Abstract
Background
Methotrexate is used in high doses to treat a number of cancers, particularly certain haematological malignancies. Monitoring of serum methotrexate concentration is important due to the potential toxicity of methotrexate and the variation in methotrexate pharmacokinetics in different patients on the same treatment regimen.
Objective
To develop a rapid liquid chromatography-tandem mass spectrometry (LC-MS/MS) method for monitoring serum methotrexate in patients on high-dose chemotherapy.
Method
Isotopically labelled internal standard was added to sample prior to protein precipitation with methanol. Diluted supernatant was injected into a Waters Acquity UPLC system linked to a TQS-Micro mass spectrometer. Separation by chromatography was achieved with a Waters Phenyl Vanguard with a retention time of approximately 0.5 min. The quantifier and qualifier transitions for methotrexate were 455.2>134.1 and 455.2>175.2, respectively.
Results
Mean recovery was 111% for three different concentrations of methotrexate spiked into seven different patient samples, with ion suppression <1%. Between-batch and within-batch coefficient of variations were <5% at three different concentrations of methotrexate in fresh frozen plasma. The lower limit of quantification was 0.02 µmol/L and the assay was shown to be linear to approximately 25 µmol/L. The LC-MS/MS assay showed a mean bias of –8.6% compared to an immunoassay, while mean bias compared to weighed in targets in external quality assessment samples was 1.6%.
Discussion
A rapid LC-MS/MS assay for methotrexate has been developed and validated. The LC-MS/MS method is likely to offer superior accuracy and specificity to more widely available immunoassays.
Introduction
Methotrexate is a therapeutic agent that inhibits folate metabolism by inhibiting the enzyme dihydrofolate reductase (DHFR), resulting in blockage of DNA synthesis and cell division. 1 High-dose methotrexate (HDMTX) therapy is defined as an intravenous dose of ≥500 mg/m2; such therapy is used for the treatment of a number of cancers including acute lymphoblastic leukaemia (ALL), osteosarcoma and lymphomas. 1 HDMTX can lead to toxicity, particularly acute kidney injury (AKI) due to crystal nephropathy when methotrexate and its metabolites crystallize in the renal tubules. 2
Measurement of methotrexate in patients receiving HDMTX is important due to the risk of toxicity and wide variations in clearance between different patients receiving the same dose. 1 It has been shown that serum methotrexate concentrations of greater than 10 µmol/L at 24 h post infusion are associated with a high risk of toxicity. 3 Consequently, it is important to measure methotrexate in serum samples from patients on HDMTX therapy to detect possible delays in its clearance. Furthermore, it is recommended that serum methotrexate is monitored daily after HDMTX therapy, until the concentration reaches less than 0.05–0.1 µmol/L, at which point leucovorin rescue therapy can be discontinued. 4
An important aspect of supportive treatment for patients on HDMTX is the drug leucovorin, which helps prevent methotrexate toxicity, with the specific regimen dependent on the condition being treated. 1 Leucovorin counteracts methotrexate toxicity by providing a supply of the tetrahydrofolates, the production of which is blocked by methotrexate. 5 Since leucovorin counteracts the effects of methotrexate, it must not be given too early following HDMTX therapy so as not to diminish the efficacy of treatment. Therefore, measurement of serum methotrexate is also important in guiding leucovorin therapy.
