Abstract
Background
CA19-9 is elevated in pancreatic cancer and other malignancies, and commonly used in clinical practice. Unfortunately, CA19-9 immunoassays are not harmonized, and reference intervals may differ between assays. The aim of this study was to establish the reference interval of the ADVIA Centaur/Atellica IM CA19-9 assay in an apparently healthy Singapore adult population.
Methods
This is a retrospective cross-sectional study. De-identified data from Health Screening participants were extracted from our database. Subjects with biochemical results suggesting anaemia, diabetes mellitus, viral hepatitis or abnormal liver, and renal and tumour markers were excluded. Outlier and subclass analyses by age and sex were performed. CA19-9 reference limits and 90% confidence intervals were then determined for candidate subclasses.
Results
Data from 12,174 subjects (5846 males and 6328 females) were available after exclusion criteria were applied. CA19-9 results did not follow a normal distribution and were higher in females compared to males (P < .001). Although CA19-9 means were statistically different between certain age groups, the evaluable 99th percentile reference limits were not statistically different. The overall 99th percentile reference limits for the Centaur/Atellica CA19-9 assay was 37 U/mL for males 21–80 years, and 60 U/mL for females 21–80 years.
Conclusions
Our results suggest that separate CA19-9 reference intervals should be applied for males and females.
Introduction
Carbohydrate antigen 19-9 (CA19-9), also known as sialyl Lewis-a, is a carbohydrate antigen first described in 1979 as a marker of colorectal carcinoma. 1 It is elevated in tissue and sera of patients with gastrointestinal tumours, especially biliary and pancreatic cancer, 2 but is also elevated in benign conditions such as chronic pancreatitis, biliary obstruction, lung infections, ovarian fibroids, thyroid nodules, and others. 3 CA19-9 is related to the Lewis blood group antigens (Le), and most individuals with the Le(a-b-) phenotype have undetectable serum levels of CA19-9. 4
Although most manufacturers use the same monoclonal antibody 1116-NS-19-9 first described by Koprowski, 1 CA19-9 immunoassays are not harmonized, and the same sample may yield variable results with different assays.5,6 Despite the lack of CA19-9 assay harmonization between different manufacturers, the original CA19-9 cut-off of ≤37 U/mL 7 is widely used as the reference interval (RI) for the general population, with a reported sensitivity of 79%–81% and specificity of 82%–90% for the diagnosis of pancreatic cancer in symptomatic patients. 2
Our laboratory had received periodic queries from clinicians regarding patients with minor elevations in CA19-9 (typically within 2× of the upper reference limit), in which further investigation yielded no significant clinical findings. The same serum sample run on an alternate CA19-9 assay (Roche Elecsys CA19-9) would yield results below that assay’s stated 97.5th percentile (34 U/mL). This led us to further investigate all samples with CA19-9 elevation but no previous history, between July 2020 and November 2021. After excluding samples with potential heterophilic interferants (via heterophilic blocking tubes and rheumatoid factor measurement), 157 samples remained. We observed that 128/157 (81.5%) samples were from female subjects, and there was a positive bias of our current assay compared to the alternate platform (Supplemental Figure 1). However, similar reference limit of ∼37 U/mL was used for both. This prompted us to review the literature regarding population reference intervals of different CA19-9 assays.
Published studies on the CA19-9 reference interval have yielded widely variable results and suggest that CA19-9 reference intervals may differ based on assay manufacturer, sex, and age.8–11
Our laboratory network has been using the Siemens ADVIA Centaur CA19-9 assay at our five sites, and recently converted to the Siemens Atellica IM CA19-9 assay, which uses the same reagents and acridinium ester technology. Internal validation studies found good correlation and low bias between the Centaur and Atellica CA19-9 assays. To our knowledge, no studies on the CA19-9 reference interval using the Centaur or Atellica assays have been published.
