Abstract
Objective
Myonectin, a newly discovered myokine, enhances fatty acid uptake in cultured adipocytes and hepatocytes and suppresses circulating concentrations of free fatty acids in mice. This study is performed to evaluate the association between serum myonectin concentrations with the presence and severity of OSAS.
Methods
This study was performed in a population of 191 patients with OSAS and 105 control subjects. Serum myonectin concentrations were measured using enzyme-linked immunosorbent assay method.
Results
Lower serum myonectin concentrations were found in OSAS patients than in the controls. Serum myonectin concentrations were associated with a reduced risk of OSAS (OR: 0.988, 95% CI: 0.984–0.993, P < 0.001). Severe OSAS patients had significantly lower myonectin concentrations compared with mild and moderate OSAS patients (P < 0.001 and P = 0.001, respectively). There are lower serum myonectin concentrations in moderate patients compared with mild patients (P = 0.024). Pearson correlation analysis revealed that serum myonectin concentrations were negatively correlated with the severity of OSAS (r = −0.344, P < 0.001). Simple linear regression analysis showed that serum myonectin concentrations in OSAS patients were negatively correlated with body mass index, fasting plasma glucose, homeostasis model assessment of insulin resistance (HOMA-IR), total cholesterol, low-density lipoprotein cholesterol (LDL-C) and apnoea hypopnea index. Multiple stepwise regression analysis shows that body mass index (β = −0.289, P = 0.03), HOMA-IR (β = −0.19, P = 0.003), total cholesterol (β = −0.155, P = 0.016), LDL-C (β = −0.176, P = 0.006) and apnoea hypopnea index (β = −0.263, P < 0.001) remained to be associated with serum myonectin.
Conclusion
Serum myonectin concentrations are inversely correlated with the presence and severity of OSAS.
Introduction
Obstructive sleep apnoea syndrome (OSAS) is a highly prevalent disease characterized by the cessation of respiration during sleep because of obstruction of the upper airway. 1 OSAS affects approximately 3–7% adult male and 2–5% adult female. 2 OSAS contributes to an increased risk for cardiovascular and cerebrovascular conditions and events, leading to increased morbidity and mortality. 3 The exact mechanism of OSAS is unclear. One potential mechanism for OSAS is the activation of inflammation. 4 OSAS has indeed been associated with elevated concentrations of various circulating inflammatory markers.
Myonectin, also called as CTRP15—C1q/TNF-related protein, promotes fatty acid uptake in cultured adipocytes and hepatocytes and inhibits circulating concentrations of free fatty acids in mice. 5 Myonectin suppressed inflammatory response to lipopolysaccharide in cultured macrophages through the S1P/cAMP/Akt-dependent signalling pathway. 6 Inflammation is one potential mechanism of OSAS. Therefore, myonectin is speculated to protect against the pathogenesis of OSAS by inhibiting inflammation.
To our knowledge, the role of myonectin in the pathogenesis of OSAS has not been examined previously. The present study is designed to determine the association between serum myonectin concentrations and OSAS.
Materials and methods
Patients
A consecutive of 191 male population with clinical symptoms of OSAS, who was examined by polysomnography (PSG) in our hospital, was enrolled in this study. As the majority of the OSAS patients in our hospital were male, we just enrolled male patients in the present study. The exclusion criteria included psychiatric disorders, history of alcohol and drug abuse, and systemic illnesses, such as cardiovascular, cerebrovascular, pulmonary or neuromuscular disease. One hundred five volunteers without clinical symptoms of OSAS were recruited as the controls. Subjects with systemic illnesses such as heart failure, chronic renal failure and chronic obstructive pulmonary disease were excluded from the control group. The control group matched with OSAS patients in age and body mass index (BMI).
The study protocol was approved by the Research Ethics Committee of our hospital, and all patients gave their informed consent before study commencement.
