Abstract
Diabetes insipidus (DI) is a group of disorders that lead to inappropriate production of large volumes of dilute urine. The three main forms are central DI (CDI), nephrogenic DI (NDI) and primary polydipsia (PP). Differentiating CDI/NDI from PP is important as patients with true DI are at risk of severe dehydration without treatment. Biochemical testing is key in the diagnosis of DI. The indirect water deprivation test (WDT) is commonly used in the investigation of DI but has drawbacks including being cumbersome and sometimes producing equivocal results. Direct measurement of AVP has theoretical advantages but has generally only been used in specialist centres. Disadvantages include the requirement to measure AVP under hypertonic stimulation and pre-analytical/analytical challenges. Copeptin (CT-proAVP) is a proxy marker for AVP that is more stable, easier to measure and has been studied more widely in recent years. Historically, the evidence supporting the diagnostic performance of these tests has been relatively poor, being based on a few small, usually single-centre studies. However more recent, well-designed prospective studies are improving the evidence base for investigation of DI. These studies have focused on the utility of copeptin measurements during stimulation tests. There is evidence that measurement of copeptin under stimulation offers improved diagnostic performance compared to the WDT. There is currently a lack of systematic, evidence-based guidelines on the diagnosis of DI, but as the quality of the evidence defining the diagnostic performance of tests for DI continues to improve, a clearer consensus on the optimal approach should become achievable.
Introduction
The definition of diabetes insipidus (DI) is the inappropriate excretion of a large volume of urine that is hypotonic or dilute. In patients presenting with polyuria (defined as urine volume >40–50 mL/kg body weight per 24 h)1–3 and polydipsia (fluid intake >∼3 L day), DI must be considered when other causes of these symptoms have been excluded (such as hyperglycaemia or hypercalcaemia). There are three main forms of DI: central (or cranial) DI (CDI), nephrogenic DI (NDI) and dipsogenic DI/primary polydipsia (PP). Although true DI is relatively rare affecting perhaps 1 in 25,000, 4 thorough investigation of primary/polydipsia and accurate diagnosis of DI is important as patients with DI may be at risk of becoming severely dehydrated if they are not treated appropriately. Improved quality of life has been demonstrated in patients with CDI receiving appropriate medical therapy.5,6
Clinical biochemistry departments play a vital role in the investigation of DI as the diagnosis is usually based on the results of biochemical investigations. Close liaison with endocrinology departments is often required when dynamic function tests such as the water deprivation test (WDT) or hypertonic saline infusion test (HST) are performed. For example, patients with DI are at risk of becoming severely dehydrated during the WDT so careful biochemical monitoring is required. Interpretation of WDT results can sometimes be challenging and clinical biochemists may be asked for assistance. Clinical biochemistry departments may also be asked for advice regarding initial investigations for patients presenting with polyuria/polydipsia in primary care. It is therefore important that clinical biochemists have a clear understanding of the utility of laboratory testing in the diagnosis of DI and that clinical biochemistry departments can offer an appropriate service for these tests.
A range of diagnostic tests has been utilized in the investigation of DI. This can broadly be divided into indirect and direct tests. Indirect tests involve assessment of appropriate production of and responsiveness to vasopressin (AVP) through determination of the patient’s ability to produce a concentrated urine. Direct tests involve measurement of either AVP itself or a marker of AVP secretion such as copeptin (CT-proAVP). Several prospective studies investigating the optimal approach to the investigation of DI have been published in recent years. The aim of this article is to provide an up-to-date review of the laboratory investigation of DI including a discussion of the evidence supporting the various approaches to diagnosis.
Pathophysiology of diabetes insipidus
Physiology of the vasopressin system
In health, AVP plays a key role in maintaining appropriate fluid balance. The synthesis and release of AVP from the posterior pituitary is regulated by various osmoreceptors and peripheral baroreceptors that detect changes in plasma osmolality and changes in circulating volume, respectively. In response to increasing plasma osmolality and decreasing circulating volume, AVP secretion is increased. There are several AVP receptors distributed in different tissues, but the most important receptors with respect to control of fluid balance are the renal V2 receptors expressed in the principal cells of the collecting ducts. Binding of AVP to the V2 receptors increases expression of aquaporin 2 (AQP2) channels and their incorporation into the apical membrane of the principal cells. The AQP2 channels allow the passive movement of water from the renal filtrate into the cells of the collecting duct along an osmotic gradient and then into the blood stream. This leads to retention of fluid and the production of smaller volumes of more concentrated urine. When an individual is fluid replete, lower plasma osmolality and increased circulating volume triggers the cessation of AVP secretion. This reduces renal AQP2 expression, reducing renal water reabsorption, leading to the production of larger volumes of relatively dilute urine. In health, this system is sensitive and prevents fluid overload or dehydration unless the individual is exposed to excessive fluid intake or an extended period of fluid deprivation. There are numerous specific pathologies that can lead to dysfunction of this system, but they can be classified into the three broader groups of CDI, NDI and PP.
Central diabetes insipidus
Causes of polyuria-polydipsia syndrome.