Glucarpidase (carboxypeptidase G2) therapy is recommended when clearance of methotrexate is delayed, with the exact serum concentration threshold for intervention depending on the particular HDMTX dosing regimen. 5 Glucarpidase is a recombinant bacterial enzyme which inactivates methotrexate by cleaving it into two non-toxic metabolites: deoxyaminopteroic acid (DAMPA) and 7-hydroxymethotrexate. These metabolites interfere in the commonly used methotrexate immunoassays, which means they can give inaccurate and potentially misleading results. 6 A case study highlighted the adverse effects on patient management that can be caused by interference in methotrexate immunoassays when glucarpidase rescue therapy is initiated and methotrexate cannot be reliably quantified due to interference from DAMPA. 7 It is therefore necessary to measure methotrexate by a more specific method, such as liquid chromatography-tandem mass spectrometry (LC-MS/MS), in patients on glucarpidase therapy. 5
Several LC-MS/MS assays have been developed in recent years for monitoring methotrexate in patients receiving HDMTX.8–11 Typically these assays use 100 µL sample with extraction by protein precipitation. We understand that currently there is no clinically validated LC-MS/MS assay in the UK for methotrexate available for use. Therefore, such an assay is needed, particularly for patients receiving glucarpidase rescue treatment following HDMTX therapy.
Materials and methods
Calibrators and internal quality control material
Calibrators and quality control material were prepared from independent certified reference material (CRM). Stock calibrator (Cerilliant®, Texas, USA) and QC (quality control) material (Sigma, Dorset, UK) were both made up at 1 mg/mL in methanol (LC-MS grade, Honeywell, New Jersey, USA) containing 0.1 mol/L sodium hydroxide. Working calibrator and QC material were prepared in fresh frozen plasma. Calibrators ranged in concentration from 0 to 10 µmol/L (
Sample preparation
Calibrator, QC material and patient samples (40 µL) were each manually pipetted into separate wells of a 96-well plate (Porvair Sciences Ltd, Leatherhead, UK) using a positive displacement pipette (Gilson, MicroMan®, Middleton, UK); internal standard (25 µL) was then added to each well, followed by 400 µL of methanol. The plate was heat sealed and vortexed for 1 min (Grant Instruments MPS-1, Cambridge, UK) and then centrifuged for 5 min at 8800
Chromatography
An Acquity® ultra-performance liquid chromatography I-class system (Waters, Manchester, UK) was used to achieve chromatographic separation. The extracted sample (3 µL) was injected into a Waters Phenyl Vanguard (inner diameter, 2.1 mm; particle size, 1.7 µm) with a flow rate of 0.5 mL/min. Mobile phases were deionized water (A) and methanol (B), each containing 0.1% formic acid (BDH, Bristol, UK) and 2 mmol/L ammonium acetate (Sigma, Poole, UK). Starting conditions were 15% B which increased linearly to 30% B over 0.5 min. The composition then changed to 100% B for 0.3 min before returning to starting conditions. Total run time injection-to-injection was 1.5 min.
Mass spectrometry
Following chromatographic separation, the eluate was directed into a Waters XEVO® TQS Micro tandem mass spectrometer in positive electrospray ionization mode. The system was controlled by MassLynx NT 4.1 software and the data was processed using the TargetLynx program.
Validation
The validation was carried out in accordance with published recommendations and acceptability criteria from the Food and Drug Administration. 12
Accuracy
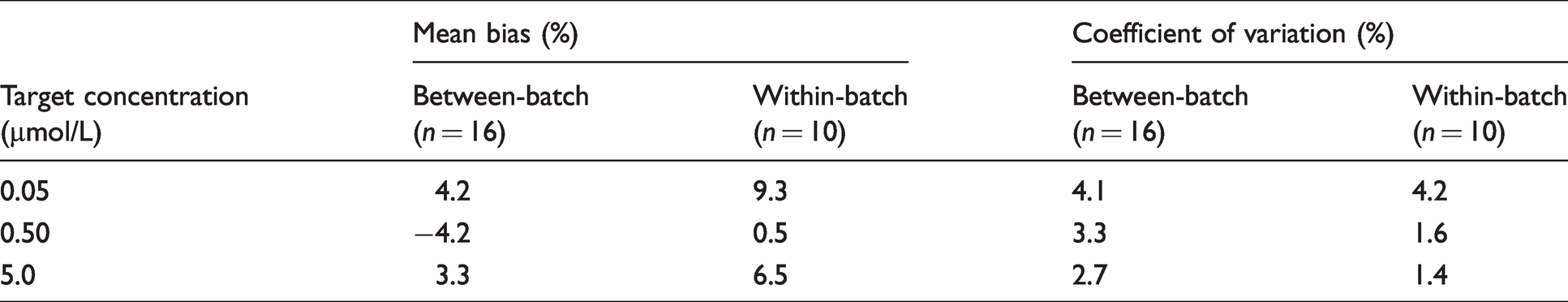
Bias was assessed using QC material which was prepared gravimetrically as described above. Mean bias <15% of the weighed-in value for both within-batch and between-batch analysis (
Imprecision
The internal quality control (IQC) material described above was also used to assess imprecision. Within-batch imprecision was determined by analysing each level of IQC 10 times on the same batch, while between-batch imprecision was determined by analysing each sample on 10 consecutive batches. Acceptance criteria was a coefficient of variation (CV) <15% at all levels of IQC.
