Abstract
Acute kidney injury is a common complication of acute illnesses and is associated with increased morbidity and mortality. Over the past years several acute kidney injury biomarkers for diagnostication, decision-making processes, and prognosis of acute kidney injury and its outcomes have been developed and validated. Among these biomarkers, tissue inhibitor of metalloproteinase-2 (TIMP-2) and insulin-like growth factor-binding protein 7 (IGFBP7), the so-called cell cycle arrest biomarkers, showed a superior profile of accuracy and stability even in patients with substantial comorbidities. Therefore, in 2014, the US Food and Drug Administration approved the use of the product of TIMP-2 and IGFBP7 ([TIMP-2] × [IGFBP7]), known as cell cycle arrest biomarkers, to aid critical care physicians and nephrologists in the early prediction of acute kidney injury in the critical care setting. To date, Nephrocheck® is the only commercially available test for [TIMP-2] × [IGFBP7]. In this narrative review, we describe the growing clinical and investigational momentum of biomarkers, focusing on [TIMP-2] × [IGFBP7], as one of the most promising candidate biomarkers. Additionally, we review the current state of clinical implementation of Nephrocheck®.
Introduction
Acute kidney injury (AKI) is a common complication of acute illnesses among hospitalized patients. It is associated with higher short- and long-term morbidity, including chronic kidney disease (CKD) 1 and cardiovascular events 2 along with higher mortality and healthcare cost. 3 , 4 Despite worldwide recognition of AKI as an epidemic, current means of preventing and treating AKI are very limited. The two main issues include inadequate understanding of its heterogeneous pathogenesis and difficulties related to its early detection.
Although Acute Dialysis Quality Initiative (ADQI), Acute Kidney Injury Network (AKIN), Kidney Disease Improving Global Outcomes (KDIGO) and European Renal Best Practice (ERBP) workgroups have endeavoured to standardize the definition and staging of AKI,5–8 the proposed criteria rely on imperfect parameters including changes in serum creatinine and urinary output, which merely reflect functional modifications rather than structural insults in the kidney. 9 , 10
