Abstract
Background
Kisspeptin is an arginine-phenylalanine amide peptide hormone critical for reproductive function. Kisspeptin is also abundantly expressed in the placenta, where it has an important physiological role in regulating placental invasion. Accordingly, plasma kisspeptin concentrations rise dramatically during normal pregnancy. However, lower plasma concentrations of kisspeptin are associated with obstetric complications such as pre-eclampsia. It is not currently known whether kisspeptin-immunoreactivity (IR) can be detected in bodily fluids not requiring invasive collection such as saliva or urine.
Aim
To determine the clinical utility of urinary and salivary kisspeptin measurement in healthy pregnant women.
Methods
Forty-nine healthy third trimester pregnant women (gestational age 34 ± 0.6 w) from a single maternity unit and 50 healthy non-pregnant women were recruited. Urine, saliva and blood were simultaneously collected from all volunteers. Kisspeptin concentrations were determined by in-house manual radioimmunoassay.
Results
Mean concentrations of plasma kisspeptin-IR were over 200-fold greater in third trimester pregnant women compared with non-pregnant women (13,783 ± 864 pmol/L, pregnant; 65 ± 13 pmol/L, non-pregnant; p < 0.0001). The urine kisspeptin:creatinine ratio was greater in pregnant women when compared with non-pregnant women (urine kisspeptin:creatinine: 37 ± 6 pmol/µmol, pregnant; 7 ± 1 pmol/µmol, non-pregnant; p < 0.0001). Mean concentrations of salivary kisspeptin-IR were not statistically different between pregnant and non-pregnant women (123 ± 34 pmol/L, pregnant; 83 ± 33 pmol/L, non-pregnant; p = 0.2).
Conclusion
We demonstrate for the first time that kisspeptin-IR is elevated in urine during pregnancy. Urinary measurement of kisspeptin-IR may, therefore, offer a non-invasive and simple method of screening for pregnancy and obstetric complications.
Introduction
Kisspeptin is an arginine-phenylalanine amide peptide hormone originally identified as an anti-metastatic factor in human melanoma cells, which plays an important physiological role in regulating placental development. 1 Kisspeptin is abundantly expressed in placental syncytiotrophoblast cells, where it regulates placental invasion into uterine tissue. Accordingly, circulating concentrations of kisspeptin rise dramatically during normal human pregnancy approximately 7000-fold when compared with non-pregnant human subjects. Reduced placental expression of kisspeptin or low circulating concentrations of kisspeptin are associated with poor pregnancy outcomes such as recurrent miscarriage, intrauterine growth restriction and pre-eclampsia. 2 Plasma kisspeptin measurement, therefore, offers a potential novel clinical marker of human pregnancy and obstetric complications.
We have previously used an in-house radioimmunoassay to quantify plasma and amniotic fluid concentrations during normal human pregnancy. 3 We have previously observed that plasma kisspeptin-IR concentrations do not fluctuate significantly during 8 h time-course studies in healthy women. 4 However, it is not known whether kisspeptin-immunoreactivity (IR) can also be detected during pregnancy in bodily fluids not requiring invasive collection, such as urine and saliva. Developing such non-invasive methods for measuring concentrations of kisspeptin-IR has the potential clinical utility of monitoring the risk of complications during pregnancy in a safe, easy and acceptable fashion. We, therefore, compared for the first time, concentrations of kisspeptin-IR in plasma, urine and saliva between pregnant and non-pregnant women.