Recovery
Three different concentrations of methotrexate were spiked into seven separate patient serum samples prior to analysis using methanolic solutions prepared from Cerilliant® CRM material to create samples with concentrations of 0.05, 0.5 and 5.0 µmol/L. Recovery of 100±20% was considered acceptable.
Matrix effects
To assess matrix effects, three different concentrations of methotrexate were spiked into seven patient samples post-extraction to create samples with the same concentrations as those used for the recovery experiments described above. Additionally, calibration material (fresh frozen plasma) was spiked with the same concentrations of methotrexate. The matrix effect was assessed by calculating the peak heights in the patient samples as a percentage of those in the corresponding calibrator material. Matrix effects of 100±15% were considered acceptable.
Linearity
Linearity was assessed by spiking calibration material with a supra-pharmacological concentration of methotrexate (approximately 10 times greater than the top standard). This was then doubly diluted four times. The five samples created were then analysed alongside a standard curve. The assay was considered linear up to the highest concentration at which the diluted samples had a measured concentration within 10% of the weighed-in concentration.
Lower limit of quantification
To determine the lower limit of quantification (LLOQ), QC material was gravimetrically diluted with fresh frozen plasma to create samples with a known low concentration of methotrexate. The LLOQ acceptability criteria were a CV and bias <20% for 10 samples analysed on the same batch (within batch) and analysis on 10 consecutive batches (between batch).
Carryover
To assess carryover, calibrator material was spiked with a supra-pharmacological concentration of methotrexate as for the linearity experiment described above. This sample was analysed followed by a blank sample of calibrator only material. Carryover was deemed acceptable if it was <20% of the LLOQ.
Analytical interference
Patient serum samples were spiked with three different concentrations of methotrexate spread across the analytical range of the assay. Known concentrations of haemolysate, bilirubin and intralipid were then added to separate aliquots of these samples to create grossly haemolysed, icteric and lipaemic samples, respectively; appropriate blanks were also set up. The spiked samples and blanks were then analysed in the same batch with acceptability criteria for no significant interference being less than 10% difference between the blanks and the spiked samples.
Method comparison
Patient samples (
Results
Following extraction and LC-MS/MS analysis, methotrexate and d3-methotrexate were found to elute at 0.48 and 0.47 min, respectively (Figure 1) with no interfering peaks present. Mean bias and CV for IQC material was <10% and <5%, respectively, at all concentrations (Table 1). The LLOQ was determined to be 0.02 µmol/L based on a mean CV and bias <20% at this concentration. When seven patient samples were spiked with three different concentrations of methotrexate before extraction, mean recovery was 111.0% (range: 103.8–116.8%). Post-extraction spiking with the same concentrations of methotrexate in seven patient samples showed a mean matrix effect of 0.2% (range: –2.6% to 11.0%). Linearity experiments suggested the assay was linear to approximately 25 µmol/L.