Therefore, this study was initiated to establish the reference interval for the ADVIA Centaur/Atellica IM CA19-9 assay for our local population as per CLSI guidelines EP28-A3c. 12
Methods
Subjects
This study was exempted from ethics review by the Parkway Independent Ethics Committee (PIEC/2022/021). De-identified data from Health Screening participants from 1 Jan 2018 to 28 Feb 2022 were extracted from the Parkway Laboratory Information System. The study flow chart is shown in Figure 1. ‘Health Screening packages’ are general health evaluation packages offered by our institution. They typically involve clinical consultation and physical examination by a general practitioner, blood tests interrogating various biomarkers, and other investigations such as vital signs, ECG, and ultrasound. These packages are not reimbursed by health insurance in Singapore, so participants pay out-of-pocket, or corporations reimburse them as part of employee health benefits. Study flow chart.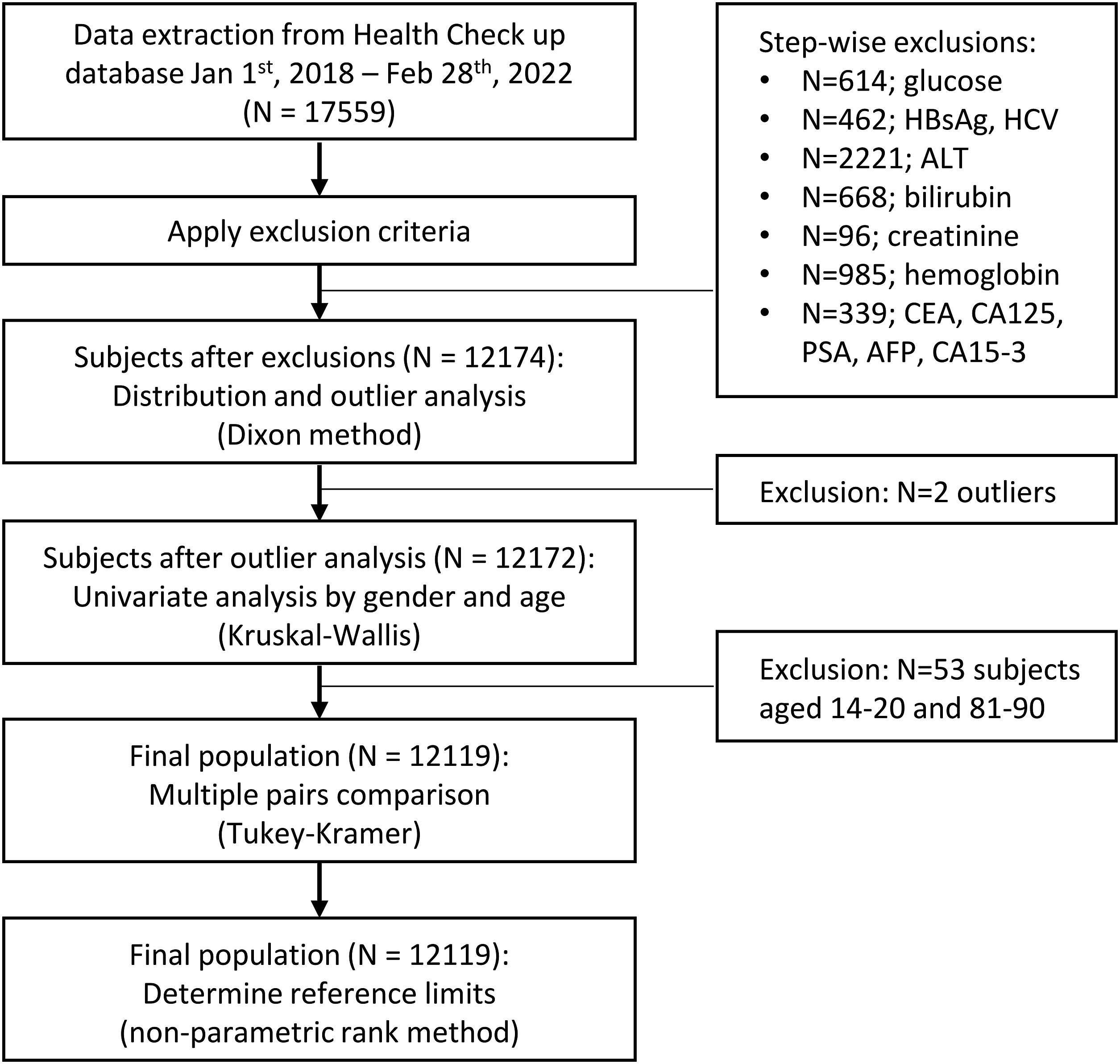
Data from all Health Screening participants from 1 January 2018 to 28 February 2022 with available Centaur/Atellica CA19-9 result were extracted. The following exclusion criteria were then applied: 1. Anaemia, defined as low hemoglobulin <13.0 g/dL (male) or <11.5 g/dL (female). 2. Potential liver or kidney abnormalities, defined as a result outside the reference interval in any of the following tests: ALT, total bilirubin, and creatinine. 3. Potential chronic viral hepatitis, defined as a reactive or equivocal result for HBsAg and/or anti-HCV. 4. Potential malignancy, defined as a result above the reference interval in any of the following tumour markers if available: CEA, CA125, PSA, AFP, and CA15-3. 5. Potential diabetes mellitus, defined as fasting blood glucose ≥7.0 mmol/L
CA19-9 measurements
CA19-9 values were measured by the chemiluminescence immunoassay analyzers ADVIA Centaur XPT and Atellica IM (Siemens Healthcare Diagnostics, Tarrytown, New York, USA) at five different clinical laboratories within the Parkway Laboratories network. Both ADVIA Centaur and Atellica IM assays have the same reagent formulations, same measurement range (1.20–700.00 U/mL), and same standardization to an internal standard. According to the manufacturer kit insert, the Atellica IM CA19-9 assay was verified to have a correlation coefficient of ≥0.95 and a slope of 1.0 ± 0.10 compared to the ADVIA Centaur CA19-9 assay. All CA19-9 values ≤1.20 U/mL were recorded as 1.20 U/mL.