Methods
Following ethical approval (West London Research Ethics Committee number 06/Q0406/12), 49 pregnant women and 50 non-pregnant women were recruited from clinics at Imperial College Healthcare NHS Trust, London. All pregnant subjects were in their third trimester of singleton pregnancy (gestational age in weeks: 34 ± 0.6, mean; 28–41, range). Subjects with renal failure or serious comorbidity were excluded. Urine, saliva and blood were simultaneously collected from all subjects in sterile containers (for urine and saliva) or lithium heparin tubes (blood, Beckton Dickson, Franklin Lakes, USA) each containing 5000 kallikrein inhibitor units of aprotinin (0.2 ml Trasylol; Bayer, UK). To reduce pre-analytical factors known to influence radioimmunoassay measurement of kisspeptin-IR, all samples were collected and stored in identical fashion. 5 Plasma, urine and saliva samples from all subjects were stored at −20℃ and then assayed together in a single experiment. Measurement of kisspeptin-IR was performed using a previously described in-house radioimmunoassay without peptide extraction.4–6 Mean recovery of kisspeptin in urine samples was 81.6%. Inter- and intra-assay coefficient of variation were 14.0% and 10.9%, respectively. Thawed plasma samples and the supernatant of thawed urine and saliva samples following centrifugation were directly assayed in duplicate. To standardize urine values for specimen concentration, a ratio of kisspeptin concentration divided by creatinine concentration was used. Pregnant and non-pregnant concentrations were compared using unpaired t test. Correlations were assessed using Pearson’s R test. Data are presented as mean ± SEM.
Results
Mean concentrations of plasma kisspeptin-IR were over 200-fold greater in third trimester pregnant when compared with non-pregnant women (13,783 ± 864 pmol/L, pregnant; 65 ± 13 pmol/L, non-pregnant; p < 0.0001, Figure 1). Mean concentrations of salivary kisspeptin-IR were not statistically different between pregnant and non-pregnant women (123 ± 34 pmol/L, pregnant; 83 ± 33 pmol/L, non-pregnant; p = 0.2, Figure 1). Mean concentrations of urine kisspeptin-IR were greater in pregnant women when compared with non-pregnant women (301 ± 59 pmol/L, pregnant; 80 ± 19 pmol/L, non-pregnant; p < 0.001, Figure 1). The urine kisspeptin:creatinine ratio was also significantly greater in pregnant women when compared with non-pregnant (37 ± 6 pmol/µmol, pregnant; 7 ± 1 pmol/µmol, non-pregnant; p < 0.0001, Figure 1). No correlation was observed between gestation of pregnancy and either concentrations of plasma, urine or salivary kisspeptin-IR (gestation vs. plasma kisspeptin-IR r = 0.05, p = 0.37; vs. urine kisspeptin:creatinine r = −0.21, p = 0.07; vs. saliva kisspeptin-IR r = −0.09, p = 0.28). A positive correlation was observed between urine kisspeptin-IR and urine creatinine concentration (r = 0.32, p < 0.05).
Plasma, urine and salivary kisspeptin-immunoreactivity (IR, pmol/L) and urine kisspeptin:creatinine ratio (pmol/µmol) in healthy women in the third trimester of pregnancy (n = 49) compared to healthy non-pregnant women (n = 50). Data presented as mean ± SEM. ***p < 0.001.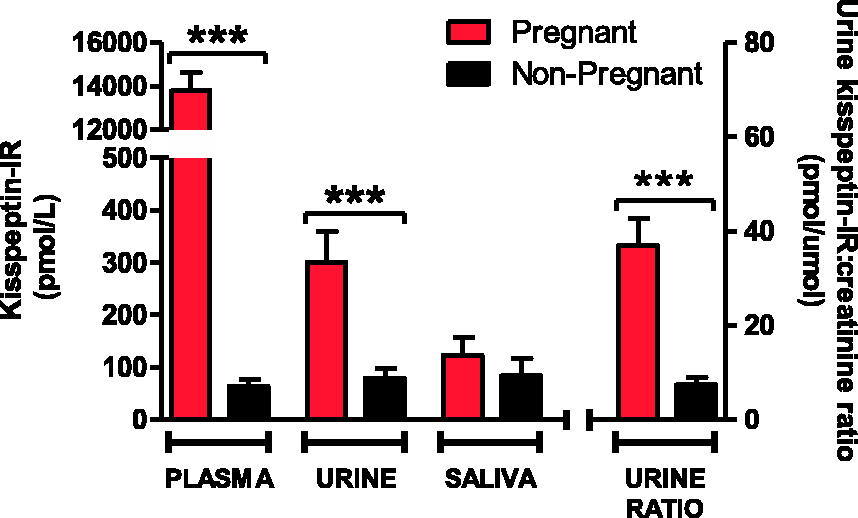
Conclusion
