Abstract
Objective
NICE recommends measurement of faecal haemoglobin (f-Hb) using faecal immunochemical test (FIT) when colorectal cancer is suspected and calprotectin (f-Cal) in the context of inflammatory bowel disease, though neither is disease specific. During the COVID-19 pandemic, f-Hb has been a requirement prior to referral for endoscopy in England; f-Cal is often performed simultaneously. The aim of this study was to investigate test performance of both tests for significant bowel disease in those patients referred.
Design
All adult patients with simultaneous measurements of f-Hb and f-Cal between April 2019 and September 2020 were included. For those referred, outcomes were determined from clinical records.
Results
650 patients with simultaneous samples for f-Hb an f-Cal were managed in Primary Care; 319 patients were referred to hospital; SBD was found in 32 (10.0%) (CRC 5, high risk adenomas 5, IBD 22). At a cut-off of 10 μg/g for f-Hb and 200 μg/g for f-Cal, the sensitivity, specificity and negative predictive value for diagnosis of SBD were 84.4%, 58.2% and 96.7% and 68.8%, 89.6% and 95.7%, respectively. Performance of both tests would have enabled diagnosis of two more cases of significant, but non-malignant, bowel disease but required over 4% more referrals for investigation.
Conclusion
Use of FIT has become established to assist prioritisation of patients for referral from Primary Care. Whilst introduced specifically for CRC, FIT performs well as a rule out for IBD in Primary Care and the use of f-Cal is not required.
Keywords
Introduction
Calprotectin is a calcium and zinc binding protein derived predominantly from neutrophils and constitutes about 60% of their cytosolic protein. It can be detected in various body fluids in proportion to the severity of any existing inflammation. In health, the concentration of calprotectin in faeces is about six times that of normal plasma, which led to the concept that measurement of faecal calprotectin (f-Cal) would be a sensitive and specific marker of gastrointestinal inflammation. 1
The National Institute for Health and Care Excellence (NICE) has recommended f-Cal testing as an option to support the differential diagnosis of inflammatory bowel disease (IBD) or irritable bowel syndrome (IBS) in adults with recent onset lower gastrointestinal symptoms for whom specialist assessment is being considered, if cancer is not suspected. 2 NICE originally suggested a cut-off for f-Cal of 50 µg/g to prompt referral but more recently has supported raising this to 100 μg/g. 3 Use of f-Cal is recommended by the British Society of Gastroenterology in patients under 40 years presenting with chronic diarrhoea in whom cancer is not suspected and a cut-off of >250 μg/g is suggested to correlate with endoscopic inflammation. 4 It is known that there are quantitative differences in calprotectin assays of up to three-fold, 5 indicating the importance of establishing cut-offs appropriate for local pathways.
More recently, NICE recommended measurement of faecal haemoglobin (f-Hb), using faecal immunochemical testing (FIT) to guide referral for suspected colorectal cancer in patients with unexplained abdominal symptoms without rectal bleeding, who do not meet criteria for urgent referral. 6 A f-Hb of ≥10 µg/g was recommended as the cut-off to prompt urgent referral.
In the context of suspected inflammatory bowel disease, f-Cal is a useful test but it is inflammation, not disease, specific and many bowel diseases, including colorectal cancer (CRC), are associated with inflammation and would therefore be expected to be associated with an elevated calprotectin concentration. Similarly, FIT detects blood in faeces which is found in conditions other than CRC. f-Hb has been shown to be reliable as an objective way of identifying patients with an extremely low risk not only of CRC but of significant bowel disease (i.e. CRC, higher risk adenomas (HRA) and IBD), thus avoiding invasive investigation. 7 Studies in countries where f-Hb measurement is freely available for CRC screening, but access to f-Cal is limited, have shown that f-Hb has a role in the management of patients with IBD. 8
In the COVID-19 pandemic, England was placed into lockdown for the first time in March 2020 and non-emergency colonoscopy was temporarily suspended. Resumption of service was associated with reduced capacity because of the necessity for new health and safety protocols, and with an accumulated backlog of patients requiring investigation. To assist triage, performance of FIT was mandated nationally as a criterion for referral. This caused us to consider the current role of f-Cal testing in Primary Care.
