Abstract
Background
Data is limited evaluating novel biomarkers in right ventricular dysfunction. Normal right heart function improves the prognosis of patients with heart failure. Therefore, this study investigates the association between the novel biomarker copeptin and right heart function compared to NT-proBNP.
Methods
Patients undergoing routine echocardiography were enrolled prospectively. Right ventricular function was assessed by tricuspid annular plane systolic excursion (TAPSE) and further right ventricular and atrial parameters. Exclusion criteria were age under 18 years, left ventricular ejection fraction < 50% and moderate to severe valvular heart disease. Blood samples were taken for biomarker measurements within 72 h of echocardiography.
Results
Ninety-one patients were included. Median values of copeptin increased significantly according to decreasing values of TAPSE (P = 0.001; right heart function grade I: tricuspid annular plane systolic excursion; TAPSE > 24 mm: 5.20 pmol/L; grade II: TAPSE 18–24 mm: 8.10 pmol/L; grade III: TAPSE < 18 mm: 26.50 pmol/L). Copeptin concentrations were able to discriminate patients with decreased right heart function defined as TAPSE < 18 mm (area under the curves [AUC]: copeptin: 0.793; P = 0.001; NT-proBNP: 0.805; P = 0.0001). Within a multivariable linear regression model, copeptin was independently associated with TAPSE (copeptin: T: –4.43; P = 0.0001; NT-proBNP: T: –1.21; P = 0.23). Finally, copeptin concentrations were significantly associated with severely reduced right heart function (TAPSE < 18 mm) within a multivariate logistic regression model (copeptin: odds ratio: 0.94; 95% confidence interval: 0.911–0.975; P = 0.001).
Conclusions
This study demonstrates that the novel biomarker copeptin reflects longitudinal right heart function assessed by standardized transthoracic echocardiography compared with NT-proBNP.
Introduction
Cardiovascular diseases represent a major health burden worldwide. Heart failure itself is the leading cause of cardiovascular death. Patients suffering from heart failure are associated with increasing risk of hospitalization related to significant expenditures in the health-care system. 1 The lifetime risk for patients suffering from heart failure aged 55 years is 33% for men and 28% for women in Europe. 2 Therefore, accurate diagnosis and therapy are essential for each individual heart failure patient.
Studies commonly focus on left-sided heart failure either with reduced or preserved ejection fraction; however, the right ventricle often remains unconsidered. In a study of 205 stable heart failure patients with left ventricular systolic dysfunction, de Groote et al. showed that right ventricular function (RVF) was an independent predictor of survival. 3 Notably, even in patients with advanced left-sided heart failure, RVF may represent a key factor for prognosis, as demonstrated by Guazzi and Naeije. 4 Postcapillary pulmonary arterial hypertension may deteriorate RVF in the presence of advanced stages of left ventricular heart failure.5–9 From the perspective of the right ventricle, the increasing afterload may lead to myocardial hypertrophy and final dilatation, resulting in right ventricular longitudinal dysfunction. Standardized transthoracic echocardiography represents the most common and accurate diagnostic technique to evaluate RVF. Tricuspid annular plane systolic excursion (TAPSE) is regarded as the parameter of choice to reflect longitudinal RVF. TAPSE can be assessed fast, easily and precisely even under bad ultrasound conditions, featuring a high interobserver reliability and correlating with right ventricular ejection fraction measured by radionuclide angiography.6,10–17
Copeptin is released in equimolar mass together with vasopressin by the hypothalamus. 18 Compared to the potent vasoconstrictive neurohormonal peptide vasopressin, copeptin, consisting of 39 amino acids, reveals a longer half-life and is therefore often used as a surrogate biomarker in patients with heart failure.19–24 In line, aminoterminal pro-B-type natriuretic peptide (NT-proBNP) is an established biomarker for heart failure. 18 However, there is only limited data available evaluating the association between copeptin, NT-proBNP and longitudinal RVF. Therefore, the present study evaluates whether copeptin is able to reflect longitudinal RVF compared to NT-proBNP in patients undergoing standardized transthoracic echocardiography.