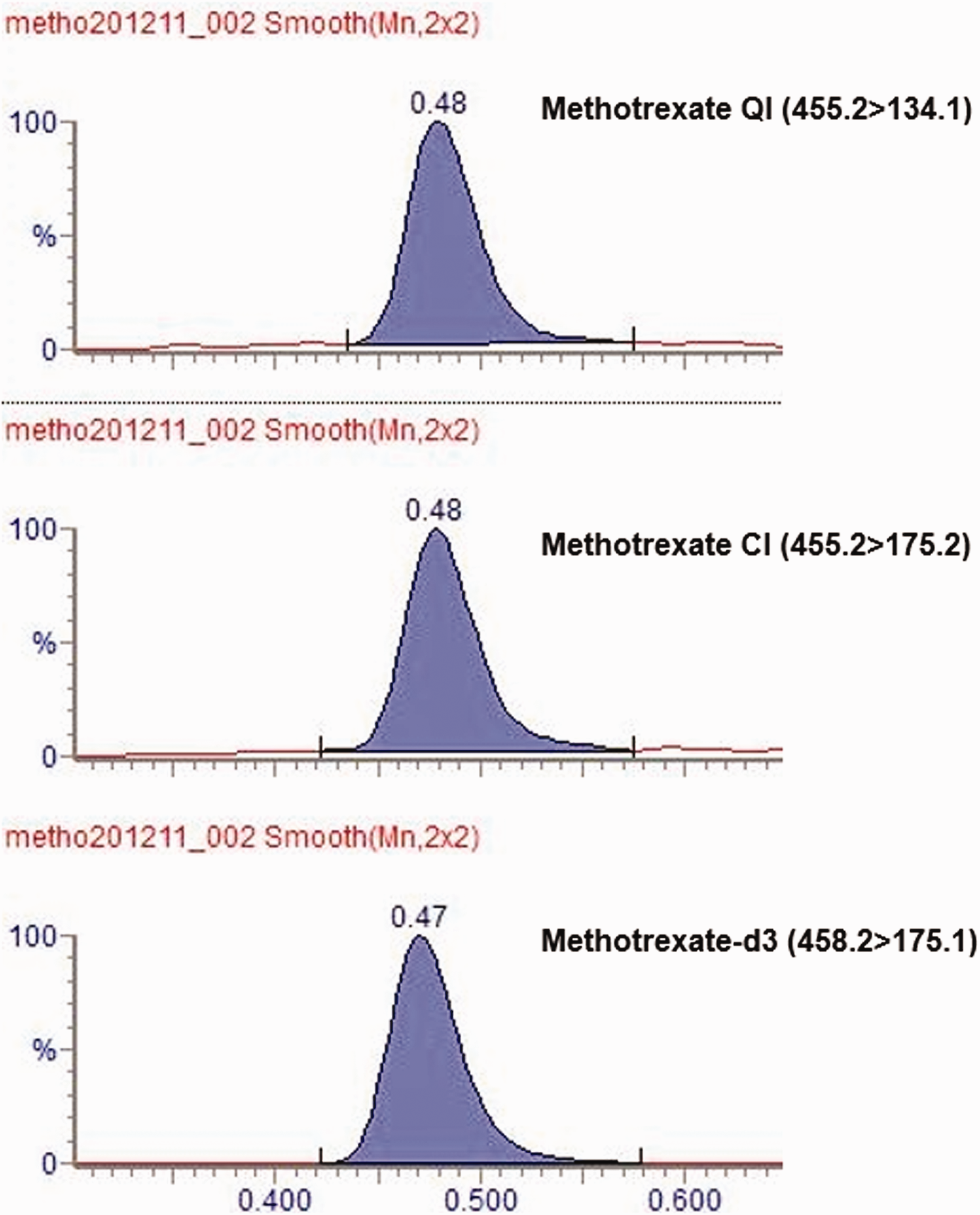
A typical chromatogram of a sample which has been analysed by the LC-MS/MS methotrexate method showing the quantifier ion (QI), confirmatory ion (CI) and methotrexate-d3 internal standard.
