Abstract
In this study, 102 patients with hypertensive intracerebral hemorrhage (52 patients in the citicoline treatment group and 50 patients in the routine treatment group) were analyzed and compared. It was found that citicoline-assisted treatment of hypertensive intracerebral hemorrhage could inhibit the release of serum glial fibrillary acidic protein (GFAP) and peptides (P < 0.05) and improve the prognosis and neurological and daily living ability of patients (P < 0.05). In addition, CDP-choline reduced the mortality of neurons, inhibited inflammatory factors, and improved the prognosis of patients (P < 0.05). A high dose of CDP-choline was also significantly effective in protecting neurons.
Introduction
Hypertensive cerebral hemorrhage is a common disease that severely impairs human health, with high rates of morbidity, disability, and case fatality. Epidemiology studies show that patients with hypertensive cerebral hemorrhage account for one-third of the patients with cerebrovascular disease, and the case fatality rate within 1 month is approximately one-fifth. 1 In particular, a series of pathological reactions of the body and partial brain tissues occur after cerebral hemorrhage, including intracerebral hematoma expansion. As a result, multiple vaso-active substances will be released, thus inducing encephaledema, coagulation, and fibrinolytic system changes; reduction of partial cerebral blood flow; and so on.2,3 To date, many new biological molecules have been identified in central nervous system disease, which facilitates the research on pathological and physiological changes of patients with hypertensive cerebral hemorrhage. 4 GFAP (glial fibrillary acidic protein) is an intermediate filament protein and is considered as a specific marker of astrocytes. It is the cytoskeleton of astrocytes. 5 Copeptin is a type of glycopeptide with a molecular weight of approximately 5 kD, and it contains 39 amino acids. Research shows that this factor can be used as an independent predictor of cerebral infarction prognosis, but it is rarely used in research on hypertensive cerebral hemorrhage. The treatment principle of hypertensive cerebral hemorrhage is to control continuous hemorrhage, prevent rehaemorrhagia, reduce intracranial pressure, and relieve encephaledema.6,7 The conventional drug mannitol can rapidly increase plasma osmotic pressure, reduce blood viscosity, and inhibit generation of cerebrospinal fluid, thus lowering intracranial pressure. However, the long-term efficacy of mannitol is poor. Citicoline can reduce the cell death resulting from excitotoxic neurotransmitter stimulation, via reducing glutamic acid concentration in brain tissues. In this study, the effects of clinical application of citicoline in the treatment of hypertensive cerebral hemorrhage on serum GFAP and copeptin level were explored. In addition, the influence of citicoline on survival rate and inflammatory factor expression after neuronal cell impairment was investigated through a cytological experiment to determine the mechanism of citicoline and improvement of prognosis.
Data and method
Ethical approval
The study was approved by the Institutional Ethics Committee of Beijing Rehabilitation Hospital of Capital Medical University, and written informed consent was obtained from all participants.
Object of study
A total of 102 patients with hypertensive cerebral hemorrhage who received diagnosis and treatment at Neurological Rehabilitation Center, Beijing Rehabilitation Hospital of Capital Medical University, from August 2014 to February 2018 were recruited into this double-blinded study. The inclusion criteria for this study, which was approved by the Ethics Committee of Beijing Rehabilitation Hospital of Capital Medical University, were as follows: conformed to diagnostic criteria of hypertensive cerebral hemorrhage and verified by computed tomography (CT) head scanning or magnetic resonance imaging (MRI); complete clinical data; age: 40–75 years old; and hospitalized within 12 h of attack. The exclusion criteria were as follows: patients with a disease that affects scoring standards, such as a tumor; pregnant women and patients in the lactation period; patients with a gastrointestinal disease that severely affects drug absorption; patients with allergic constitution; and those who did not cooperate, had poor obedience, and could not guarantee completion of the experiment. The 102 patients were divided into an observation group with citicoline treatment (52 cases) and a control group without citicoline (50 cases). The age, body mass index (BMI), the duration of attack to hospitalization, gender, bleeding amount, and bleeding part did not differ significantly among the groups (P > 0.05), as shown in Table 1.
Baseline data and total effective rates comparison of both groups.