The aim of this study was to examine retrospectively those patients in whom f-Hb and f-Cal had been requested simultaneously in Primary Care in order to investigate test performance in those patients referred.
Methods
Patients
All adult patients who had paired requests for FIT and f-Cal requested from Primary Care between 1st April 2019 and 30th September 2020 were included. Due to the potential variables of disease and of pre-analytical factors on faecal measurements, paired measurements were restricted to those in whom samples were received within 3 days of each other. Until April 2019, FIT was offered in line with NICE DG30 6 ; subsequently it became a requirement for referral on the urgent lower gastrointestinal cancer referral pathway. The majority of locations used electronic requesting and were only able to order f-Cal in adults aged ≤60 years. There was no automatic restriction on the minority who requested using paper forms.
Outcome definition
Clinical outcomes were collected until 31st May 2021 and diagnoses confirmed from clinical notes, radiological reports and endoscopy and histology findings. HRA were defined using British Society of Gastroenterology guidance 2020. 9
Sample analysis
Samples were taken at home and returned to Barts Health NHS Trust laboratory at the earliest opportunity. Those for f-Cal were taken into a screw top pot and stored frozen prior to analysis which took place within 1 week of receipt. F-Hb samples were taken into an Eiken specimen collection device using the sampling probe in the lid and stored at 4°C until analysis, which took place within a week of receipt and 2 weeks of sampling. The laboratory is accredited by the UK Accreditation Service to ISO 15189 standards. F-Cal was measured using the Accusay enzyme-linked immunosorbent assay (Launch Diagnostics, Longfield, UK). F-Hb was measured by immunoturbidimetry using a single OC Sensor (Eiken Chemical Co Ltd, Tokyo, Japan). The coefficients of variation were <12% for f-Cal and 2.8% at 14 μg/g and 3.0% at 91 μg/g for f-Hb. External quality assurance was achieved via satisfactory performance in the relevant United Kingdom National External Quality Assessment Service schemes. The lower limits of quantification were 12.5 μg/g and 4 μg/g and the upper limits of the measuring range were 2800 μg/g and 200 μg/g for f-Cal and f-Hb, respectively. Samples with a concentration above the upper analytical limit were not diluted and re-assayed but reported as ≥2800 μg/g and ≥200 μg/g, respectively.
Results were reported using a cut-off of f-Hb of 10 μg/g, based on NICE guidance 6 and 200 μg/g, based on in-house evaluation, to prompt referral for consideration of a diagnosis of CRC and IBD, respectively.
Statistical considerations
Data were summarised and tabulated; population characteristics were summarised by appropriate descriptive statistics by data type. Continuance measures (e.g. sensitivity, specificity, PPV and NPV) were described by average and the exact 95% confidence interval (CI) for each measure using MedCalc (MedCalc Software Ltd, Ostend Belgium).
Ethical considerations
Data were gathered during routine patient care therefore ethical approval was not required but the project was registered with the Clinical Effectiveness Unit ID 11774.
Results
Patients
In total, 972 patients returned samples for f-Hb and f-Cal within 3 days in the study period. Of these 650 (66.9%) were managed in Primary Care and 319 (32.8%) were referred. Three patients were already under the care of hospital teams elsewhere.
The 650 patients managed in Primary care, had an age range of 18–82 years (median 42), with eight patients aged over 60 years and 342 (42.6%) being female. The most common presenting symptoms were change in bowel habit (40.6%), abdominal pain (21.9%), rectal bleeding (20.2%), weight loss (7.1%) and anaemia (2.5%). The 319 patients referred were aged between 18 and 87 years (median 45), 11 patients were older than 60 years and 164 (53.8%) were female. Amongst referrals, the most common presenting features were change in bowel habit (53.8%), rectal bleeding (30.3%), abdominal pain (24.1%), weight loss (8.9%) and anaemia (2.5%).