Nephrogenic diabetes insipidus
As is the case in CDI, NDI has both acquired and inherited causes. NDI is caused by reduced renal sensitivity to the effects of AVP rather than hormone deficiency. Acquired NDI is more common than hereditary and is often relatively mild. Lithium therapy is the most common cause of NDI. A study of 1,105 patients on long-term lithium therapy found that 19% had polyuria (defined as >3 L/day). 13 The mechanism behind lithium induced NDI is thought to be related to inhibition of intra-cellular signalling pathways that control AQP2 expression.14,15 Other drugs that have been reported to induce reversible NDI include demeclocycline, 16 foscarnet 17 and amphotericin B. 18 Hypercalcaemia/hypercalciuria, hypokalaemia and obstructive uropathies can also be associated with transient NDI. The most common cause of inherited NDI is a mutation in AVPR2, leading to X-linked NDI which results in severe AVP insensitivity in affected males. 19 Autosomal dominant NDI can be caused by mutations in AQP2. 20
Primary polydipsia
PP is caused by excessive fluid intake, which leads to appropriate suppression of AVP secretion and production of a large volume of dilute urine. This contrasts with the secondary polydipsia that occurs due to AVP insufficiency or insensitivity in the case of CDI or NDI, respectively. Patients with compulsive water drinking appear to have a normal AVP response to changes in plasma osmolality but exhibit abnormalities in thirst appreciation. 21 PP can occur in chronic schizophrenia where it appears to be related to disruption of hypothalamic function and an abnormal thirst response. 22 Rarely, PP may be associated with structural abnormalities affecting the neurohypophysis such as hypothalamic sarcoidosis or craniopharyngioma. 2
Gestational diabetes insipidus
A presentation resembling CDI can occur during pregnancy due to placental production of the enzyme vasopressinase. 23 Increased circulating vasopressinase leads to increased degradation of AVP, although plasma AVP concentrations often remain normal due to increased AVP secretion.
Diagnosis of diabetes insipidus
Importance of laboratory testing
Reliable identification of the cause of polyuria/polydipsia cannot usually be achieved based on clinical features alone. Patients with CDI, NDI and PP can present with similar complaints of excessive urination (including nocturia) and thirst. Although features in the patient’s history may lead to an increased suspicion of DI (e.g. family history, history of neurosurgery or head trauma, and medications associated with DI), these are not always present. Urine volume, random urine osmolality and serum osmolality or sodium results may also be similar in patients with different underlying pathologies. Diagnosis or exclusion of DI therefore often depends on careful investigation. The diagnosis of DI and discrimination of CDI and NDI is largely dependent on biochemical investigations. Radiological investigations are of limited utility in this respect. A distinct area of intensity known as the pituitary bright spot is usually observed in the posterior pituitary of healthy individuals. This bright spot is thought to reflect stored AVP in neurosecretory granules in the posterior pituitary. Although the pituitary bright spot is often absent in patients with CDI, 24 this is not invariably the case. Studies have shown that the bright spot may be absent in some healthy individuals 25 and conversely, present in some patients with confirmed CDI. 26 The pituitary bright spot may also be absent in some patients with NDI. 27 The presence or absence of this bright spot on pituitary MRI is therefore not a reliable test in the diagnosis of DI and discrimination of CDI/NDI. However, once CDI has been confirmed, pituitary imaging may be useful in determining the underlying pathology by identifying evidence of masses involving the pituitary, infiltrative diseases or an empty sella. Similarly, genetic testing is of limited use in the initial diagnosis or exclusion of DI as there is a wide range of potential primary and secondary causes but may be useful in identifying an underlying inherited cause for CDI or NDI once a biochemical diagnosis has been secured.
There is no single ‘gold standard’ test or criteria for the diagnosis of DI. This can make study of the diagnostic performance of new and existing tests for DI difficult. Recent prospective studies investigating the performance characteristics of laboratory tests for DI have used blinded assessment by independent experts in the field, who based a reference diagnosis on routine biochemistry results, significant features in the patient’s history, non-laboratory tests/findings and response to treatment.28,29
Initial investigations
When a patient presents with complaints of polydipsia/polyuria, there are a number of straightforward investigations that can be performed to determine which specialist investigations are required, if any. It is important to confirm that the patient is producing an excessive amount of urine (rather than suffering from increased frequency for example) by documenting a 24-h urine volume. Polyuria is defined as >50 mL/kg/day in adults and older children, >100 mL/kg/day in children ≤2 years of age and >150 mL/kg/day in neonates.1–4 Once polyuria has been confirmed, common causes such as hyperglycaemia, hypercalcaemia and hypokalaemia should be excluded. Paired serum and urine samples should be taken for measurement of sodium and osmolality, ideally early in the morning (patients without DI are more likely to demonstrate adequate urinary concentration with an early morning urine rather than a random sample). In cases where the random/early morning osmolality is > 750 mOsm/kg, the patient has demonstrated that they can adequately concentrate their urine, which makes a diagnosis of DI unlikely. Conversely, if the patient was evidently dehydrated with a raised serum sodium and/or osmolality and a paired urine that is clearly inappropriately dilute (urine osmolality is lower than serum osmolality for example), the patient is likely to have DI. Further investigations are required to determine if they have CDI or NDI, although this may not be necessary if there is an obvious underlying cause, for example, a history of neurosurgery or a family history of an inherited form of DI.