Methods
Study design and population
The Cardiovascular Imaging and Biomarker Analyses study (clinicaltrails.gov identifier: NCT 03074253) is a clinically prospective, controlled and mono-centric study conducted at the University Medical Center Mannheim, Germany. The present study incorporated a population subset derived from a patient cohort who underwent routine echocardiography at the University Medical Center Mannheim, Germany between 2014 and 2016. Written informed consent was obtained from all participants or their legal representatives. For our study, the Declaration of Helsinki was applied, and it was approved by the local ethics committee (the medical ethics commission II of the Faculty of Medicine in Mannheim, University of Heidelberg, Germany [local ethics reference number: 2014–615 N-MA]). Patients coming in for routine checkup underwent transthoracic echocardiography, and blood was drawn for measuring natriuretic peptides as well as copeptin and other laboratory markers. Also, relevant clinical data of each patient were ascertained, such as medical history and laboratory work-up. Patients under the age of 18 years or those with a left ventricular ejection fraction < 50% or moderate to severe valvular heart disease were excluded from the study. As this is a non-interventional, observational study, the diagnostic procedures and treatment of the patients have not been modified.
Echocardiography
A detailed routine transthoracic echocardiography was performed for all patients and examined by independent doctors. Standard techniques were applied in order to acquire reproducible images. The assessed structural indices included systolic function with left ventricular ejection fraction (LVEF) using Simpsons’ biplane mode, left atrial (LA) size, end-systolic and -diastolic volumes and diameters, as well as mild valvular heart diseases, right atrial (RA) size and RVF measured by TAPSE. The apical four-chamber view was adjusted to obtain TAPSE by using the M-mode cursor optimally aligned along the direction of the tricuspid lateral annulus. 6 The area as well as the volume of the RA was measured end-systolic in a four-chamber view, immediately before opening the tricuspid valve. Transthoracic echocardiography was performed during clinical routine by specialized operators (operation count each >2000 echocardiographies in the past) and in addition by supervision by advanced attending operators (operation count >20,000 echocardiographies in the past). Specifically, before data analysis, all studies were finally reviewed by an independent echo specialist (operation count >20,000 echocardiographies in the past), who reviewed and in case of inconsistent results re-assessed echo measurements based on the original echocardiographic study and measurements obtained. Echocardiographic analyses were investigated while blinded to the results of final biomarker measurements. The echocardiographic inclusion criteria specified that patients had a preserved LV function (LVEF ≥ 50%). Consequently, patients with an LVEF < 50% were excluded. Additionally, patients with moderate and severe heart valve disorders, classified as either stenosis or regurgitation, were excluded. Furthermore, patients with existing pulmonary arterial hypertension (PAH) were excluded, defined as mean pulmonary arterial pressure above 50 mmHg, as assessed by echocardiography indicating no evidence of relevant PAH.
Laboratory analysis
At a single assessment point upon study, inclusion blood samples by venipuncture were collected within 72 h after routine echocardiography. The samples were taken into serum monovettes or EDTA containing tubes and centrifuged within 30 min at 2500 g for 10 min afterwards. The plasma and serum were separated, cooled liquid nitrogen and stored at –80°C for further analysis. For generating quantitative data of copeptin, the copeptin proAVP assay (BRAHMS, Thermo Fisher Scientific Inc., Hennigsdorf, Germany) was used on the Kryptor compact PLUS analyzer. The sample matrix was EDTA plasma which was centrifuged at 2700 g for 15 min after thawing. Following the CLSI EP17-A2 guideline the Limit of Detection (LOD) has been assessed as being 0.69 pmol/L and the Limit of Quantification (LOQ) as beeing 1.23 pmol/L. The mean in healthy controls is 5.10 pmol/L with a SD of 3.7. The intra-assay CVs are < 15% (concentration range 2.0–4.0 pmol/L) and < 3% (concentration > 50 pmol/L). The inter-assay CVs are < 18% (concentration range 2.0–4.0 pmol/L) and < 5% (concentration > 50 pmol/L). The direct measuring range is 0.7–500 pmol/L. Due to no availability of local performance data, all data is taken from the instructions for use (IFU). The serum concentration of NT-proBNP, used as a reference biomarker, was measured using a proBNP II STAT assay on a Cobas e 602 analyser (Roche Diagnostics, Mannheim, Germany). After thawing, the serum was centrifuged at 2500 g for 10 min. The age-dependent reference values in an apparently healthy reference group according to the IFU are as follows. In the age range from 18 to 44 years, the mean is 35.6 ng/L with a SD of 30.2 and >75 years 211 ng/L with a SD of 62.4. The CVs for repeatability are 2.0% (concentration of 142 ng/L) and 2.0% (concentration 6552 ng/L). The CVs for intermediate precision are 3.5% (concentration 59.3 ng/L) and 2.3% (concentration 6552 ng/L). The measuring range is 5–35,000 ng/L. Serum creatinine concentrations were measured using the Creatinine Jaffe Gen.2 assay on a Cobas c 702 analyzer (Roche Diagnostics, Mannheim, Germany), and the glomerular filtration rate (eGFR) was estimated using the modification of diet in renal disease (MDRD) formula. 17 These samples were also centrifuged at 2500 g for 10 min. The gender-dependent reference values are in females 44-80 µmol/L and males 62 -- 106 µmol/L. The CVs for repeatability are 2.3 % (concentration 72.9 µmol/L) and 1.0 % (concentration 1812 µmol/L). The CVs for intermediate precision are 3.7 % (concentration 51.3 µmol/L) and 2.1 % (2005 µmol/L). The measuring range is 15 -- 2200 µmol/L. All data is taken from the IFU.
