Abstract
Background
Early recognition of sepsis and prompt treatment improves patient outcome. C-reactive protein is a sensitive marker for tissue damage and inflammation, but procalcitonin has greater specificity for bacterial infection. Limited research exists regarding the use of C-reactive protein and procalcitonin at term pregnancy and the immediate postpartum period.
Aim
This study sought to define reference values for C-reactive protein and procalcitonin at term and the early postnatal period.
Methods
A prospective cross-sectional study was performed in a university teaching hospital. Venous blood was collected from healthy women (n = 196), aged between 19 and 45 years with an uncomplicated singleton pregnancy, at term (37–40 weeks’ gestation) and on day 1 and day 3 postpartum for the measurement of C-reactive protein and procalcitonin.
Results
The reference population comprised of 189 participants: term pregnancy (n = 51), postpartum day 1 vaginal delivery (n = 70) and caesarean section (n = 38) and day 3 (caesarean section, n = 30). The maximum procalcitonin value at term pregnancy was 0.1 μg/L. On day 1 postpartum, 90% and 86.8% of procalcitonin results for vaginal delivery and caesarean section, respectively, were below the decision-threshold of 0.25 μg/L. The specificity of procalcitonin to rule out infection in the reference population was 91.5%.
Conclusions
Reference values for procalcitonin were established in a well-characterized population of healthy pregnant women at term and immediately postpartum. The variability of C-reactive protein limits its clinical utility in the assessment of systemic sepsis. Application of the procalcitonin cut-off of 0.25 μg/L in this population will be a valuable adjunct to clinicians ruling out infection in pregnancy and postpartum.
Introduction
Sepsis is a leading cause of maternal deaths, and pregnancies complicated by sepsis are associated with increased rates of fetal infection and preterm delivery. 1 Healthy pregnancies have the potential to be complicated by bacterial infection that may fulminate in sepsis with outcomes influenced by early recognition and prompt treatment. 2 Diagnosis of sepsis is challenging as clinical diagnostic criteria overlap with non-infective causes of systemic inflammation. 3 C-reactive protein (CRP) and procalcitonin (PCT) are non-specific acute-phase reactants and in clinical practice, are useful adjuncts to detect occult sepsis and infection.4,5 CRP is produced by the liver,6,7 with blood concentrations increasing within 4–6 h of stimulation onset, peaking between 35 and 60 h and normalizing two to three days post restoration of homeostasis. 8 Normal CRP concentrations vary, higher levels are observed with increasing age, body mass index (BMI), in pregnancy, and in different ethnic groups. 9 In healthy pregnancies, CRP increases as the pregnancy progresses, with further increases reported during labour.10–13 A rise in maternal CRP concentrations has been correlated with pregnancy complications such as preeclampsia, preterm delivery, fetal growth restriction, pregnancy loss, preterm premature rupture of membranes (PPROM) and chorioamnionitis. 14 CRP concentrations in the puerperium, particularly after instrumental or operative delivery are not established.
PCT is the pro-hormone of calcitonin produced in the C cells of the thyroid gland under normal physiological conditions. 15 In response to severe systemic inflammation, particularly bacterial infection, PCT is released into the circulation from a variety of extrathyroidal sources. 16 PCT rises more rapidly than CRP and can be detected in serum as early as 2–4 h after bacterial invasion peaking within 6–24 h with a half-life of approximately 24 h. 3 In health, PCT values are normally lower than 0.1 μg/L, 17 while concentrations <0.5 μg/L make systemic infection unlikely. 18 PCT has a sensitivity of 94% as an indicator of sepsis and is an effective biomarker for infection.18–20 A cut-off concentration of 0.25 μg/L has been proposed to guide antibiotic treatment. 21 PCT has primarily been studied in intensive care settings in risk assessment of critically ill and immunocompromised populations.22,23 The predictive value of PCT for the diagnosis of subclinical intrauterine infection in patients with premature rupture of membranes has been demonstrated to be superior to CRP. 24 In pregnancy, emerging evidence suggests that PCT complements clinical findings used to diagnosis sepsis in a more robust and timely fashion.25,26 This study sought to establish reference values for CRP and PCT at term pregnancy and in the immediate postpartum period.
Materials and methods
This prospective cross-sectional study of pregnant women was conducted over 12 months in a large tertiary teaching hospital with approximately 8000 births per annum. Participants were recruited at routine obstetric clinics in Cork University Maternity Hospital (CUMH). Sample size was based on the work of Paccolat et al. 27 These authors assessed PCT in pregnancy, delivery and the postpartum in 60 women without signs of clinical infection (31 vaginal deliveries [VDs], 29 caesarean sections [CSs]) recruited over a 10-month period. This research study was approved by the Clinical Research Ethics Committee of the Cork Teaching Hospitals, University College Cork, Ireland (ECM:4 (k) 04/2/2014). Informed written consent was obtained from all participants at time of recruitment. Participants consented to provide a blood sample and allow access to their clinical information.
