Abstract
Objective
Carbohydrate antigen 72-4 (CA72-4) is widely used in the diagnosis and monitoring of many cancers. However, there are few studies on the differences of CA72-4 concentrations in terms of age and gender.
Methods
A total of 10,957 healthy subjects were divided into two groups according to gender and three age groups. The serum CA72-4 were detected. Statistical analysis was performed by SPSS.
Results
The CA72-4 concentration in female group was significantly higher than that in male group. The concentration of CA72-4 gradually decreased with age. Compared with the age >60 group, the CA72-4 concentrations were increased in the age 46–60 group and 16–45 group (P >0.05, respectively). To better observe the age difference, the age 16–45 and 46–60 groups were combined into the age 16–60 group. In comparison to the age >60 group, the CA72-4 concentration of age 16–60 group was significantly increased (P = 0.000). In the age >60 group, there was no difference between genders. Nevertheless, the difference between the sexes in the age 16–60 group was significant (P = 0.023).
Conclusions
The reference interval of CA72-4 for local healthy population was established. CA72-4 concentrations gradually decreased with the increase of age, and CA72-4 concentration in females aged 16–60 years (0–18.0 U/mL) was higher than in males (0–14.5 U/mL); however, there was no gender difference in the age group above 60 years old (0–14.5 U/mL). Moreover, in male CA72-4 there was no significant difference among all age groups, while the potential mechanism of female changes with age needs further study.
Introduction
Tumour markers are closely related to the occurrence and development of malignant tumours and play an important role in prognosis assessment, treatment observation and recurrence prediction of tumors. 1 , 2 Carbohydrate antigen 72–4 (CA72-4), a tumour marker, is a high molecular glycoprotein antigen, which is low expressed in benign tumour tissues, body fluids and normal tissues. However, it is highly expressed in gastrointestinal tumours, pancreatic cancer, endometrial cancer and ovarian cancer. Therefore, it is widely used in the diagnosis and postoperative monitoring of digestive tract tumours, ovarian cancer, endometrial cancer and other cancers.3–5 The CA72-4 reference interval used in our laboratory was determined based on the European and American population. However, some studies had reported that the expected values of many tumour markers varied greatly in different regions, which may be affected by race, dietary habits, lifestyle, regional environment. 6 , 7 Moreover, through communication with clinicians, we found that many people in our hospital had CA72-4 test results exceeding the upper limit of the reference interval without any clinical symptoms or manifestations. Subsequently, in the process of establishing the CA72-4 reference interval for the local population, we found that the concentration of CA72-4 differs in terms of age and gender.
Materials and methods
Instrument and reagents
All tests were run on Roche Cobas 8000 e602 electrochemical luminescence analyser (Roche Diagnostics GmbH, Mannheim, Germany). CA72-4 reagent kits, calibrators and control materials were also purchased from Roche Diagnostics GmbH (Mannheim, Germany). All tests were run with the same instrument and the same batch of reagents (Lot number: 344248). The smaller variation of internal quality control (less than 5%) ensures the precision of the test results.
Subjects
The study was conducted at the Clinical Laboratory Center of Xiyuan Hospital, China Academy of Chinese Medical Sciences, Beijing, China, starting on 6 March 2019 and ending on 10 July 2019.
In our study, a total of 10,957 reference individuals (63.0 ± 14.5 years) were enrolled, including 4841 males (64.0 ± 14.9 years) and 6116 females (63.0 ± 14.2 years).
According to the Clinical and Laboratory Standards Institute (CLSI) EP28-A2,
8
the exclusion criteria for reference individuals were as follows: Basic physical signs:(1) Abnormal blood pressure (systolic pressure > 140 mmHg, and/or diastolic pressure > 90 mmHg); (2) body mass index (BMI) > 28 kg/m2, or BMI <18.5 kg/m2; (3) Women in pregnancy, lactation or miscarriage less than three months. Abnormal specimens (haemolysis, lipemia, jaundice, etc.). Fasting blood glucose >7.0 mmol/L or various types of diabetes. Elevated blood sugar can lead to an increase in CA72-4 concentrations.
9
Gout also can cause an increase in CA72-4.
10
Diagnosis of acute and chronic infections, cardiovascular diseases, respiratory diseases, liver diseases, kidney diseases, digestive diseases, metabolic diseases, rheumatic diseases, endocrine diseases, mental or genetic diseases. These may result in abnormal CA72-4 concentrations.
11
Benign and malignant tumours can cause the increase of CA72-4 results.