Statistical analysis
The statistical analysis was performed using SPSS (version 25.0 optimized for macOS). RVF was measured by TAPSE and also indirectly by RA area. The study population was diverted into three subgroups: TAPSE >24 mm, TAPSE 18–24 mm, TAPSE < 18 mm. Continuous variables are presented as mean or median and interquartile ranges (IQRs; 25th to 75th percentile), whereas category variables are presented as percentages. Two-tailed P < 0.05 was considered statistically significant. Kolmogorov-Smirnov test was used to check whether the neurohormonal measurements were normally distributed or not. Non-normally distributed data were calculated as logarithmic for univariable correlation. When data was not normally distributed with significant deviations from the Gaussian distribution, Kruskal-Wallis was assessed to compare differences in copeptin and NT-proBNP concentrations among the different subgroups of RVF. Correlation between copeptin and baseline variables, such as NT-proBNP and echocardiographic parameters, was assessed using Pearson correlation. To identify variables that influence RVF a backward stepwise multivariable linear regression analysis was performed. The area under the curves (AUC) of receiver operating characteristics (ROC) of copeptin and NT-proBNP were generated to assess the ability to predict right ventricular (RV) dysfunction grade III given with 95% confidence intervals. ROC curves were generated for TAPSE < 18 mm and TAPSE < 14 mm. Comparisons of AUCs were performed by the method of Hanley et al. 25 Multivariable linear regression analysis was performed in order evaluate superiority for the association of copeptin with TAPSE over NT-proBNP and clinical variables.
Results
Study population
A total of 91 patients were enrolled. The baseline characteristics are described (Table 1). Mean age was 66 years (range 22–97 years); sex was almost equally distributed (54% vs. 46% females). Longitudinal RVF was assessed by TAPSE, and the population was divided into three subgroups: TAPSE > 24 mm (n = 23), TAPSE 18–24 mm (n = 55), TAPSE < 18 mm (n = 13). The cardiovascular risk factors arterial hypertension (87%) and hypercholesterinemia were most common (47%). No evidence of relevant chronic kidney disease was found in the present cohort (median eGFR of 67 ml/min/m2, median creatinine 88.00 μmol/L). The distribution of echocardiographic parameters in the overall cohort and in TAPSE subgroups is shown (Table 2). No relevant differences of echocardiographic parameters were evident in between these groups.
Baseline characteristics of study population.

IQR: interquartile range.
Distribution of echocardiographic indices according to TAPSE sub-groups (median (IQR)).
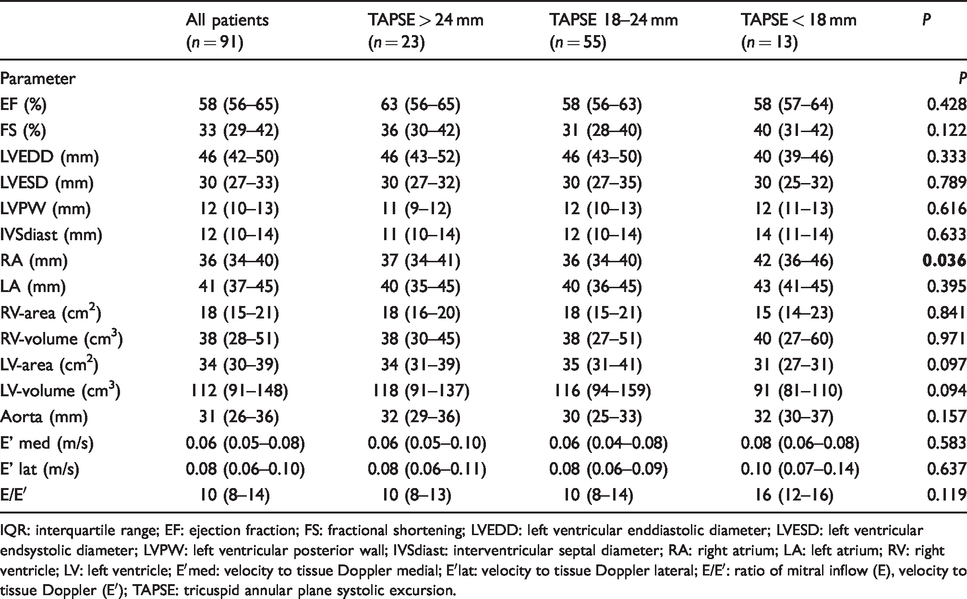
IQR: interquartile range; EF: ejection fraction; FS: fractional shortening; LVEDD: left ventricular enddiastolic diameter; LVESD: left ventricular endsystolic diameter; LVPW: left ventricular posterior wall; IVSdiast: interventricular septal diameter; RA: right atrium; LA: left atrium; RV: right ventricle; LV: left ventricle; E′med: velocity to tissue Doppler medial; E′lat: velocity to tissue Doppler lateral; E/E′: ratio of mitral inflow (E), velocity to tissue Doppler (E′); TAPSE: tricuspid annular plane systolic excursion.