The water deprivation test (indirect testing)
Typical protocol and interpretation for the water deprivation test.

One of the advantages of the WDT is that it does not require the performance of any specialist biochemistry tests and in theory is relatively straightforward to perform. Combining a period of water deprivation with desmopressin challenge allows the diagnosis or exclusion of DI and discrimination of CDI and NDI during a single diagnostic procedure. There are, however, some practical problems that may be encountered during the performance of the WDT. It is possible for patients with severe DI to become dehydrated very rapidly when denied fluids. Monitoring of serum electrolytes and osmolality as well as body weight is important to prevent severe dehydration (the test should be stopped if body weight drops by more than 5% or the patient develops marked hypernatraemia).1–3 Serum sodium and osmolality should also be measured before the start of the test to ensure that the patient is not dehydrated from the outset. Successful performance of the WDT relies on strict deprivation of fluids during the test. This can be challenging for patients with severe DI or abnormalities in thirst perception as they may suffer from extreme thirst and water craving. Close supervision of such patients during the test is required to ensure that surreptitious drinking does not take place. Monitoring of fluid balance may also be required in the interest of patient safety following administration of desmopressin. 2 Some patients may be hypernatraemic at the end of the period of water deprivation. If large volumes of fluids are consumed after the period of water deprivation has ended and desmopressin has been administered, there is the potential for a rapid decrease in serum sodium concentrations, which can be dangerous. In a study that documented side-effects and adverse events in 141 patients undergoing WDT, 98% experienced thirst, 39% vertigo, 60% headache, 36% nausea and 59% malaise. 29 Symptomatic hypernatraemia was experienced in 1 case, headache severe enough to require pain medication occurred in 1 case, shivering in 1 case and hyponatraemia due to excess water retention after administration of desmopressin occurred in 4 cases with 1 of these cases requiring hospitalization.
Although the WDT is widely regarded as the standard diagnostic test for the investigation of DI, in practice the test has some significant diagnostic limitations. A detailed discussion of published studies investigating the diagnostic performance of the WDT and other tests can be found later in this article, but it is widely understood that results which are difficult to interpret or equivocal are quite common.2,3,33,34 In cases of complete (severe) CDI or complete NDI, the results from the WDT and desmopressin challenge are usually straightforward. Patients with severe DI are often dehydrated and therefore hypernatraemic/hyperosmolar at the end of the period of water deprivation. If