Sample size
CLSI-EP28 guidelines recommend at least 120 subjects per subclass for reference interval establishment. We estimated that about 3000 subjects per sex would be required to obtain at least 120 males and 120 females in each decade of age. Therefore, we aimed to extract data for about 7000–10,000 subjects (before applying exclusion criteria).
Statistical analyses
The following analysis was performed as per CLSI EP28 guidelines.
12
Analyze-It for Microsoft Excel 5.90 (Analyze-it Software Ltd, Leeds, United Kingdom) was used for all analyses. 1) Outlier analysis: • Histograms of CA19-9 distribution for males and females were constructed. • Visual inspection of the histograms was performed, and analysis of visually apparent outliers was performed using the Dixon method/one-third rule.
12
Briefly, if D/R ≥1/3 the outlier is rejected, where D = absolute difference between the outlier and the next largest (or smallest) observation; R = range of all observations. 2) Partitioning/subclass analysis: • The following variables were investigated: sex (M/F) and age (by decade). • Normality was assessed by the Anderson-Darling test, which found that the distribution of CA19-9 was not normal (P < .0001 for both males and females). Hence, non-parametric methods were used for all subsequent analyses. • Univariate analysis was performed using the Kruskal–Wallis test, to determine if significant difference in CA19-9 exists between different subclasses (cut-off: 5% significance