12
Positive samples of HBsAg, anti-HCV or anti-HIV may interfere with the detection of CA72-4.
Exclusion criteria are based on medical records and laboratory test results. The exclusion criteria included benign and malignant tumours, acute stroke, hepatorenal failure, acute infection, digestive system diseases, hyperglycaemia, hyperuric acid, hyperlipidaemia, and so on. 9 Subjects who did not suffer from the above-mentioned diseases that led to elevated concentrations of tumour markers were included in the study.
Sample collection and processing
All venous samples were centrifuged at 1500 g for 10 min. The serum of the specimen had a clear appearance without haemolysis, blood lipids, jaundice and other abnormalities. Subsequently, the serum samples were detected by automatic electrochemiluminescence analyser.
Statistical analysis
Statistical analyses were performed by SPSS. The data were presented as median ± 25th percentile. Differences between two or more groups were assessed by Mann-Whitney rank sum test or Kruskal-Wallis rank sum test, respectively. The Nemenyi rank sum test was used for pair-wise comparison among multiple groups. A P-value less than 0.05 was considered statistically significant.
Results
Distribution of serum CA72-4
In this study, the serum CA72-4 concentration of 10,957 individuals ranged from 0.20 to 144.10 U/mL and all data showed a skewed distribution (Figure 1). There were 4841 males and 6116 females. There were 1307, 3238 and 6412 subjects in the 16–45, 46–60 and > 60 years old groups, respectively (Table 1).
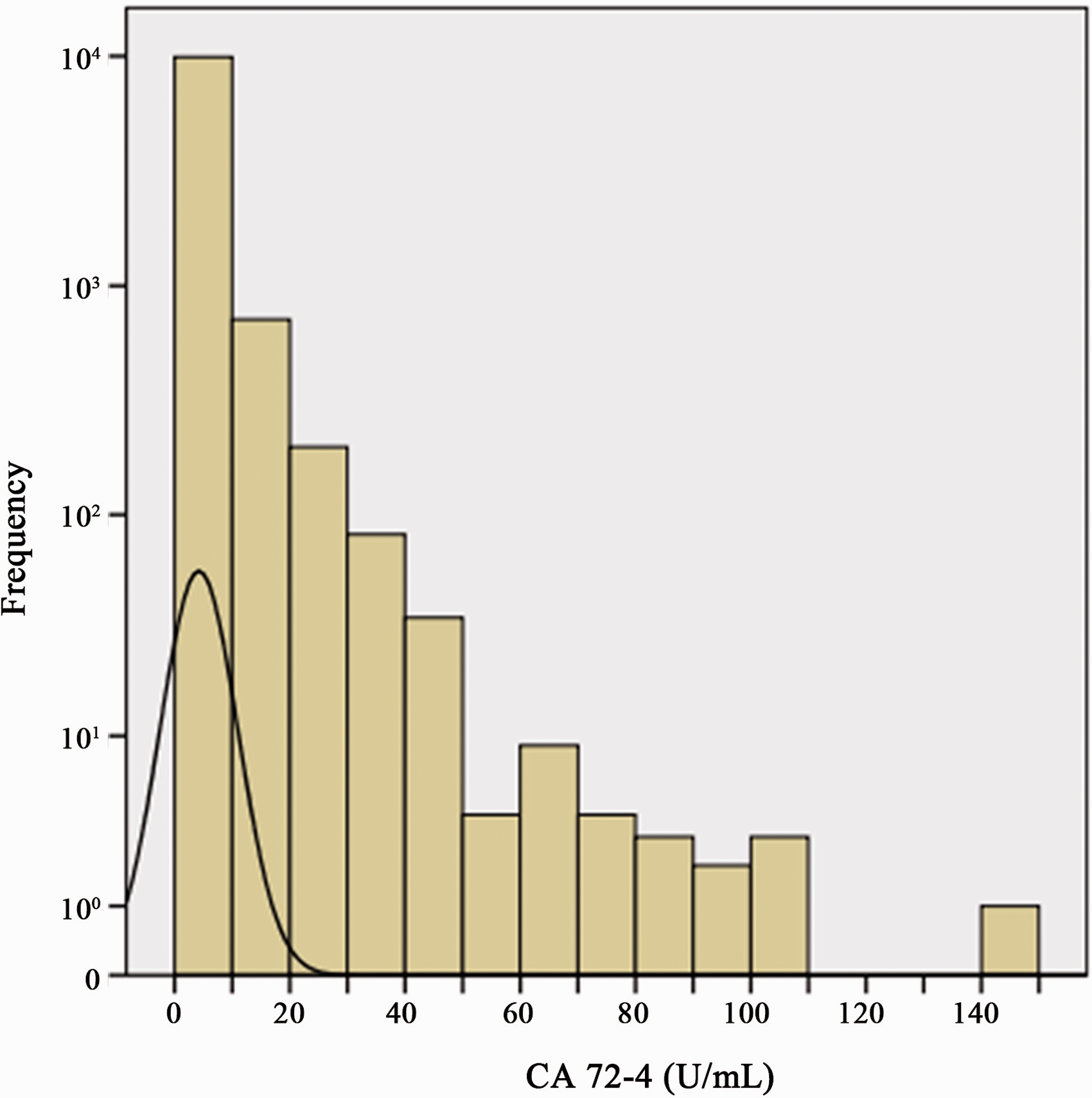
Concentrations of serum CA72-4 and distribution of healthy subjects.