Further investigations and final diagnoses
The study flow chart is shown in Figure 1 Study flow chart. CRC, colorectal cancer; HRA, higher risk adenoma; IBD, inflammatory bowel disease.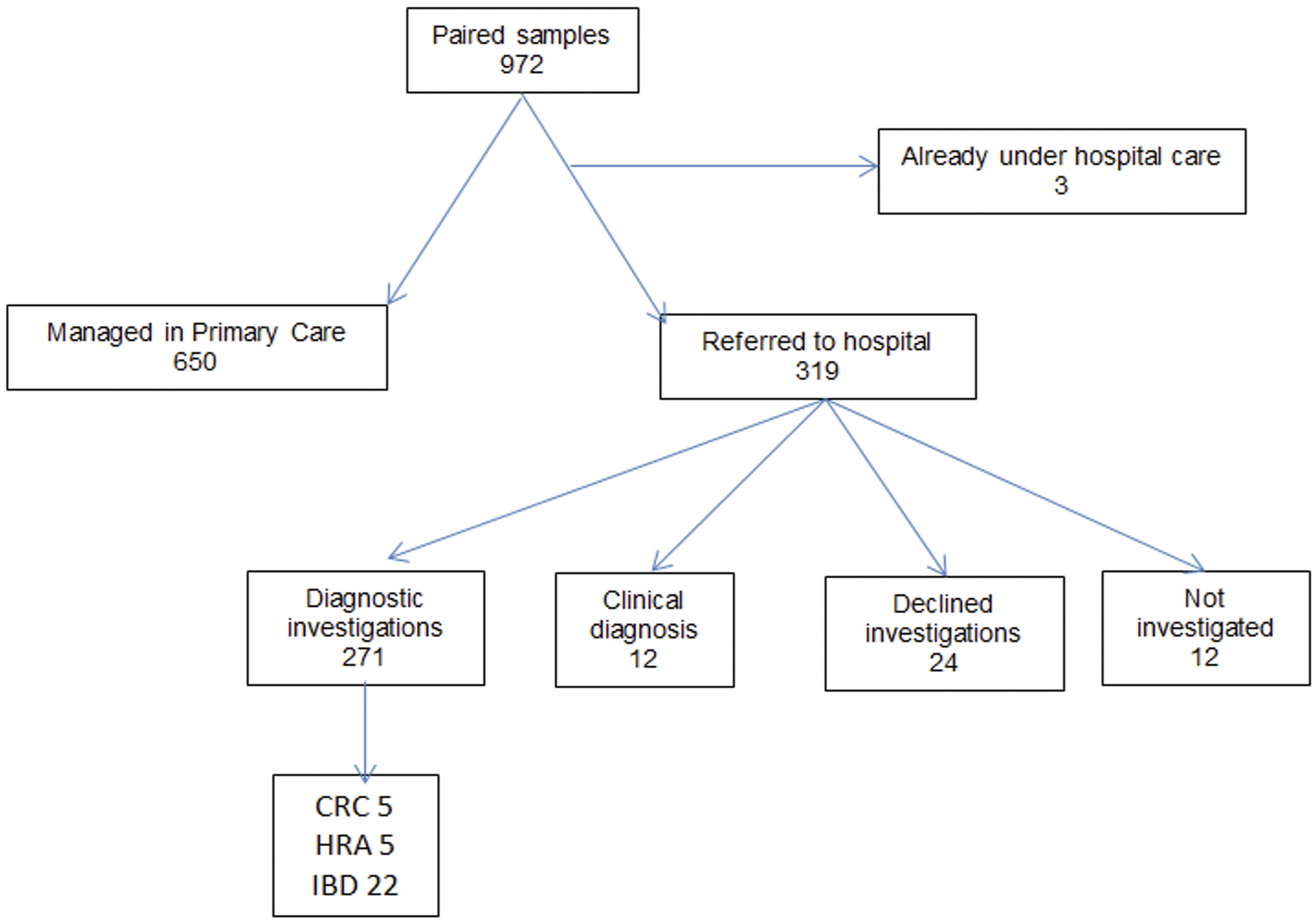
Of the 319 patients referred, a final diagnosis was made in 283. In 12 patients, in whom both f-Hb and f-Cal were below the limits of quantification, a diagnosis of IBS was made on clinical grounds. 271 underwent further investigations to establish a definitive diagnosis. The final investigation used to establish the diagnosis was colonoscopy in 216, CT colonography in 12, CT abdomen and pelvis in 25 (with flexible sigmoidoscopy performed in addition in one), flexible sigmoidoscopy alone in 14 and MRI in four. The following diagnoses were made CRC (5), HRA (5), IBD (22), LRA (24), diverticular disease (16), coeliac (1), NSAID enteropathy (1), perianal abscess (1) and threadworms (1). 11 patients had minimal non-specific inflammation on histology but this was not thought to represent serious pathology. 137 patients had normal findings at colonoscopy, a further 47 had normal findings except for the presence of haemorrhoids.
