Abstract
Background
Trimester-specific reference intervals (RIs) for thyroid function tests are lacking for Beckman Dxl 800 analysers. We aimed to establish RIs for thyroid stimulating hormone (TSH), free thyroxine (fT4) and to track intraindividual changes in thyroid function throughout pregnancy.
Methods
One hundred and thirty healthy women without antithyroid peroxidase antibodies were followed longitudinally. Thyroid function was determined at trimester-1 (T1): 9–13 weeks; trimester-2 (T2): 22–26 weeks; trimester-3 (T3): 35–39 weeks and postpartum (PP): 8–12 weeks. A subgroup (n = 47) was used to track intraindividual changes using PP as non-pregnant state (baseline).
Results
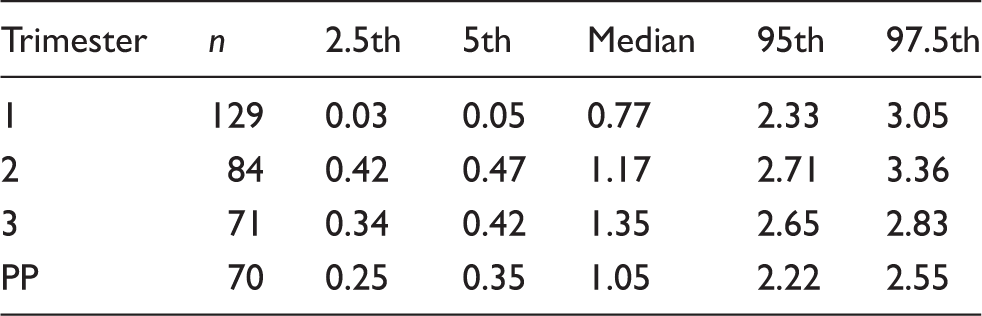
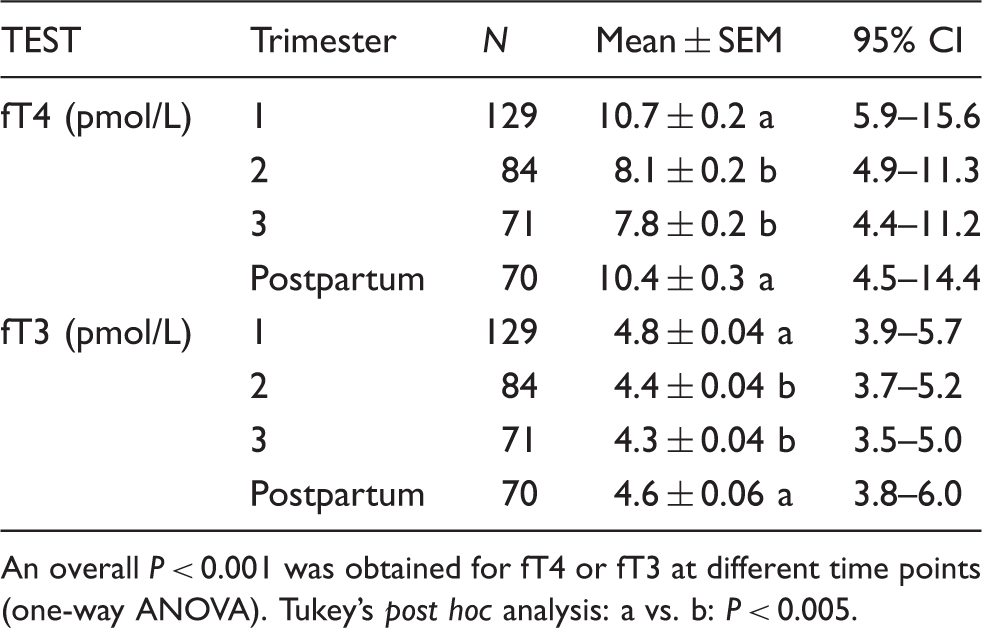
For trimesters 1–3, TSH (median (2.5th, 5th, 95th and 97.5th percentile)) was 0.77 (0.03, 0.05, 2.33, 3.05), 1.17 (0.42, 0.47, 2.71, 3.36) and 1.35 (0.34, 0.42, 2.65, 2.83) mIU/L, respectively. Free T4 (mean (95%CI)) was 10.7 (5.9–15.5), 8.1 (4.9–11.3), 7.8 (4.5–11.0) pmol/L, respectively. In T2 and T3, 36% and 41% of the fT4 values, respectively, fell below the non-pregnancy lower normal limit. In the subgroup assessed for longitudinal changes, of the women with baseline TSH ⩽ median, 71–75% remained at or below the corresponding median for trimesters 1–3. Of the women with baseline fT4 ⩽ median, 69–81% also remained at or below the corresponding median for trimesters 1–3. High correlation was observed at different trimesters and baseline for TSH (Spearman’s r: 0.593–0.846, P < 0.001) and for fT4 (r: 0.480–0.739, P < 0.001).