Serum CA72-4 concentrations and percentiles (U/mL).
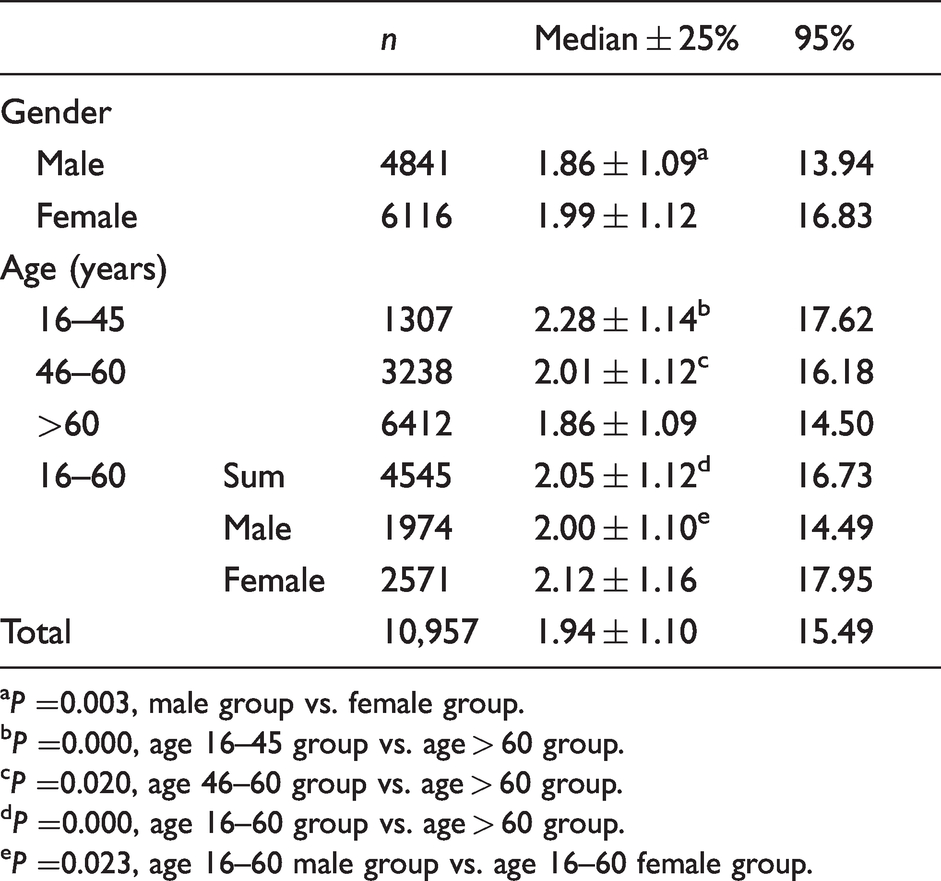
aP =0.003, male group vs. female group.
bP =0.000, age 16–45 group vs. age > 60 group.
cP =0.020, age 46–60 group vs. age > 60 group.
dP =0.000, age 16–60 group vs. age > 60 group.
eP =0.023, age 16–60 male group vs. age 16–60 female group.
Differences of serum CA72-4 concentrations on gender and age
Many studies have suggested that concentrations of tumour markers may vary by age and sex. Therefore, all healthy subjects were first analysed by gender and divided into male and female groups. The CA72-4 concentrations were 1.86 ± 1.09 U/mL in male group and 1.99 ± 1.12 U/mL in female group (Table 1). The results showed that CA72-4 concentration in male group was significantly lower than that in female group (P =0.003, Figure 2(a)).