urine osmolality is obviously inappropriately low (less than around 300 mOsm/kg) in this context, this is strong evidence of DI. When there is then an obvious response to administration of desmopressin (an increase of urine osmolality of 50% or more), the underlying cause is clearly a central pathology. Conversely, if there is a complete lack of an increase in urine osmolality following desmopressin, the underlying cause is obviously nephrogenic. There are also cases where DI can be confidently excluded following WDT. Where the patient demonstrates an ability to maximally concentrate the urine (urine osmolality above 750 mOsm/kg) during the period of water deprivation, DI can be confidently excluded. Difficulties in the interpretation of the WDT arise when peak urine osmolality lies in the range 300–750 mOsm/kg so that the urine is not unequivocally inappropriately dilute but also not maximally concentrated. In this situation it may be difficult to distinguish less severe cases of CDI or NDI and primary polydipsia.35,36 The range of maximum urine osmolality observed during WDT overlaps between patients with partial DI and a normal ability to produce AVP.
31
This overlap of post-WDT urine osmolality concentrations between partial DI and primary polydipsia may be in part due to impairment of the renal interstitial solute gradient following prolonged polydipsia, which can lead to an impaired ability to maximally concentrate the urine (effectively due to partial resistance to the anti-diuretic action of AVP).2,33 A diagram illustrating this potential overlap of WDT results between partial CDI/NDI and primary polydipsia is shown in Figure 1. This problem of equivocal results in the WDT has the potential to lead to misclassification of some patients as demonstrated by studies that have identified discrepancies in diagnosis between WDT and direct tests for DI.29,32,35 A diagram illustrating typical changes in urine osmolality during WDT followed by desmopressin challenge in complete nephrogenic DI (cNDI), complete central DI (cCDI), partial nephrogenic DI (pNDI), partial central DI (pCDI), primary polydipsia with impaired renal osmotic gradient (PP) and normal secretion of and responsiveness to AVP (NORMAL).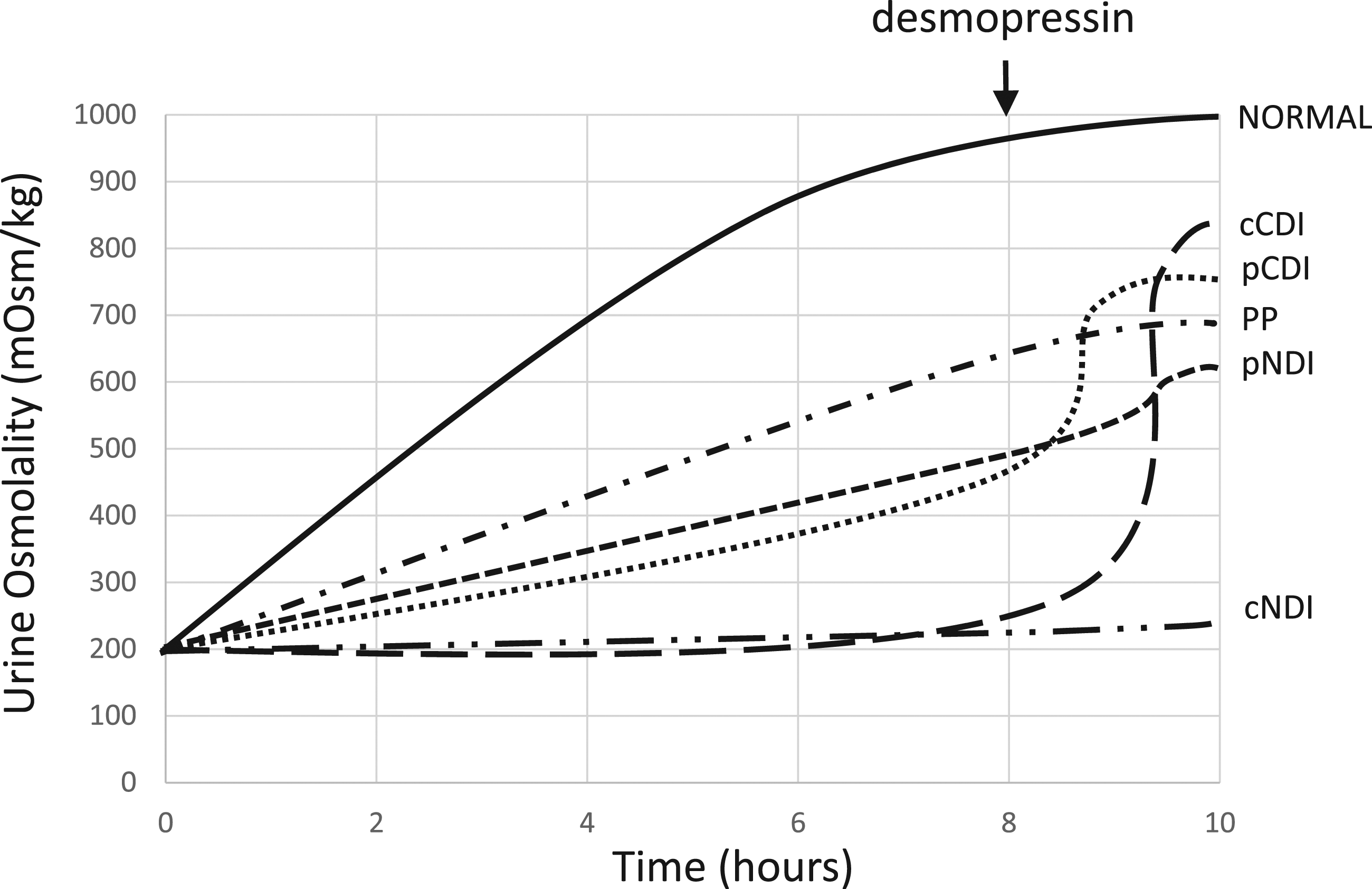
Direct measurement of vasopressin
An alternative to the indirect WDT is direct measurement of plasma AVP under osmotic stimulation. It is necessary to measure plasma AVP when patient serum osmolality/sodium is elevated as there is overlap in plasma AVP concentrations between healthy individuals and patients with CDI in the context of euvolaemia with normal serum osmolality. Therefore, plasma AVP has typically been measured during hypertonic saline infusion, which produces a progressive rise in serum osmolality and sodium. In healthy individuals, rising serum osmolality stimulates secretion of AVP so that it is possible to discriminate appropriate and subnormal AVP production at higher serum osmolality (Figure 2). Patients with primary polydipsia or NDI have plasma AVP responses to increasing serum osmolality that are appropriate and comparable to healthy individuals32,35,37–39 (patients with NDI may display an exaggerated AVP response as a result of AVP resistance). Those with central DI have subnormal concentrations of AVP at higher serum osmolality compared to healthy individuals. AVP concentrations provide an indication of the severity of the defect in AVP secretion, with severe cases associated with very low or undetectable AVP throughout hypertonic saline infusion and milder cases associated with a detectable but blunted AVP response.38,39 The theoretical advantage of the measurement of AVP under osmotic stimulation compared to WDT is that the hormone of interest is being measured, so that there is direct evidence of the ability or inability to secrete appropriate concentrations of AVP. In the WDT, a failure to maximally concentrate the urine is assumed to be due to a lack of AVP activity when other factors can also explain a failure of urinary concentration, such as impairment of the renal interstitial solute gradient (secondary to chronic polydipsia) or an insufficient degree of dehydration to stimulate maximal urinary concentration by the end of the WDT. Expected plasma AVP response to increasing plasma osmolality in healthy individuals/patients with primary polydipsia and patients with central diabetes insipidus (Thompson 1986, Baylis 1980, Baylis 1977).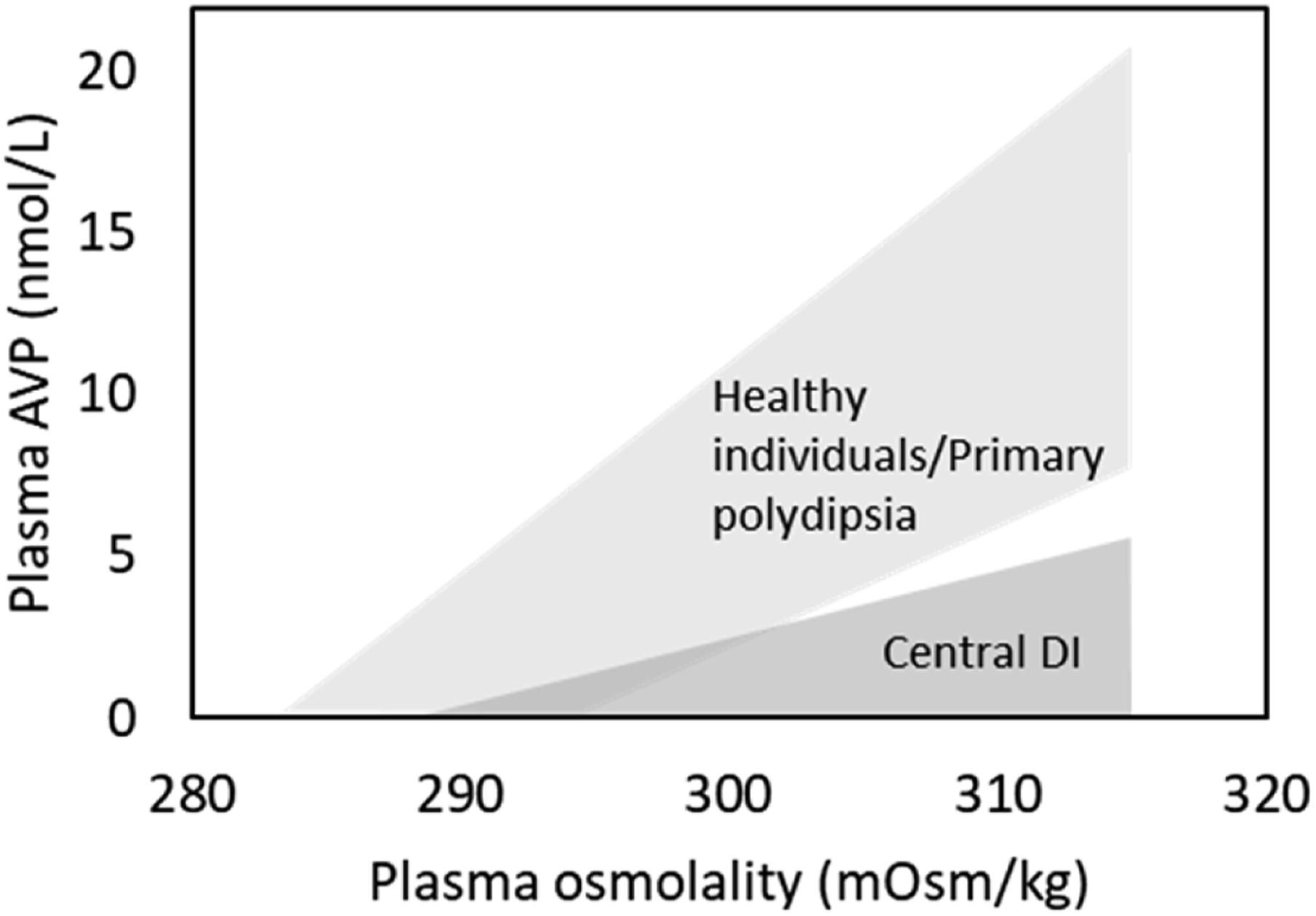
One of the practical disadvantages of direct measurement of AVP is that hypertonic saline infusion is typically required to allow assessment of the response of plasma AVP to an increased plasma osmolality. Infusion of hypertonic saline is an invasive procedure and carries the risk of excessive hypernatraemia if not performed carefully (target serum sodium/osmolality is typically ∼150 mmol/L or 305–310 mOsm/kg, respectively). It is usually recommended that patients are monitored post saline infusion, with serum sodium often checked 1 h after the procedure to ensure sodium has returned to normal. Side effects are common. In a study of 141 patients undergoing a diagnostic hypertonic saline procedure for the investigation of DI, 100% experienced thirst, 68% vertigo, 67% headache, 50% nausea and 69% malaise. 29 More serious adverse events were symptomatic hypernatraemia in 2 cases, shivering in 3 cases, diarrhoea in 1 case and prolonged hypernatraemia after saline infusion in 2 cases. All subjects in this study underwent separate WDT and hypertonic saline infusion and interestingly, 62% of subjects indicated a preference for hypertonic saline infusion rather than WDT. In the study, the mean duration of the WDT was 20 h (fluid restriction commenced at midnight in patients with a low degree of suspicion for DI) while this was only 3.1 h for hypertonic saline infusion, which may be a significant factor influencing patient acceptability of the tests.