Conclusions
Use of trimester-specific RIs would prevent misclassification of thyroid function during pregnancy. In the majority of women, TSH and fT4 tracked on the same side of the median distribution, from a non-pregnant baseline, throughout pregnancy.
Keywords
Introduction
There is increased interest in identifying changes in thyroid function during pregnancy due to the role of the thyroid gland in maintaining a viable pregnancy and contributing to healthy offspring. It is known that overt hypothyroidism is associated with adverse effects on pregnancy and fetal development. 1 However, adverse effects of maternal subclinical hypothyroidism on pregnancy2–5 or neurological outcome in the fetus is less clear.6,7
Thyroid stimulating hormone (TSH) concentration is known to decrease throughout the pregnancy, especially in the first trimester (T1), under the influence of the placental human chorionic gonadotropin (βhCG). 8 Recent guidelines recommend to use method and trimester-specific reference intervals (RIs) for TSH.9,10 When these are not available in a particular laboratory, the following common RIs can be used: 0.1–2.5, 0.2–3.0 and 0.3–3.0 mIU/L in trimester 1 (T1), trimester 2 (T2) and trimester 3 (T3), respectively,9,10 based on the assumption that different TSH methods are relatively consistent and reliable. 9 For free thyroxine (fT4) in pregnancy, however, common RIs cannot be recommended 9 due to wide variations among different fT4 methods. 11 Pregnancy is associated with an increase in serum thyroid binding globulin, free fatty acid concentrations and a decrease in albumin concentration, resulting in a method-dependent decrease in serum fT4 concentrations seen with most common immunoassays. 11 Thus, method-specific and trimester-specific intervals are requested for accurate assessment of fT4 during pregnancy.9,10
Over the past decade, there have been a number studies seeking to determine RIs for thyroid function in pregnancy using different analytical systems8,12–15 and most have been cross-sectional in design. Prior to commencement of this study, pregnancy associated RIs for thyroid function from either cross-sectional or longitudinal studies were not available for the Beckman Dxl 800, a common analytical system for immunoassays in Australia. This current prospective, longitudinal study was therefore conducted to address the absence of trimester-specific RIs for thyroid function tests for the Beckman Dxl 800. Furthermore, few prospective studies have assessed intraindividual variation of maternal thyroid function (i.e. whether women’s thyroid function ‘track’ during pregnancy)16,17 and none have compared tracking of thyroid function in pregnancy to non-pregnant values. A further aim in this study was therefore to examine the tracking of TSH and fT4 values from the non-pregnant baseline and throughout pregnancy.
Materials and methods
Between May 2006 and February 2011, women presenting to Mercy Hospital for Women (MHW), a tertiary obstetric hospital in Melbourne, at or prior to 13 weeks gestation were recruited. Exclusion criteria included; past history of thyroid disease, thyroid hormone replacement therapy, type 1 diabetes, past history of intravenous drug abuse or the presence of major systemic illness. Women who were Thyroid Peroxidase Antibody (TPOAb) positive initially (weeks 9–13) were also excluded. Blood samples (20 mL each) were taken at 9–13 weeks, 22–26 weeks and 35–39 weeks of gestation to represent T1, T2 and T3. An additional blood test was taken between 8 and 12 weeks postpartum (PP) to represent the non-pregnant state.
Serum TSH, fT4 and fT3 concentrations were measured at all time points. Spot urinary iodine concentration (UIC) was also measured initially to classify the iodine status of this study population. The Human Research Ethics Committee at MHW approved the study and written informed consent was obtained from each study participant.
All serum specimens were frozen after collection and analysed as a batch for TSH, fT4 and fT3 at the conclusion of the study. However, to ensure that unexpected abnormal results were not missed during pregnancy, all initial samples were also analysed on the day of collection for TSH, fT4 and fT3 and these results were available to the treating doctors. At the time when this study was conducted, it was a routine practice at MHW to use a first trimester target TSH of ⩽2.5 mIU/L as recommended by international guidelines. 9 Thus, of the four patients with TSH values between 2.5 and 3.6 mIU/L in this study, three were commenced on thyroxine. The initial (pre-thyroxine therapy) blood samples of these patients were included in the analysis but once commenced on thyroxine, no subsequent samples from these patients were included. At the PP time point, results from five women were excluded from analysis due to biochemical evidence of PP thyroiditis (four had hyperthyroidism with TSH ⩽0.01 mIU/L and one had hypothyroidism TSH = 14 mIU/L). All five women had negative TPOAb, so results from other time points were kept for the analyses.
Longitudinal assessment for tracking intraindividual changes in TSH and fT4 was performed on a subgroup of 47 women who had a full set of four samples available across each trimester and PP. Data from postpartum bloods were used to represent non-pregnant state (baseline) for each woman. These women were grouped according to whether they were above or below (i) the baseline median TSH or (ii) the baseline median fT4. We examined whether TSH and fT4 values track, in the same side of the median, from the non-pregnant state to T1 and from T1 throughout the pregnancy to T2 and T3. We also examined the relative effects of β-hCG and baseline TSH on TSH values in T1.