Another aspect to keep in mind is the emergence of new concepts related to AKI, i.e. subclinical AKI, defined as a syndrome with elevations of tubular damage biomarkers alone, which might or might not evolve into a clinically manifested syndrome. 11 Recent evidence suggested that 15–20% of patients who do not fulfil the consensus criteria for AKI are nevertheless likely to have the acute tubular injury and suffer from its adverse outcomes; 12 thus, biomarkers have been gaining more momentum as research and clinical priorities (Figure 1).
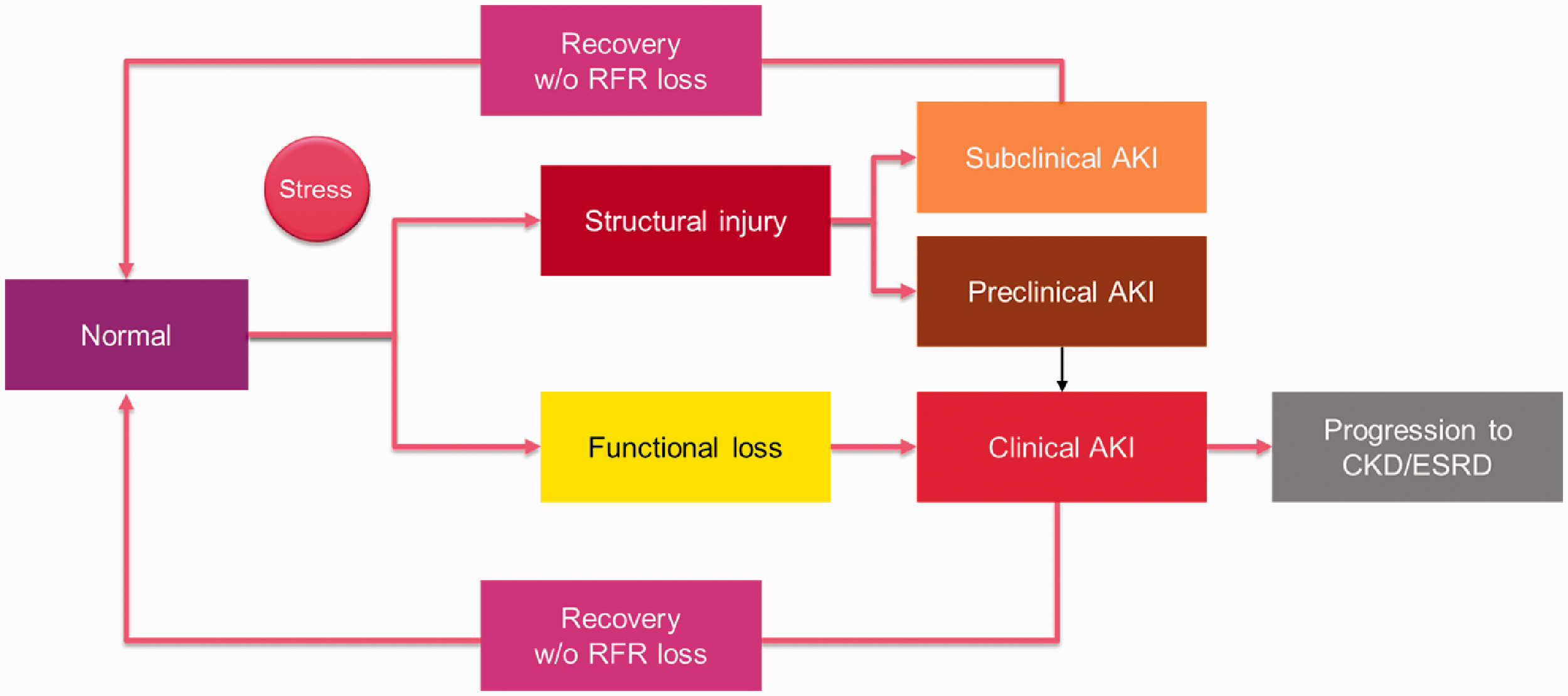
Pathophysiological process to AKI.
With the development of proteomic technologies, 13 novel biomarkers have been identified. Different molecules, such as neutrophil gelatinase-associated lipocalin (NGAL), 14 , 15 kidney injury molecule-1 (KIM-1), 16 liver-type fatty acid-binding protein (L-FABP), 17 interleukin 18 (IL-18), 18 tissue inhibitor of metalloproteinase 2 (TIMP-2) and insulin-like growth factor-binding protein 7 (IGFBP7) (Table 1) 19 have been investigated, in order to improve diagnostication, decision-making processes and prognostication of AKI and its outcomes. 20 , 21 The ideal set of biomarkers could potentially provide substantial additional information not only regarding the risk of AKI but also about the intensity, location, chances of recovery and kidney response to current preventive measures or future therapeutic options (Figure 2). Among all these biomarkers, TIMP-2 and IGFBP7 showed a superior profile of accuracy and stability even in patients with substantial comorbidities. 22 As a result, in September 2014, the United States Food and Drug Administration (FDA) approved the use of the product of TIMP-2 and IGFBP7 ([TIMP-2] × [IGFBP7]), known as cell cycle arrest biomarkers, to aid critical care physicians and nephrologists in the early prediction of AKI in the critical care setting. 23 To date, Nephrocheck® is the only commercially available test for [TIMP-2] × [IGFBP7].
Acute kidney injury biomarker characteristics.