Results of bias and imprecision studies in IQC material.
A method comparison of the LC-MS/MS method to a UKAS-accredited immunoassay method was performed using 45 patient samples. A Shapiro-Wilk test showed that both immunoassay and LC-MS/MS results had a non-Gaussian distribution (p<0.0001). Passing-Bablok analysis showed a linear relationship between the two methods (Figure 2) with an equation of LC-MS/MS methotrexate = immunoassay methotrexate – 0.01. Mean bias of the LC-MS/MS method compared to the immunoassay method was –8.7% (Figure 3). Mean bias of the LC-MS/MS method compared to weighed-in targets in UKNEQAS samples (
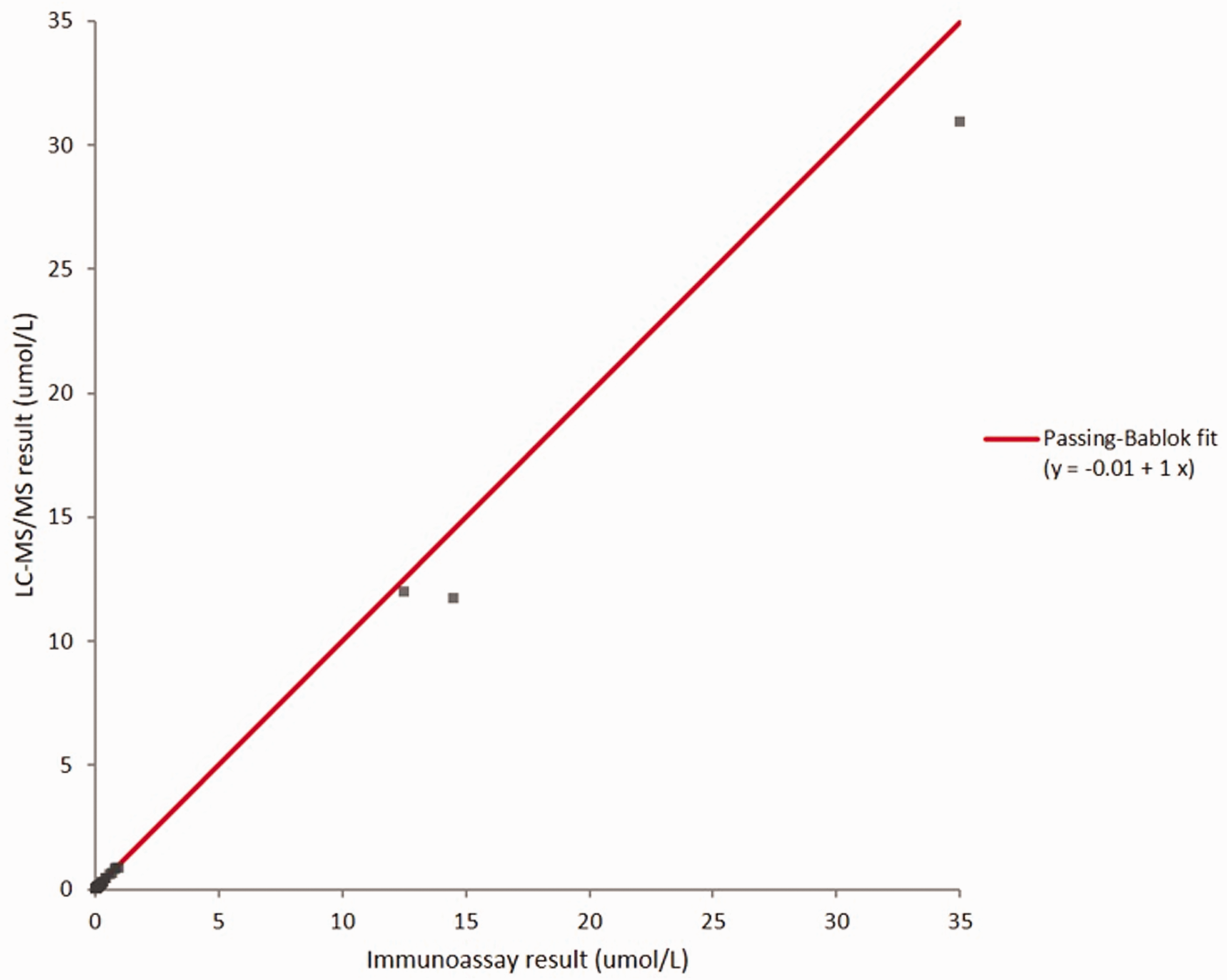
Passing-Bablok linear regression plot of the LC-MS/MS method compared to the established immunoassay method.