Assay methods
Serum TSH, fT4, fT3 and TPOAb were measured by paramagnetic particle, chemiluminescent immunoassay using a Beckman Dxl 800 analyser. Urine iodine concentration was measured by the inductively coupled plasma mass spectrometry method using the Agilent 7500 CE instrument with an octopole reaction cell. The non-pregnancy normal limits quoted by the manufacturer over the study period were 0.35–5.60 mIU/L for TSH, 7.5–21.1 pmol/L for fT4, and 3.3–6.4 pmol/L for fT3. An internal validation of the manufacturer’s recommended lower limit for fT4 was obtained by means of a data extraction of all the thyroid function tests performed over the study period; between May 2006 and Feb 2011. Of 13,570 men and women with TSH between 0.5 and 2.5 mIU/L, the fT4 2.5th percentile was 7.3 pmol/L, similar to the manufacturer’s recommended lower limit of 7.5 pmol/L. A serum TPOAb ⩾ 10.0 IU/mL was considered as positive. The limit of detection for TSH was 0.01 mU/L. The between run coefficients of variation were 7% for TSH, 8% for fT4, 11% for fT3, 4% for TPOAb and 2.3% for urinary iodine at 92 µg/L.
Statistical analysis
All the parameters were tested for normality. Due to non-Gaussian distribution of TSH values, TSH data were presented as percentiles (2.5th, 5th, 50th, 95th and 97.5th).
The RI for TSH was defined as the 2.5th–97.5th percentile values calculated using Excel 2007. fT4 and fT3 were normally distributed, so data are shown as mean ± SEM, and the RIs were defined as the 95% confidence intervals. All other statistical analysis was performed using SPSS for Windows (Version 17, Chicago, IL, USA). One-way analysis of variance (ANOVA) followed by Tukey’s post hoc analysis was used to assess the differences in trimester-specific results for TSH (log-transformed), fT4 and fT3. A P value < 0.05 was considered statistically significant.
Spearman rank correlation was used to examine various relationships between TSH, fT4 and β-hCG during pregnancy versus the non-pregnant state. Logistic regression analysis was performed to examine the influence of different factors on TSH values in T1. The independent variable (dichotomous) was whether the T1 TSH value was below the median value, while the dependent variables were β-hCG at T1, baseline TSH and maternal age.
Results
One hundred and fifty four women were recruited into the study. Seven were excluded due to miscarriage prior to 20 weeks gestation, one due to a history of intravenous drug abuse, and two due to twin pregnancy. Of the remaining 144 women, a further 14 were TPOAb positive thus were also excluded, leaving 130 women in the final study. Overall, data were obtained from 129 women in T1 (one missing datum), 84 in T2, 71 in T3 and 70 in PP.
At recruitment, the mean age was 31 ± 4.7 years. Urine iodine concentration (median (2.5th–97.5th)) from 124 women was 146 (34–967) µg/L. There were no statistically significant differences found in the TSH (log-transformed) or fT4 values of women with UIC of <100 µg/L versus those with concentrations of ⩾100 µg/L. Therefore, women who had UIC of <100 µg/L were not excluded in the analysis.
Reference intervals
Trimester-specific TSH values (2.5th, 5th, 50th, 95th and 97.5th) for pregnant women with TPOAb negative are shown in Table 1. The mean ± SEM as well as trimester-specific 95% confidence intervals for fT4 and fT3 are shown in Table 2. An overall P < 0.001 was obtained when comparing TSH (log-transformed), fT4 and fT3, in the different trimesters and PP, by one-way ANOVA. The longitudinal changes in TSH, fT4 and fT3 across each trimester and the PP state are demonstrated in Figure 1. Post hoc analysis showed that the mean TSH was lower in T1 compared to either T2, T3 or PP (P < 0.005). Conversely, fT4 and fT3 were significantly higher (P < 0.005) in T1 compared to T2 or T3 (Figure 1).