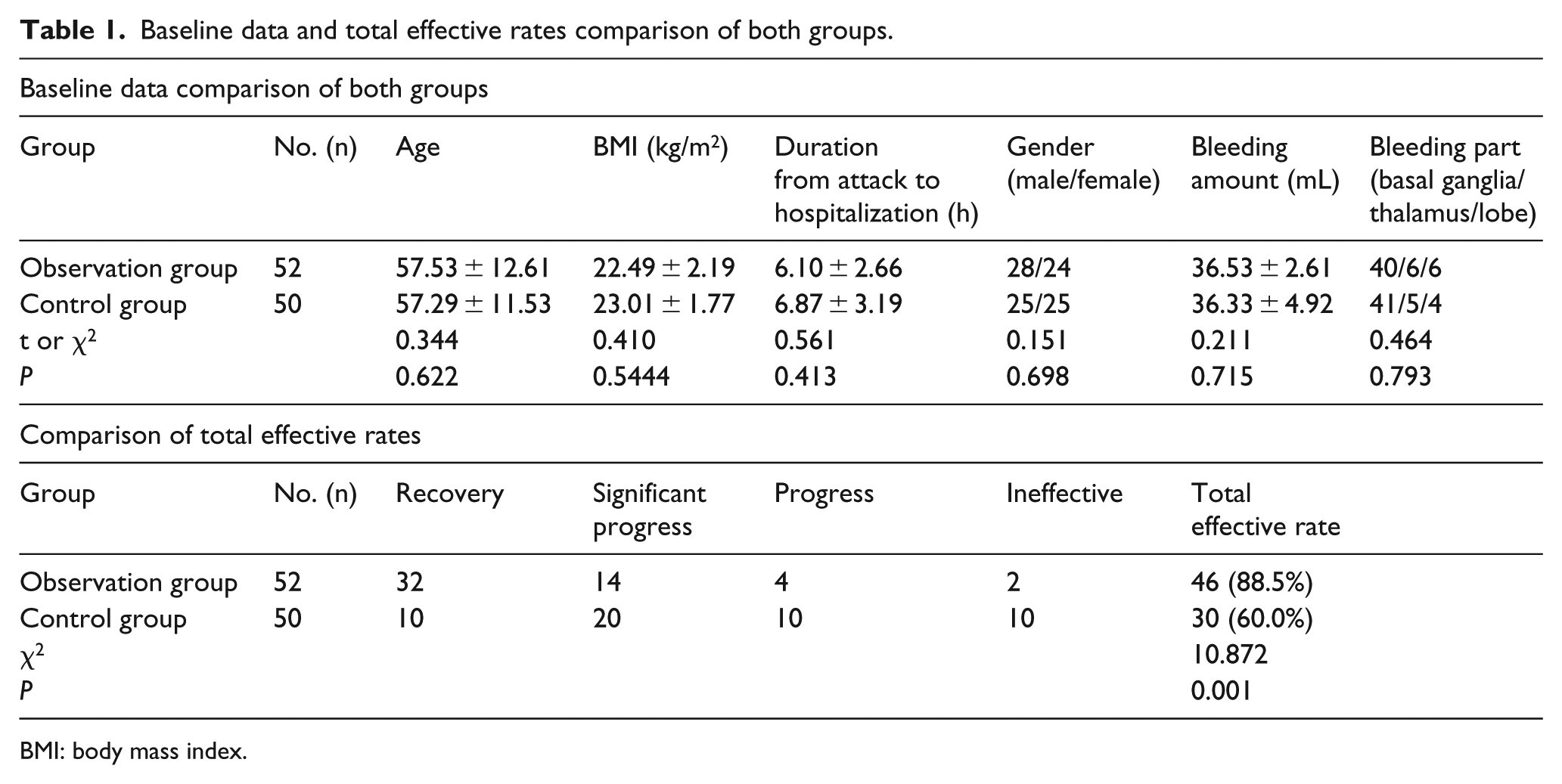
BMI: body mass index.
Therapeutic method
Conventional treatment was administered to both groups, including maintaining water, electrolyte and acid-base equilibrium, bed rest, dehydration, and reduction of intracranial pressure and blood pressure. For the antihypertensive strategy, in the acute phase of intracerebral hemorrhage (ICH), we gave priority to patients with moderate hypertension who have sublingual nifedipine as an antihypertensive agent and use Labellol as an antihypertensive agent. Labellol is easy to measure and has a fast antihypertensive effect, but it has no effect on intracranial pressure, cerebral blood flow, and cerebral circulation autoregulation. For patients with severe hypertension, left ventricular failure, and acute coronary insufficiency, intravenous drip of sodium nitroprusside was used to reduce blood pressure. The objective of blood pressure control is to set individualized blood pressure lowering targets according to the patient’s personal conditions. Generally, the mean arterial pressure (MAP) is maintained at 100–130 mmHg, or a systolic blood pressure (SBP) of 140–160 mmHg, to ensure that sufficient cerebral perfusion pressure is maintained while lowering blood pressure, regulating blood glucose, and keeping respiratory tract unobstructed.
