Abstract
There are limited and conflicting data on the value of serum calprotectin (sCp) in discriminating active from inactive disease activity in ulcerative colitis (UC). Faecal calprotectin (fCp), sCp, serum C-reactive protein (sCRP) and platelets were compared in patients with UC who had clinically active (n = 29) and clinically inactive (n = 42) disease. Serum calprotectin was measured with Bühlmann® (BMN sCp) and ImmunodiagnostikTM (IDK sCp) assays. Median (interquartile range) fCp was higher in active than inactive disease [1004 (466–1922) versus 151 (55–280) µg/g; p < 0.0001). BMN sCp [4534 (3387–6416) versus 4031 (2401–5414) ng/mL; p = 0.1825], IDK sCp [4531 (2920–6433) versus 3307 (2104–4789) ng/mL; p = 0.1065], sCRP [ 4 (2–8) versus 2 (1–4) mg/L; p = 0.0638) and platelets [269 (233–331) versus 280 (227–325) ×10−9/L; p = 0.8055] were similar in active and inactive disease respectively. The area under the receiver operator characteristics curves with 95% confidence limits were 0.85 (0.76–0.94) for fCp, 0.61 (0.47–0.74) for BMN sCp, 0.61 (0.48–0.75) for IDK sCp, 0.69 (0.56–0.81) for sCRP and 0.52 (0.38–0.66) for blood platelets. Faecal calprotectin is the optimum biomarker for discriminating between active and inactive UC. The diagnostic performance of sCp, irrespective of assay, and systemic biomarkers was poor; of these sCRP performed best.
Introduction
Ulcerative colitis (UC) is a chronic inflammatory condition limited to the mucosal layer of the colonic epithelium and often with continuous extension to the colon for a variable distance. It is characterized by episodic symptomatic relapses and remissions,1–4 associated with extra-colonic manifestations and carries increased risk of chronic complications, including strictures, dysplasia and colorectal cancer. Key in the management of UC is identification of patients who may benefit from early intervention and therapeutic escalation to prevent chronic complications.3,4
While endoscopy is the ‘gold standard’ for assessing intestinal inflammation and mucosal healing, it is an expensive and invasive procedure with associated risk. A robust non-invasive biomarker of intestinal inflammation to reduce or replace endoscopy would carry many advantages.
Faecal calprotectin (fCp), a sensitive and specific non-invasive marker of intestinal inflammation, correlates well with endoscopic inflammation in UC and therefore is often used as a non-invasive alternative to endoscopy to confirm active disease and guiding therapy 4 or to exclude patients who do not require further investigation, 5 thereby reducing the necessity for endoscopic or radiological investigations. Faecal calprotectin, however, is limited by difficulty in patient sampling, delays in laboratory testing, intra-individual variability in active UC and optimal timing for sampling.6–8
Serum biomarkers are, therefore, appealing for patient convenience, cost and being quick to result. Routinely used systemic biomarkers markers of inflammation such as serum C-reactive protein (sCRP), full blood count, platelet count, white cell count and erythrocyte sedimentation rate correlate with endoscopic activity in monitoring disease activity in patients with UC. 4 They, however, lack sensitivity and specificity and correlate poorly with disease activity indices limiting their prognostic utility in UC.9,10 Although commercial kits for measurement of serum calprotectin (sCp) are available for monitoring inflammatory bowel disease, a paucity of studies investigating sCp in assessing disease activity in UC reported conflicting results.3,11–13
Given the limitations of routine systemic and faecal biomarkers of inflammation, we investigated the utility of sCp to replace or supplement fCp and compared its performance to conventional inflammatory blood biomarkers, sCRP and platelets, in discriminating between active and inactive disease activity in UC in an outpatient setting.
Materials and methods
In an institution approved and registered service evaluation project, we assessed the utility of sCp in discriminating between active and inactive UC. As part of their routine clinical care, stool and blood samples were collected from patients with UC attending an inflammatory bowel disease clinic. Blood samples were collected soon after the clinic appointment for serum biochemistry (including sCRP) and a full blood count (including platelets). Following completion of requested tests, serum samples were retrieved within 2 h of collection and surplus serum tipped off and stored at −80°C until analysed for sCp, using two different commercial assays in single batches. Stool samples were provided by patients within 2 weeks of their clinical appointment and prior to any changes in medical therapy. Matched patient faecal and serum samples were included in this service evaluation. Samples were excluded where there was evidence of infection or where there had been recent use of non-steroidal anti-inflammatory drugs and antibiotics. Classification of patients into active or inactive UC was obtained from the patient medical records and was based on clinical assessment and where available endoscopy.