TSH, fT4 and fT3 across each trimester and postpartum. Trimester-specific reference intervals for TSH (mU/L) using the Beckman Dxl 800 assay in women with negative antithyroid peroxidase antibody. Trimester-specific reference intervals for fT4 and fT3 in women with negative antithyroid peroxidase antibody. An overall P < 0.001 was obtained for fT4 or fT3 at different time points (one-way ANOVA). Tukey’s post hoc analysis: a vs. b: P < 0.005.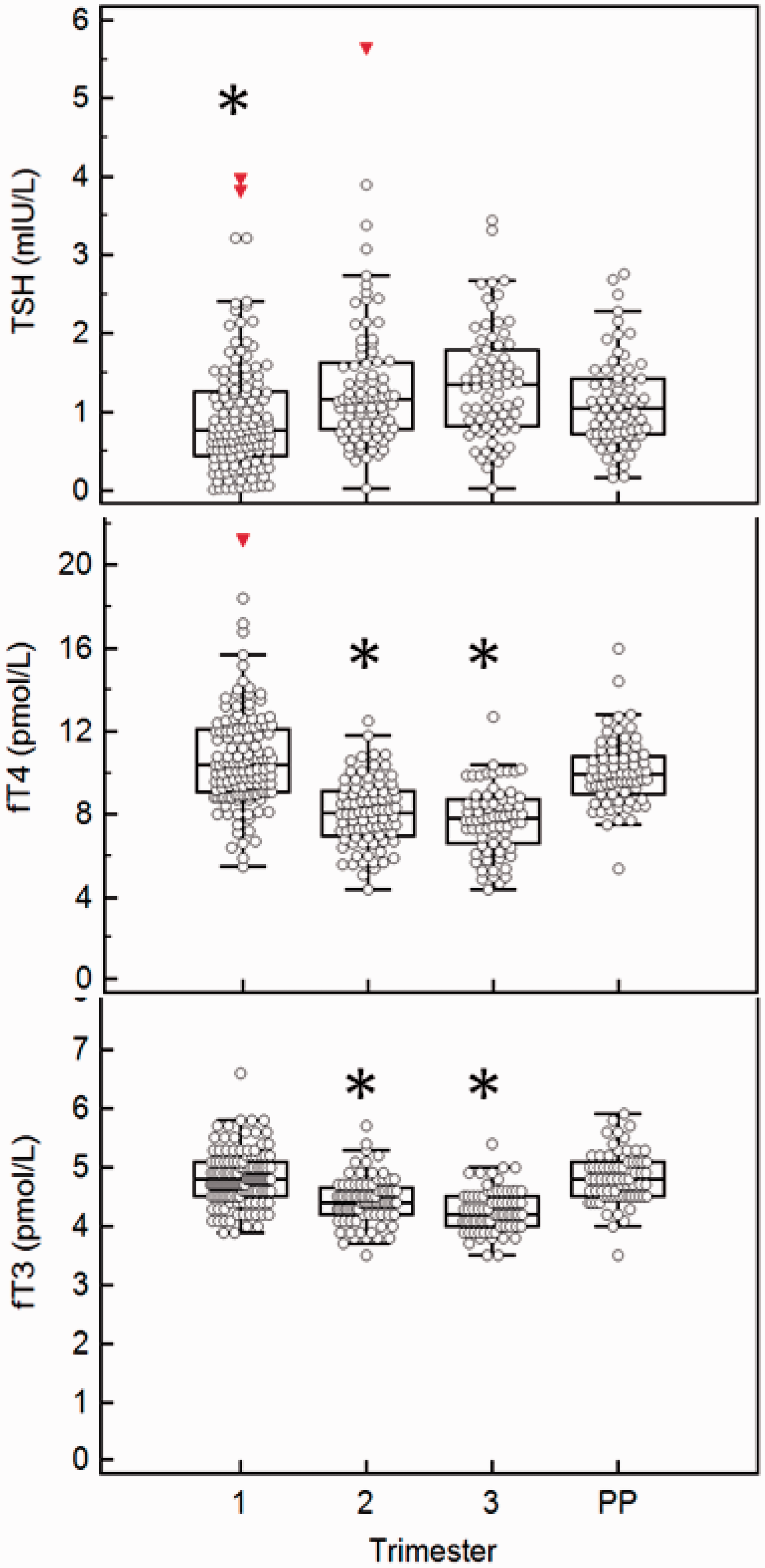
Proportion of women with negative antithyroid peroxidase antibodies whose TSH, fT4 or fT3 values in different trimesters would be below the non-pregnancy normal limits quoted by the laboratory.