Reference population
Healthy women, aged 19–45 years with an uncomplicated singleton pregnancy without any signs of clinical infection were recruited. Women having an Elective Lower Segment Caesarean Section (ELCS) or Emergency Lower Segment Caesarean section (EMCS) received one dose of prophylactic antibiotics intravenously before delivery. Those women who had normal or instrumental VD did not receive prophylactic antibiotics. Women were excluded if they had one or more of the following risk factors; prolonged labour, prolonged rupture of membranes (>18 h), temperature in labour >38°C, use of antibiotics in labour, maternal tachycardia >100 beats per min (bpm), fetal tachycardia >160bpm, maternal pyrexia >38°C, manual removal of placenta, third/fourth degree tear, offensive lochia, wound infection, bladder injury or prolonged catheterization, multiple pregnancy, known fetal congenital anomalies and neonatal sepsis.
Data collection
At the initial booking visit and following informed written consent, eligible participants had an early dating scan for an agreed estimated due date (EDD), BMI (kg/m2) calculated from weight and height, maternal age, parity, ethnicity and gestation of pregnancy in weeks recorded. Additionally, the following clinical data was recorded for the delivery cohort: onset of labour, mode of delivery, cord pH at birth, infant birth weight and Apgar score at 1 and 5 min. On the day of phlebotomy, gestational age (weeks and days) was confirmed sonographically. Venous blood (5 mL) was collected into appropriate specimen tubes (VACUETTE® Serum tubes – Greiner Bio-one, France) from study participants booked for induction or elective CS, at term (37–40 weeks’ gestation) and on days 1 and 3 postpartum. All samples for CRP and PCT analysis were transported immediately to the laboratory for processing (centrifugation 4000 g for 10 min and serum separated and frozen at –20°C within 4 h of blood draw) prior to analyses in a laboratory accredited to ISO15189:2012 standard for medical testing laboratories.
Analytical methods
CRP was measured in serum using the Beckman Coulter latex immune-turbidimetric assay on the Olympus AU640 Automated Clinical Chemistry analyser (Olympus Corporation, Tokyo, Japan), according to the manufacturer’s instructions. The assay principle is based on the formation of immune complexes during the antigen–antibody reaction that scatter light in proportion to the concentration of CRP in the sample. Traceability is to the International Federation of Clinical Chemistry standard CRM 470. Inter-assay precision at a mean CRP concentration of 6.0 mg/L, 64.8 mg/L and 137.7 mg/L was 1.85%, 3.35% and 1.64%, respectively.
PCT was measured in serum after a single freeze–thaw cycle using the BRAHMS PCT two-step sandwich immunoassay, a biotinylated anti-PCT capture antibody and a ruthenium-labelled anti-PCT tag antibody, on the Roche E-170 single modular analyser (Roche Diagnostics, Basel, Switzerland). The concentration of PCT in the sample is directly proportional to the chemiluminescent emission measured by the system photomultiplier. The method has been standardized against the BRAHMS PCT luminescence immunoassay. Inter-assay precision at a mean PCT concentration of 0.5 μg/L and 10.0 μg/L was 4%, and 2.7%, respectively.
Statistical analyses
Microsoft Excel 2016 and Minitab 2017 were used for data recording and statistical analyses. Continuous parametric data were represented as mean (±standard deviation) and non-Gaussian data, as median (interquartile range). Comparison of means was made using Student’s independent t-test or analysis of variance (ANOVA). For non-parametric data, the Mann-Whitney U test and Kruskal–Wallis test were used. Categorical data are summarized with frequencies (in percentage). For statistical purposes, CRP and PCT values were reported to the lower limit of detection of the assay, 0.2 mg/L and 0.01 μg/L, respectively. A P value <0.05 was considered statistically significant. Boxplots were used to illustrate the data for CRP/PCT in healthy pregnant women according to (a) mode of delivery and (b) day postpartum. The box portion of the box and whisker plot includes 50% of the data, the lower, median (represented by a line) and upper quartile. The whiskers extend to the minimum and maximum values. Disconnected points are potential outliers. Diagnostic specificity for PCT was calculated using the decision-threshold of 0.25 μg/L.
Results
A total of 191 pregnant women without signs of clinical infection were prospectively recruited to this study. Of these, two were excluded as they failed to meet the study inclusion criteria: (1) preterm delivery (at 32 weeks’ gestation), (2) Prolonged labour (maternal pyrexia in labour and fetal tachycardia). The reference population comprised of 189 pregnant women, 59 (37.1%) were primigravida and 33 (20.8%) had a history of previous CS. The study cohort was stratified into three groups: term pregnancy (37–40 weeks’ gestation, n = 51), day 1 postpartum (n = 108) and day 3 postpartum (n = 30), as shown in Figure 1.