24 patients declined investigations, in the majority of cases because of patient choice to defer for factors associated with the COVID-19 pandemic. Further investigations were not carried out in five patients with complex medical histories and multiple co-morbidities, or in seven patients where it was not felt to be indicated (newly diagnosed thyrotoxicosis, complete resolution of upper gastrointestinal symptoms on treatment for Helicobacter pylori, lower urinary tract symptoms (2), recent gastrointestinal infection (3)).
Faecal haemoglobin and calprotectin
Distribution of f-Hb and f-Cal results.
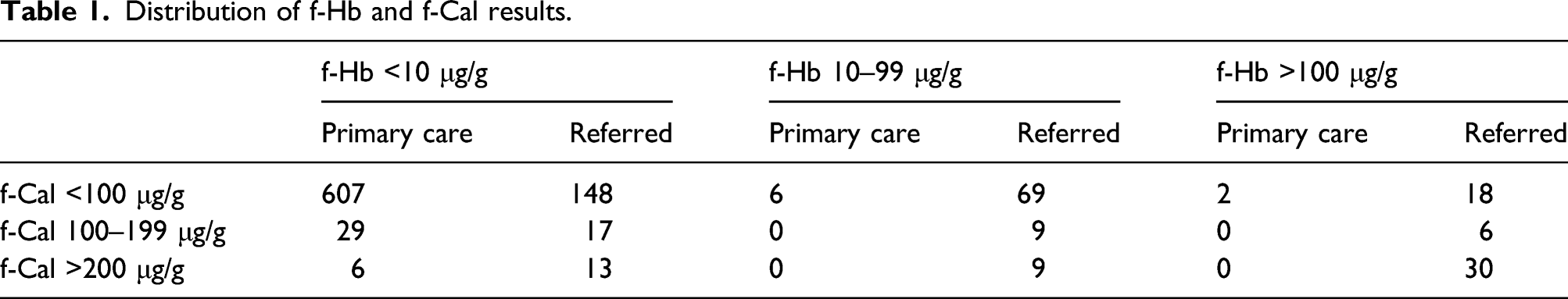
93 (14.4%) of the patients managed in Primary Care had f-Cal between 50 and 200 μg/g. The eight patients with f-Hb >10 μg/g who were not referred were all aged below 45 years. Six had f-Hb between 11 and 20 μg/g and, of these, three had undergone colonoscopy with normal findings between 3 and 12 months prior to faecal testing. Two of the patients had a repeat test with a result <6 μg/g and the remaining patients received a pre-existing diagnosis of haemorrhoids. Two patients had f-Hb > 200 μg/g and were not referred; one had the test repeated within 3 days with a result of <6 μg/g and the other, who had multiple co-morbidities had resolution of her symptoms of acute change in bowel habit.
Diagnostic test performance of faecal haemoglobin and faecal calprotectin in 283 patients who were referred and in whom a final diagnosis was made.