Copeptin and echocardiographic parameters
Copeptin correlated significantly (Table 3) with TAPSE (r = –0.412; P = 0.0001), RA diameter (r = 0.275; P = 0.010), RA area (r = 0.256; P = 0.016), RA volume (r = 0.375; P = 0.001), RV volume (r = 0.218; P = 0.039) and E/E′ (r = 0.378; P = 0.0001). Median copeptin concentrations were significantly higher in patients with RA area > 18 cm2 compared to ≤18 cm2 (15.4 pmol/L vs. 10.6 pmol/L; P = 0.002) (data not shown). Finally, copeptin correlated significantly with NT-proBNP (r = 0.646; P = 0.0001). No correlation in between both biomarkers and LVEF was found.
Univariable correlations between echocardiographic parameters and biomarkers.
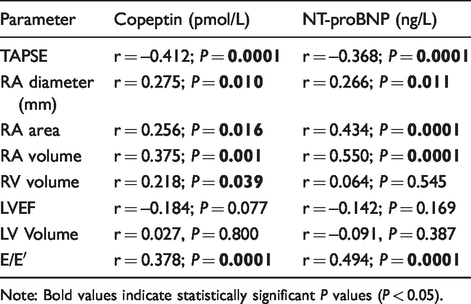
Note: Bold values indicate statistically significant P values (P < 0.05).
Copeptin reflects different stages of RV dysfunction according to TAPSE
Copeptin concentrations increased according to decreasing stages of TAPSE (P = 0.001) (Figure 1(a)). Accordingly, NT-proBNP concentrations increased significantly (P = 0.002) (Figure 1(b)). The mean value of copeptin in RVF grade I (TAPSE > 24 mm) was 7.5 pmol/L (median: 5.2 pmol/L), in RVF grade II (TAPSE 18–24 mm) was 12.7 pmol/L (median: 8.1 pmol/L) and in RVF grade III (TAPSE < 18 mm) was 32.3 pmol/L (median: 26.5 pmol/L).