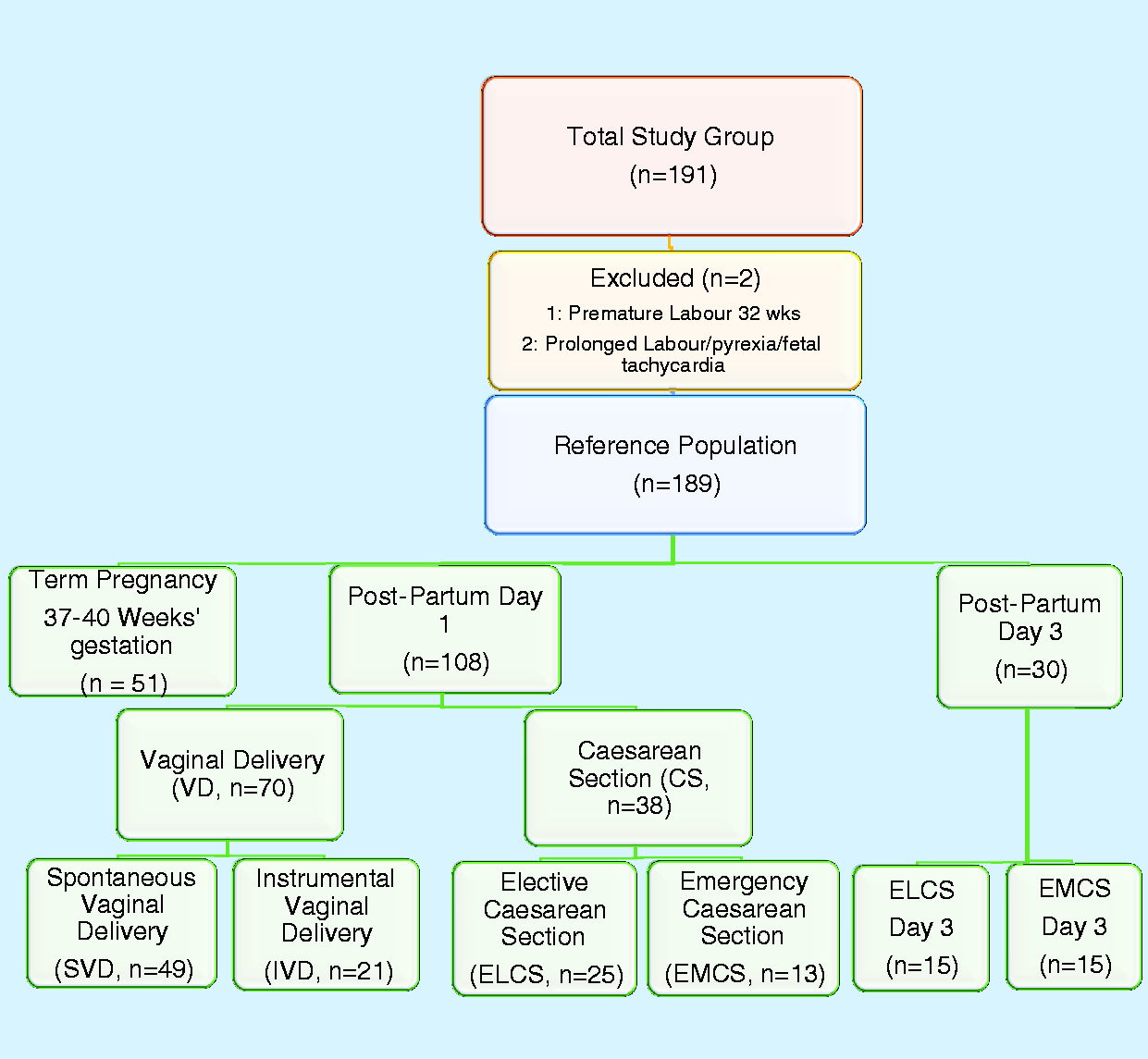
Study recruitment schematic stratified by mode of delivery and day postpartum.