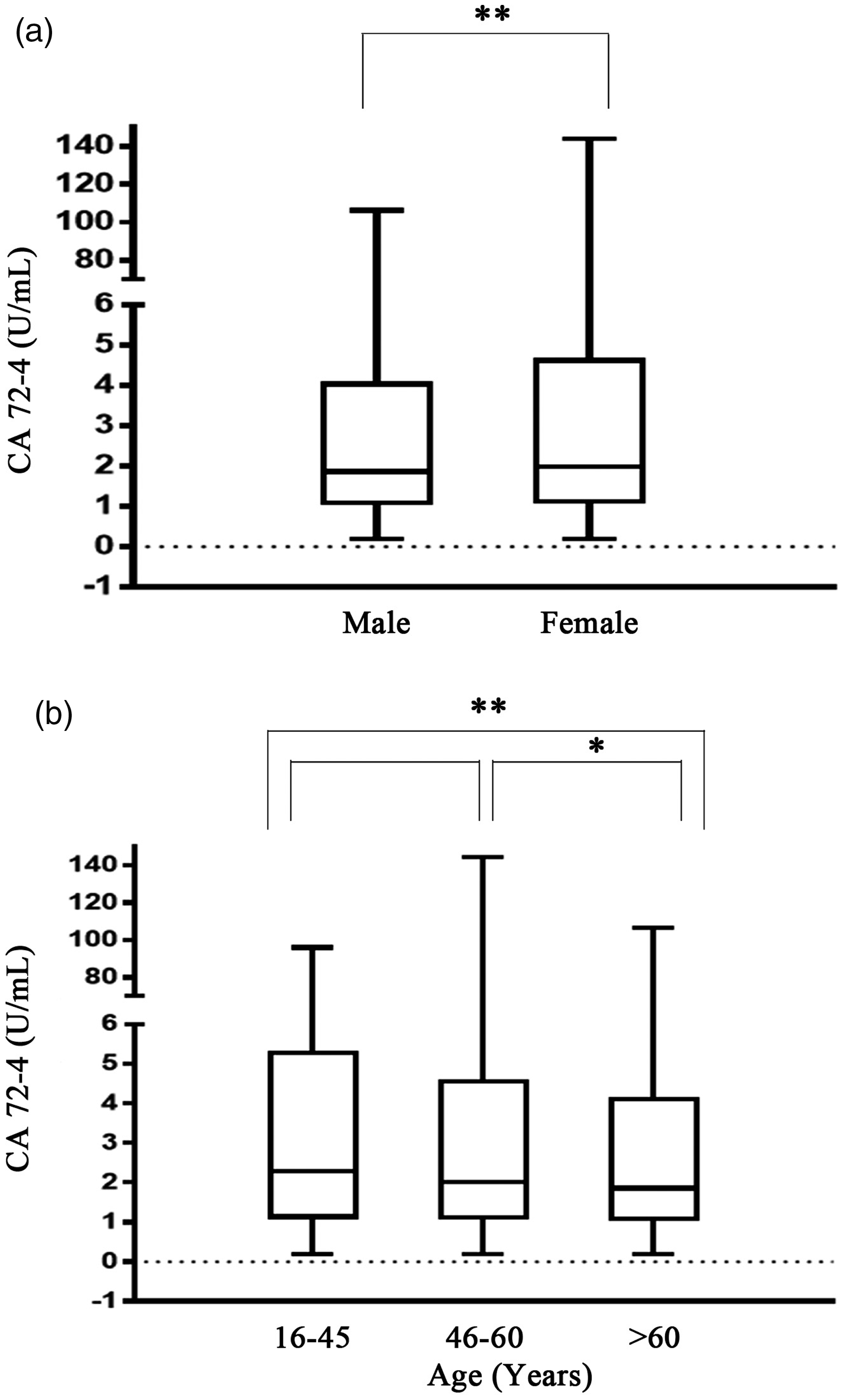
Serum CA72-4 concentrations in both gender and age groups. (a) Comparison of serum CA72-4 concentrations between male and female groups. (b) Comparison of serum CA72-4 concentrations between age 16–45 group, age 46–60 group and age > 60 group. Mann-Whitney rank sum test was used for differences between the two groups, Kruskal-Wallis rank sum test was used for multiple groups and Nemenyi rank sum test was used for pair-wise comparison between multiple groups. **P < 0.01, *P < 0.05.