Sleep study
Full PSG monitoring was performed in all participants. OSAS was diagnosed according to clinical symptoms such as excessive daytime sleepiness, unexplained daytime fatigue, choking or gasping during sleep and an apnoea hypopnea index (AHI) of ≥5 events/h on PSG. The cessation of breathing for at least 10 s was considered as apnoea, and a 50% decrease in nasal airflow with at least a 4% decrease in oxygen saturation was considered as hypopnea. AHI was defined as the average frequency of apnoea and hypopnea in 1 h. OSAS patients were then divided into three subgroups: mild OSAS (AHI ≥5 and <15, n = 46), moderate OSAS (AHI ≥15 and <30, n = 94) and severe OSAS (AHI ≥30, n = 51) according to AHI. Subjects with AHI <5 were included in the control group.
Measurements
During the initial examinations, the height, weight and blood pressure of the subjects were measured. Venous blood was collected after a minimum of 10 h of fasting. Fasting plasma glucose (FPG) was assayed by using the glucose oxidase method (7600–120; Hitachi, Tokyo, Japan). Serum triglycerides (TG), total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C) and low-density lipoprotein cholesterol (LDL-C) were determined by applying standard enzymatic methods using a biochemical analyser (7600–120; Hitachi, Tokyo, Japan). Serum myonectin concentrations were determined using an enzyme-linked immunosorbent assay (Code Number: SK00393-15, Aviscera Biosciences, Santa Clara, CA). BMI was calculated as weight in kilograms divided by height squared in meters (kg m−2). Homeostasis model assessment of insulin resistance (HOMA-IR) was calculated as insulin reading (µIU/mL) multiplied by plasma glucose concentration (mmol/L) and divided by 22.5.
Statistical analysis
Statistical analysis was carried out using SPSS version 17.0 software program (SPSS Inc., Chicago, Illinois). The results were expressed as means ± standard deviation or median (interquartile range). The characteristics of OSAS patients were compared with the controls through unpaired t-test or Mann–Whitney U test. Logistic regression analysis was utilized to calculate the odds ratio values (OR) and 95% confidence intervals (CI) for the presence of OSAS. ROC curve analysis was used to determine the cut-off value serum myonectin to distinguish OSAS from the controls. Kruskal–Wallis test was used to compare the differences in serum myonectin concentrations among the controls, and patients with mild, moderate and severe OSAS. The correlation between serum myonectin and the severity of OSAS were analysed using Pearson correlation analysis. Simple and multiple linear regression analysis was performed to identify whether there was a correlation between serum myonectin and other variables. Statistical significance was set as P < 0.05.
Results
Baseline clinical characteristics
OSAS patients had elevated systolic blood pressure (SBP), diastolic blood pressure (DBP), FPG, HOMA-IR, TG and AHI, as well as lower HDL-C compared with the controls (Table 1).
Clinical and biochemical characteristics of OSAS patients and healthy controls.
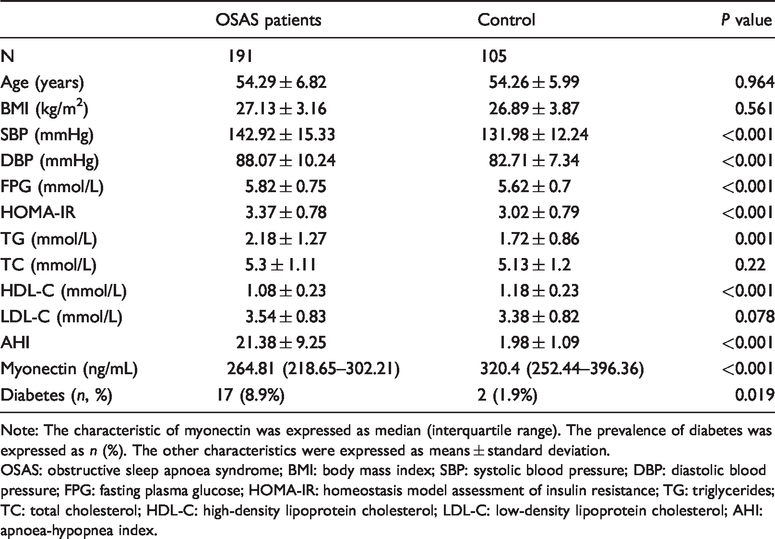
Note: The characteristic of myonectin was expressed as median (interquartile range). The prevalence of diabetes was expressed as n (%). The other characteristics were expressed as means ± standard deviation.