In control group, mannitol was used for treatment. An intravenous drip of 250 mL 20% mannitol (Sichuan Kelun Pharmaceutical Co., Ltd., GYZZ H20043784) was administered at 8-h intervals (3 times per day).
In observation group, based on the treatment of the control group, citicoline treatment was given. A 0.5 g citicoline injection (Changchun Dazheng Pharmaceutical Co., Ltd., GYZZ H22026208) was used for intravenous drip once per day.
Both groups were treated and observed for 28 days.
Observation indicators
(1) Criteria of curative effect were as follows: recovery—after the treatment, reduction ratio of NIHSS (National Institutes of Health Stroke Scale) score 91%–100%; significant progress—after the treatment, reduction ratio of NIHSS score 46%–90%; progress—after the treatment, reduction ratio of NIHSS score 18%–45%; ineffective—failed to achieve the above criteria or deteriorated. Total effective rate = (basic recovery + significant progress)/total number of cases × 100%. (2) Before and after the treatment, NIHSS scoring and Barthel index scoring were carried out. A higher NIHSS score indicated a more serious neurologic impairment; a higher Barthel index score indicated a stronger daily living capacity. (3) Before and after the treatment, patients’ venous blood was collected, left to sit for 30 min, and then centrifuged at a low temperature (4°C, 3000 rpm/min, 10 min). Then, the upper serum was separated and refrigerated at −80°C. Enzyme-linked immunosorbent assay method was employed to detect serum GFAP and copeptin level. The operation was conducted in strict accordance with the specifications of the kit (purchased from American RB Corporation and GSK).
Steps of cell experiment
Cell treatment
The sample was divided into five groups: (1) normal group, (2) lipase (LPS) group, (3) low dose group, (4) moderate dose group, and (5) high dose group. After neuronal cell (from Beijing Rehabilitation Hospital of Capital Medical University) passage, 200,000 cells were inoculated in each pore. After the cell growth value reached 80%–90%, 2% FBS (fetal bovine serum) was used for hunger treatment for 24 h. The drug or compound that was able to be dissolved in the medium was dissolved with 1% DMSO (dimethyl sulfoxide) serum-free medium. The drug or compound that was difficult to dissolve was dissolved with analytically pure DMSO and then diluted to half concentration of the inhibition ratio by the serum-free medium. The primary medium was sucked out, and all kinds of compounds and drugs prepared with the medium were sucked in. After co-incubation in the CO2 cell incubator for 24 h, the protein was split, and protein expression level detection was conducted.
Cell lysis
Original cell culture fluid of cells in each group was sucked out and discarded. After immersion and cleaning once with 1× PBS (phosphate-buffered saline), 300 μL/pore of RIPA (radioimmunoprecipitation assay) was added and shaken under 4°C for 30 min. Then, the pipettor was used to blow and beat cells in each pore repeatedly. The cell lysis buffer was moved to a 1.5-mL centrifuge tube to gain cell protein samples for protein quantification.
Main reagents of MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromidefor) experiment
Steps of MTT experiment
The acellular medium was inoculated in a 96-pore plate with 100 μL per pore with a cell density of 3000/pore. There was 100 μL of complete medium in each pore. Three side-pores were set for each group. The marginal pore was filled with sterile PBS and placed in a 5% CO2 incubator with a constant temperature of 37°C for 24 h;
The fresh medium was changed. According to the requirements of each group, each group of cells was stimulated by LPS or a low concentration of citicoline, moderate concentration of citicoline, or high concentration of citicoline. The blank control group was not treated. Then, the cells were placed in a 5% CO2 incubator with a constant temperature of 37°C for culturing;
After culturing for 24 h, 20 μL MTT solution (5 mg/mL) was added to each pore. The cells were continuously cultured for 4 h in the incubator;
The medium was discarded, and 100 μL DMSO was added to each pore and shaken with the shaking table to fully dissolve the crystal substance;
The absorption value of each pore was measured at 490 nm. The experimental group served as the horizontal coordinate and the absorption value served as the longitudinal coordinate to draw the histogram (Figure 1).

Cell survival rates of each group.
Main detection reagents of Western blot (WB)

WB detection method
Protein quantification: cryopreserved supernate was taken for protein quantification.