Faecal calprotectin was measured using Bühlmann fCALTM ELISA Calprotectin (Bühlmann Laboratories AG, Baselstrasse 55, CH – 4124 Schönenbuch, Switzerland). Serum Cp was measured using ELISA assays supplied by Bϋhlmann Laboratories (Bϋhlmann Laboratories, AG, CH – 4124 Schönenbuch, Switzerland) and ImmunodiagnostikTM Laboratories (ImmunodiagnostikTM Laboratories AG, D–64,625 Bensheim, Germany). Both manufacturer kit inserts state that sCp frozen at −20°C is stable for at least 6 months. Serum C-reactive protein was measured by immunoturbidimetry using methods and reagents supplied by Abbott diagnostics on the Abbott ARCHITECT c16000 analyser (Abbott Diagnostics, Abbott Park, IL, USA). Platelets were counted by flow cytometry (Sysmex XN-10®, Sysmex Corporation, Kobe, Japan). The intra-assay CVs are 3.1% for Bühlmann serum calprotectin (BMN sCp) and 2.9% for ImmunodiagnostikTM calprotectin (IDK sCp). The respective intra-assay and inter-assay CVs for fCp are 5.6% and 8.2%, and for sCRP 0.59% and 0.65%. The inter-assay CV for platelet count is 4.29%.
The Kolmogorov–Smirnov and Shapiro–Wilks tests assessed the normality of data. Comparisons of continuous variables between groups were made using either unpaired t-tests for parametric data or Mann–Whitney U test for non-parametric data. Spearman tests rank-order correlation measured the degree of association between non-parametric variables. Comparisons of categorical variables between groups were assessed using Chi-squared test. p values < 0.05 were considered statistically significant. The area under the receiver operator characteristic curve (AUROCC) assessed diagnostic performance of the inflammatory biomarkers. A Passing–Bablok test and Bland–Altman plots assessed between method bias of BMN®-Cp and IDK®-Cp assays. Parametric data are presented as mean with standard deviation and non-parametric data as median with interquartile ranges.
Data processing and statistical analysis were performed using GraphPad Instat version 3.00 for Windows 95 (GraphPad Software, San Diego, California, USA), Microsoft Excel® XLSTAT (Addinsoft, New York, NY 10,001, USA; https://www.xlstat.com/en/), Analyse–it Software (Analyse–it Software Ltd., The Tannery, 91 Kirkstall Road, Leeds LS3 1HS, United Kingdom) and SPSS Statistics version 25 (IBM SPSS Statistics Software, Armonk, New York, USA).
Results
Patient characteristics and results of faecal and serum biomarkers in those with active and inactive ulcerative colitis.
