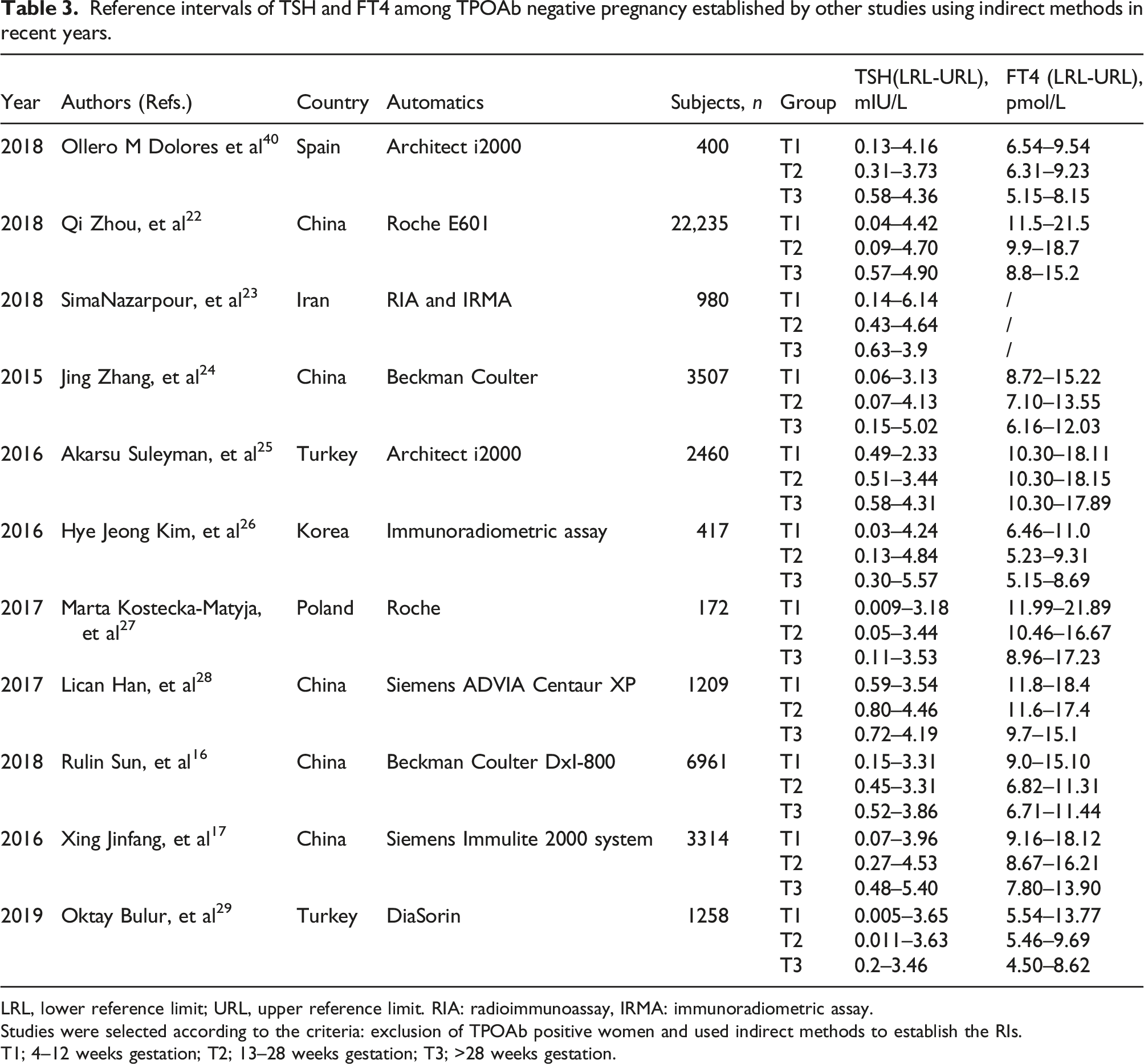
LRL, lower reference limit; URL, upper reference limit. RIA: radioimmunoassay, IRMA: immunoradiometric assay.
Studies were selected according to the criteria: exclusion of TPOAb positive women and used indirect methods to establish the RIs.
T1; 4–12 weeks gestation; T2; 13–28 weeks gestation; T3; >28 weeks gestation.
Discussion
Since the thyroid-related hormones and antibodies have a vital impact on the health of pregnant women and fetuses, at present, the establishment of specific reference intervals for pregnancy women has attracted worldwide interest. In China, the clinical laboratory should verify whether the reference intervals provided by the manufacturer’s documentation in the test kit are applicable to the specific patient population in the local area or local hospital. Hence, we conducted this study to establish specific thyroid hormones RIs for pregnant women in Southwest China using clinical “big data.”