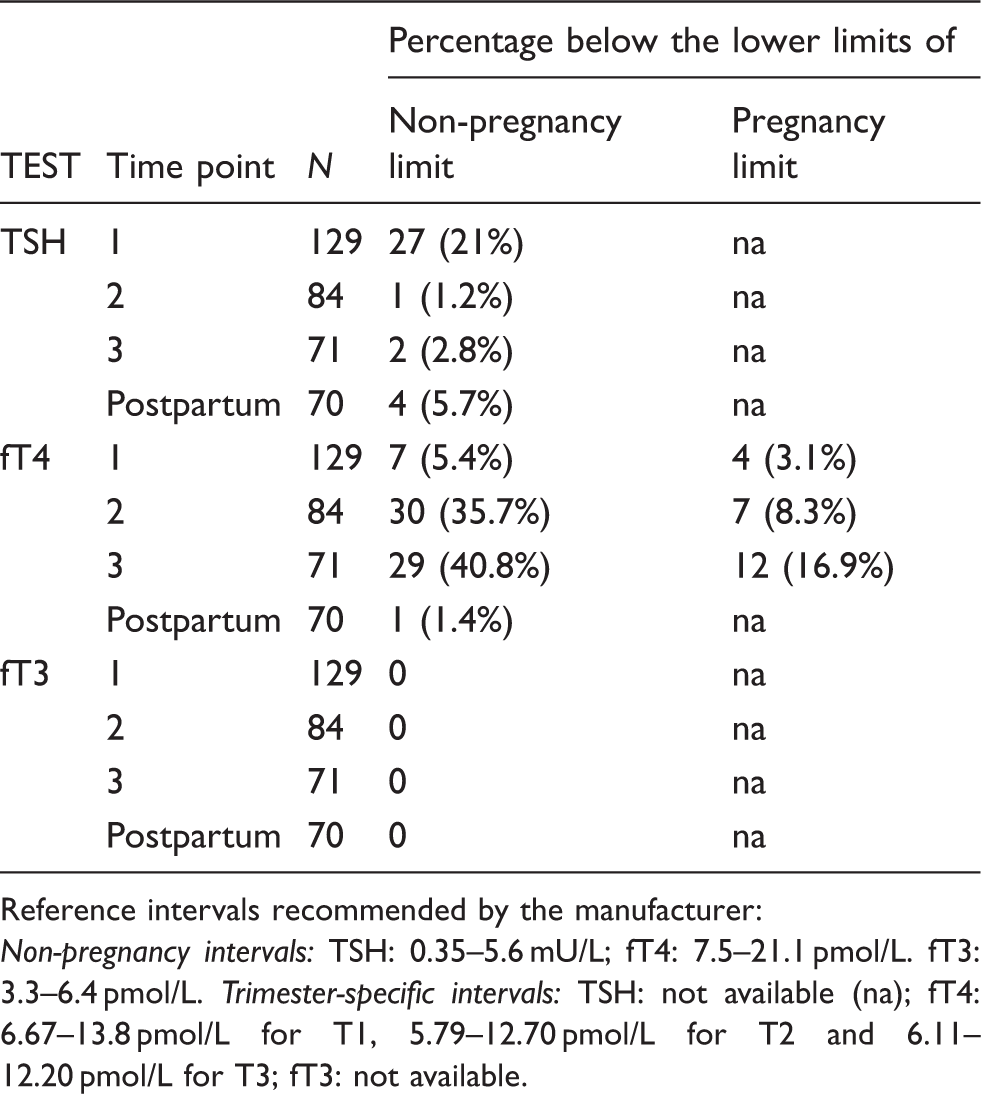
Reference intervals recommended by the manufacturer:
Non-pregnancy intervals: TSH: 0.35–5.6 mU/L; fT4: 7.5–21.1 pmol/L. fT3: 3.3–6.4 pmol/L. Trimester-specific intervals: TSH: not available (na); fT4: 6.67–13.8 pmol/L for T1, 5.79–12.70 pmol/L for T2 and 6.11–12.20 pmol/L for T3; fT3: not available.
Longitudinal analysis
In the longitudinal subgroup analysis of 47 subjects, TSH changes in each woman over time are presented in Figure 2 where women were grouped according to baseline TSH of below median (Figure 2(a)) or above median (Figure 2(b)). Of the 23 women with baseline TSH below the median of 1.04 mIU/L (Figure 2(a)), 16 (70%) also had T1 TSH below its respective median of 0.85 mIU/L (grey lines). Conversely, of the 23 women with baseline TSH above the respective median of 1.04 mIU/L (Figure 2(b)), 16 (70%) also had T1 TSH above its respective median of 0.85 mIU/L (grey lines). Tracking TSH values from T1 to T2 and T3, it was found that of the 23 women with a T1 TSH below the median of 0.85 mIU/L, 88% in T2 and 83% in T3 remained below the respective median TSH values and these proportions were not significantly different (χ2 = 4.13, P = 0.3).