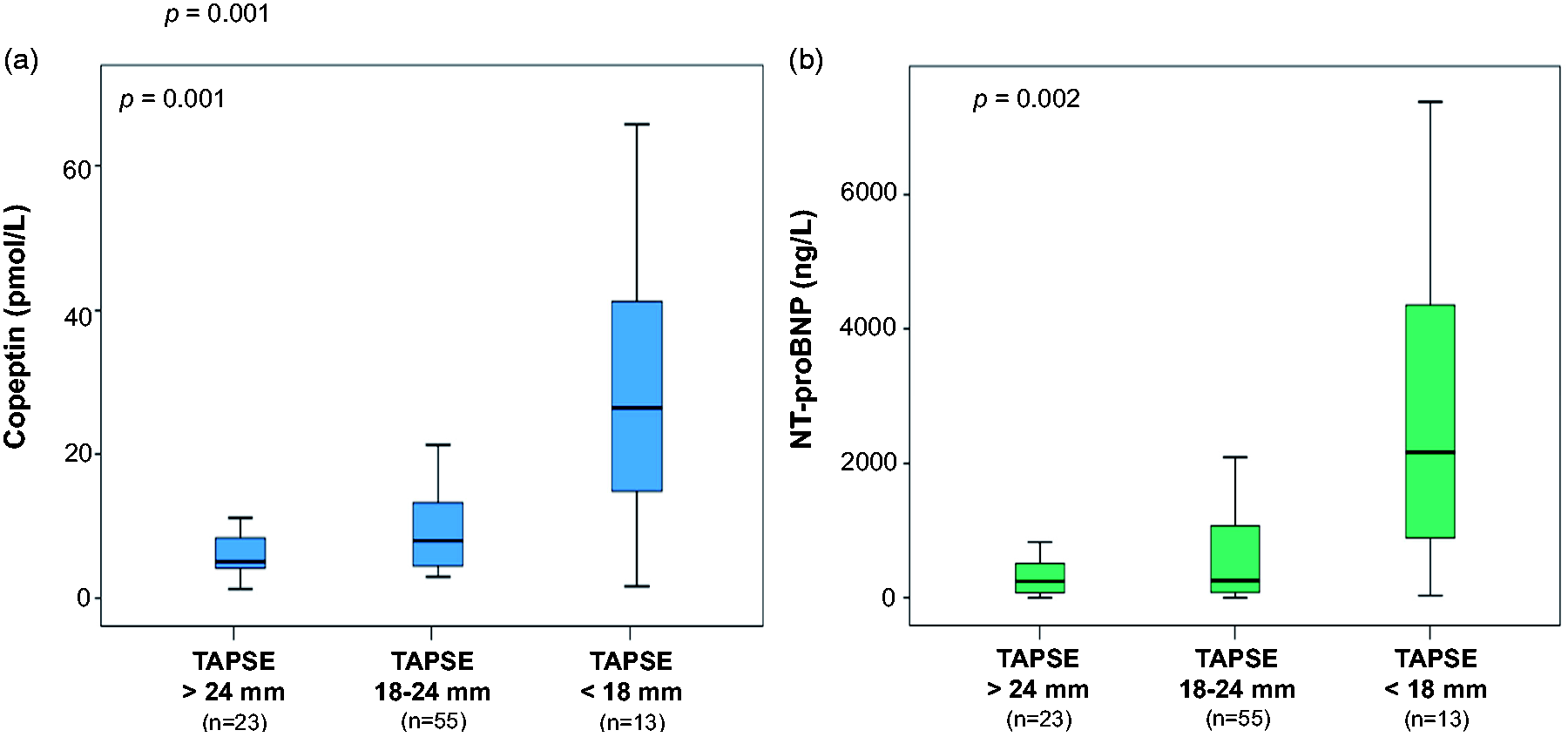
Distribution of Copeptin (a) and NT-proBNP (b) serum concentrations according to subgroups of reduced TAPSE. Data are presented as medians with 25th and 75th percentiles (boxes) and 5th and 95th percentiles/whiskers).
As demonstrated by C statistics (Figure 2), copeptin as well as NT-proBNP were able to discriminate the diseased right ventricle with TAPSE below 18 mm (copeptin: AUC: 0.793; 95% CI: 0.641–0.944; P = 0.001; NT-proBNP: AUC: 0.805; 95% CI: 0.676–0.934; P = 0.001). Furthermore, both copeptin and NT-proBNP were able to discriminate TAPSE <14 mm (Figure 3), (copeptin: AUC: 0.801; 95% CI: 0.608–0.994; P = 0.003; NT-proBNP: AUC: 0.794; 95% CI: 0.622–0.965; P = 0.004). However, no significant difference was observed between the AUCs of copeptin and NT-proBNP (Hanley method; TAPSE <18 mm, P = 0.911; TAPSE <14 mm, P = 0.938).25
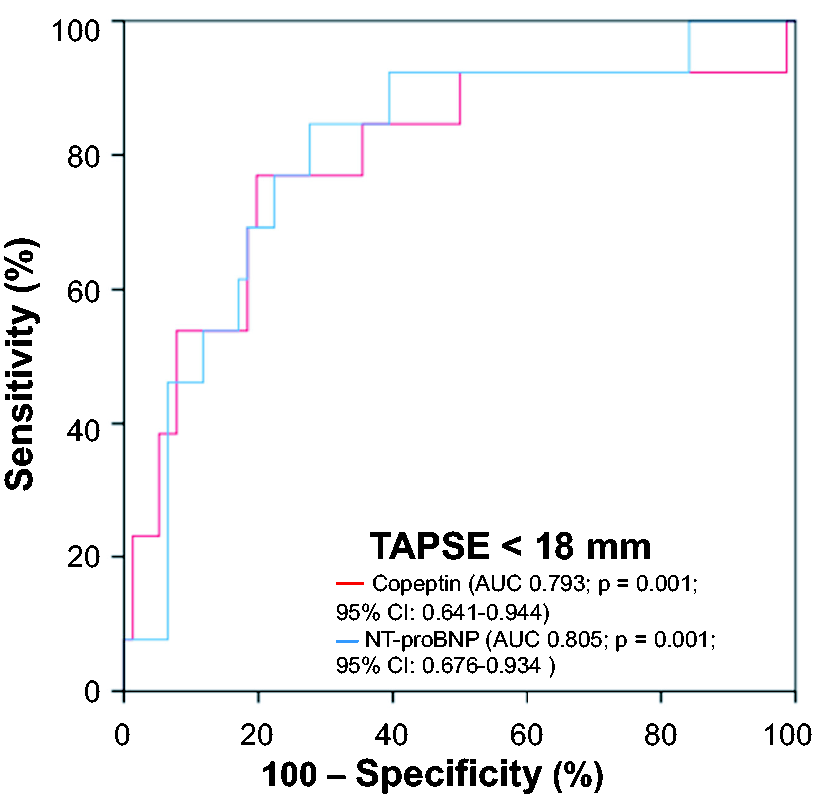
Receiver-operating characteristic (ROC) curves revealing valuable discrimination of patients with reduced TAPSE of < 18 mm by serum concentrations of copeptin and NT-proBNP.