In addition, all subjects were divided into three groups according to age. The concentrations of CA72-4 in the age 16–45 group, age 46–60 group and age > 60 group were 2.28 ± 1.14 U/mL, 2.01 ± 1.12 U/mL and 1.86 ± 1.09 U/mL, respectively (Table 1), with statistically significant differences (P = 0.000). To better observe the differences between age groups, pair-wise comparisons were made between the three age groups. The results showed that there was difference in CA72-4 concentration between the age 46–60 group and the age > 60 group (P = 0.020), while there was a more significant difference between the 16–45 group and the age > 60 group (P = 0.000, Figure 2(b)). However, there was no significant difference between the age 16–45 group and the age 46–60 group (P = 0.220, Figure 2(b)).
Distribution and difference of serum CA72-4 concentrations in age 16–60 group and age >60 group in genders
In order to more accurately observe the age distribution of serum CA72-4 concentration, the age 16–45 group and the age 46–60 group were combined to form an age group of 16–60 group (2.05 ± 1.12 U/mL, Table 1). After the combination, subjects in age 16–60 group and age > 60 group were 4545 and 6412, respectively (Table 1). The serum CA72-4 concentration of age 16–60 group was significantly higher than that in age > 60 group (P =0.000, Figure 3(a)). In age > 60 group, there was no statistically significant difference between the sexes (P = 0.050, Figure 3(c)), but in the 16–60 age group, the serum CA72-4 concentration in the male group was 2.00 ± 1.10 U/mL (1974 subjects), compared with the CA72-4 concentration in the female group (2.12 ± 2.16 U/mL, 2,571 subjects), the difference was significant (P =0.023, Table 1, Figure 3(b)).
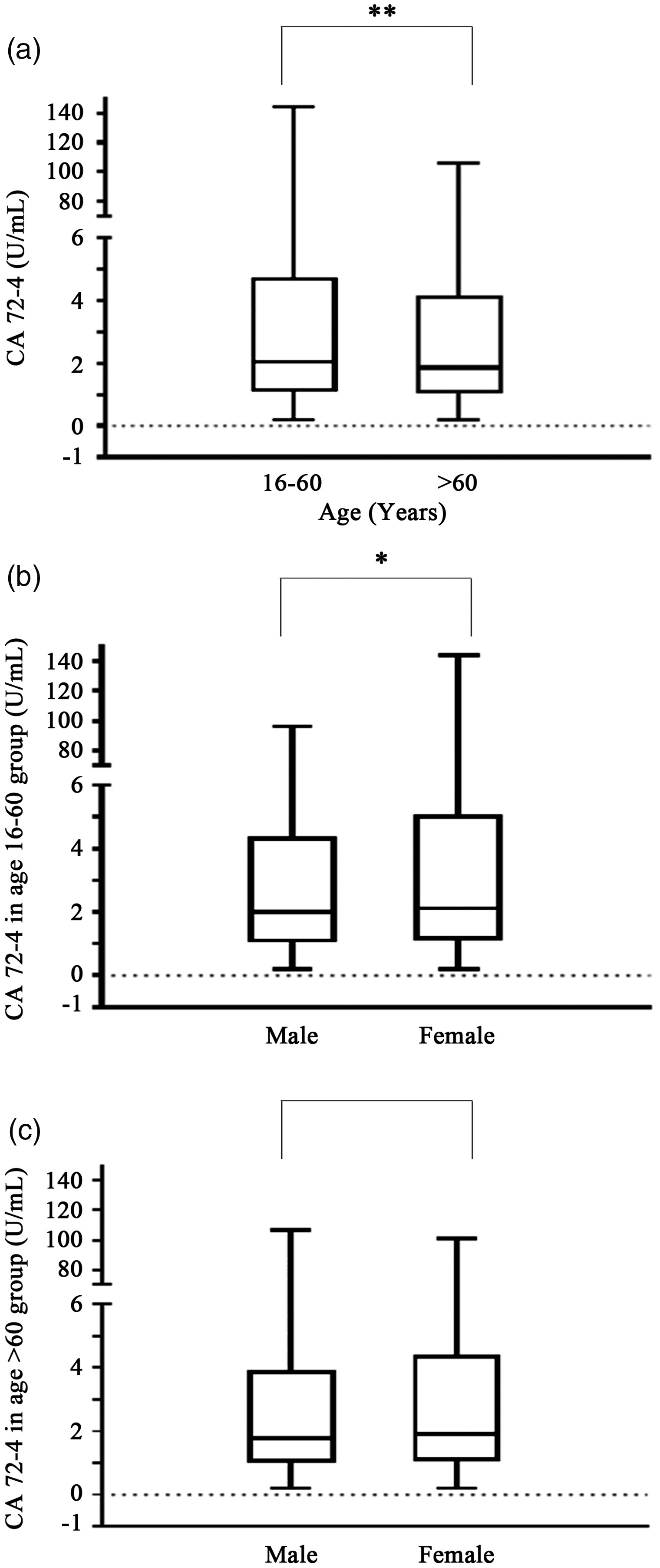
Difference of serum CA72-4 concentration between age 16–60 group and age > 60 group. (a) Difference in serum CA72-4 concentrations between age 16–60 group and age > 60 group. (b) Gender differences in serum CA72-4 concentrations between the age 16–60 group and the age > 60 group, respectively. Mann-Whitney rank sum test was used for the difference between the two groups. **P < 0.01, *P < 0.05.