level). • Additionally, Tukey-Kramer multiple pairs comparison was performed within each sex, for potential subclass partitioning by decade of age. 3) Reference interval determination • CA19-9 reference limits were determined by the non-parametric rank (N+1)p method, as described in CLSI EP28.
12
Results
Data from 17,559 subjects were extracted, and 12,174 subjects (5846 males and 6328 females) remained after applying exclusion criteria (Figure 1).
The CA19-9 frequency histograms (Figure 2) for males and females were generated. By visual inspection and the Anderson-Darling test, it was observed that the CA19-9 distribution was not normal (P < .0001). There were two extreme outlier points (1 male and 1 female), which were excluded after applying the Dixon test. CA19-9 distribution in males and females. Frequency histogram of CA19-9 in (a) males and (b) females. The x-axis has been truncated at 100 U/mL in order to better show the distribution at the low end range. All CA19-9 values ≤1.20 U/mL were recorded as 1.20 U/mL.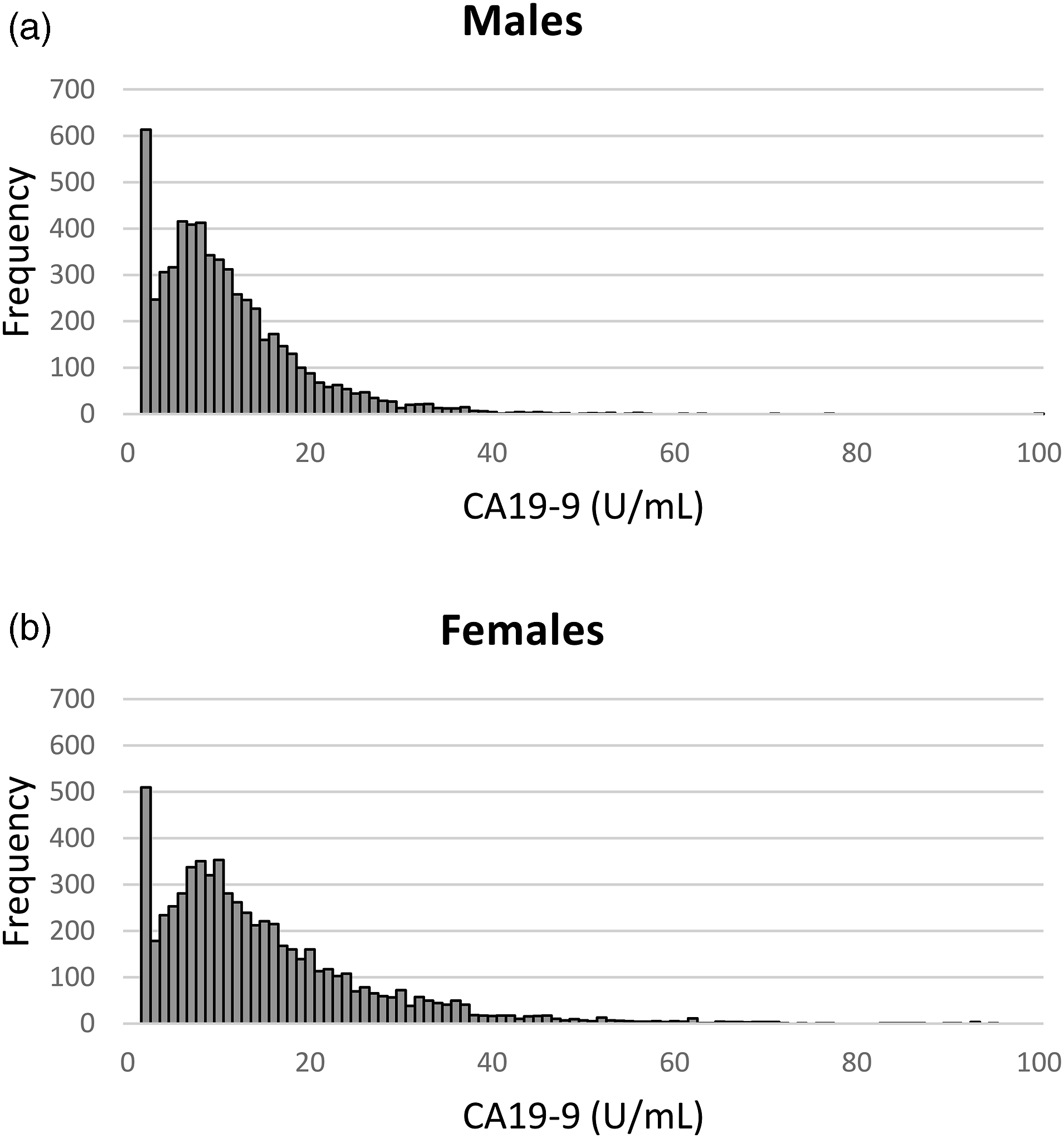
In total, 400/6328 (6.3%) of females and 482/5846 (8.2%) of males had CA19-9 results below the measuring range of the assay (<1.2 U/mL), which likely reflects low levels of CA19-9, and/or the Lewis Le(a-b-) phenotype. For analysis, all CA19-9 values ≤1.20 U/mL were recorded as 1.20 U/mL.
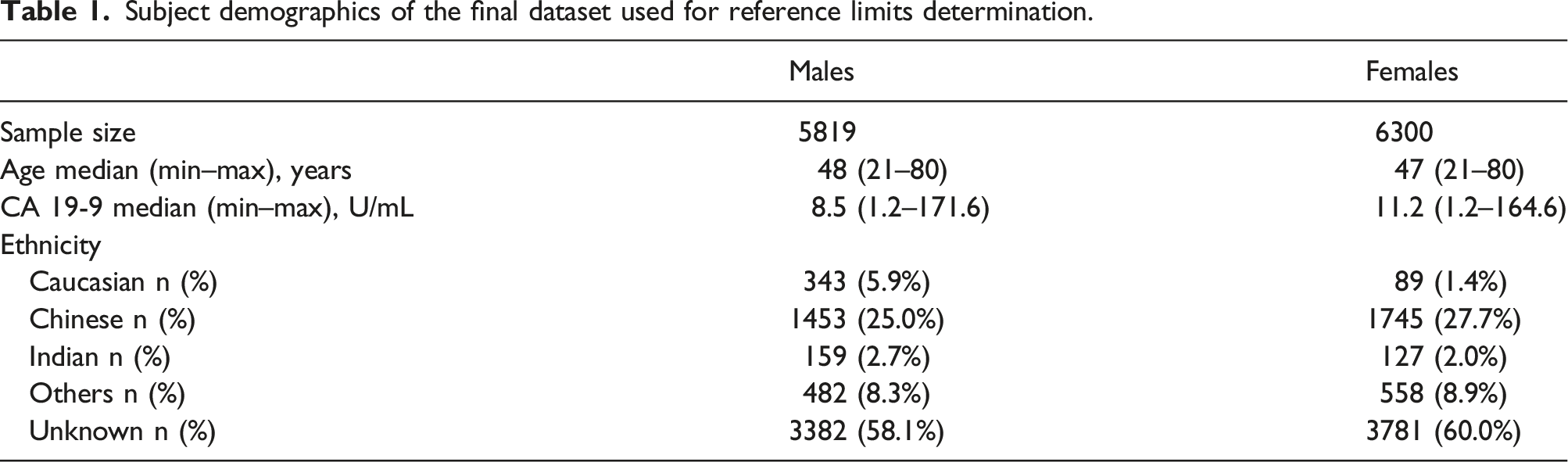
Subject demographics of the final dataset used for reference limits determination.