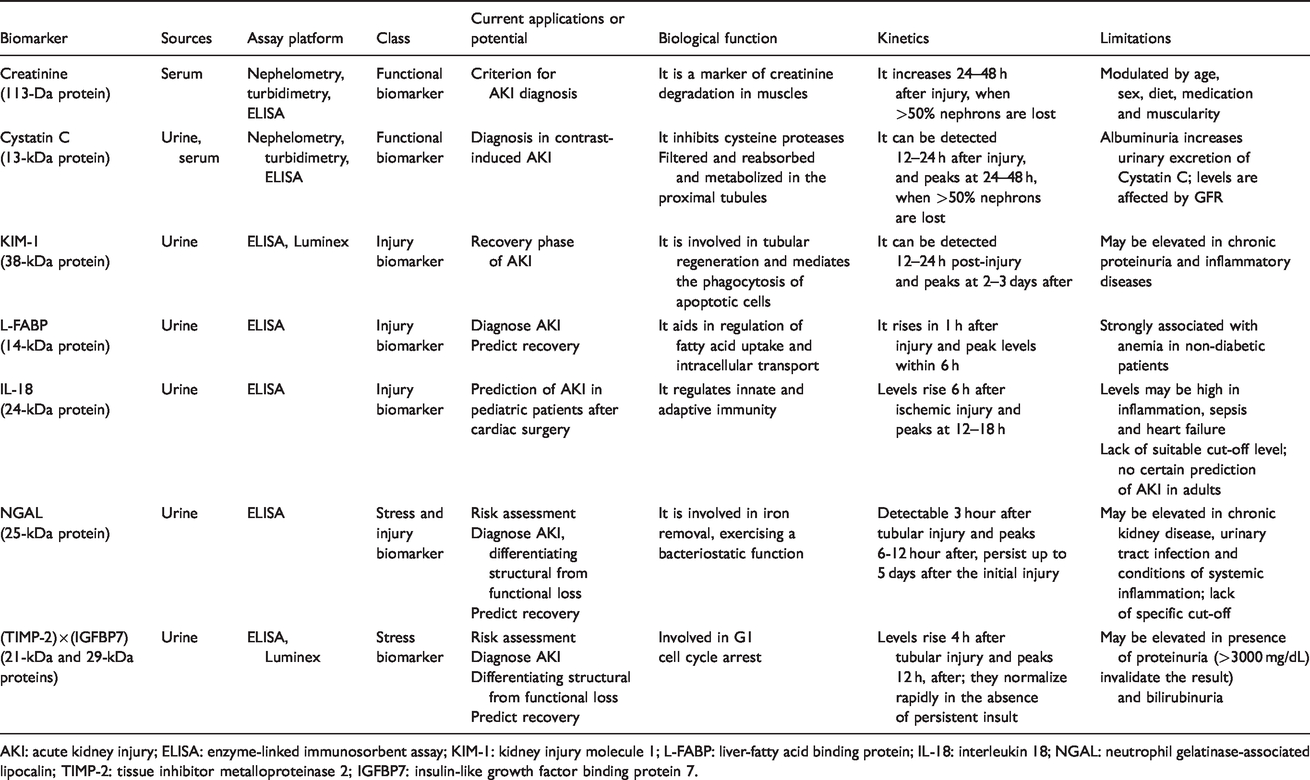
AKI: acute kidney injury; ELISA: enzyme-linked immunosorbent assay; KIM-1: kidney injury molecule 1; L-FABP: liver-fatty acid binding protein; IL-18: interleukin 18; NGAL: neutrophil gelatinase-associated lipocalin; TIMP-2: tissue inhibitor metalloproteinase 2; IGFBP7: insulin-like growth factor binding protein 7.
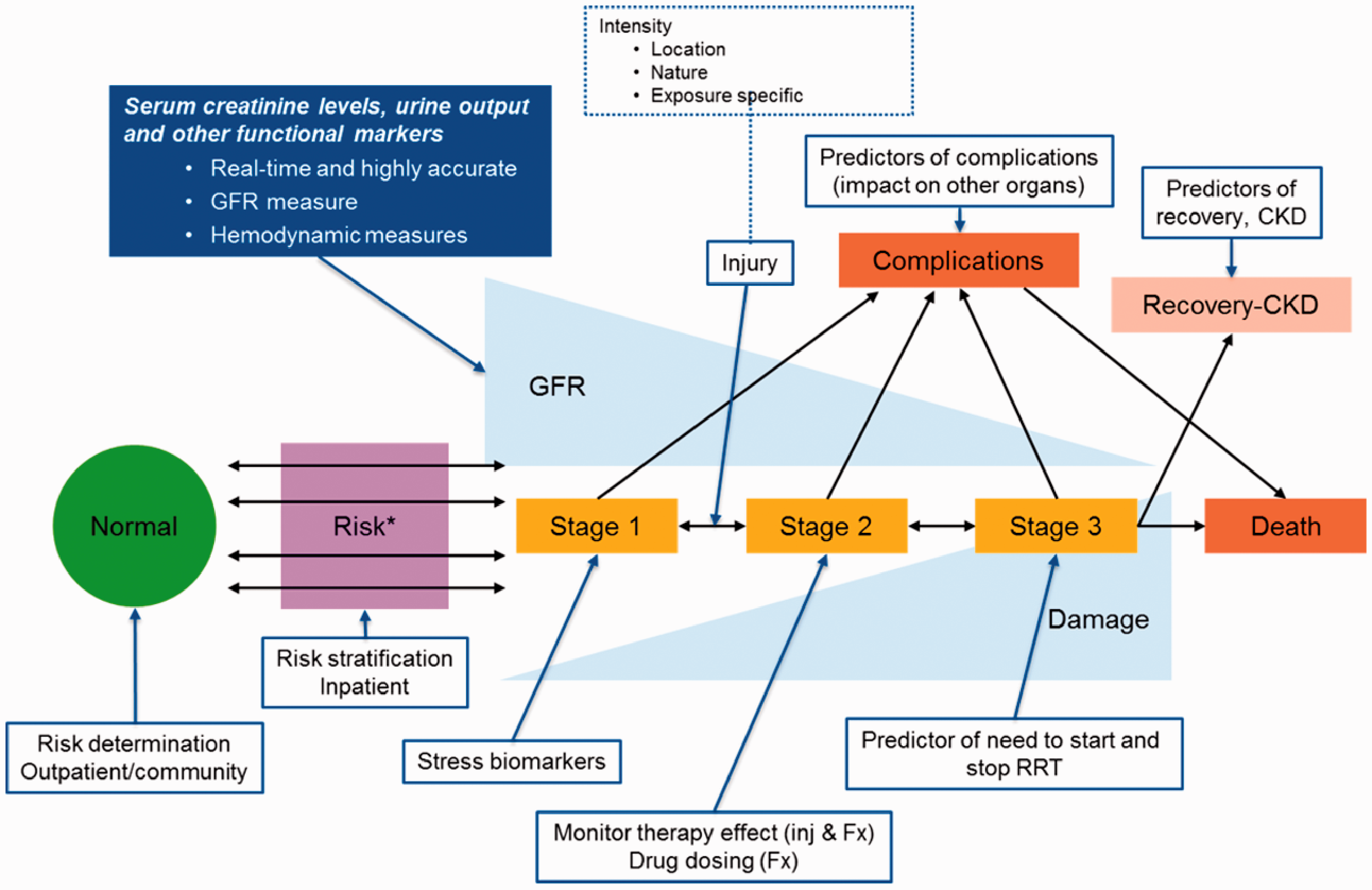
Conceptual model for acute kidney injury development indicates multiple steps, including AKI risk to different levels of injury, which leads to changes in stages of acute kidney injury assessed based on kidney function. 29 This figure shows the potential available or future biomarkers that could assist with decision-making processes, including both changes in kidney function and evaluation of intensity and nature of the injury (solid blue box indicates potential functional biomarkers and boxes outlined with blue indicate potential biomarkers that can assist with decision-making processes).
In this narrative review, we describe the growing clinical and investigational momentum of biomarkers, focusing on [TIMP-2] × [IGFBP7], one of the highest promising candidate biomarkers. In this article, we review the current state of the clinical implementation of Nephrocheck®.
Importance of AKI biomarkers