Prepared SDS (sodium dodecyl sulfate) degenerated 10% polyacrylamide gel (substratum separation gel, single face).
Prepared SDS degenerated 5% polyacrylamide gel (substratum spacer gel, single face).
After the protein sample was mixed with 5× supernate sample buffer (containing β-mercaptoethanol), it was boiled and degenerated for 5 min and ice-bathed for 5 min. The appropriate amount of protein supernate samples was taken for SDS degenerated 10% polyacrylamide gel electrophoresis (SDS-PAGE). After the target protein was effectively separated, electrophoresis was terminated.
After electrophoresis, the gel was taken out and put in the specialized sandwich clamp for membrane transfer. The gel was placed at the negative electrode, and polyvinylidene difluoride (PVDF) membrane was put in the positive electrode. Membrane transfer was conducted for 2 h under 4°C at constant current of 350 mA in the membrane transfer buffer to make protein in the gel transfer to PVDF and form the imprint.
The membrane was put in the 1× Blotto, shaken, and sealed for 2 h under room temperature.
The membrane was cut off according to the imprint position of protein; put in Blotto containing JAK2 (1:1000), p-JAK2 (1:1000), STAT3 (1:1000), and p-STAT3 (1:1000) primary antibodies; and shaken overnight under 4°C.
The membrane was put in 1× TBST (tris-buffered saline, 0.1% Tween 20) solution, shaken, and rinsed for 5 min for 4 times.
The membrane was put in Blotto containing corresponding secondary antibody for 1.5 h under room temperature.
The membrane was put in 1× TBST solution, shaken, and rinsed for 5 min for 4 times.
The membrane was put in the color-developing agent Western Lightning™ Chemiluminescence Reagent for 30 s.
The membrane was put in the exposure box immediately and the photographic film was exposed for 1 min in the darkroom. Then, development and fixation were conducted.
The film was shot with LabWorks™ gel-imaging and analytic system, and brightness values of the protein strips in each group were analyzed. The specific value between brightness values of protein strips of each sample and brightness values of corresponding β-actin (internal reference) strip was calculated to gain the corrected brightness values of protein strips. The control group was used as the standard value 1, and a histogram was drawn (Figure 2).
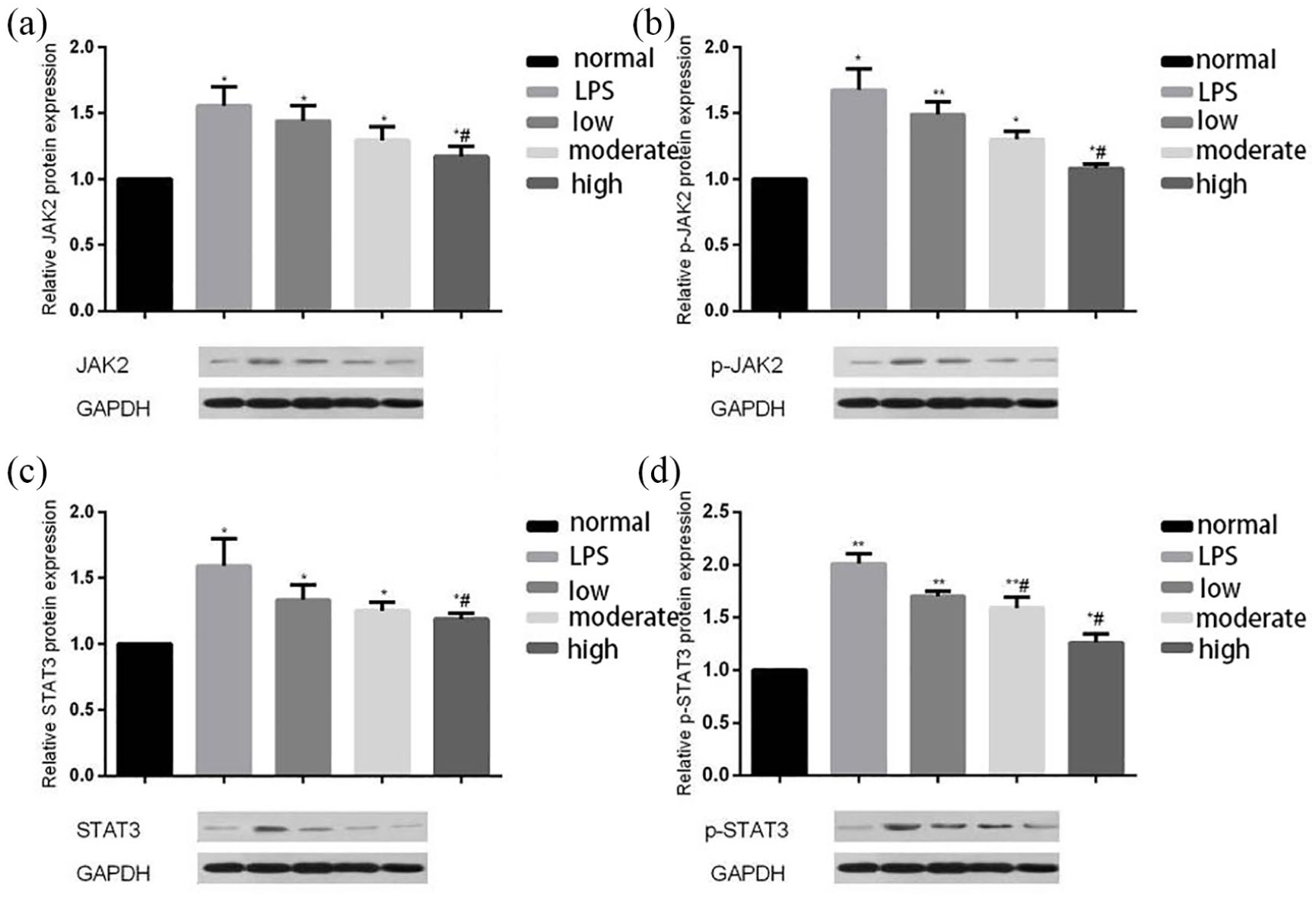