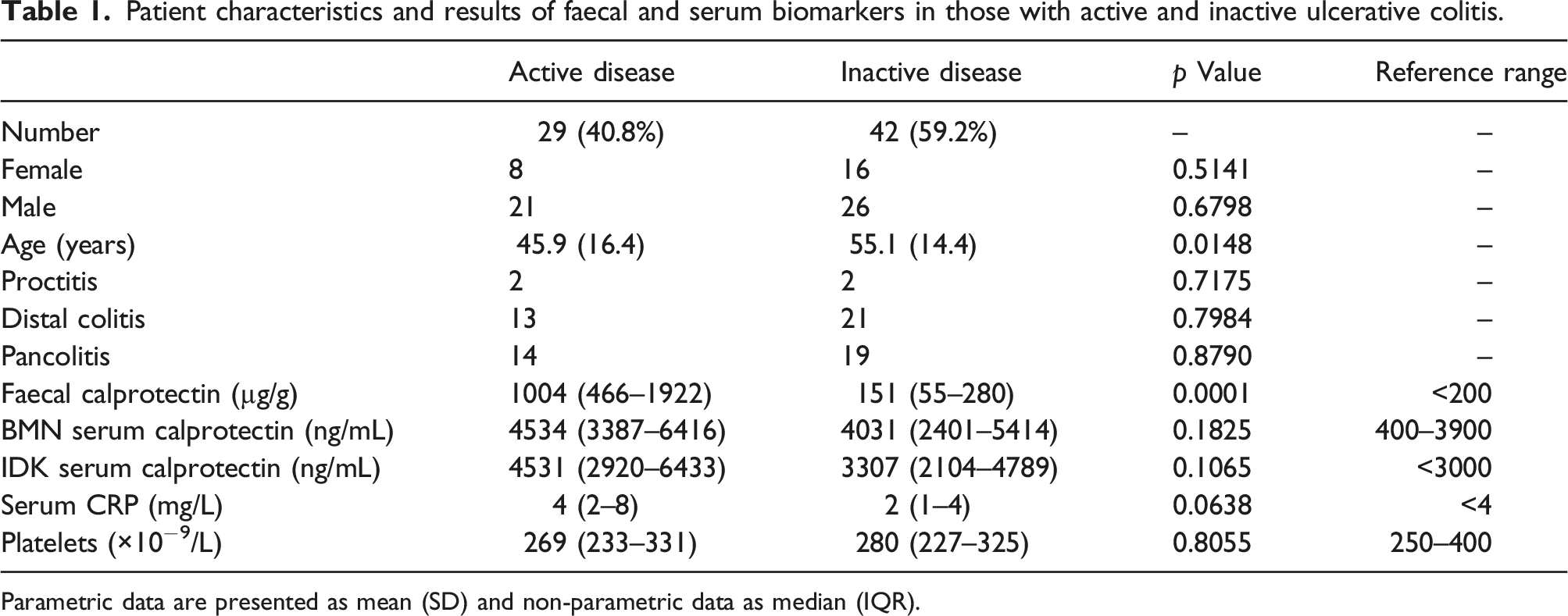
Parametric data are presented as mean (SD) and non-parametric data as median (IQR).
Spearman’s rank correlations between inflammatory biomarkers.
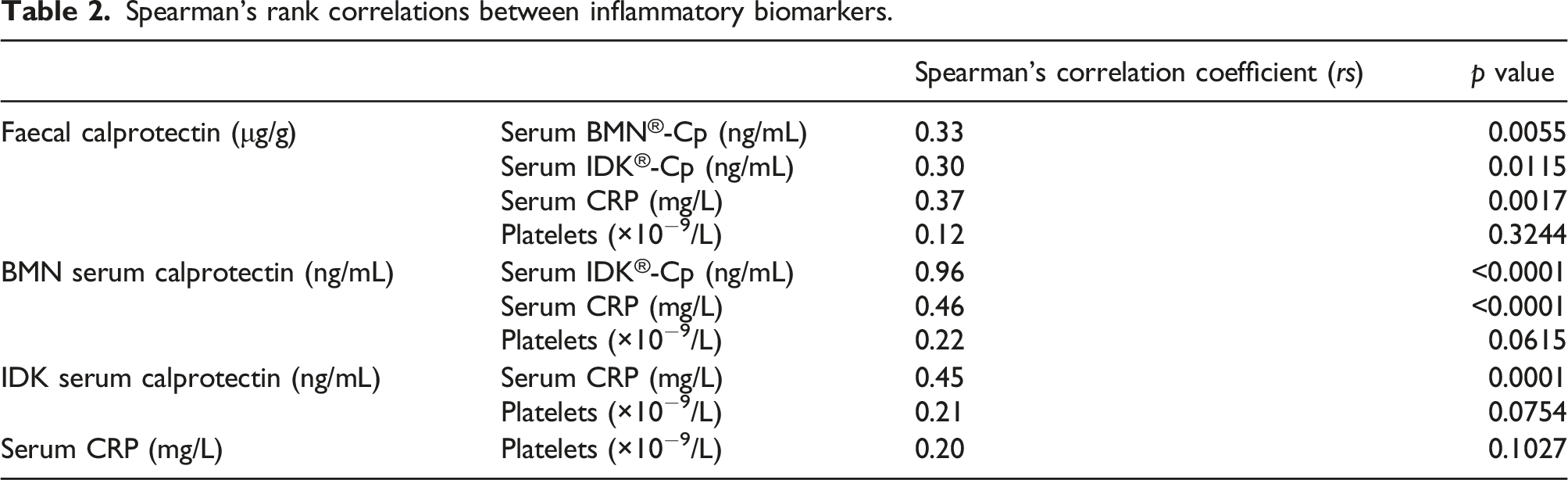
A Passing–Bablok test and Bland–Altman plots demonstrated that BMN sCp assay had a constant 14.5% positive proportional bias relative to the IDK sCp assay.