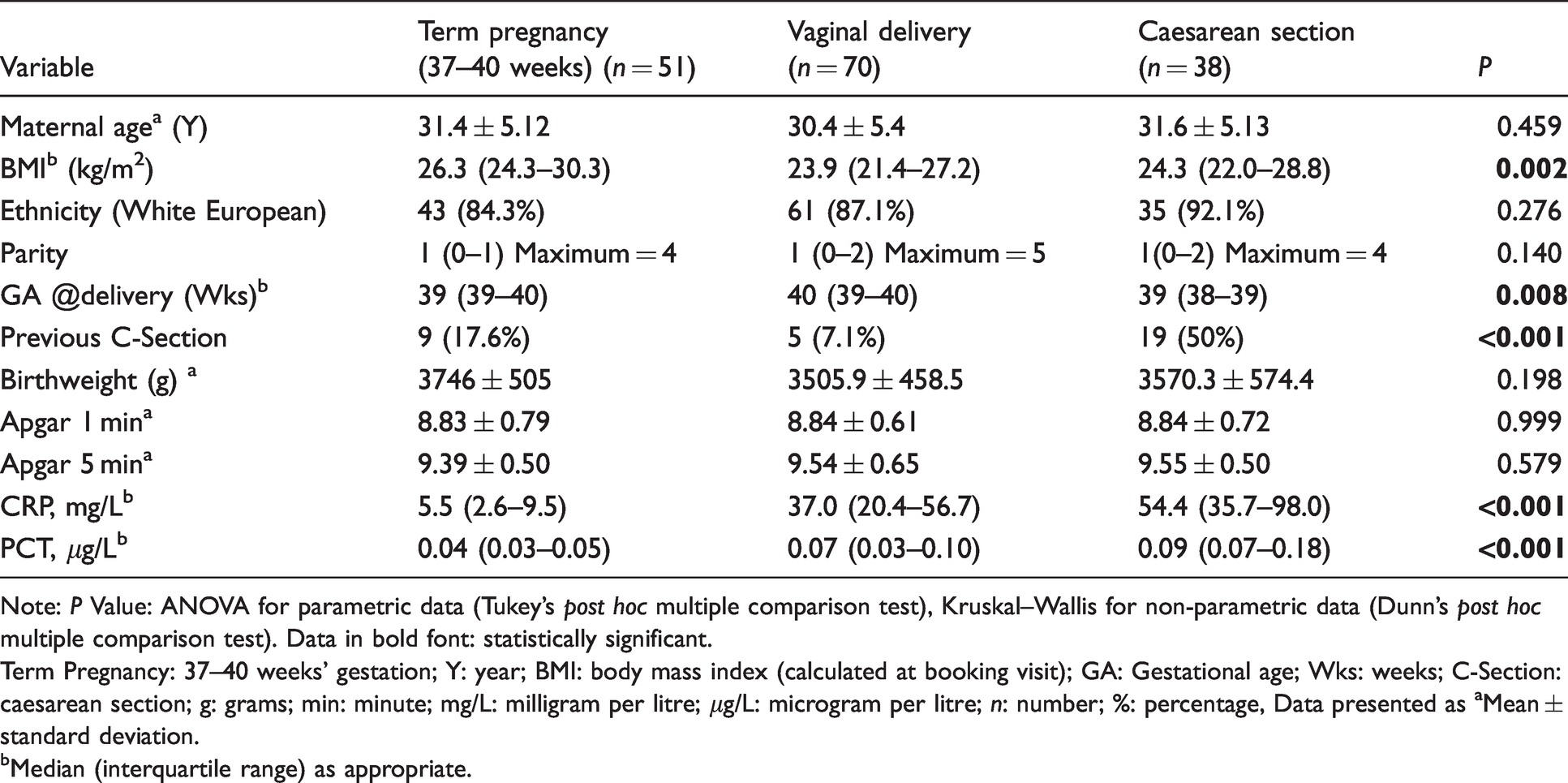
The postpartum day 1 delivery group was categorized according to mode of delivery: VD (n = 70) and CS (n = 38). The VD group was further subdivided into spontaneous vaginal delivery (SVD, n = 49) and instrumental vaginal delivery (IVD, n = 21) and the CS group into ELCS (n = 25) and EMCS (n = 13). The postpartum day 3 delivery group all had a CS and were further classified into ELCS (n = 15) and EMCS (n = 15). The main demographic and clinical characteristics of the study population according to mode of delivery are detailed in Table 1.
Demographic and clinical characteristics of the study population.
Note: P Value: ANOVA for parametric data (Tukey’s post hoc multiple comparison test), Kruskal–Wallis for non-parametric data (Dunn’s post hoc multiple comparison test). Data in bold font: statistically significant.
Term Pregnancy: 37–40 weeks’ gestation; Y: year; BMI: body mass index (calculated at booking visit); GA: Gestational age; Wks: weeks; C-Section: caesarean section; g: grams; min: minute; mg/L: milligram per litre; μg/L: microgram per litre; n: number; %: percentage, Data presented as aMean ± standard deviation.
bMedian (interquartile range) as appropriate.
BMI, gestational age (weeks), history of CS, CRP and PCT concentrations differed significantly between the study participants. BMI between the delivery cohort, VD and CS was non-significant (P = 0.408). BMI was significantly different between those at term pregnancy and VD (P≤0.001) and CS (P = 0.044). Gestational age in weeks was only significantly different between the VD vs. CS (P = 0.002). The history of CS was not significant between VD and term pregnancy (P = 0.076), but was highly significantly different between VD and CS (P < 0.001) and between CS and term pregnancy (P = 0.001).