The establishment of CA72-4 reference intervals
We used the 95th percentile method to establish the reference interval of CA72-4 in Beijing area. Among them, the reference interval for male population aged 16–60 years was 0–14.5 U/mL, and that for female population was 0–18.0 U/mL. However, there was no statistically significant difference between men and women in >60 years old, so the reference interval was combined to 0–14.5 U/mL. It was clear that there was a significant difference between the reference interval provided by the manufacturer (0–6.9 U/mL) and the reference interval derived from our research. Moreover, we also found that the reference interval of male population was almost the consistent between those under 60 years old and those over 60 years old (Table 2).
The newly established and manufacturer-supplied CA72-4 reference intervals.
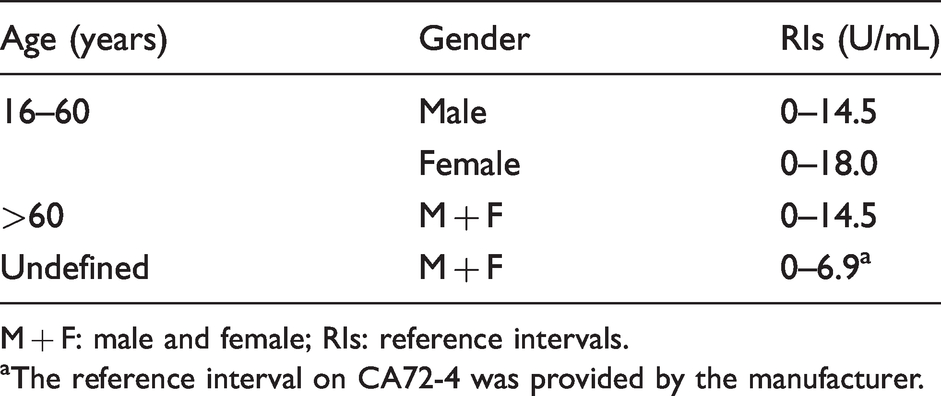
M + F: male and female; RIs: reference intervals.
aThe reference interval on CA72-4 was provided by the manufacturer.
Difference of serum CA72-4 concentration on age in male group and female group
To further confirm that CA72-4 may be no significant age difference in male population, we analysed differences of CA72-4 concentrations about male and female population in three age groups. As predicted, there was no statistically significant difference in age among males (Figure 4(a)). Nevertheless, the results of the female group showed that the concentration of CA72-4 decreased in age > 60 group compared with the age 16–45 group or the age 46–60 group, respectively (P =0.030, P =0.040, Figure 4(b)), while there was no significant difference in the concentration of CA72-4 between the 16–45 age group and the 46–60 age group (P =0.690, Figure 4(b)).