OSAS: obstructive sleep apnoea syndrome; BMI: body mass index; SBP: systolic blood pressure; DBP: diastolic blood pressure; FPG: fasting plasma glucose; HOMA-IR: homeostasis model assessment of insulin resistance; TG: triglycerides; TC: total cholesterol; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; AHI: apnoea-hypopnea index.
Serum myonectin concentrations in OSAS patients
Serum myonectin concentrations were significantly lower in OSAS patients compared with the controls (P < 0.001) (Table 1). Simple logistic regression analysis indicated that SBP, DBP, FPG, HOMA-IR, TG, HDL-C and serum myonectin showed a trend toward an association with the presence of OSAS (Table 2). All these parameters were then entered into a multivariate logistic regression model. Serum myonectin concentrations remained to be adversely associated with the presence of OSAS (OR: 0.988, 95% CI: 0.984–0.993; P < 0.001) (Table 2).
Logistic regression analysis for the presence of OSAS.
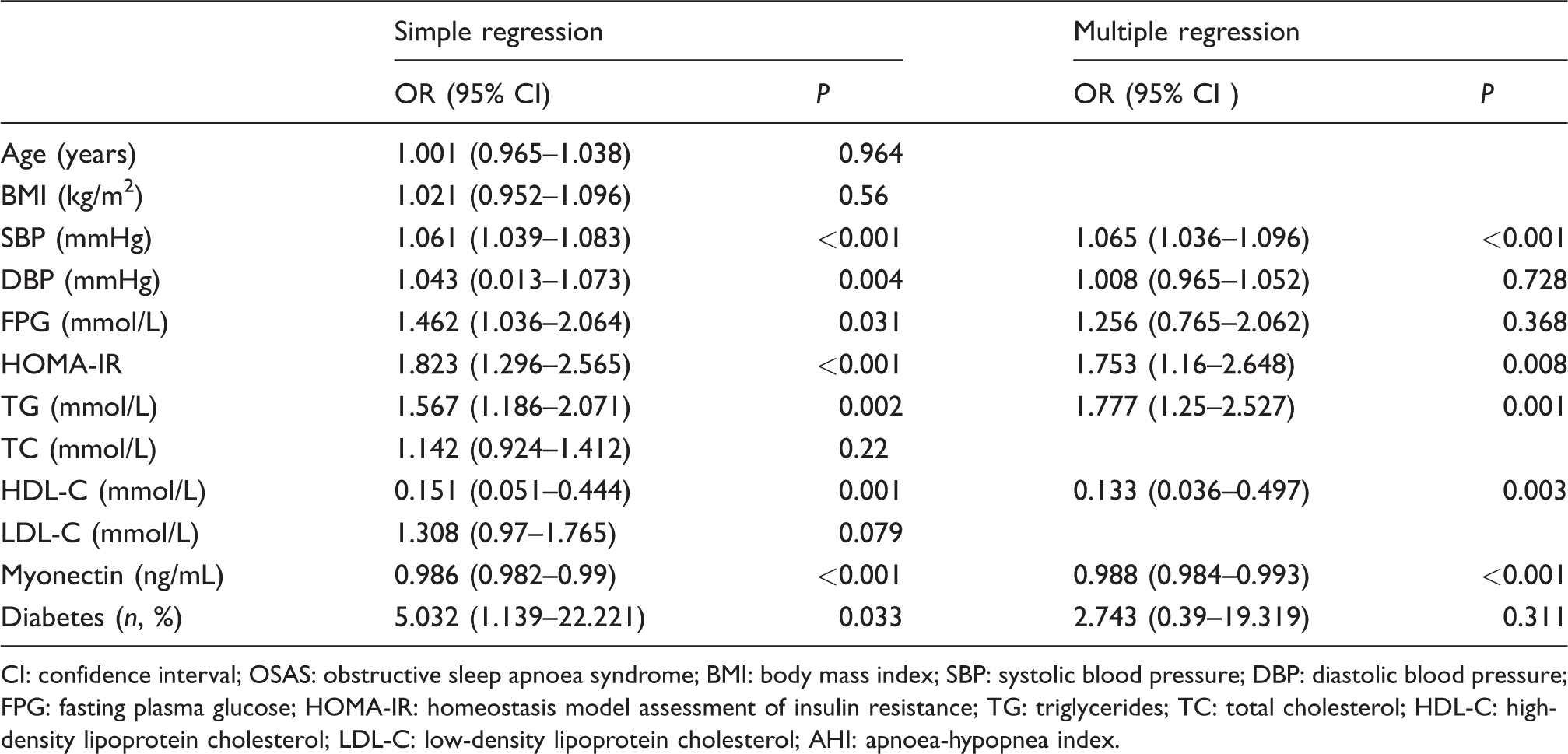
CI: confidence interval; OSAS: obstructive sleep apnoea syndrome; BMI: body mass index; SBP: systolic blood pressure; DBP: diastolic blood pressure; FPG: fasting plasma glucose; HOMA-IR: homeostasis model assessment of insulin resistance; TG: triglycerides; TC: total cholesterol; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; AHI: apnoea-hypopnea index.
The cut-off value of serum myonectin to diagnose OSAS
The ROC curve analysis showed that serum myonectin concentration of 318.55 ng/mL could be used as a cut-off value to distinguish OSAS from the controls (Figure 1), responding a sensitivity of 52.4% and a specificity of 86.9% (area under the ROC curve (AUC) = 0.723, 95% CI: 0.659–0.787, P < 0.0001).
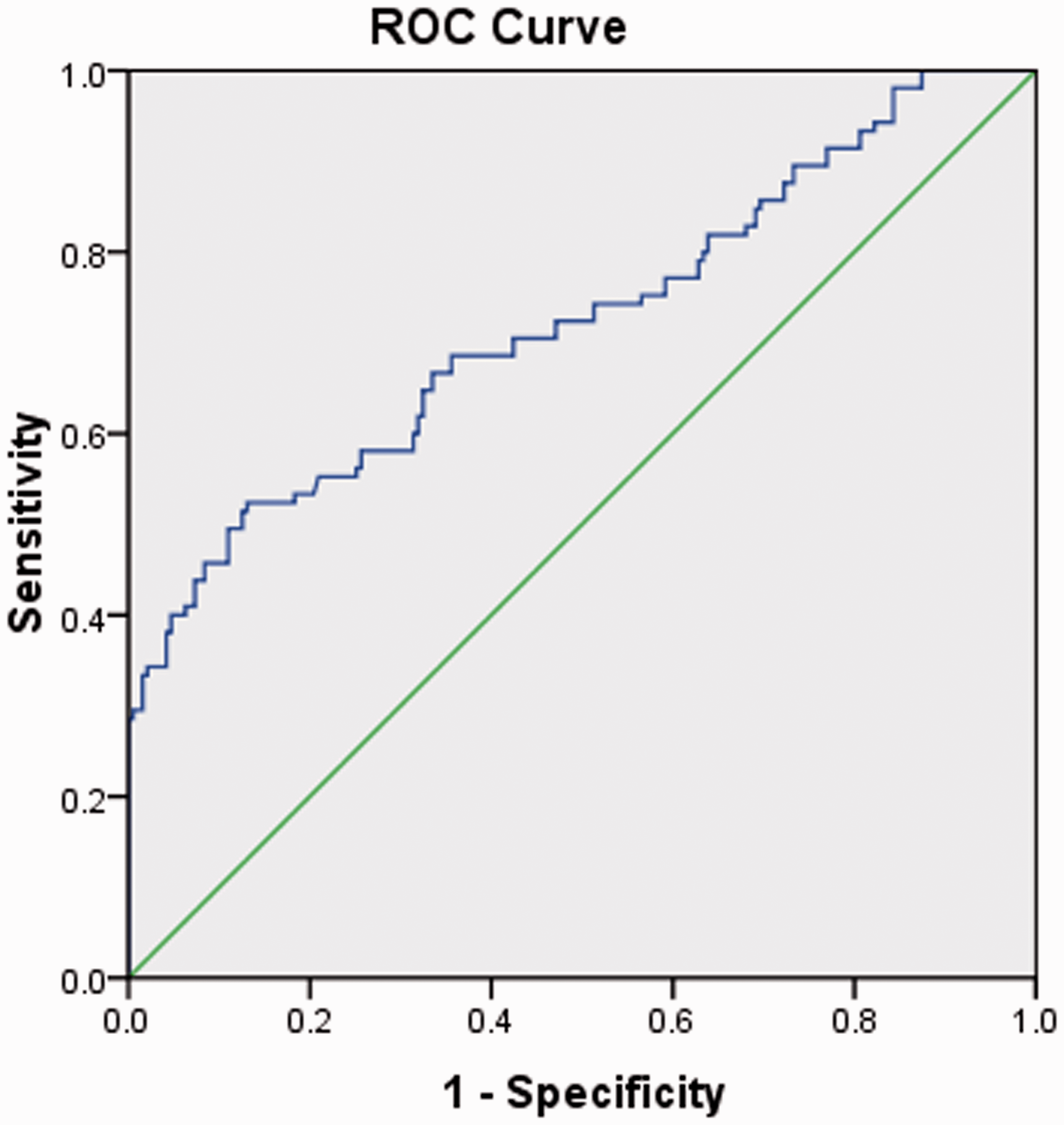
The ROC analysis showing serum myonectin concentration to be used to distinguish OSAS patients from the controls.
The correlation of serum myonectin concentrations with the severity of OSAS