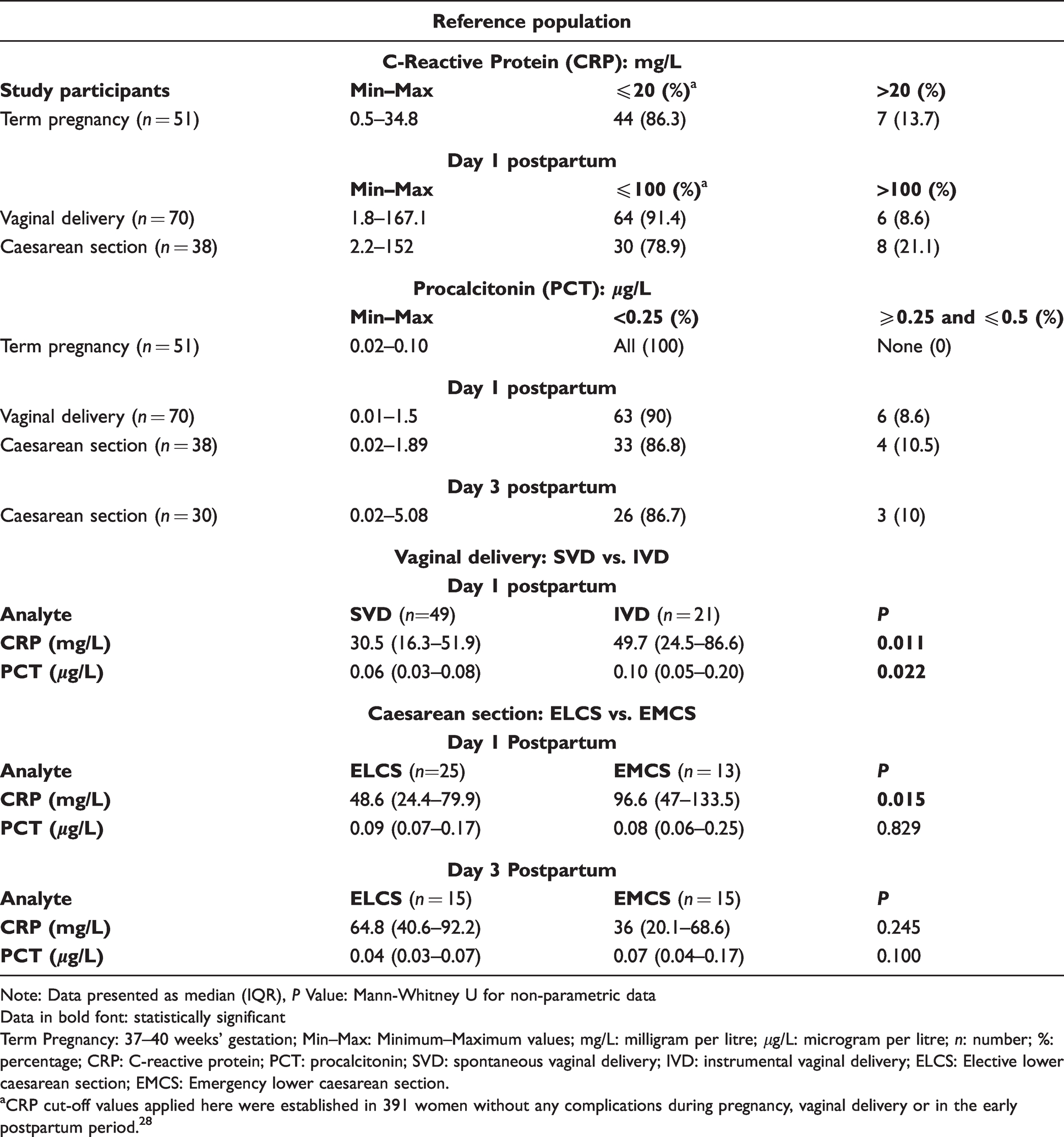
Reference values for CRP at term (37–40 weeks’ gestation) ranged from 0.5 to 34.8 mg/L as shown in Table 2. In the day 1 postpartum group, the maximum CRP concentration observed was 167 mg/L for VD compared with 152 mg/L for CS with concentrations <100 mg/L in 91.4% of VD and 78.9% of CS (P = 0.013). A significant difference in CRP values was also observed (P = 0.015) between women who underwent ELCS compared with EMCS, with higher values recorded in the EMCS cohort.
CRP and PCT in the reference population stratified according to mode of delivery and day postpartum.