A further difficulty with diagnostic tests relying on direct measurement of plasma AVP is that it is analytically challenging. Regardless of the analytical methodology used to measure AVP, there are pre-analytical factors that can strongly influence measured AVP concentrations. AVP is relatively unstable ex vivo, particularly in whole blood. In whole blood, AVP concentrations increase significantly within 2 h at 25°C and within 6 h at 4°C,37,40 so rapid centrifugation is required following sample collection. Stability in plasma is improved at up to 24 h at 4°C. Although AVP is stable for up to 1 month at −20°C (4 months at −80°C), repeated freeze-thaws have a deleterious effect. 40 A large proportion of circulating AVP is bound to platelets via V1 receptor binding. 41 Centrifugation speed has been demonstrated to strongly influence plasma AVP measurements.42,43 A study investigating the influence of the relative centrifugal force on AVP concentrations found that AVP concentrations were 81% lower at 2000 g relative to 800 g, 40 demonstrating the importance of applying an appropriate and consistent centrifugation method to blood samples for AVP measurement. Measurement of AVP has largely been performed using radioimmunoassay (RIA) methods,37,44–49 although ELISA methods have also been developed. 50 Accurate measurement of AVP by immunoassay is challenging due to the significant problem of interference from other plasma proteins or other constituents of plasma. 44 An extraction step is therefore required prior to immunoassay of AVP. Various extraction methods have been used including C18 or Sephadex column chromatography and solvent extraction. These methods are therefore quite cumbersome and technically demanding and so have only been performed in a small number of specialist centres. More recently, LC–MS/MS methods for AVP have been developed, which may have some advantages compared to RIA51,52 including the addition of an internal standard, which accounts for incomplete recovery of AVP during extraction. These assays do not yet appear to have been widely applied in a clinical setting, however.
Measurement of copeptin
Relatively recently copeptin has been used as an alternative marker of AVP secretion. Commercial immunoassays are available.53,54 Copeptin, a 39-amino acid peptide, 55 is the C-terminal portion of the prohormone proAVP and is secreted from the posterior pituitary alongside AVP on an equimolar basis. Studies of serum/plasma copeptin during hypertonic saline infusion and water loading have shown that circulating copeptin concentration is correlated to plasma osmolality in a similar fashion to AVP itself.29,56,57 Measurement of copeptin has therefore been investigated as an alternative to measurement of AVP in direct testing for DI.28,29,36 As is the case with AVP, measurements of copeptin are more likely to be diagnostic when they are made under osmotic stimulation such as during hypertonic saline infusion.28,29,36 Two prospective studies investigating the diagnostic performance of copeptin in the investigation of DI have identified an optimal diagnostic threshold of >4.9 pmol/L for exclusion of CDI, when serum sodium is at least 150 mmol/L.28,29 Conversely, patients with NDI included in these studies have relatively high basal copeptin compared to patients with PP or CDI. 28 Other studies have investigated alternative methods of stimulating copeptin secretion for diagnostic purposes, for example, using arginine 58 or insulin infusion. 59 A more detailed discussion of the evidence for the diagnostic performance of these different tests is included later in this article.
The practical advantages of using copeptin as a marker of vasopressin secretion rather than AVP itself are more favourable stability in whole blood and serum/plasma and the availability of commercial copeptin immunoassays, which are more practical for use in routine clinical laboratories than available AVP assays. While AVP has limited stability in whole blood and plasma at room temperature, copeptin is stable in whole blood for at least 24 h at 25°C or 4°C 40 and in serum or plasma for up to 7 days at 25°C or 4°C. 53 Repeated freeze/thaw cycles have a much smaller impact on copeptin concentrations compared to AVP, with up to 4 cycles having no significant effect. 40 Copeptin is not bound to platelets as is the case with AVP and so variations in centrifugal force do not affect measured copeptin concentrations (using a relative centrifugal force of 800, 2000 or 5000 g has no significant impact on copeptin concentrations). 40 This higher degree of stability for copeptin means that the more stringent sample collection and handling requirements that apply for AVP are not necessary for copeptin. Measurement of copeptin is also more straightforward than AVP as commercial CE marked immunoassays are available. BRAHMS have marketed two copeptin assays; a coated-tube immunoluminometric assay 53 (LIA) and an automated immunofluorescent immunoassay on the Kryptor platform. Non-CE marked or ‘research only’ ELISAs are also available. It seems that while the BRAHMS LIA and Kryptor assays appear to give similar results, at least one of the ELISA kits on the market yields significantly different results compared to the BRAHMS assays. 54 There is currently no established reference preparation available for copeptin. Most of the recent published studies investigating the diagnostic performance of copeptin in the investigation of DI have been based on results produced using the BRAHMS Kryptor assay.28,29,58
Evidence for the diagnostic performance of tests for DI
Summary of studies investigating the diagnostic performance of indirect and direct tests for diabetes insipidus.