The receiver operator characteristics curves for fCp, BMN sCp, IDK sCp, sCRP and platelets are shown in Figure 1. Area under the receiver operator characteristic curve and 95% confidence limits were 0.85 (0.76–0.94) for fCp, 0.61 (0.47–0.74) for BMN sCp, 0.61 (0.48–0.75), for IDK sCp, 0.69 (0.56–0.81) for sCRP and 0.52 (0.38–0.66) for blood platelets. The diagnostic sensitivity and specificity biomarkers for discriminating active and inactive disease in UC at in-house cut-off values derived from guidelines for fCp
4
and manufacturer supplied cut-off values for systemic biomarkers are shown in Table 3. Receiver operator characteristics curves for (a) faecal calprotectin (FCp), (b) serum BMN calprotectin (BMN sCP), (c) serum IDK sCp (IDK sCp), (d) serum C-Reactive Protein (sCRP) and (e) blood platelets (platelets). Test performance of faecal and systemic inflammatory biomarkers in discriminating between active and quiescent disease. ain-house cut-off values. bManufacturer.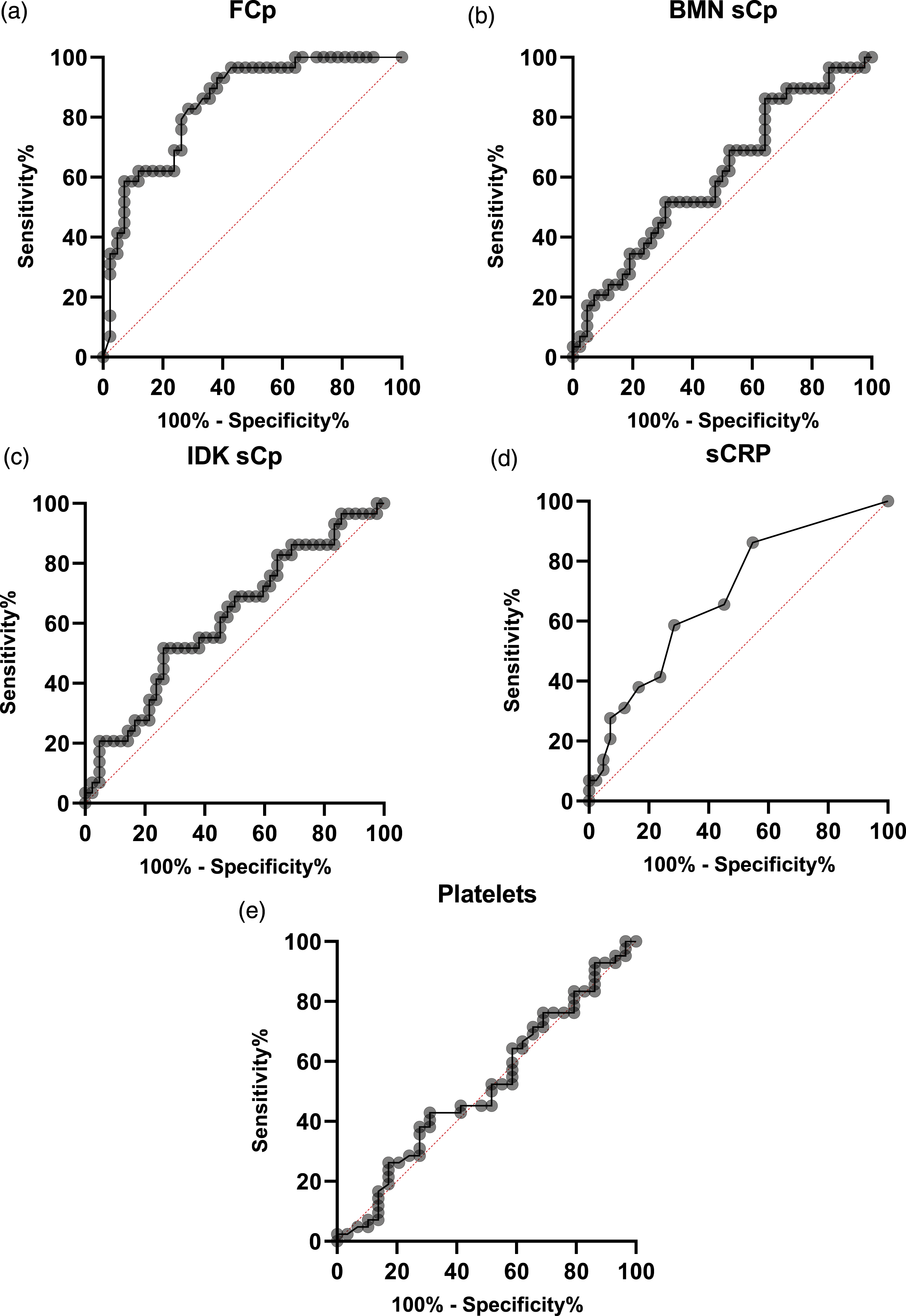

Discussion
Faecal calprotectin, in this study, was higher in active than inactive UC and had very good diagnostic performance consistent with previous studies reporting fCp to be a useful surrogate marker of inflammatory activity in UC,4,13–17 and therefore a non-invasive alternative to flexible sigmoidoscopy to guide therapy. 4
Despite very good diagnostic performance of fCp in monitoring disease activity in UC, patients may have difficulty collecting stool samples. We, therefore, investigated the diagnostic performance of blood biomarkers for patient convenience. Systemic inflammatory biomarkers, including sCp, were similar in clinically active and inactive UC, consistent with a study reporting similar sCp levels in active and inactive UC. 18 Compared to fCp (AUC 0.85), ROC curve analysis confirmed the poorer diagnostic performance of systemic inflammatory biomarkers in discriminating between active and inactive UC, including both sCp assays (AUC 0.61) and of these sCRP (AUC 0.69) performed best consistent with previous