Box plots of CA19-9 by decade of age for each sex are shown in Figure 3. We observed that CA19-9 values decreased with age in females, but had a slight U-shaped trend in males, with a nadir at the 41–50 years age group. Due to the non-normal distribution, we did not perform multivariate analysis by ANOVA. Instead, Tukey-Kramer all pairs comparison was performed, and males and females were analysed separately. The test compared the means of each age group with all other age groups, to identify groups which were statistically different from each other. The results suggested that statistically significant subclasses were 21–30 years versus 31–80 years for males, and 21–40 years versus 41–80 years for females (Supplemental Tables S1 and S2). Since only sample means were evaluated by the Tukey-Kramer test, these candidate subclasses were further evaluated by comparing the 90% confidence interval of the reference limits, for each candidate subclass compared to the overall male or female population. Box plot of CA19-9 by decade of age in (a) males and (b) females. Boxes represent median and interquartile range. A line connecting the medians of each subpopulation is drawn to illustrate the trend of CA19-9 by age. N, sample size.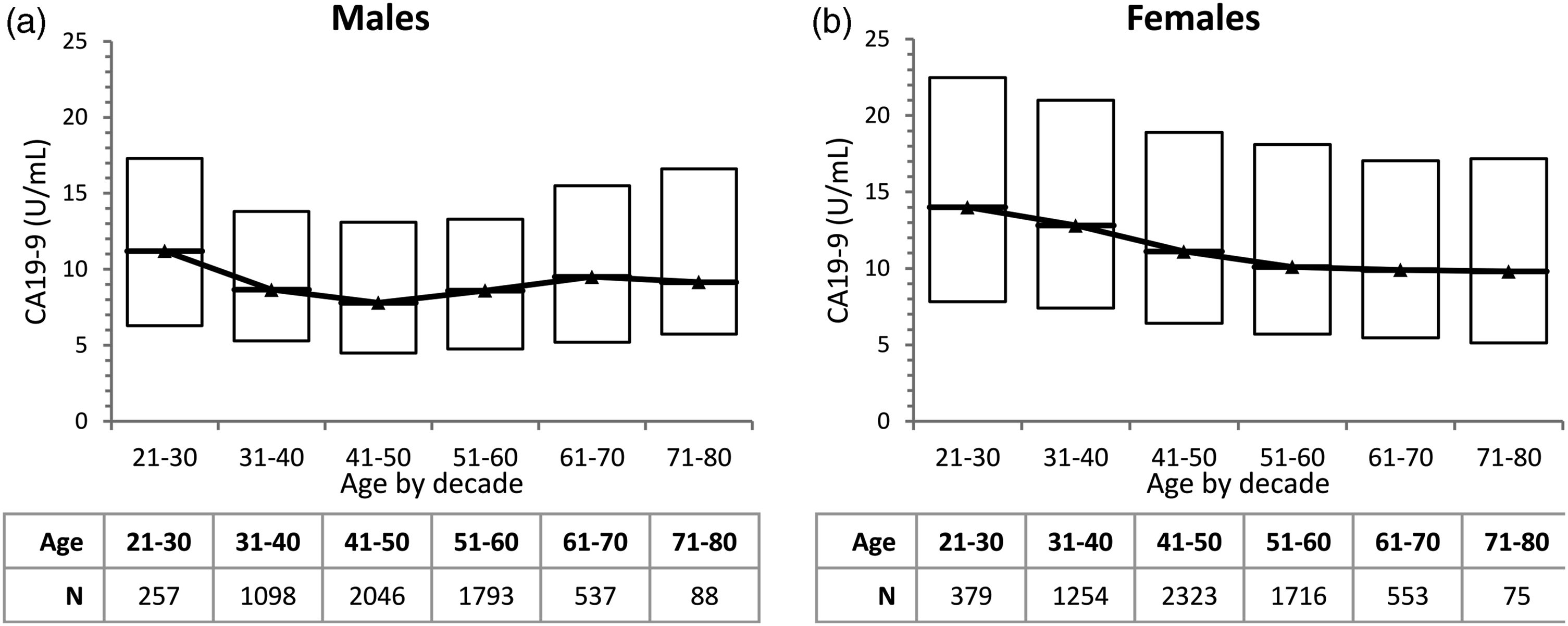
CA19-9 reference limits and 90% confidence intervals.
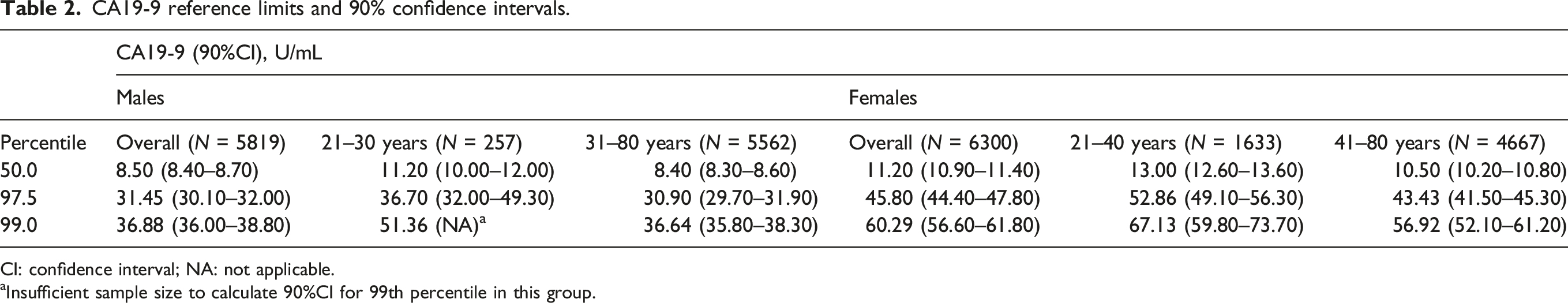
CI: confidence interval; NA: not applicable.