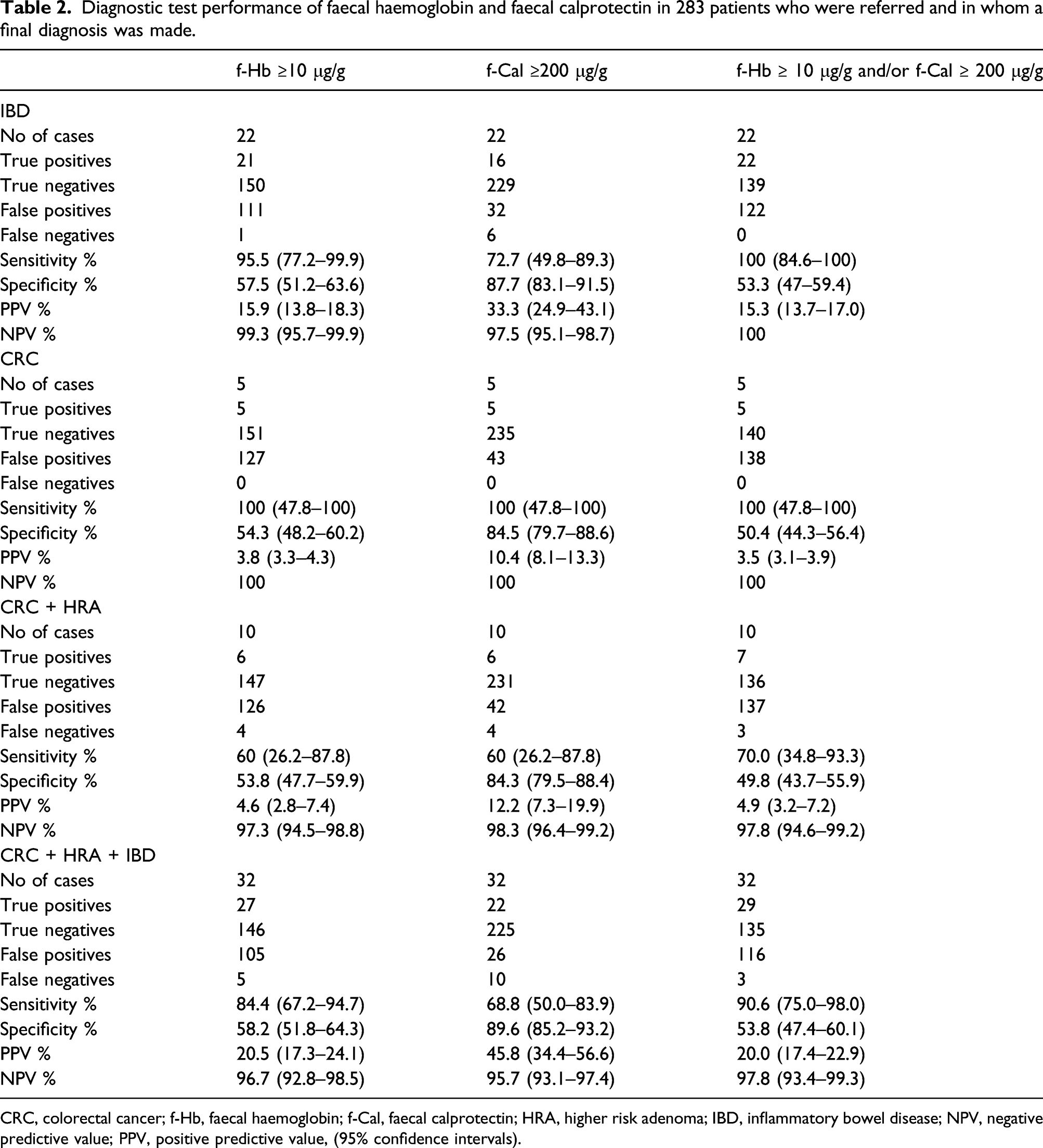
CRC, colorectal cancer; f-Hb, faecal haemoglobin; f-Cal, faecal calprotectin; HRA, higher risk adenoma; IBD, inflammatory bowel disease; NPV, negative predictive value; PPV, positive predictive value, (95% confidence intervals).