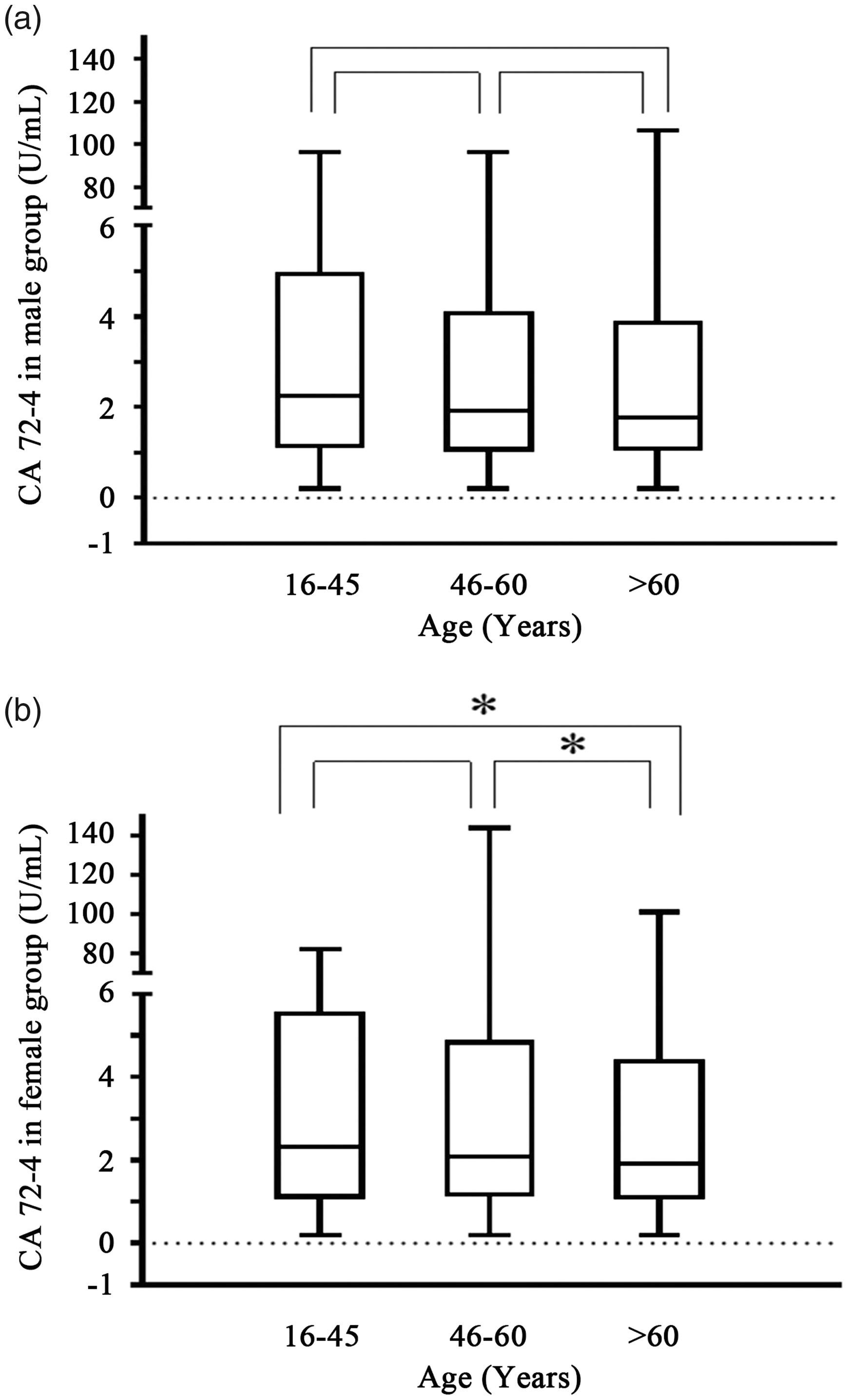
Serum CA72-4 concentrations in males and females were different in different age groups. (a) Differences in serum CA72-4 concentrations among males in different age groups. (b) Serum CA72-4 concentrations in females in different age groups. Differences in three age groups were assessed by Kruskal-Wallis rank sum test. The Nemenyi rank sum test was used for pair-wise comparison among three groups. *P < 0.05.
Discussion
With the increasing incidence and mortality, cancer has not only become one of the main causes of death at this stage but also become a serious public health problem worldwide, posing a serious threat to human health. 13 , 14 Pathology, cytology, imaging and laboratory tests are important means for tumour diagnosis, efficacy monitoring and prognosis assessment.15–17 Tumour marker detection in serum or other body fluid specimens is widely used in clinical practice due to its non-invasive and easy to be dynamically observed.
Due to the influence of race, diet and lifestyle, the incidence of cancer in different regions is significantly different. For example, the incidence of gastric cancer is higher in areas with high intake of salt and preserved food, and the risk of gastric cancer of smokers is also significantly higher than that of non-smokers.18–20 As one of the important markers of gastric cancer, CA72-4 is a high molecular weight glycoprotein identified by two monoclonal antibodies CC49 and B72.3, and its abnormal increase is common in many other cancers. 21 It is often used in combination with other tumour markers to improve the sensitivity and specificity of tumour diagnosis. 22 In addition, some health foods, such as ganoderma spore powder, have been found to cause a significant increase in serum CA72-4 concentrations. 23 This may be related to the fact that traditional Chinese herbal medicines and health food directly stimulates the immune cells of the body and regulates the immune function by enhancing the function of NK cells and T cells. 24 Therefore, CA72-4 concentrations in different regions are also different due to various factors.