Longitudinal changes in TSH and fT4 throughout pregnancy and postpartum.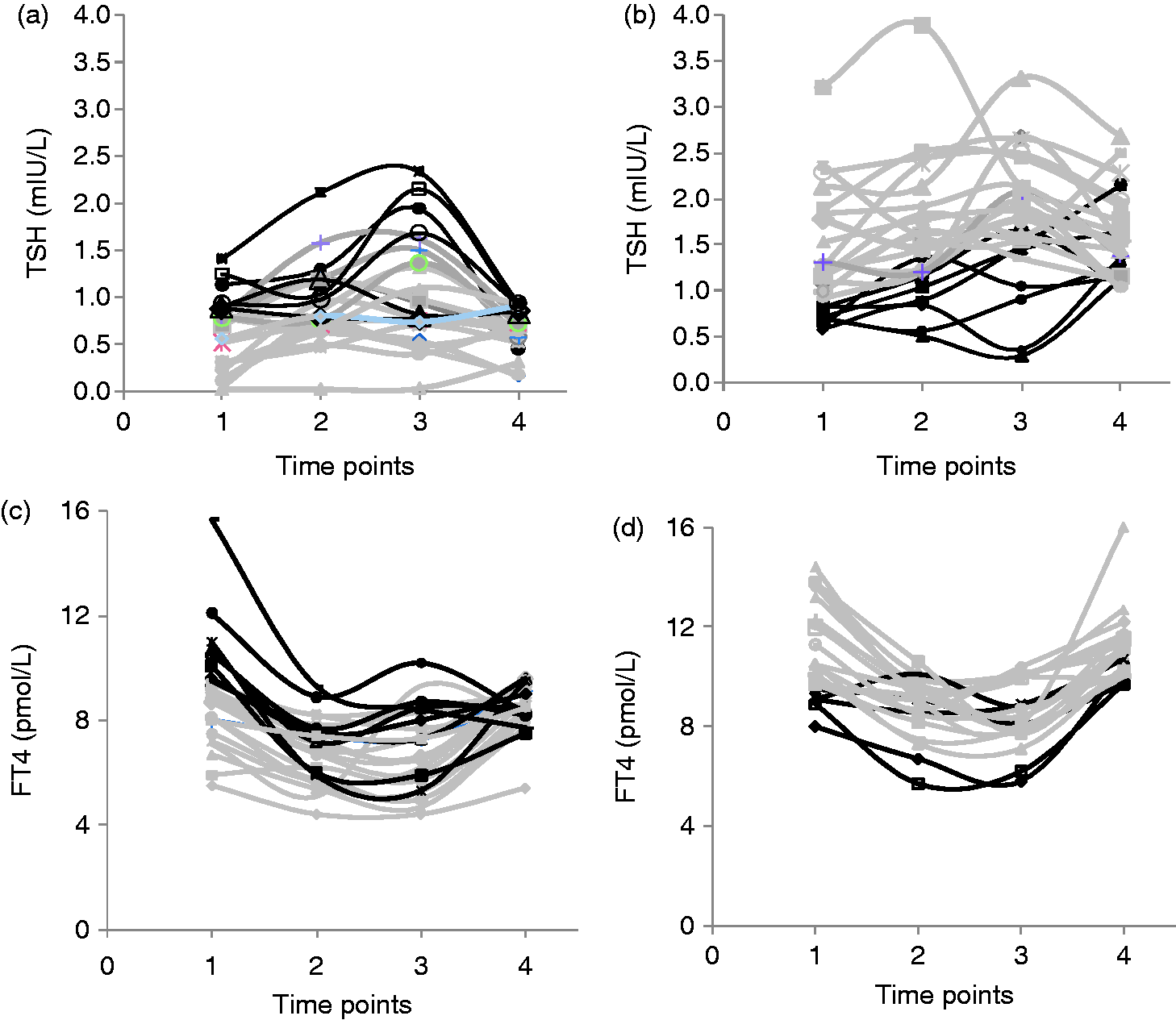
Similarly, Figure 2 shows fT4 changes in each woman over time where women were grouped according to baseline fT4 of below the median (Figure 2(c)) or above the median (Figure 2(d)). Of the women with baseline fT4 at or below the median value of 9.6 pmol/L (Figure 2(c)), 73% remained at or below the corresponding median of 9.5 pmol/L in T1 (grey lines). Conversely, of the women with baseline fT4 above the median (Figure 2(d)), 76% remained above the corresponding median in T1 (grey lines).
Spearman rank correlation of TSH, fT4 and β-hCG values at each trimester and postpartum.
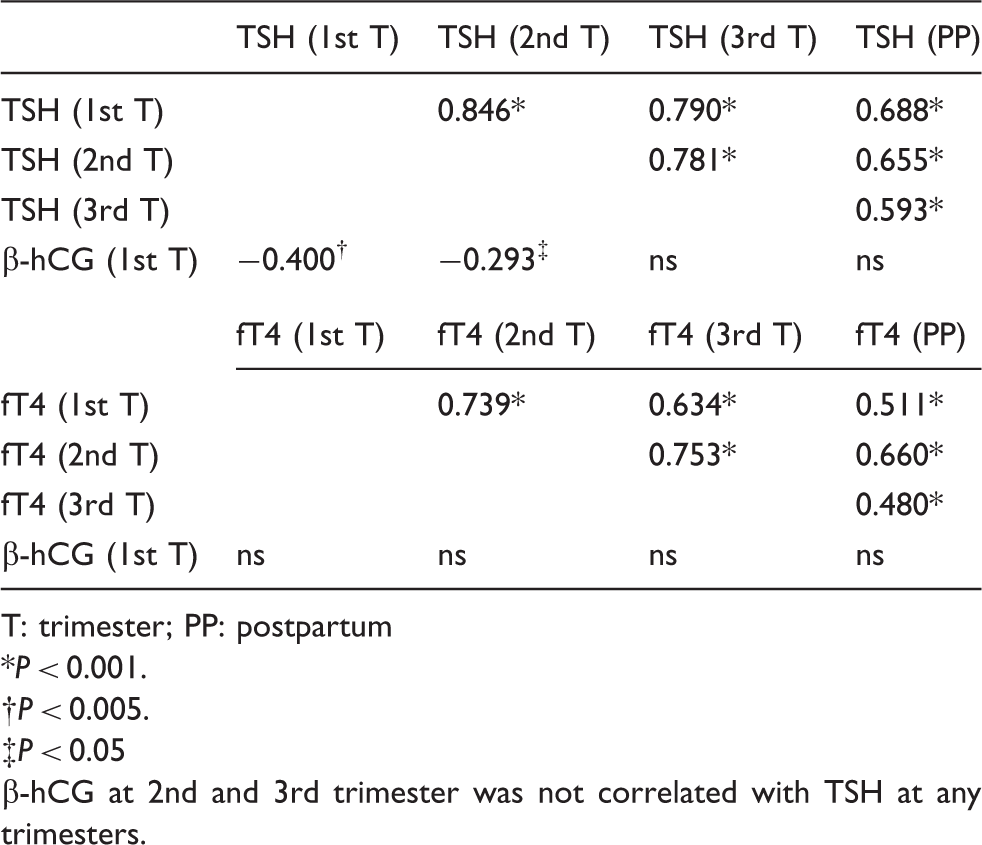
T: trimester; PP: postpartum
P < 0.001.