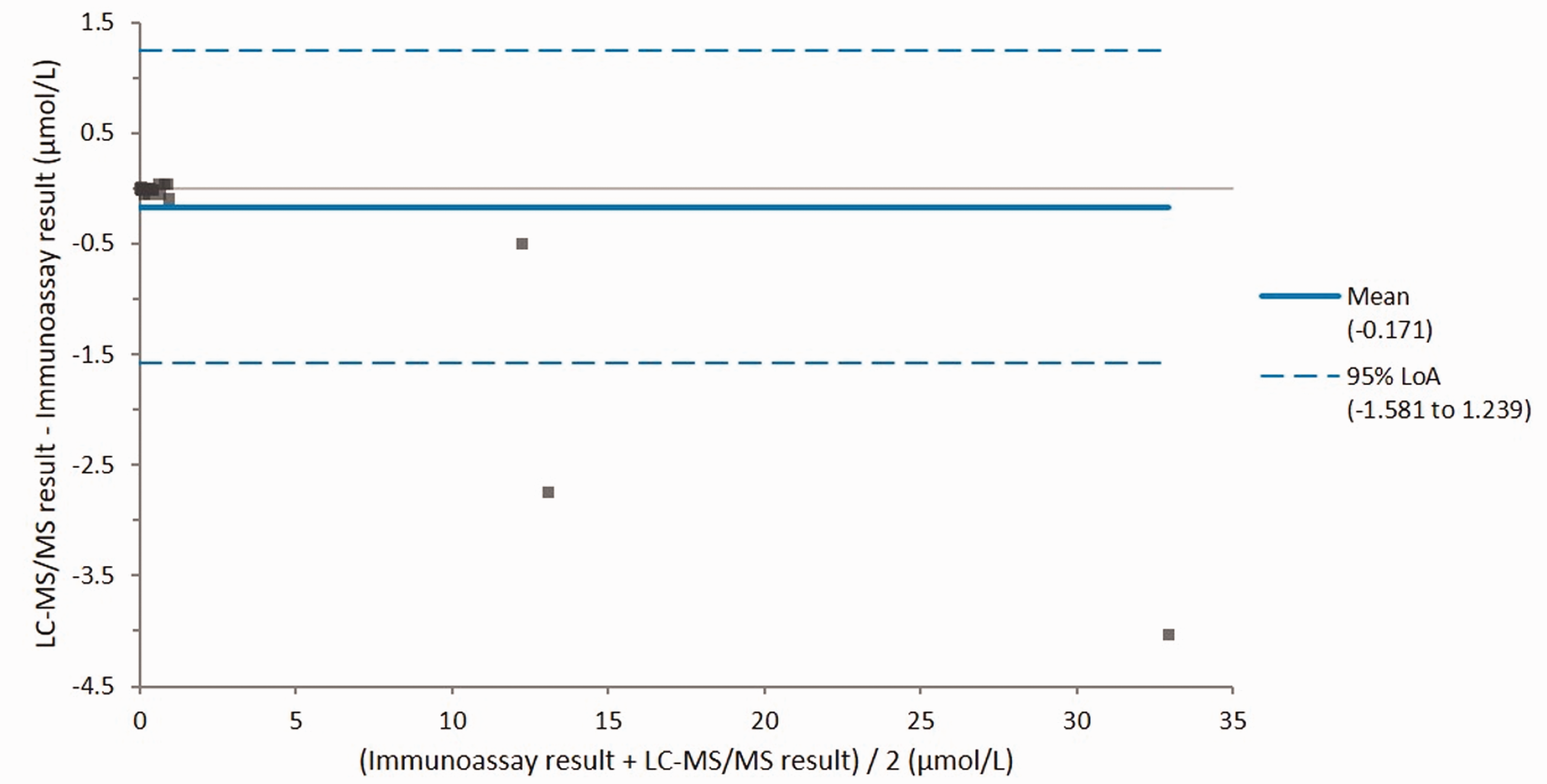
Altman-Bland difference plot of the LC-MS/MS methotrexate method compared to the immunoassay method.