Mild, moderate and severe OSAS patients had significantly lower serum myonectin concentrations compared with the controls (P = 0.009, P < 0.001 and P < 0.001, respectively). Severe OSAS patients showed significantly lower serum myonectin concentrations compared with mild and moderate OSAS patients (P < 0.001 and P = 0.001, respectively) (Figure 2). Furthermore, there were lower serum myonectin concentrations in moderate patients compared with mild patients (P = 0.024) (Figure 2). Pearson correlation analysis suggested that serum myonectin concentrations were negatively correlated with the severity of OSAS (r = −0.344, P < 0.001).
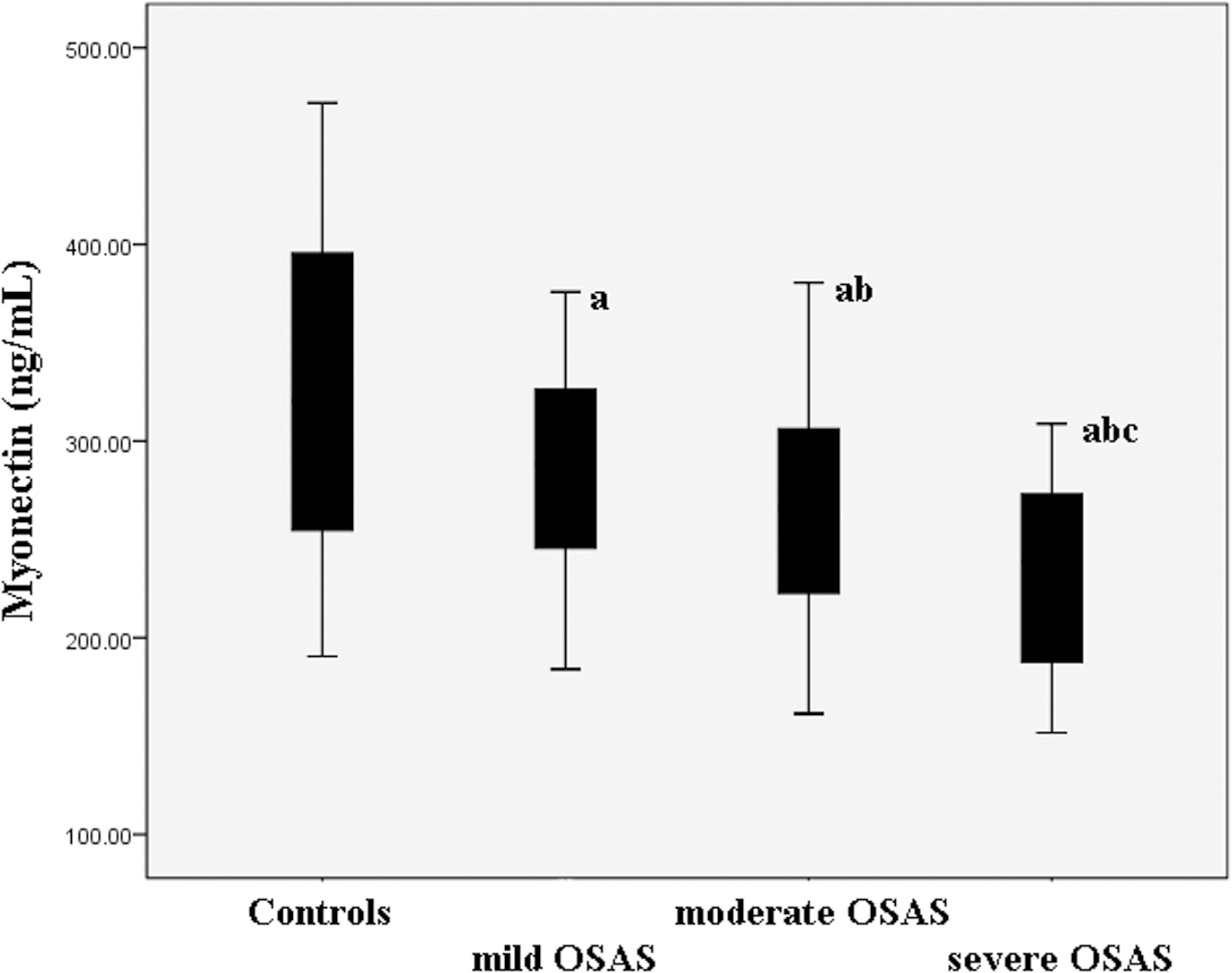
Serum myonectin concentrations in the controls as well as mild, moderate and severe OSAS patients. Mild, moderate and severe OSAS patients had significantly lower serum myonectin concentrations compared with the controls (P = 0.009, P < 0.001 and P < 0.001, respectively). Severe OSAS patients showed significantly lower serum myonectin concentrations compared with mild and moderate OSAS patients (P < 0.001 and P = 0.001, respectively). Furthermore, there are lower serum myonectin concentrations in moderate patients compared with mild patients (P = 0.024).
The association of serum myonectin concentrations with other clinical characteristics
Simple linear regression analyses showed that serum myonectin concentrations in OSAS patients were negatively correlated with BMI (r = −0.315, P < 0.001), FPG (r = −0.162, P = 0.025), HOMA-IR (r = −0.232, P = 0.001), TC (r = −0.193, P = 0.007), LDL-C (r = −0.189, P = 0.009) and AHI (r = −0.297, P < 0.001) (Table 3). Then variables including BMI, FPG, HOMA-IR, TC, LDL-C and AHI were incorporated into the stepwise linear regression model. Multiple stepwise regression analysis shows that BMI (β = −0.289, P = 0.03), HOMA-IR (β = −0.19, P = 0.003), TC (β = −0.155, P = 0.016), LDL-C (β = −0.176, P = 0.006) and AHI (β = −0.263, P < 0.001) remained to be associated with serum myonectin.