JAK2, p-JAK2, STAT3, and p-STAT3 expression levels in primary neuronal cells in Western blot test. (a) JAK2 expression levels in primary neuronal cells in Western blot test. (b) p-JAK2 expression levels in primary neuronal cells in Western blot test. Protein expression levels of JAK2, p-JAK2, STAT3 and p-STAT3 in the LPS group, low citicoline dose group, moderate citicoline dose group, and high citicoline dose group increased, compared to the normal group. Compared to the LPS group, protein expression levels of JAK2, p-JAK2, STAT3 and p-STAT3 in the low citicoline dose group, moderate citicoline dose group, and high citicoline dose group decreased, with the decline of the high citicoline dose group being most obvious (p<0.05). (c) STAT3 expression levels in primary neuronal cells in Western blot test. (d) p-STAT3 expression levels in primary neuronal cells in Western blot test.
Statistical method
SPSS 22.00 statistical software was used for analysis. The measurement data were expressed with mean ± standard deviation, and enumeration data were expressed with percentage values. The t tests and χ2 tests were used to analyze data at a significance of α = 0.05.
Results
Comparison of total effective rates
After treatment, the total effective rate of the observation group was 88.5%, which was higher than that of control group (60.0%) (P < 0.05), as shown in Table 1.
Comparison of changes in NIHSS score and Barthel index score
After the treatment, NIHSS scores of both groups were lower than those before the treatment, and Barthel index scores were higher than those before the treatment (P < 0.05). Differences between groups were statistically significant. In addition, scores between the observation group and control group after treatment also differed significantly (P < 0.05), as shown in Table 2.
Comparison of changes in NIHSS score and Barthel index score (score, mean ± standard deviation) and comparison of serum GFAP and copeptin content before and after treatment (ng/mL, mean ± standard deviation).

NIHSS: National Institutes of Health Stroke Scale; GFAP: glial fibrillary acidic protein.
Comparison of serum GFAP content
After the treatment, serum GFAP content and copeptin content of both groups were significantly lower than those before the treatment, and the observation group was lower than the control group (P < 0.05), as shown in Table 2 and Figure 1.
Comparison of serum copeptin content
After the treatment, serum copeptin content of both groups was significantly lower than that before the treatment, and the observation group was lower than that of the control group (P < 0.05), as shown in Table 2 and Figure 2.
Results of MTT experiment
LPS group presented the lowest survival rate of neuronal cells, while this measure rose successively in the low citicoline dose group, moderate citicoline dose group, and high citicoline dose group. The cell survival rates of the moderate citicoline dose group and high citicoline dose group were significantly higher than that of the LPS group (P < 0.05; P < 0.01).