Note: Data presented as median (IQR), P Value: Mann-Whitney U for non-parametric data
Data in bold font: statistically significant
Term Pregnancy: 37–40 weeks’ gestation; Min–Max: Minimum–Maximum values; mg/L: milligram per litre; μg/L: microgram per litre; n: number; %: percentage; CRP: C-reactive protein; PCT: procalcitonin; SVD: spontaneous vaginal delivery; IVD: instrumental vaginal delivery; ELCS: Elective lower caesarean section; EMCS: Emergency lower caesarean section.
aCRP cut-off values applied here were established in 391 women without any complications during pregnancy, vaginal delivery or in the early postpartum period. 28
The maximum PCT value at term (37–40 weeks’ gestation) was 0.1 μg/L. At day 1 postpartum, 90% and 86.8% of PCT values in the VD and CS groups, respectively, were <0.25 μg/L with two results >0.5 μg/L (VD:1.5 μg/L and CS:1.89 μg/L). On day 3 postpartum in the CS group, 86.7% of PCT results were <0.25 μg/L and in agreement with that found on day 1 postpartum (86.8%). Of these, two PCT results were greater than 0.5 μg/L (ELCS: 5.08 μg/L and EMCS: 4.12 μg/L). PCT concentrations were not impacted to the same extent as CRP by mode of delivery despite the fact that PCT concentrations in the postdelivery cohorts were higher compared with the term pregnancy cohort (P < 0.001). Concentrations of PCT were also significantly higher in women who had IVD compared with SVD (P = 0.022). Applying the common decision-thresholds for sepsis (CRP <20 mg/L, PCT <0.25 μg/L) to the SVD cohort, 29% (14/49) and 94% (46/49) of women, respectively, had values below these thresholds. Applying the cut-offs to the IVD cohort, 10% (2/21) and 81% (17/21) of women, respectively, satisfied the criteria. In the CS cohort, 16% (6/38) and 87% (33/38) women met the decision-thresholds. Women who had CS deliveries without any evidence of infection demonstrated the highest PCT concentrations when compared with VD (P = 0.009) as shown in Figure 2.
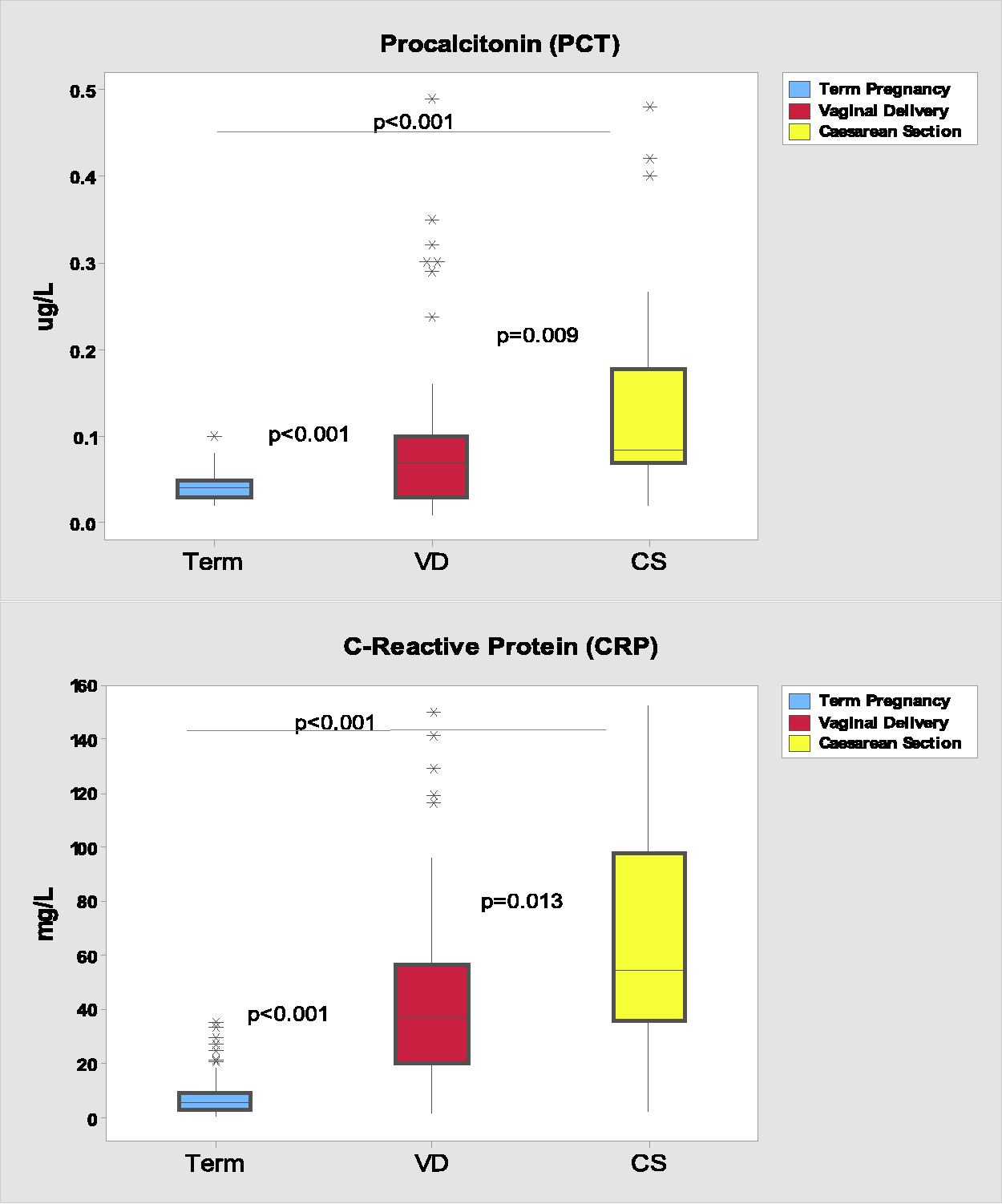
CRP and PCT values at term pregnancy and postpartum according to mode of delivery.