P < 0.005.
P < 0.05
-hCG at 2nd and 3rd trimester was not correlated with TSH at any trimesters.
Discussion
This study, for the first time, has presented data for thyroid function in pregnancy on the Beckman system and provided a guide to the RIs expected. The current study was conducted because of an absence of pregnancy and trimester-specific RIs for thyroid function tests for the Beckman Dxl 800, a common analytical system in Australia. The current study also addresses a common clinical problem of misclassifying pregnant women with abnormal thyroid function, which would not have occurred if pregnancy and trimester-specific RIs were used. In addition, the subgroup analysis of tracking of thyroid function in pregnancy, as well as correlation with non-pregnant thyroid status has demonstrated that in the majority of women, TSH and fT4 concentrations tend to remain in the same side of median from the non-pregnant baseline and throughout pregnancy.
Reference intervals
In the present study, the 2.5th percentile for TSH obtained in T1 was 0.03 mIU/L for the Beckman method, which is comparable with the results from cross-sectional studies reporting 0.01 mIU/L for the Immulite 2000, 8 0.04 mIU/L for the Roche, 12 and 0.07 for Abbott Architect methods. 13 In T2 and T3, the 2.5th percentile for TSH was 0.42 and 0.34 mIU/L respectively, which is also comparable to the recent international guidelines. 9 In the studies that were used for the development of the American Thyroid Association (ATA) guidelines, the lower limit for TSH in T3 reported varied from 0.1 to 0.7 mIU/L in the third trimester depending on whether the 2.5th or 5th percentile value was used in the study. In the ATA guidelines, of the six studies used to derive the recommended RIs, three studies used 2.5th to 97.5th percentiles whereas the other half used 5th to 95th percentiles as RIs. The trimester-specific TSH values derived from our study are comparable to those of previous cross-sectional studies 8 and longitudinal studies. 18
When compared to the non-pregnancy RIs quoted by the manufacturer, 36% and 41% of women, in T2 and T3 respectively, had fT4 values that fell below the non-pregnancy lower normal limits (Table 2). The finding of a large proportion of pregnant women having fT4 values below the manufacturer’s RI has been demonstrated in several studies12,19 and has been shown to be as high as 60% with some fT4 immunoassay methods. 12 In contrast, and although infrequently required in pregnancy, fT3 measurements were less affected by pregnancy in comparison to the fT4 immunoassay.
In most instances, testing of thyroid function in pregnancy will be performed to screen for overt or subclinical hypothyroidism and for this common scenario screening is most appropriate with TSH concentrations alone.9,10 However, fT4 measurement during pregnancy will, on occasions, be necessary for the diagnosis and monitoring of hyperthyroid conditions. This study highlights the potential confusion that could occur if fT4 is ordered during pregnancy but not referenced to the trimester-specific intervals. Therefore we strongly agree with Endocrine Society & American Thyroid Association guidelines which recommend that each institution should aim to obtain and utilize a method- and trimester-specific RI.9,10
Results of fT4 measurement by immunoassays are influenced by alterations in the matrix of patients’ serum and vary in a method-specific manner.19–21 Therefore, there is no single fT4 cut-off value that can define hypothyroxinaemia across different methods 22 and this reinforces the need for each laboratory to establish method-specific RIs for each trimester of pregnancy.9,10 Equilibrium dialysis and gas chromotography mass spectrometry (the reference methods) give the best estimate for fT4. Using these methods, the decrease in mean fT4 during T3 as compared to non-pregnant controls is usually less than 10%.19,23 However equilibrium dialysis is impractical to perform for routine use. Total T4 measurements adjusted for pregnancy have also been suggested to be a more reliable method for estimating free T4 in pregnancy, 19 as it avoids the matrix effect. Although total T4 assays are not currently routinely available in Australia, measurement of total T4 during pregnancy may provide a practical alternative and would be logistically easier to perform than equilibrium dialysis.