Linear and multiple regression analyses between serum myonectin and other clinical parameters.
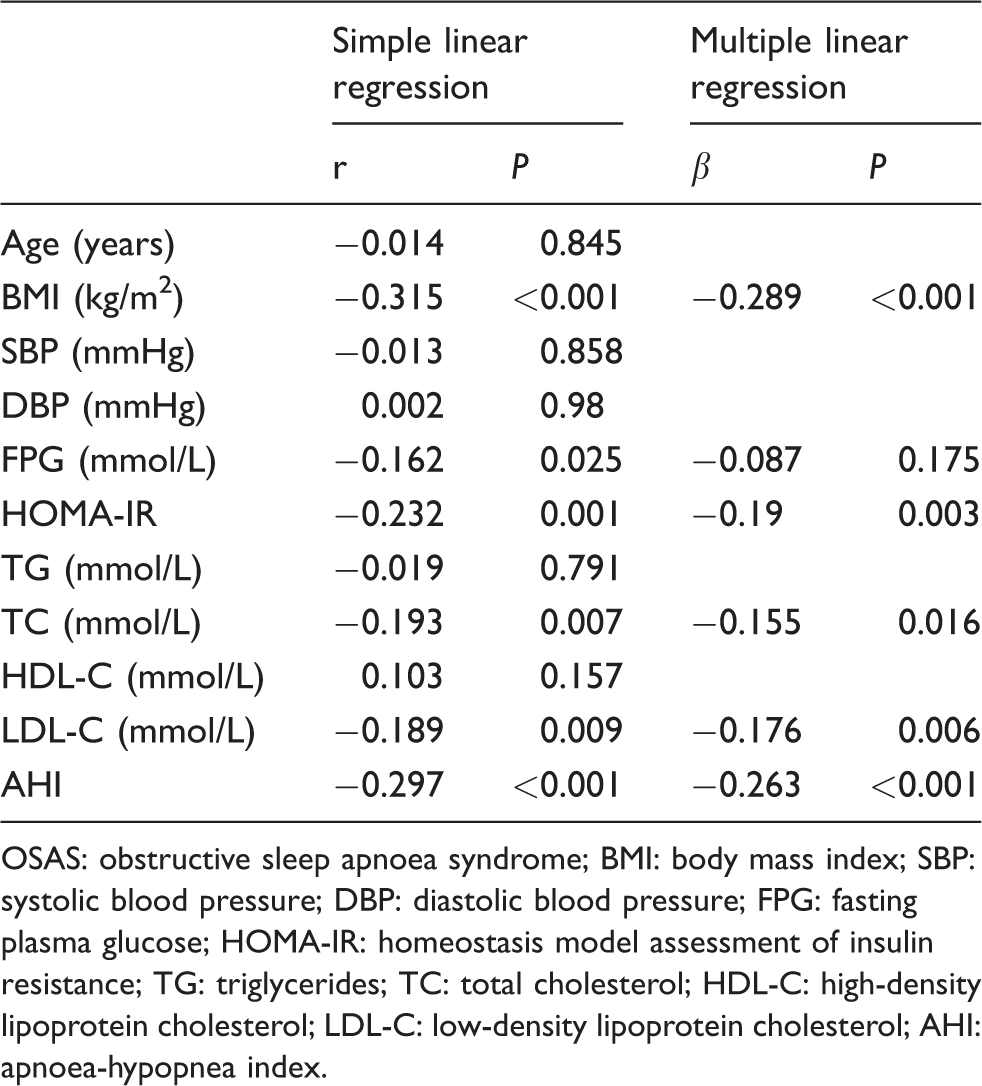
OSAS: obstructive sleep apnoea syndrome; BMI: body mass index; SBP: systolic blood pressure; DBP: diastolic blood pressure; FPG: fasting plasma glucose; HOMA-IR: homeostasis model assessment of insulin resistance; TG: triglycerides; TC: total cholesterol; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; AHI: apnoea-hypopnea index.