Discussion
This prospective study sought to define reference values for CRP and PCT in a well-characterized cohort of healthy pregnant women at term pregnancy (37–40 weeks’ gestation) and on day 1 and day 3 postpartum. However, CRP demonstrated high variability limiting its clinical utility in the assessment of systemic sepsis in this population. BMI was calculated at the booking visit and gestational age sonographically confirmed at time of blood sampling. Strict exclusion criteria were applied and 191 women were recruited with only two women requiring subsequent exclusion. Reference values for CRP in our study ranged from 0.5 to 34.8 mg/L at term and from 1.8 to 167 mg/L postpartum and were markedly higher than those cited for both the pregnant and non-pregnant adult. 9 Only 43.1% of women had a CRP value of <5 mg/L, the concentration expected in healthy adults. 28 Klajnbard et al. published reference values for CRP with a cut off in the third trimester (35–42 weeks’ gestation, n = 352) of 20 mg/L and at day 1 postpartum of 100 mg/L (n = 260). 29 Applying these CRP cut-offs to our cohort, determined that only 86.3% and 87% of results, respectively, met this criterion.
The term pregnancy group (37–40 weeks’ gestation) had a median PCT of 0.04 μg/L (IQR, 0.03–0.05). This is broadly in agreement with a recent study by Dockree et al., who established an upper reference limit (URL) for PCT of 0.05 μg/L in healthy women of gestational age 31–38+6 weeks’ selected from the Oxford Pregnancy Biobank. 30 PCT values in our term cohort ranged from 0.02 to 0.10 μg/L and are consistent with those determined by Paccolat et al. 27 (0.01–0.11 μg/L) in women at 36–40 weeks’ gestation. At term, 96% of PCT results were ≤0.10 μg/L, the URL quoted for health. 17 Using the decision-threshold of 0.25 μg/L to rule out infection provided 100% specificity at term. At day 1 postpartum, PCT values for VD and CS ranged from 0.01 to 1.89 μg/L, with 88.9% of results <0.25 μg/L, 98.1% of results <0.5 μg/L and all results <2.0 μg/L. 17 Based on these results, we obtained a diagnostic specificity of 91.5% in our reference population.
Traditional laboratory indicators such as cholesterol, white blood cell (WBC) count, leucocyte count, erythrocyte sedimentation rate (ESR) and lactate can lack sensitivity and specificity to identify bacterial infection/sepsis early and guide antibiotic treatment regimens. Use of blood culture alone to establish bacterial sepsis is slow and can take several days to confirm sepsis. CRP and PCT reference values in our study population differed markedly according to mode of delivery. Women who underwent CS had CRP and PCT values that were significantly higher when compared with those who had VD.