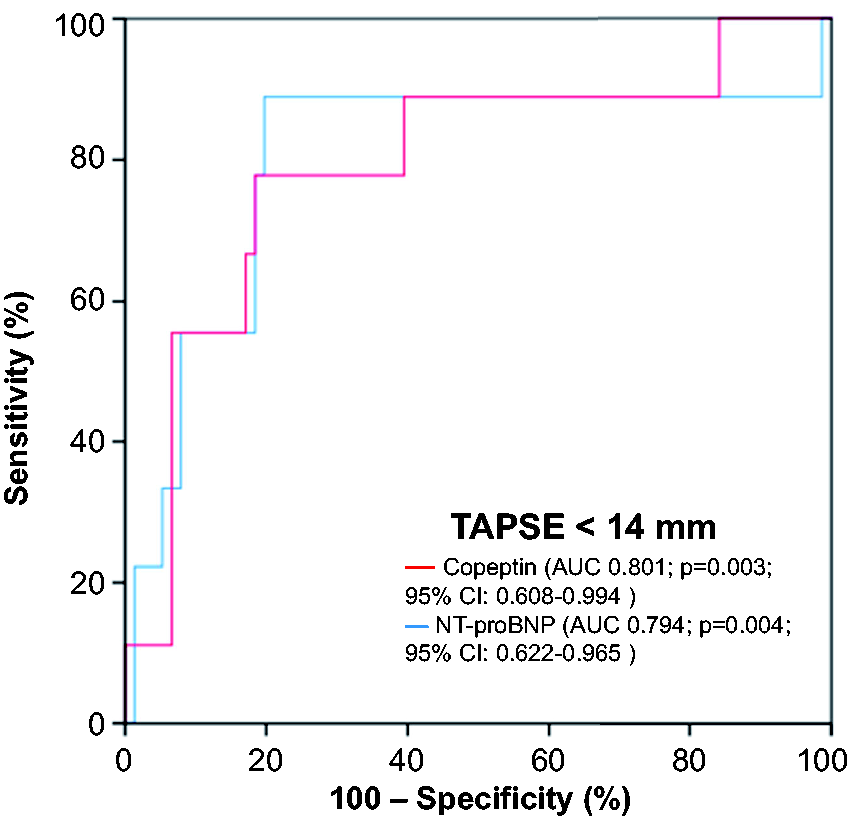
Receiver-operating characteristic (ROC) curves revealing valuable discrimination of patients with reduced TAPSE of <14 mm by serum concentrations of Copeptin and NT-proBNP.
Multivariable linear regression model
A backward stepwise multivariable linear regression model adjusted for sex, age, creatinine and NT-proBNP revealed that was copeptin independently associated with TAPSE (copeptin: regression coefficient B: –0.017; standard error: 0.004; Beta –0.429; T –4.43; P = 0.0001), while being superior to NT-proBNP (regression coefficient B: –0.038; standard error: 0.0001; Beta: –0.145; T: –1.21, P = 0.23) and further clinical variables (Table 4).
Multivariable linear regression model to evaluate associations with TAPSE.
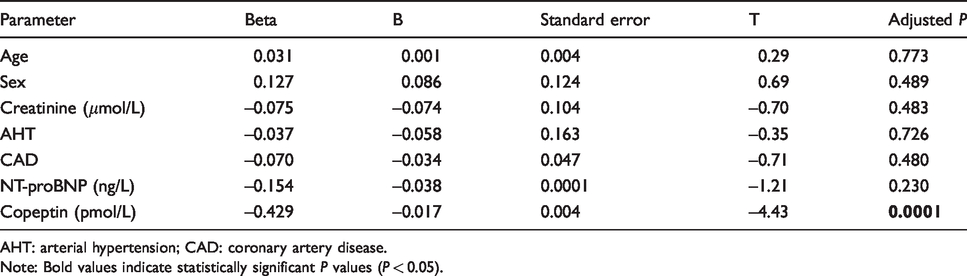
AHT: arterial hypertension; CAD: coronary artery disease.