Discussion
OSAS is a highly prevalent disorder which causes great medical morbidity and mortality to millions of people worldwide. 7 Therefore, it is necessary to assess the risk of OSAS earlier and then to target strategies to prevent or treat OSAS. Our results revealed that serum myonectin concentrations were significantly lower in patients with OSAS compared with the controls. Serum myonectin concentrations were negatively correlated with the presence and severity of OSAS. OSAS is a low-grade inflammation disease which is characterized by both local and systemic inflammation. 8 Inflammation is correlated with the upper airway narrowing and collapsibility, as well as inspiratory pharyngeal muscle dysfunction. 9 This is confirmed by the evidence that upper airway tissues from OSAS patients showed significantly elevated inflammatory cell infiltration. 10 Snoring stimulation induced the human bronchial epithelial cells to secrete more pro-inflammatory molecules. 11 Recent evidence also suggested the occurrence of systemic inflammation in OSAS. Circulating inflammatory mediators including intercellular adhesion molecules-1, C-reactive protein (CRP) and tumour necrosis factor-α (TNF-α) were found to be elevated in OSAS patients. 12
Ischemia–reperfusion in myonectin-knockout mice led to enhancement of myocardial infarct size, cardiac dysfunction, apoptosis and proinflammatory gene expression including TNF-α, interleukin-6 (IL-6) and monocyte chemoattractant protein 1 (MCP-1) compared with wild-type mice. 6 Expression concentrations of TNF-α and IL-6 in ischemic heart were significantly reduced in myonectin-transgenic mice compared with wild-type mice. 6 Similarly, myonectin treatment attenuated lipopolysaccharide-stimulated expression of TNF-α, IL-6 and MCP-1 in cultured macrophages. 6 Serum myonectin concentrations were significantly negatively correlated with CRP in diabetic patients. 13 Those findings indicated the anti-inflammatory role of myonectin. Myonectin may be involved in the mechanism of OSAS development and progression by its anti-inflammatory effects.
Our results indicate that serum myonectin was correlated with BMI which is a parameter for obesity. Li also reported that obese non-diabetic controls had significantly lower serum myonectin concentrations compared with lean non-diabetic controls. 13 The mRNA and circulating concentrations of myonectin were reduced in a diet-induced obese state. 5 Myonectin-deficient mice had larger gonadal fat pads and developed mild insulin resistance when fed a high-fat diet. 14 Myonectin deletion contributed to greater fat storage in adipose tissue. 14 Serum myonectin concentrations were significantly negatively correlated with BMI, visceral fat area and subcutaneous fat area. 13 Obesity is considered a key risk factor for OSAS. 15 Obese or severely obese patients showed approximately two times of prevalence of OSAS compared with normal-weight adults. 16 Therefore, it is hypothesized that myonectin may serve as a protective role in the pathogenesis of OSAS by inhibiting obesity development.
The limitation of the present study should be considered. First, the sample size was relatively small. Further studies with great numbers are warranted. Second, this study was a cross-sectional design, so our findings should be validated in long-term prospective studies. Third, we enrolled only male patients in the present study. This may have some bias on the results. Further studies are required to confirm the association between myonectin concentrations and OSAS in female patients. Last, we did not assess the effects of continuous positive airway pressure treatment on serum myonectin concentrations. We will test this hypothesis in the next study. And the results may help to determine whether serum myonectin could be used to assess the response to treatment of OSAS.
In conclusion, serum myonectin concentrations were negatively correlated with the presence and severity of OSAS.
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The study protocol was approved by the Research Ethics Committee of Tangdu Hospital, Air Force Medical University (No: TDH0535).
Guarantor
YL.
Contributorship
Yan Liu conceived and designed the research. Huili Zou and Weiyi Yang collected data and conducted the research. Huili Zou wrote the initial paper. All authors read and approved the final manuscript.