aInsufficient sample size to calculate 90%CI for 99th percentile in this group.
Examination of the candidate subclasses compared to the overall male and female population showed significant differences in the 50th percentiles, but overlapping 90% confidence intervals of the 97.5th and 99th percentiles (Table 2). The 99th percentile of females 21–40 years (59.8–73.7 U/mL) and females 41–80 years (52.1–61.2 U/mL) overlapped with the 99th percentile of the overall female population (56.6–61.8 U/mL). Due to insufficient sample size (n = 257) in the male 21–30 years group, it was not possible to determine 90%CI for the 99.0th percentile. However, the 97.5th percentile of males 21–30 years (32.0–49.3 U/mL) and males 31–80 years (29.7–31.9 U/mL) overlapped with the 97.5th percentile of the overall male population (30.1–32.0 U/mL), suggesting that the 99th percentiles would not be statistically different as well.
Discussion
We performed a retrospective analysis of a large dataset of apparently healthy individuals, and determined the population reference limits of the Centaur/Atellica CA19-9 assay as per CLSI guidelines. Due to the large dataset, we were able to analyse the effect of age and sex. Visual inspection of our CA19-9 frequency histograms (Figure 2) showed very similar CA19-9 distributions for males and females. Females had a slightly higher median value, and a longer ‘tail’ at the high end, resulting in significantly different 97.5th and 99th percentile reference limits for each sex. We also found a statistically significant effect of age in both males and females. In females, CA19-9 decreased with older age. Males had a slight U-shaped trend of CA19-9 with age, with a nadir at the 41–50 year age group (Figure 3).
Based on the final dataset of N = 12,119 subjects, the 99th percentile reference limit for the Centaur/Atellica CA19-9 assay in our Singapore-based population was ∼37 U/mL for males 21–80 years and ∼60 U/mL for females 21–80 years.
Comparison of CA19-9 adult reference interval studies.
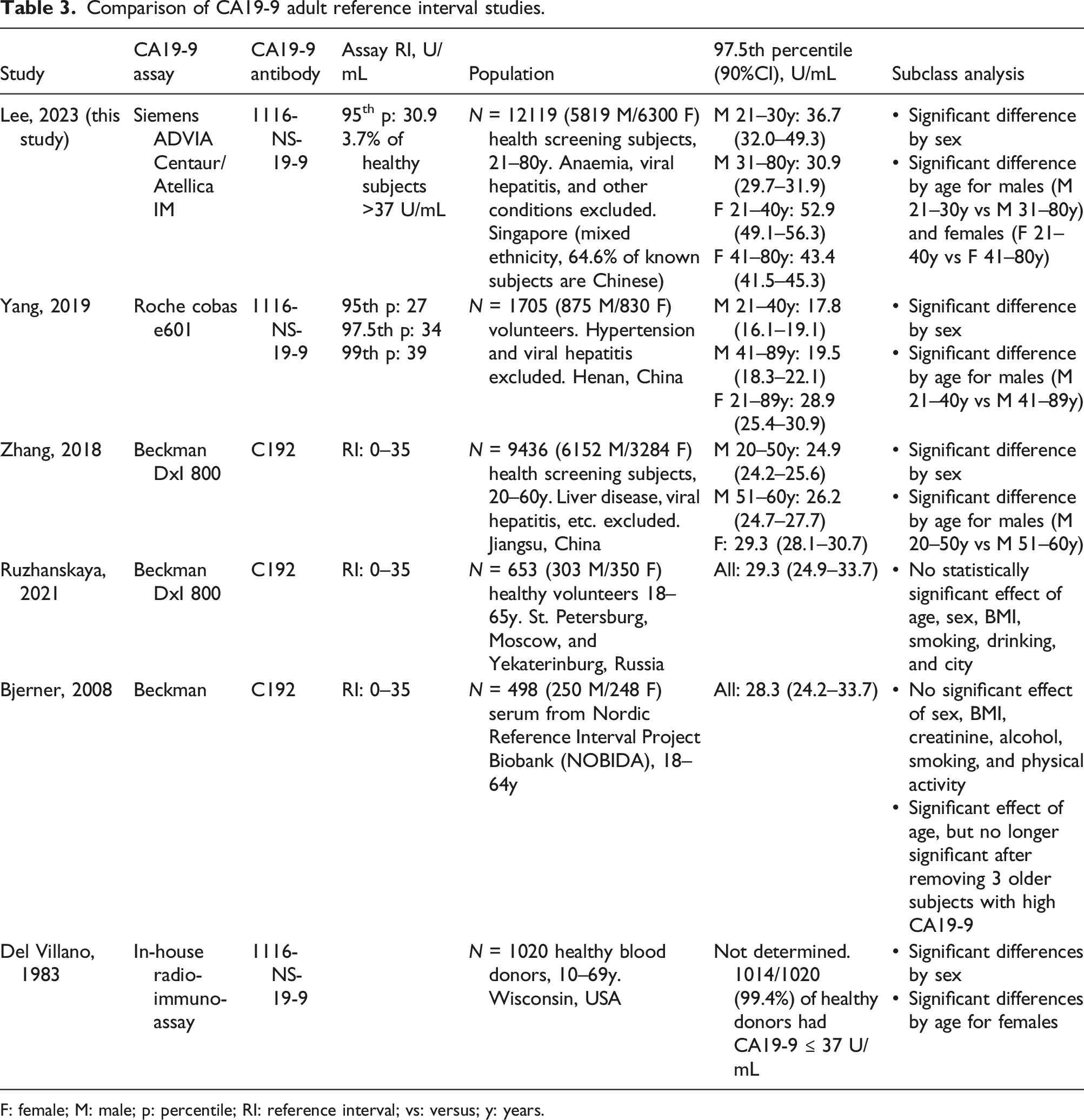
F: female; M: male; p: percentile; RI: reference interval; vs: versus; y: years.
The 97.5th percentile reference limits reported by the studies differed by sex, age, and CA19-9 assay (Table 3). All five previous studies ignored the impact of the Le(a-b-) (CA19-9 negative) population and did not exclude this subset in the determination of the population reference limits. When we excluded all values ≤1.20 U/mL and re-calculated the population reference limits, there was negligible impact on the results. The overall male 99th percentile increased from 36.9 to 37.1 U/mL, and the female 99th percentile increased from 60.3 to 60.9 U/mL. Hence, we included all values ≤1.20 U/mL, since it was not possible for us to distinguish which had low levels of CA19-9, and which had no CA19-9 due to the Le(a-b-) genotype. Our observed prevalence of undetectably low CA19-9 (6.3% of females and 8.2% of males) was similar to the commonly quoted prevalence of 7%–10%. 