The CA72-4 concentration of 10,957 subjects showed a significant skewed distribution, so non-parametric rank sum test was chosen for statistical analysis (Figure 1). For the non-parametric test method, Mann-Whitney rank sum test was used for the difference between two independent samples, and Kruskal-Wallis rank sum test was used for the difference between multiple independent samples. In order to further determine the difference sources among multiple independent samples, we refer to previous studies and adopt Nemenyi rank sum test to conduct pair-wise comparison among multiple groups.25–27
In many studies, age and sex are often used as criteria for grouping to find differences between groups.6,28 Therefore, this study also used it as a grouping strategy. The data showed that the number and proportion of subjects were relatively balanced in age and gender grouping (Table 1), while the serum CA72-4 concentration did show differences in age and gender (Figure 2). It should be noted that in the study of the same Chinese population in Henan region, there were no significant differences in the concentrations of CA72-4 among all healthy volunteers in terms of gender and age. 6 The results suggest that although the race is the same, the reference individuals in different living areas may still be different.
Refer to Clinical and Laboratory Standards Institute (CLSI) recommendations, composed of 95% health reference population CA72-4 reference interval, at the same conditions of instruments and reagents, the concentration CA72-4 was not only significantly higher than the reference value based on the European and American population (Table 2) but also different from that of Asian population including Chinese Henan (0–7.14 U/mL) and Kuwaitis (0–2.4 U/mL).6,29 In terms of age groups, CA72-4 was the highest in the age 16–45 group, followed by the age 46–60 group and finally the age >60 group (Table 2). Although there was no statistical difference in CA72-4 concentrations between the age 16–45 group and the age 46–60 group, the results still showed a trend of gradual decline with age (Figure 2). When the age 16–45 group and the age 46–60 group were combined to form a new age 16–60 group, the CA72-4 concentration was still higher than the age >60 group (Figure 3(a)). The analysis results pointed out that CA72-4 may be affected by aging and immune function decline in physiological conditions, so the secretion concentration of CA72-4 gradually decreases. In addition, according to gender analysis, both in the overall population and in the age 16–60 group, females were higher than males (P = 0.003, P =0.023, Table 1, Figures 2(a) and 3(b)); however, there was no significant difference in the concentration of CA72-4 between females and males in the age >60 group (Figure 3(c)).
Subsequently, the results in different age groups differentiated by gender also confirmed that the concentration of CA72-4 in males did not change significantly with the increase of age, while the concentration of CA72-4 in females was consistent with the overall age groups and gradually decreased with the growth of age (Figure 4). The above data indicated that the CA72-4 concentration of young and middle-aged women was higher than that of men in the same period, and oestrogen may be involved in the synthesis and regulation of CA72-4 concentration. 30 Some studies have found that oestrogen is an important factor in inducing cancer, and the high expression of oestrogen will also reduce the survival rate of cancer patients.31–32 This evidence suggests that oestrogen is closely related to cancer and changes in its concentrations may also lead to changes in the concentration of tumour markers in the body. In the future, we will further explore the relationship between women's menstrual cycle, sex hormone concentrations and CA72-4.
It is worth mentioning that, in order to minimize the impact of the error of the analysis system on the test results, all the measurements in this study are carried out on the same instrument, and a single batch of reagents are used and the coefficient of variation is controlled within 5%. In this case, however, a smaller CV may rule out the general validity of the results.
Conclusion
Based on the observation of 10,957 healthy reference population in Beijing, the different reference interval of CA72-4 in local region was established. As for the overall distribution, CA72-4 concentration decreased with age. In the age group of 16–60, CA72-4 concentration was higher in females than in males; however, there was no gender difference in people over the age of 60. In addition, there was no significant difference in male CA72-4 among all age groups, while the potential mechanism of female changes with age remains to be further studied.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Innovation Plan of Xiyuan Hospital (Grant number: XYKY-MP (2013)–41).
Ethical approval
This study was approved by the ethics committee of Xiyuan Hospital, China Academy of Chinese Medical Sciences (Ethical approval No.: 2015XL038).
Guarantor
XC.
Contributorship
XC and JW provided the concept for the article. TD wrote the first draft of the article with contribution from XC. JW wrote the study protocol and RL gained ethical approval. HG and JD examined the samples and participated in the data analysis. JG contributed to data interpretation and writing of the article. All authors corrected and approved the final version of the article.