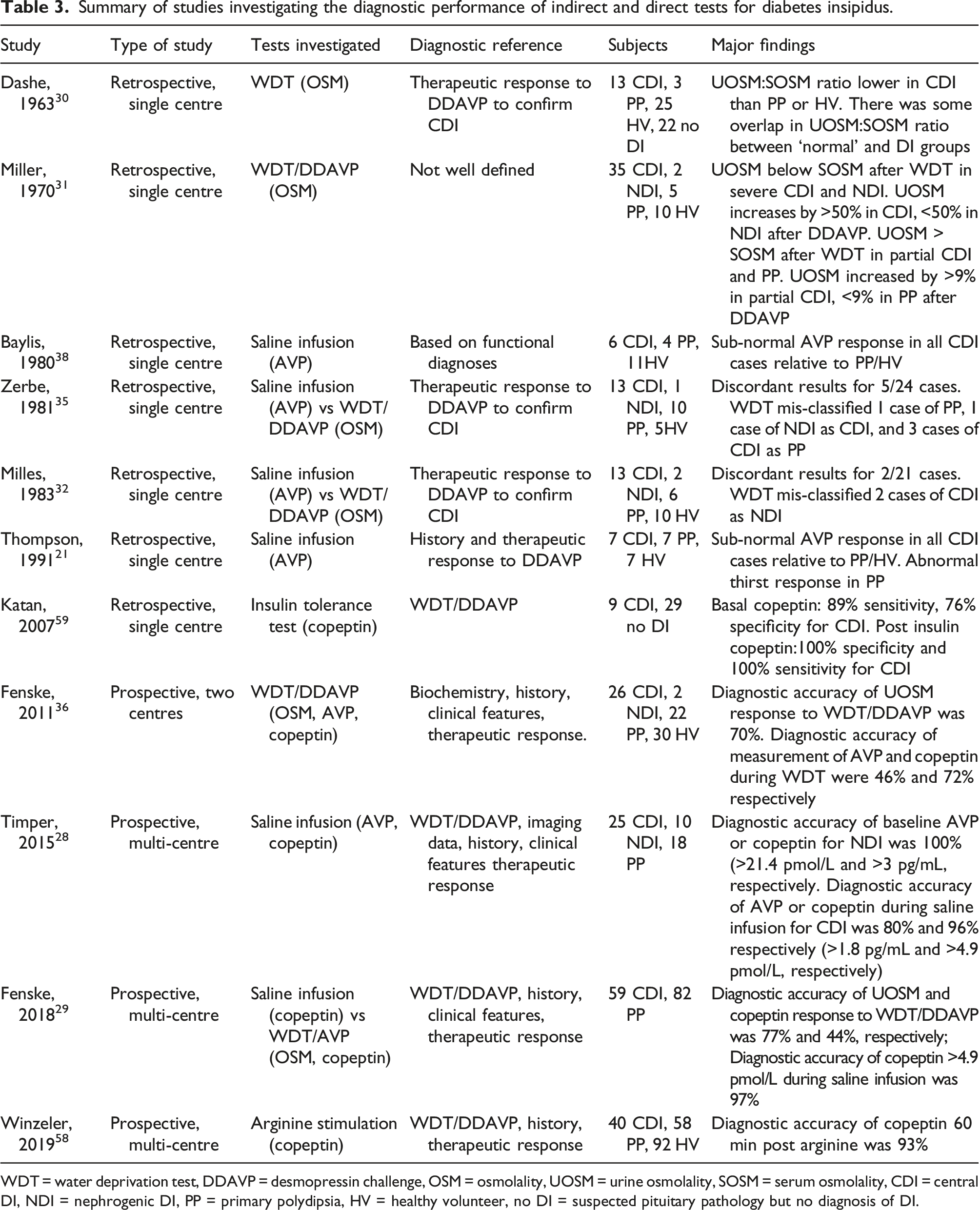
WDT = water deprivation test, DDAVP = desmopressin challenge, OSM = osmolality, UOSM = urine osmolality, SOSM = serum osmolality, CDI = central DI, NDI = nephrogenic DI, PP = primary polydipsia, HV = healthy volunteer, no DI = suspected pituitary pathology but no diagnosis of DI.
The lack of an agreed ‘gold standard’ for the diagnosis of CDI or NDI is a significant problem when defining the diagnostic performance of any test for DI. There is no single test or set of clinical criteria that provide a definitive diagnostic reference. Some of the studies detailed in Table 3 do not provide well defined criteria for classifying the participants as CDI, NDI or PP (or indeed complete/severe or partial/mild DI). More recent multi-centre, prospective studies have attempted to standardize the classification of the participants using a combination of biochemical data, patient history, clinical features and follow up to determine response to therapy with the final diagnosis being decided by experts in the field.28,29,36,58 This is not a perfect diagnostic reference for studies of diagnostic performance, as these criteria are still subject to a certain degree of subjectivity. However, these criteria may be the best achievable diagnostic reference in the absence of definitive biochemical, imaging, histological or genetic tests for CDI or NDI. Despite these difficulties, a number of studies published in recent years have significantly improved the body of evidence defining the diagnostic performance of biochemical tests for DI. The studies published from 1960 to 2000 were all relatively small and some did not characterize their subjects with rigorous diagnostic reference criteria. Despite the relatively poor evidence defining the diagnostic performance of WDT/DDAVP, this has become the most widely applied test used in the investigation of polyuria/polydipsia and the diagnosis of DI. Some authors have argued that direct measurement of AVP during hypertonic saline infusion leads to a more accurate diagnosis than the indirect WDT/DDAVP test, but the published evidence to support this view is also limited. However, some more recent published evidence is based on prospective, multi-centre studies that have recruited a larger number of subjects.28,29,58 These more recent larger studies have generally focused on investigating the role of copeptin in the diagnosis of DI and the results have supported the measurement of copeptin under an osmotic stimulus (achieved by hypertonic saline infusion) or other stimulus (such as arginine infusion). Data from these studies suggest that measurement of copeptin during these stimulation tests can provide a high degree of diagnostic accuracy.