Using f-Cal at the manufacturer’s recommended cut-off of 50 μg/g would have resulted in a sensitivity and specificity of for SBD of 84.4% (67.2–94.7%) and 64.9% (58.7–70.8%) with 21 cases of IBD detected; and at 100 μg/g sensitivity and specificity of 81.3% (63.6–92.8%) and 80.5% (63.6–92.8%) with 20 cases of IBD detected.
Details of inflammatory bowel disease patients not detected by faecal calprotectin and faecal haemoglobin at cut-offs of 200 μg/g and 10 μg/g, respectively.
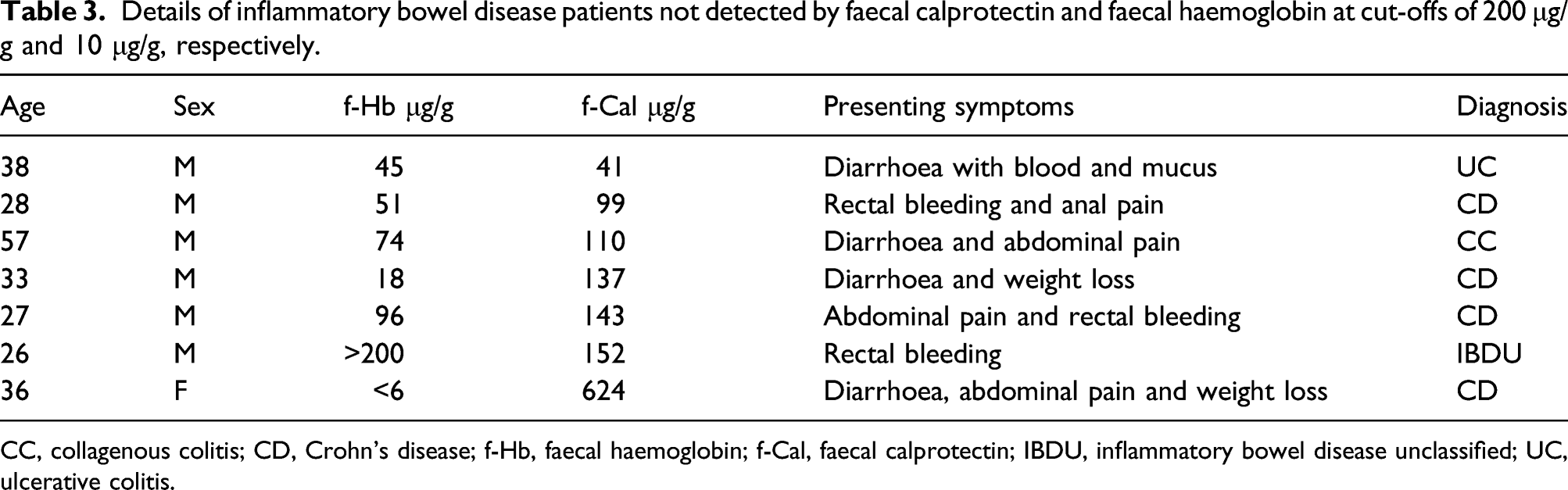
CC, collagenous colitis; CD, Crohn’s disease; f-Hb, faecal haemoglobin; f-Cal, faecal calprotectin; IBDU, inflammatory bowel disease unclassified; UC, ulcerative colitis.
Discussion
This study demonstrates that patients presenting to Primary Care with lower gastrointestinal symptoms can be assessed for the presence of significant bowel disease using measurement of f-Hb. Using f-Hb at a cut-off of 10 μg/g would have led to the referral of 141 patients for colonoscopy based on the test result alone, whereas use of both tests would have led to the referral of 154, with the detection of only two more cases of significant, but non-malignant, bowel disease. There are limitations to this work likely to affect test performance, but this study was designed to review the actual use of the tests in practice. However, it is assumed that those patients managed in Primary Care did not have SBD, and those with SBD are highly likely to have been referred. For the reasons already stated, we only considered patients whose paired samples were received in the laboratory within 3 days of each other, though a number of patients had sequential samples sent. Even with this constraint, the laboratory-based cost of the additional f-Cal measurement would have been of the order of £30,000.