studies.3,11 In addition, fCp correlated weakly with both sCp assays and sCRP and not at all with platelets. These results support studies reporting little or no correlation between fCp and sCp, sCRP or platelets in UC.11,12,17 Our results, therefore, indicate that sCp and sCRP do not have a significant role in predicting exacerbations in UC and unlikely to replace or supplement fCp, which remains the biomarker of choice in monitoring outpatients with UC.
Our results, however, differ from a study reporting sCp and not fCp predicted colectomy in adult patients with UC. 11 We suggest that these differences between faecal and systemic biomarkers in UC reflect the underlying disease process and severity of disease. Ulcerative colitis is an intestinal disease limited to the mucosa; thus, disease exacerbations are typified by intestinal specific inflammatory response increasing fCp and, unless in patients hospitalised with acute and very severe UC, unlikely to provoke a systemic inflammatory response with unremarkable sCp and sCRP levels.
In conclusion, fCp in non–hospitalised patients remains the optimum laboratory biomarker for discriminating between active and inactive UC. In comparison to fCp, the diagnostic performance of sCp, sCRP and platelets in assessing disease activity was inferior but of these sCRP performed best. Serum Cp has little or no role in assessing disease activity in UC, if systemic inflammatory biomarkers are required, sCRP is more robust, easily available and less expensive.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
An institution approved service evaluation project ID 5948 registered with the Royal Wolverhampton NHS Trust.
Guarantor
RG.
Contributorship
MU researched the literature, designed the study, collected data, analysed serum samples, analysed the data, and wrote the first draft. HSC supervised calprotectin analyses and helped analysed the data. ST and BA collected clinical and endoscopic data. MJB, CF and RG helped design the study. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