14 We hypothesize that the higher prevalence of CA19-9 ≤1.20 U/mL in males was likely due to low CA19-9 expression at levels below the assay limit of detection. However, we cannot exclude the impact of the ethnic differences potentially leading to higher prevalence of Le(a-b-) in our male population.
Four studies (including this study) found significantly higher CA19-9 levels in females compared to males. Two studies (Ruzhanskaya 2021 and Bjerner 2008) did not detect a significant difference between males and females, but it may be due to the smaller sample size in those studies, which only recruited 250–300 males and 250–350 females. A larger study using the same assay (Zhang 2018 with 6152 males and 3284 females) found significantly higher CA19-9 in females compared to males.
We detected significant changes in CA19-9 by age in both sexes, although the trend by age had different patterns in males versus females. In females, we observed decreasing CA19-9 with increasing age, until about age 50, when CA19-9 values remain constant. This trend is not well described in recent studies with automated CA19-9 assays, but matches the finding of Del Villano in 1983 using the first CA19-9 immunoassay. 7 It has been described that benign uterine or ovarian conditions can lead to CA19-9 elevations. 3 Hence, it is tempting to speculate that CA19-9 can be elevated due to the higher levels of female reproductive hormones in younger females. Indeed, a case report of exceedingly high CA19-9 levels (∼1000 U/mL) in a subject on hormone replacement therapy has been described. 15 We were unable to exclude pregnancy or hormone replacement therapy in our subjects, which may have impacted results. Notably, the three studies using the Beckman assay, based on the C192 antibody, did not find a significant difference in CA19-9 by age for females. But two of the three studies using assays based on the 1116-NS-19-9 antibody observed this phenomenon. Therefore, it is possible that the finding is antibody specific, reflecting a difference in epitope or in susceptibility to an unknown interferant.
In males, we observed a slight U-shaped trend of CA19-9 with age. A similar trend is observed in the data of Zhang et al., although their analysis concluded that the oldest age group (51–60 years) was statistically different, while our analysis indicated that the youngest age group (21–30 years) was statistically different. 9 In both studies, the absolute difference between age groups was small and may not be clinically significant.