Although NICE recommends FIT for CRC and f-Cal for IBD, the tests are not disease specific and studies have investigated the use of f-Hb in IBD and f-Cal in CRC. For example, an early study of the use of FIT in IBD showed that f-Hb reflected the mucosal status of ulcerative colitis at colonoscopy, 10 and in a study of 74 children with abdominal symptoms who underwent colonoscopy, all patients with normal histology had f-Hb < 4 μg/g and f-Hb >20 μg/g was associated with significant inflammation. 11
In a meta-analysis of seven studies involving 2661 patients’ f-Cal had a sensitivity of 36% and specificity of 71% for CRC and its routine use was not recommended. 12 In our study, the subset of patients in whom both f-Hb and f-Cal had been performed prior to referral had a median age of 45 years, which is younger than those referred with suspected CRC overall, with a lower percentage CRC and relatively greater number with IBD. 13 The five patients with CRC were detected by both tests but these patients all had f-Hb >200 μg/g. A recent large study showed that 9% of CRC would have been missed using the cut-off for f-Hb of 10 μg/g recommended by NICE and using the test at a lower cut-off and/or in combination with specific symptoms can improve this figure. 14
Various studies have compared the performance of the two tests. The CEDAR study evaluated 266 patients median age 61 years, presenting to Primary Care with bowel symptoms and concluded that measurement of f-Hb at point of care, together with clinical information, could exclude SBD, therefore avoiding unwarranted endoscopy in one third of patients and that the addition of f-Cal was of limited value. 15 In 755 patients referred with bowel symptoms using f-Hb at a cut-off of 10 μg/g had an NPV of 94.4%, sensitivity of 68.6% and specificity of 83.6%, compared with 89.4%, 43.6% and 77.0%, respectively, for f-Cal at a cut-off of 200 μg/g. 7
FIT has been recommended as a test to support the diagnosis of colorectal cancer, and thus broadening its scope would require reconstruction of the current pathway that mandates an urgent lower gastrointestinal cancer referral for those where tests have shown faecal haemoglobin above the agreed cut-off. We found that 148 of 319 patients referred (46.4%) had a f-Hb <10 μg/g and f-Cal <200 μg/g, of whom only one patient was found to have non-malignant SBD. This suggests that perhaps the tests were not being used to guide referral and that there may be an existing need for refinement and further guidance with respect to clinical pathways and use of faecal testing following on from changes in policy occasioned by the COVID-19 pandemic.
Conclusion
F-Cal has a confirmed place in optimising management of patients with established IBD and measurement of f-Hb is recommended by NICE for use in lower gastrointestinal cancer referral pathways. However, during the COVID-19 pandemic FIT has become mandatory to assist with triage for colonoscopy and is now embedded in the Primary care test repertoire with wider uses than those for which it was originally recommended. This study has shown that, as currently used in Primary Care, FIT can be used to assist in the diagnosis of SBD without the need for f-Cal. We suggest that FIT can be used alone to aid triage for both CRC and IBD in Primary Care without the need for access to f-Cal. As currently performed, FIT is a more acceptable test to patients than f-Cal and more easily analysed within the laboratory and the use of a single test has the potential to save money and simplify patient pathways.
Footnotes
Acknowledgements
No other acknowledgements.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Data were gathered during routine patient care therefore ethical approval was not required but the project was registered with the Barts Health Clinical Effectiveness Unit ID 11774.
Guarantor
RMA.
Contributorship
Conception of the Study by Dr Ruth Ayling. All authors contributed to the data collection, data analysis and drafting of the original and final manuscript.