CRP is a universal marker of any inflammation 17 and non-specific for bacterial infection. 4 It is used in the diagnostic algorithm for sepsis with concentrations <5 mg/L 28 (or <3 mg/L) 9 considered normal in the non-pregnant adult. In early pregnancy (gestation age <18 weeks), median CRP values have been reported as 3.2 mg/L (interquartile range 0.3–12.1). 9 Gestational-age-specific reference intervals for CRP based on 2.5th–97.5th percentiles have been reported in women with uncomplicated pregnancy, at delivery and immediately postpartum. 29 However, reference values for CRP in the puerperium are not well established, although postpartum infection is common and difficult to recognize. 31 Applying previously established CRP cut-off values 29 to our term pregnancy group, only 43% women (22/51) had CRP results <5 mg/L and 86% women (44/51) had CRP results <20 mg/L. These results support the findings of a systematic review by Trochez-Martinez et al., which acknowledges an association between CRP concentrations and histological chorioamnionitis but finds no clear evidence to support its use as an early diagnostic marker of sepsis and suggests that use of commonly accepted CRP thresholds might be misleading. These authors suggest that using serial CRP estimations, concentrations ≥20 mg/L may be more predictive of infection. 32 A recent systematic review evaluating CRP at the 20 mg/L cut-off for chorioamnionitis in PPROM reports low sensitivity and specificity, 59% (95% CI: 48–69) and 83% (95% CI: 74–89), respectively. 33 Adoption of the 20 mg/L decision-threshold in the current study would result in 14% of healthy pregnancies being exposed to unnecessary investigations and interventions. We also determined that CRP concentrations were influenced by the mode of delivery. CRP concentrations increase concomitantly with the normal physiological changes that ensue on progression of healthy pregnancy, with further increases observed in the puerperium, and this complicates its interpretation and limits its use as a marker of infection in this cohort.10–13
During normal pregnancy, extra villous trophoblast and decidual stromal cells synthesize PCT. 34 In the days before and after delivery, physiological and immune changes in pregnant women trigger the induction of inflammatory markers such as PCT. High concentrations of PCT have also been associated with abdominal and heart surgery, while small surgeries or slight trauma has demonstrated low or absent PCT serum concentrations. 35 In the current study, healthy women who underwent CS deliveries had the highest PCT concentrations when compared with VD. It is likely that surgical intervention stimulates the inflammatory cascade and this may explain the higher PCT concentrations observed post IVD and CS compared with SVD. Serum PCT concentrations have been shown to decrease faster than other inflammatory biomarkers after surgery which is important for an accurate diagnosis of bacterial infection. 36 PCT concentrations of 0.5–2.0 μg/L are indicative of possible systemic infection, with values between 2.0 and 10 μg/L highly indicative of sepsis. 4 It has been mooted that PCT is especially useful for detecting gram-negative bacteraemia. Of note, gram-negative E. coli is the most commonly implicated pathogen in blood cultures in severe obstetric infections. 1
Paccolat et al., in a study of 60 clinically well pregnant women, determined that the PCT decision-threshold of 0.25 μg/L could be used during the third trimester, at delivery and the immediate postpartum to rule out infection. 27 In our prospective study, PCT values in all 51 healthy term pregnancies were ≤0.1 μg/L as expected in health. 17 , 37 The calculated diagnostic specificity of PCT was 91.5% and almost identical to that reported by Paccolat et al. (91.1%) in a smaller patient cohort. 27 Our findings further substantiate the use of the PCT decision-threshold of <0.25 μg/L to rule out infection at term and in the early postpartum period.
In this study, CRP and PCT cut-offs were defined using the Beckman Coulter and BRAHMS assays, respectively. Notwithstanding, it must be acknowledged that currently there is no international reference standard for PCT and that despite the availability of the CRP Certified Reference Material (ERM/DA472/IFCC), CRP assays do not always produce equivalent values with some demonstrating a significant negative bias. 38 Together, these factors have the potential to influence the diagnostic performance of the respective suggested cut-offs and mandates the use of method-specific decision-thresholds for both PCT and CRP.
A potential limitation of the current study is that data was not collected from pregnant women with infectious complications, prohibiting the assessment of diagnostic sensitivity. In addition, collection of day 3 bloods from women post VD was not possible as women were discharged within two days of giving birth. We acknowledge that the kinetics of PCT are of particular diagnostic and prognostic importance, and that serial PCT measurements might be more useful for diagnosing infection than a once off measurement.4,39 Serial measurements of CRP and PCT were only available in the CS group.
Conclusion
This study demonstrates that CRP values vary widely in healthy uncomplicated pregnancies and in the early postpartum period. This variability together with the diversity of published CRP cut-off values to predict infection in pregnancy and the puerperium, limit its clinical utility in the assessment of systemic sepsis.32,33,40 In contrast, quantitation of PCT has greater diagnostic specificity and is therefore less likely to produce false positive results with the potential to lead to inappropriate intervention. The findings of this study support the use of a PCT decision-threshold of 0.25 μg/L to rule out infection at term pregnancy and in the immediate postnatal period. Further research is required to validate the clinical utility of PCT to diagnose pregnancies complicated by sepsis.
Footnotes
Acknowledgements
We wish to express our gratitude to the women, nursing and medical staff of Cork University Maternity Hospital and to the scientific staff in Cork University Hospital who made this study possible. Special thanks to Mary Stapleton, consultant clinical biochemist for her valuable advice and helpful discussion on this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This research study was approved by the Clinical Research Ethics Committee of the Cork Teaching Hospitals, University College Cork, Ireland (ECM:4 (k) 04/2/2014).
Guarantor
KOD.
Contributorship
KOD and CJ: study concept and design. HA: study conduct, protocol development. KOD: supervision of patient recruitment and study conduct. YL: patient recruitment and data acquisition. SD: laboratory analysis and data acquisition. CJ: responsibility for laboratory analysis and data integrity. POS: Data analysis and interpretation. CJ and KOD: First draft of this article. CJ, POS and KOD critically reviewed and edited the article. All authors approved the final version of the article.