A number of recent studies have demonstrated that low concentrations of plasma kisspeptin are associated with serious adverse obstetric outcomes. 2 Kisspeptin is, therefore, a potential novel marker to identify women at high risk of obstetric complications. However, until now the only method of measuring kisspeptin concentration during pregnancy has been plasma collection or amniocentesis. Urinary measurement of β-human chorionic gonadotropin is an established marker of pregnancy. We, therefore, investigated whether urinary measurement of kisspeptin could provide an additional urinary obstetric biomarker. Urine kisspeptin-IR as well as urine kisspeptin:creatinine ratio was significantly greater in pregnant compared to non-pregnant women. Our data suggest that kisspeptin-IR is elevated in the urine, but not saliva, during pregnancy. Urinary measurement of kisspeptin-IR may, therefore, offer a non-invasive and simple method of screening, which is particularly suited to busy clinical settings such as obstetric clinics. Furthermore, the positive correlation between urine kisspeptin-IR and urine creatinine suggests that measuring urine kisspeptin:creatinine ratio may be preferable to account for inter- and intra-individual differences in concentrating urine.
Concentrations of kisspeptin-IR were much lower in the urine of pregnant women when compared with plasma concentrations in the same subjects. This might reflect incomplete filtration of kisspeptin by the kidney, or might reflect breakdown following urinary filtration. We also observed that in non-pregnant subjects, mean kisspeptin-IR was slightly higher in the urine when compared with the plasma (80 vs. 65 pmol/L); this could be due to the accumulation of low circulating concentrations of kisspeptin in the bladder. However, it is possible that a degree of assay interference may have accounted for the slightly increased concentrations of kisspeptin-IR in the urine of non-pregnant subjects when compared with plasma of non-pregnant subjects. It would also be interesting to subsequently determine the stability of kisspeptin-IR in urine, in addition to the dynamic changes of kisspeptin-IR in urine during different trimesters of pregnancy.
This pilot study was performed in women during the third trimester of pregnancy, when concentrations of plasma kisspeptin-IR are known to be highest. Unsurprisingly, no significant correlation between kisspeptin-IR and the narrow range of gestational age studied was observed. However, it would be interesting to compare concentrations of urine and plasma kisspeptin-IR during all gestational ages.
In summary, our data suggest for the first time that kisspeptin-IR is elevated in urine during the third trimester of human pregnancy. Further work is needed to determine if urinary kisspeptin measurements could be used to as a novel screening tool in obstetric healthcare.
Footnotes
Author contributions
Channa N Jayasena and Alexander N Comninos are joint first authors.
Declarations of conflicting interests
The authors declare that there is no conflict of interest.
Funding
The department is funded by an Integrative Mammalian Biology Capacity Building Award and NIHR Biomedical Research Centre Funding Scheme. CNJ is supported by an NIHR Clinical Lectureship and an AMS/Wellcome Starter Grant for Clinical Lecturers. ANC and AA are supported by Wellcome Trust Training Fellowships. GMNK is supported by an NIHR Clinical Lectureship. WSD is supported by an NIHR Career Development Fellowship.
Ethical approval
The West London Research Ethics Committee approved this study (REC Number: 06/Q0406/12).
Guarantor
WSD.
Contributorship
CJ and WSD conceived the study and wrote the first draft of the manuscript. SN and ZM collected the samples. CJ, AC, MC, MG and SB optimized the manual radioimmunoassay. CJ, AC, SN, AA, GN, MC and ZM carried out the laboratory analysis. AC performed the data analysis and drafted the results and figure. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