Diagnostic testing for DI in paediatrics
The studies of the diagnostic tests for DI described above and in Table 3 are based on adult subjects. The evidence for the performance of these tests in infants and children is limited. Most of the published information on the biochemical investigation of DI in paediatrics is based on case reports and retrospective analysis of data from cohorts of patients with a previous diagnosis of DI. Some recent small studies have investigated the utility of copeptin in paediatrics. Tulli et al. 60 measured copeptin in 53 children following WDT and found that this distinguished CDI from PP with a diagnostics sensitivity of 75% and specificity of 83.3% (only 6 patients with a diagnosis of CDI were included in the study). In another study, Bonnet et al. 61 measured copeptin in the morning following an overnight fast in 278 children (aged 2 months to 18 years) which included 40 patients investigated for polyuria/polydipsia (21 had CDI, 3 NDI and 16 PP). In this study fasted copeptin concentrations <3.5 pmol/L were diagnostic of CDI with 100% sensitivity and 87.4% specificity. There is a lack of published data on the measurement of AVP or copeptin under osmotic stimulus, although one article demonstrated the potential utility of AVP during saline infusion in a series of 5 paediatric cases 62 and the validation population for a study of copeptin during arginine infusion included some paediatric subjects. 58
Diagnosis of severe DI in paediatrics is often not particularly challenging. When the patient becomes spontaneously dehydrated and markedly hypernatraemic while producing a clearly inappropriately dilute urine, a diagnosis of DI can often easily be made. In cases where the underlying aetiology is not clear, measurement of copeptin may potentially be useful. Two cases studies have reported very high copeptin concentrations in infants with NDI.63,64 These cases are consistent with local experience in Newcastle upon Tyne Hospitals, where we have seen several cases of severe CDI in children with inappropriately low copeptin concentrations (usually <5 pmol/L) and several cases of severe NDI associated with high copeptin concentrations, typically greater than 100 pmol/L (unpublished data). In cases of milder DI, where differentiation from PP is more difficult, there is currently little evidence from prospective studies that the approaches to biochemical diagnosis applied in adults can be transferred to paediatrics, although WDT is widely used in practice. Some diagnostic procedures such as hypertonic saline infusion may be more challenging to administer, particularly in infants and younger children.
Current status of diagnostics for DI and future perspectives
As described above, the principal tests that have been investigated as diagnostic tests for DI are the indirect WDT, measurement of AVP under hypertonic saline infusion and measurement of copeptin (also under hypertonic saline infusion, with emerging evidence on arginine infusion). All these tests are relatively cumbersome to perform so it is important to ensure all the basic initial investigations are performed before progressing to these more complex diagnostic procedures. Investigations that should be completed before proceeding to more formal testing include measurement of 24-h urine volume to confirm polyuria, determination of U + E (including potassium), calcium and glucose to rule out other common causes of polyuria and measurement of osmolality on paired serum and urine samples (ideally on early morning samples). Consistently performing these investigations in patients complaining of polyuria/polydipsia will obviate the requirement for further investigation in some cases, for example, those without true polyuria, those demonstrating adequate urinary concentration with a morning sample and patients with unequivocal evidence of DI based on paired morning serum/urine osmolality. For those patients requiring further investigation to rule out or diagnose DI, the clinician can choose between indirect and direct testing. The indirect WDT has been the most widely used test in clinical practice, but recent studies have suggested that the diagnostic accuracy of the WDT is limited. Direct testing can be performed by measuring AVP itself or by using copeptin as a surrogate marker. AVP/copeptin should be measured under osmotic stimulus (or potentially arginine stimulus as indicated in a recent study) to achieve a high degree of diagnostic performance. Measurement of copeptin is more straightforward, practical and less susceptible to pre-analytical factors than AVP so is likely to become preferred to measurement of AVP itself. A number of reviews on the topic of the diagnosis of DI have been published in recent years. Some of these reviews have suggested that direct testing is preferable to indirect WDT and increasingly suggest that measurement of copeptin (under appropriate conditions) could become the recommended test for diagnosis of DI.33,34,65 It is difficult however to find systematic, evidence-based guidelines on this subject. This is likely due to the relatively modest amount of evidence supporting any of the approaches to the diagnosis of DI, although this situation is improving with further prospective trials underway including a multi-centre prospective study comparing the diagnostic performance of copeptin measurement during hypertonic saline infusion with copeptin measurement during arginine infusion. 66 As more well-designed studies are published, a clearer consensus on the optimal approach to the diagnosis of DI should become achievable.
Footnotes
Acknowledgements
This article was prepared at the invitation of the Clinical Sciences Reviews Committee of the Association for Clinical Biochemistry and Laboratory Medicine.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
CB.
Contributorship
CB wrote the article.