Note: Bold values indicate statistically significant P values (P < 0.05).
Discussion
The present study aimed to determine the relationship between copeptin and longitudinal RVF reflected by the echocardiographic parameter TAPSE. It was demonstrated that copeptin concentrations significantly increased alongside decreasing stages of longitudinal RVF and valuably discriminated RV dysfunction classified by TAPSE < 18 mm. Copeptin significantly increased according to three different stages of RVF reflected by TAPSE. Copeptin was able to discriminate patients with TAPSE < 18 mm as well as < 14 mm, which are both known predictors of adverse clinical outcome. 11 , 18 , 22 Furthermore, copeptin correlated with TAPSE, RA parameters as well as with RV volume as well as with NT-proBNP. Even after multivariable adjustment in a linear regression model, copeptin revealed superior association with TAPSE compared to NT-proBNP and other clinical variables. The present study therefore delivers novel insights about the diagnostic potential of the biomarker copeptin to diagnose RVF after exclusion of patients with LV dysfunction and PAH, which may become useful for clinical routine diagnostics, even in combination with NT-proBNP.
Transthoracic echocardiography represents the standard diagnostic technique to assess RVF, since it is commonly available, easily to perform and highly cost efficient. Further diagnostic imaging modalities, e.g. cardiac magnetic resonance imaging and real-time three-dimensional or speckle-tracking echocardiography represent rather advanced techniques and are therefore not suitable to assess RVF during clinical routine in every patient. 12 , 14 TAPSE represents the main parameter for assessing RVF by transthoracic echocardiography. Due to the anatomy of the right ventricle, ventricular stroke volume is mainly generated by longitudinal shortening rather than by the reduction of the diameter of the radial cavity. 26 This explains the strong correlation between RVF and TAPSE, as recently demonstrated by Vizzardi et al. 14 This concept was proven by further studies, thereby proposing TAPSE as the predominant surrogate parameter for RVF. 11 , 15 , 16 The present study further demonstrated that copeptin also correlated with RA area and volume, indicating further impairment of RVF.
RV dysfunction very often reflects the beginning of end-stage heart failure after years of predominantly left ventricular systolic dysfunction. Therefore, persistent biventricular failure is limiting the prognosis of each patient suffering from heart failure. 4 , 27 , 28 Moreover, an acute myocardial infarction (AMI) not only represents acute myocardial cell death but can also be followed by RA impairment as a distinct feature leading to an impaired RVF. 29 In line, the therapeutic options for RV dysfunction are limited as well, and supportive scientific evidence is rare.30–32 Although surgical tricuspid valve replacement or annuloplasty, interventional tricuspid valve repair, as well as right ventricular assist devices exist as therapeutic options, they merely aim to delay global heart failure and do not represent causal therapies even as a consequence of left-sided heart failure.33–35
The evaluation of novel cardiac biomarkers to assess RVF is strongly needed. In order to avoid potential bias from left-heart pathologies, e.g. LVEF < 50% or moderate to severe aortic or mitral valve disorders, it is essential to exclude these conditions from biomarker studies evaluating the diagnostic value of candidate biomarkers for RVF. Copeptin was initially investigated in patients suffering from AMI.36–38 Thereafter, copeptin was further evaluated in the context of heart failure. Within the biomarker in acute heart failure (BACH) study, Maisel et al. demonstrated the valuable prognostic capacity to predict 90-day all-cause mortality. 39 The prognostic value of copeptin was proven by further studies in patients suffering from chronic heart failure. 24 , 27 ,40–42 Copeptin already has been analysed in left-sided heart failure or PAH. However, the diagnostic value of copeptin for exclusive RVF has rarely been investigated and therefore was the primary aim of the present study.