For the purpose of diagnosing pancreatic adenocarcinoma, 37 U/mL was proposed by Del Villano as a cut-off for the first CA19-9 immunoassay. 7 This was based on the observation that 99.4% of healthy blood donors, and 98.5% of subjects with benign gastrointestinal conditions, had CA19-9 ≤37 U/mL, while 78.7% of subjects with pancreatic adenocarcinoma had CA19-9 >37 U/mL. 7 Current guidelines recommend CA19-9 for the prognostication and monitoring of pancreatic cancer but not for pancreatic cancer screening.14,16,17 Although CA19-9 >37 U/mL was reported to have reasonable sensitivity (79%–81%) and specificity (82%–90%) for pancreatic cancer in symptomatic patients, 2 7–10% of the population has the Le(a-b-) phenotype and do not have detectable CA19-9. 14 CA19-9 has poor positive predictive value (PPV) for pancreatic cancer (PPV 0.5%–0.9%) in asymptomatic subjects, and PPV remains low in asymptomatic subjects even if all other cancer types are taken into account.2,18
Given the poor PPV of CA19-9 in asymptomatic patients, ongoing clinician concern about false positive results, and historical observation of 37 U/mL as approximating the 99th percentile of healthy blood donors, we favoured the use of the 99th percentile instead of the 97.5th percentile as the CA19-9 reference interval for our institution. Since there is significant difference in 99th percentile for males versus females, but overlap in 90% confidence intervals of the 99th percentile for subclasses within each sex, we decided to adopt the overall male and female 99th percentile values (37 U/mL and 60 U/mL, respectively) as the upper limit of normal. Internal investigations of clinician-queried samples with CA19-9 results above 37 U/mL found that 128/157 (81.5%) samples were from female subjects, and 117/128 (91.4%) female samples were below 60 U/mL. All tested samples with Atellica IM CA19-9 results between 37 and 60 U/mL gave values within the reference interval when run on an alternate CA19-9 assay. This gave us confidence to implement separate reference intervals by sex, and to adopt a 60 U/mL cut-off for females going forward.
We expect negligible impact of this change for our Oncology colleagues, since it is a persistently rising/falling trend in CA19-9 or an extremely high result that informs clinical management for pancreatic cancer.14,17 For our General Practice colleagues who manage asymptomatic Health Screening participants, we expect to reduce queries by ∼90%, based on internal review of queried results between 37 and 60 U/mL compared to >60 U/mL. Since Health Screening packages at our institution include clinical evaluation, additional tests, and investigations, we expect that participants with any concerning signs and symptoms would be followed up accordingly.
The strengths of our study include the large sample size and ability to exclude subjects with potential clinical confounders using available laboratory data. Our exclusion criteria were designed to rule out common pathological conditions that are easily identifiable by abnormal blood test results (e.g. anaemia, liver disease, kidney disease, diabetes mellitus, and malignancies). However, the reference intervals used in our exclusion criteria may not have adequate sensitivity to detect early disease, which is a limitation of the study. For example, fasting blood glucose ≥7.0 mmol/L at best excludes uncontrolled diabetes, and the other tumour markers may not reliably exclude malignancy. Also, ALT and total bilirubin were applied as exclusion criteria, but AST, ALP, and GGT were not used. Since most of our reference intervals are based on the central 95% of the normal population, we also expect that application of these criteria will exclude healthy normal subjects.
The main weakness of the study is that we did not have further clinical information on our subjects and were not able to determine if other potentially confounding comorbidities or clinical conditions exist. In addition, our population consist of various ethnicities and we were not able to obtain ethnicity data for 59% of subjects. Of subjects with available data, the majority identified as Chinese (59% of males and 69% of females with self-declared ethnicity information). Since Health Screening packages from our institution require patients to pay out-of-pocket or have a certain level of employee health benefits, our population is likely not reflective of the general Singapore population in terms of income level and ethnicity. However, it is representative of the population that we serve at our institution.
In conclusion, we observed in our study and literature review, that CA19-9 reference intervals differ significantly by assay manufacturer and sex. It is recommended for laboratories to establish or validate reference intervals for the method used, in the context of the local population.
Supplemental Material
Supplemental Material - Establishment of CA19-9 reference intervals in an apparently healthy adult population in Singapore
Supplemental Material for Establishment of CA19-9 reference intervals in an apparently healthy adult population in Singapore by Joanne Mee Yin Lee, Pearline Teo, and Leslie Choong Weng Lam in Annals of Clinical Biochemistry
Footnotes
Acknowledgements
We would like to thank Ronelio Tan Magdurulang for valuable administrative assistance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study was exempted from ethics review by the Parkway Independent Ethics Committee (PIEC/2022/021).
Guarantor
LL.
Contributorship
L.L. and J.L. conceived the study. All authors were involved in literature review and protocol development. J.L. submitted and obtained ethical waiver for the study. P.T. performed the statistical analysis and wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
Nomenclature
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