The documents CLSI EP28-A3C recommend the direct method to establish the RI. However, to produce high quality reference values using the direct way according to the recommendations of CLSI EP28-A3C for all relevant analytics is far beyond the capacity of a single laboratory.12,31 With the development of information technology, the method of using the indirect method to establish the RI by using a large amount of data stored in the LIS has also been accepted by EP28-A3C and used in many studies.32,33 Therefore, we used an indirect method, which avoids some of these hurdles and uses data stored in our laboratory information system, to establish TSH and FT4 RIs.
Our results showed that TSH levels increased across the pregnancy period, with the prolongation of pregnancy, the FT4 level exhibited a gradually decreasing trend, which is consistent with other research studies.19,34-36 The TSH levels of pregnant women in the TPOAb positive group were higher than those in the TPOAb negative group at all stages, and the trend of change was consistent with the TPOAb negative pregnant women. Due to the negative impact of TPOAb positive on pregnancy,37,38 some studies have excluded TPOAb positive people when establishing pregnancy-specific reference intervals for TSH or FT4. Our study did not exclude TPOAb positive pregnant women but divided them into separate groups to establish the reference intervals of TSH and FT4 according to the non-parametric method. Interestingly, the RIs of TSH and FT4 in the TPOAb positive group were narrower than those in the TPOAb negative group. The reason for this results may be that the distribution of TSH levels in antibody-positive pregnant women was less discrete than in the antibody-negative group (QP=1.5<QN=2.09), and the former group ought to be less susceptible to the influence of hCG. 39 The antibody levels did not significantly change during the entire pregnancy but were all significantly lower than the RIs proposed by the manufacturers. This may be due to the fact that pregnancy is a state of subtle immunosuppression. 40 Therefore, a true positive antibody may be undetected when we use the criteria proposed by the manufacturer to assess the level of antibodies, thereby increasing the rate of misdiagnosis of subclinical hypothyroidism (SCH). Since TPOAb is also an important parameter in the diagnosis and treatment of hypothyroidism in pregnancy, this parameter should be taken into account when laboratories establish regional reference intervals for thyroid hormones,and should not only establish the regional-specific references intervals for thyroid hormones but also those for thyroid-related antibodies.
The reference intervals of TSH and FT4 are influenced by different regions, races, detection systems and other factors.4,18,24,26-29,41 Similar to our research, for women in the same stage of pregnancy, reference intervals determined by different detection systems differed but exhibited the same trends. Our results show that the upper limits of the RIs for TSH are slightly higher than recommended by the American Thyroid Association (ATA) 13 in the first, second, and third trimesters of pregnancy (i.e. 5.23,5.24, and 5.68 mIU/L, compared to the recommended 2.5 mIU/L in the first trimester of pregnancy and 3 mIU/L in the second and third trimesters, respectively. This is consistent with the results of other researchers who developed the reference intervals for pregnancy24,26,28 and confirm the consensus that RIs determined by a variety of factors.
Some limitations to this study must be addressed. First, despite our strict exclusion criteria, it would have been ideal if we could exclude those women who were healthy at the time of recruitment but developed complications during pregnancy. Second, in this study, we did not collect the iodine level of pregnant women, which may bias the true results of TSH and FT4 to some extent. Sichuan is a province in which the external environment is generally in a state of iodine deficiency, after the adjustment of the iodized salt concentration in 2012, the iodine nutritional level of key populations has generally maintained an appropriate state, and it can be considered that this bias has little effect on the results. Third, we were not able to recruit a non-pregnant group of women which could provide better comparisons for our results.
Conclusion
This study focused on reference intervals in gestational thyroid function and reported, for the first time, the differences between TPOAb positive and negative from the same platform in China. The RI is the standard of making a medical diagnosis, therapeutic assessment, or other physiological assessment. Our findings suggest that the TSH and FT4 reference intervals in TPOAb positive women during pregnancy are different from those of TPOAb negative individuals and should be differentiated for diagnosis.
Supplemental Material
sj-pdf-1-acb-10.1177_00045632211063142 – Supplemental Material for Establishment of trimester-specific reference intervals for thyroid stimulating hormone and free thyroxine during pregnancy in southwest China by indirect method
Supplemental Material, sj-pdf-1-acb-10.1177_00045632211063142 for Establishment of trimester-specific reference intervals for thyroid stimulating hormone and free thyroxine during pregnancy in southwest China by indirect method by Ying Guo, Bin Wei, Wei Dai, and Hongjian Xie in Annals of Clinical Biochemistry
Footnotes
Acknowledgements
We give thanks to LIS staff for providing data for the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study protocol was approved by the Ethics Committee of West China Second Hospital of Sichuan University and was performed in accordance with the Declaration of Helsinki.
Guarantor
YG.
Contributorship
All the authors were involved in drafting the article. YG and BW designed the structure of the article and performed the statistical analysis. YG wrote the first draft of the manuscript and WD revised it and helped in writing its final section. All authors approved the final version.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