JAK2, p-JAK2, STAT3, and p-STAT3 expression levels in primary neuronal cells in WB test
Protein expression levels of JAK2, p-JAK2, STAT3, and p-STAT3 in the LPS group, low citicoline dose group, moderate citicoline dose group, and high citicoline dose group increased, compared to the normal group. Compared to the LPS group, protein expression levels of JAK2, p-JAK2, STAT3, and p-STAT3 in the low citicoline dose group, moderate citicoline dose group, and high citicoline dose group decreased, with the decline of the high citicoline dose group being most obvious (P < 0.05) (Figure 2).
This study showed that after the treatment, the total effective rate of the observation group was 88.5%, significantly higher than that of the control group (60.0%) (P < 0.05). After the treatment, NIHSS scores of both groups were lower than those before the treatment, and Barthel index scores were higher than those before the treatment (P < 0.05). The differences were statistically significant. The scores between the observation group and control group after the treatment also differed significantly (P < 0.05), indicating that the combined use of both can improve therapeutic effect and improve patients’ prognostic nerve and daily activity capacity.
Discussion
Hypertensive cerebral hemorrhage is a common disease severely impairing human health, and mostly attacks people between the ages of 40 and 70. It is also one of the primary reasons for disability. Citicoline can reduce cerebral vascular resistance through the blood brain barrier, and thus improve brain tissue metabolism, promote cerebral function, increase cerebral blood flow, enhance reticular activating system function, and facilitate recovery of limb disorder. 8 The results of this study suggest that the treatment of hypertensive ICH with citicoline can improve the prognosis of patients and the effects of therapy. In addition, CDP-choline can reduce the mortality of neurons and inhibit inflammatory factors. High doses of CDP-choline have been shown to be significantly more effective in protecting neurons.
Research shows that serum GFAP of patients with cerebral hemorrhage maintains a positive correlation with encephaledema volume. A more substantial cerebral hemorrhage leads to more damaged spongiocytes and a greater GFAP release. 9 Citicoline can protect cerebral neurons through inhibiting generation of soluble phosphatidylecithin and effectively weakening activity of phosphatidase A2. This is consistent with our findings, indicating that the application of citicoline can facilitate serum GFAP content.
Copeptin is a C-end fragment of vasopressin and can be used as a biological marker to replace vasopressin. Brain cell impairment can stimulate the hypothalamus–hypophysis–epinephrine axis to synthesize glucocorticoids and release copeptin, thus aggravating nervous system impairment. Copeptin maintains a significant positive correlation with the degree of cerebral injury. 10 This study showed that after the treatment, serum copeptin content of both groups was significantly lower than that before the treatment, and the observation group was lower than that of the control group (P < 0.05), indicating that the combined application of both drugs can more effectively inhibit copeptin release. Although citicoline has a good clinical effect, the specific biological mechanism of citicoline is unclear. In the cytological experiment, we found that citicoline could lower the expression levels of inflammatory proteins such as JAK2, p-JAK2, STAT3, and p-STAT3. Meanwhile, the application of citicoline can increase the survival rate of neuronal cells. High-dose citicoline was more effective. It is believed that one main function of citicoline is to stimulate cephalin synthesis and increase cephalin content. The mechanism may be related to inhibiting the expression of pro-inflammatory cytokines, such as TNF-α (tumor necrosis factor alpha) and IL-6 (interleukin 6), which may be the result of JAK2/STAT3 pathway participation. In addition, the neutrophil-to-lymphocyte ratio (NLR) has been proposed as an easy parameter to assess individual inflammatory status; its predictive value has also been suggested in patients with ICH. Blood-derived inflammatory cells strongly contribute to secondary brain injury following ICH. The neutrophil-induced neurotoxicity is related to a multitude of pathways, including the secretion of cytotoxic mediators and proinflammatory cytokines such as TNF-α and IL-1b, the upregulation of matrix metalloproteinases, the excessive generation of reactive oxygen species, and macrophage activation. 11 For patients with hypertensive cerebral hemorrhage, the application of citicoline is associated with better clinical prognosis. The rise in blood pressure is common after ICH, and it results from the variable combination of multiple mechanisms including premorbid hypertension, increased intracranial pressure, activation of neuro-vegetative signaling, and neuro-endocrine pathways. The analysis of blood pressure management strategies can provide a more comprehensive analysis of factors affecting prognosis, which needs to be explored in future research. 12
Limitations
This study also has some limitations. The sample size is small, and there was no targeted grouping for citicoline application dose. There are many prognostic factors influencing patients with cerebral hemorrhage. These factors will be examined further in future research. Inflammatory factors should be included in future studies to more comprehensively analyze the relationship between CDP-choline and the prognosis of brain injury. An additional limitation of this study is that we did not measure the blood pressure of the patients.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