Tracking of intraindividual thyroid function in pregnancy
The longitudinal analysis of data in this study has shown baseline and pregnancy concentrations for both TSH and fT4 values to be highly correlated. The pregnant women tended to maintain a similar ranked concentration of thyroid function from the non-pregnant baseline into each trimester of pregnancy. If women started the pregnancy above or below the median, the majority would remain in the same side of median for the entire pregnancy.
Consistent with the finding reported by Glinoer, 24 a significant inverse association was also found in our study between T1 TSH and T1 β-hCG by the Spearman rank correlation. However, this association was not as strong as that seen between the T1 TSH and baseline TSH of each woman by logistic regression.
The strong relationship between TSH and fT4 during pregnancy and the non-pregnant state is a new and important finding of this study. These data suggest that the TSH and fT4 concentrations during pregnancy are strongly influenced by a woman’s baseline concentration of thyroid activity and less influenced by β-hCG concentrations. As thyroid function in the majority of pregnant women track within the same median distribution, TSH concentrations which deviate outside this narrow band, despite remaining within the current RIs,9,10 may represent a subtle but impaired thyroid response to pregnancy. Further studies are required to confirm this hypothesis.
Limitations of the current study are recognized. Only serum from 71 and 70 (after exclusion of patients with PP thyroiditis) subjects were available for the third trimester and PP time points, respectively. There were no differences in the first trimester thyroid function in those who completed the study and those who did not complete the study. Despite a dedicated nurse who contacted all the participants and co-ordinated for them to have their blood tests taken, as well as all patients being offered a pathology nurse home visit PP, many participants did not complete the study. The third trimester sample size was contributed to by delivery prior to the T3 blood sample with six patients delivering at or prior to 35 weeks and 14 patients delivering between 35.0 and 38.0 weeks gestation. The high dropout rate in the current study demonstrates some of the inherent difficulties associated with conducting such prospective studies in pregnant women. The major strength of the study is its prospective and longitudinal nature. Although many other studies have used cross-sectional samples randomly taken in each trimester, longitudinal studies of thyroid function in pregnancy are scarce. This is due to the need to follow the patients throughout their entire pregnancy which is more expensive to conduct and therefore such studies have a smaller sample size.16,18
In the longitudinal analysis, a further limitation was that a true pre-pregnancy baseline sample was not available and the PP sample, taken between 8 and 12 week PP, with exclusion of obvious PP thyroiditis, was used. Lastly, UIC was measured only in T1 which may not represent the iodine status throughout the whole pregnancy. However, the iodine status in our cohort of pregnant women compared favourably with previous studies. In our cohort of pregnant women, the median UIC was 146 µg/L. much higher than two recent Australian studies which reported a median UIC in pregnant women of 96 µg/L 25 and 81 µg/L. 26 When categorizing according to the WHO classification for women during pregnancy, 9 10% of our cohort had moderately severe iodine deficiency (<50 µg/L) which was less than previously reported of 17% 25 and 26%, 26 while 44% of our cohort had mild iodine deficiency (50–150 µg/L) and 46% were iodine sufficient (>150 µg/L).
In conclusion, in response to the recent recommendations for the use of method-specific pregnancy RIs,9,10 we present trimester-specific RIs of thyroid function for the Beckman method. Trimester- and method-specific fT4 RIs are important for accurate interpretation of thyroid function tests in pregnancy. Pregnancy and trimester-specific RIs for fT4 will enhance patient care by preventing unnecessary referrals for fT4 concentrations below the non-pregnancy specific RI, preventing the misdiagnosis of ‘isolated hypothyroxinaemia’, and finally by reducing the risk of a patient receiving unnecessary thyroxine treatment.
Footnotes
Acknowledgements
We would like to acknowledge Ms Vasantha Ragunathan for assisting with patient recruitment, Dr Sujiva Ratnaike for her intellectual contribution and Melbourne Pathology for assisting in home visits for the collection of the postpartum bloods.
Declaration of conflicting interests
None declared.
Funding
Medical Research Foundation for Women and Babies and Beckman Coulter Research Grant.
Ethical approval
The ethics committee of Mercy Hospital for Women approved this study (MHAC HREC Project Reference Number: R06/R07).
Guarantor
Dr Christine Houlihan is the guarantor of this paper.
Contributorship
EIE did intellectual project planning, patient recruitment, intellectual analysis of data and writing of the paper; ZXL did intellectual project planning, intellectual analysis of data and writing of the paper; KS did intellectual project planning, intellectual analysis of data and writing of the paper; IB did experimental work and intellectual analysis of data; KYC conducted experimental work; QL did intellectual analysis of data; NC did intellectual analysis of data; CAH did intellectual project planning, patient recruitment, intellectual analysis of data and writing of the paper