Discussion
We have developed and validated a rapid LC-MS/MS method for methotrexate which is suitable for routine use. The assay showed a slight negative bias compared to a UKAS-accredited immunoassay, but there was minimal bias compared to weighed-in targets in UKNEQAS samples, suggesting that the LC-MS/MS method is likely be more accurate than the immunoassay.
These findings are consistent with publications that have shown immunoassays are subject to interference from methotrexate metabolites, 7-hydroxymethotrexate and DAMPA. 6 , 7 Interference in methotrexate measurement is particularly important in patients on glucarpidase rescue therapy as this drug cleaves methotrexate into its inactive metabolites.5–7 Immunoassays for measuring methotrexate are unsuitable for monitoring methotrexate therapy for 48 h after glucarpidase is given due to the analytical interference from methotrexate metabolies. 5 In contrast, high performance liquid chromatography (HPLC) methods are able to separate methotrexate from its metabolites and so specifically measure the active drug. 5
While LC-MS/MS methods for methotrexate analysis have been developed previously,8–11 these tend to require larger sample volumes and longer chromatography times. To the best of our knowledge there are no clinically validated LC-MS/MS methods for methotrexate in the UK. Therefore, our method could have an important clinical role in monitoring patients on HDMTX, particularly those who have received glucarpidase rescue therapy who are likely to be the most at risk of methotrexate toxicity.
Our method is particularly suitable for routine clinical use when urgent results are required as it has a rapid run time of 1.5 min injection-to-injection due to efficient separation of methotrexate from other analytes and interfering substances on a small but efficient guard column, as opposed to a full chromatography column. The method is very robust with the in-use column having been subjected to over 1000 injections with no deterioration in peak shape or retention time. Given the important role of HDMTX therapy in certain paediatric haematological malignancies, 13 the low sample volume of 40 µl used in our LC-MS/MS method is also beneficial, as other methods use much higher sample volumes.8–11 The LLOQ of our assay (0.02 µmol/L) is ideal for monitoring methotrexate post-HDMTX therapy as it allows accurate and precise measurement around the threshold (0.05–0.1 µmol/L) at which it is considered safe to discontinue antidote treatment. 4
Conclusion
We have developed and clinically validated a rapid LC-MS/MS method for methotrexate suitable for routine use in patients who have received HDMTX therapy and it is particularly important for patients receiving glucarpidase rescue therapy due to methotrexate toxicity.
Footnotes
Acknowledgements
We would like to thank Dr Suzanne Armitage (Principal Clinical Scientist, Clinical Biochemistry, Alder Hey Children’s Hospital) for providing patient samples and data for a method comparison. We would also like to thank Mr James Hawley (Principal Clinical Scientist, Clinical Biochemistry, Wythenshawe Hospital) for advice on sample preparation.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
MPM.
Contributorship
BGK developed the method. MPM performed the validation work. MPM wrote the first draft of the manuscript. BGK and MPM edited the manuscript before submission.