Pathophysiologically, decreased renal blood flow as a consequence of low cardiac output during systolic heart failure activates the renin–angiotensin–aldosterone system (RAAS) leading to peripheral vasoconstriction. Increased plasma concentrations of angiotensin II stimulate the nucleus supraopticus and nucelus paraventricularis of the hypothalamus to increase the vasopressin secretion. 43 Furthermore, in the presence of heart failure, activation of baroreceptors due to volume overload in both atria leads to increased vasopressin secretion. The so-called Gauer-Henry-reflex is fed back via afferents leading from both atria to the hypothalamus. 43 Since copeptin is secreted in equimolar mass as vasopressin by the hypothalamus, both biomarkers are increased in the presence of heart failure. 24 , 44 , 45 The vasoconstrictive effects of vasopressin have recently been reported in the context of cardiogenic shock, where the vasopressin-mediated endothelin release increased both cardiac and cerebral blood flow. 43 Together with the RAAS-mediated vasoconstriction, more reflecting the increasing afterload during worsening congestive heart failure either within systemic or pulmonary circulation, both pathophysiologic concepts may explain the potential clinical diagnostic value of copeptin even in isolated RV dysfunction. 8 , 27 , 43
Biomarker research primarily focuses on the evaluation of novel candidate markers in rare disease conditions, especially by evaluating their comparative diagnostic capacity. This combined approach reveals the chance to detect novel pathways, which are obviously present, as shown in the present population of patients with isolated longitudinal RVF assessment. The evaluation of the novel biomarker copeptin showed comparable diagnostic potential to NT-proBNP, which is the main finding of the present research. Furthermore, copeptin might bear the potential to provide additional diagnostic information to NT-proBNP because of different pathophysiological pathways. As mentioned in the study limitations, this needs to be evaluated in larger randomized controlled studies.
Taken together, the present study demonstrated that copeptin concentrations inversely correlate with decreasing longitudinal RVF. Furthermore, stages of clinically relevant RV dysfunction as defined by TAPSE below 18 or 14 mm were demonstrated to be reliably discriminated by copeptin concentrations. Until today, the diagnosis of heart failure poses a challenge and needs improvement with its clinical handling. Especially the right ventricle has been neglected much too often, although RV dysfunction is strongly associated with clinical outcomes – as emphasized in the guidelines. 6 Copeptin as a screening biomarker could potentially allow an earlier detection of right ventricular impairment – even in the presence of preserved LV function in the present study. Revealing information about RVF could help to adjust heart failure therapy earlier and therefore improve the prognosis of each heart failure patient. Implementing both biomarkers – with comparable diagnostic capacities for RVF, i.e. copeptin and NT-proBNP – in a multimodal approach may reveal further benefits. These may consist in evaluating patients with combined right and left heart failure, where either copeptin or NT-proBNP may outweigh the weaknesses of each other due their different pathophysiology and stimuli for synthesis. Future larger, prospective, randomized and controlled studies need to re-evaluate the diagnostic yield of copeptin compared with NT-proBNP. Using multimodal imaging approach, e.g. by echocardiography together both biomarkers may allow a more precise assessment of right heart function and failure. Moreover, sequential follow-ups with serial blood measurements would potentially reveal even more insights. Before implementing copeptin into clinical routine, also the cost–benefit ratio needs to be investigated further.
Limitations of the study
This was a single-centre study with a limited sample size and therefore results cannot easily be transferred to the general population. Echocardiographic assessment was performed using a standard 2D ultrasound device, while 3D or 2D speckle-tracking echocardiography was not applied. Measurements of Tei-Index and radial RVF were beyond the scope of the present study, since both represent less confirmatory markers of RVF. Future and large-scaled multicentre studies are needed to re-evaluate our findings and further translate them into patients with global heart failure in relation to adequate assessment of both right and left ventricular function.
Conclusion
Circulating plasma copeptin concentrations were significantly elevated in patients suffering from isolated right heart failure and were able to discriminate reduced RVF defined as TAPSE < 18 and 14 mm. The novel biomarker copeptin may bear the potential to become a useful additional biomarker in the assessment of chronic heart failure patients, especially in those suffering from isolated RV or end-staged global heart failure, while accomplishing the clinical and echocardiographic evaluation by pathophysiological biochemical aspects.
Footnotes
Acknowledgements
The copeptin assay kits were kindly provided by BRAHMS (Thermo Fisher Scientific Inc., Hennigsdorf, Germany).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by the local ethics commission II of the Faculty of Medicine Manheim, University of Heidelberg.
Guarantor
MBe.
Contributorship
MH participated in data acquisition, data analysis, statistical analysis, literature research and drafted the first manuscript. MN, MBe conceived the study, participated in its design and coordination, participated in data and statistical analysis and interpretation and helped to draft and revise the manuscript for important intellectual content. SHK, JM, UA, MZ participated in data acquisition, data analysis and literature research and helped to draft the first manuscript. AH, SL and MF revised the article for important intellectual content. UH, MK participated in the study design and critically revised the article for important intellectual content. MBo and IA critically revised the article for important intellectual content. TR, TB participated in data analysis, carried out the immunoassays and revised the article for important intellectual content. MBe conceived the study, participated in its design and coordination and revised the article for important intellectual content. All authors read and approved the final version of the article.