An AKI biomarker is a substance that is measurable in blood and/or urine during the pathophysiological process that leads to AKI (Figure 1). From kidney stress 24 , 25 or injury, through reduction and loss of function to renal recovery or chronic kidney disease, an AKI biomarker is an indicator of exposure, effect, susceptibility, clinical situation or response to intervention (Figure 2). 21 In the clinical field, biomarkers could be used for risk assessment, diagnosis, prognosis and evaluation of response to therapy.
In the past two decades, several molecules have been identified and investigated in the AKI field. These molecules have ranged from proteins (e.g. exosomal proteins) and more recently messenger ribonucleic acid (mRNA) released by the damaged kidney to molecules upregulated in response to injury and tissue products that are filtered, reabsorbed, or secreted by the kidney. 26 Table 1 lists examples of biomarkers that are described for AKI in humans. To date, no kidney-specific biomarker has been universally accepted for routine use in clinical practice, even though some of them are locally available for clinical use (NGAL in Europe, L-FABP in Japan, [TIMP-2] × [IGFBP7] in most of the developed countries). 27
Current literature supports the role of biomarkers as a research priority to improve outcomes. Hence, many investigations are focused on showing that the availability of new biomarkers might offer the opportunity for improved evaluation and management of AKI.28–30 However, despite the potential benefits of biomarkers in processes of care and patient outcomes, the high cost of these biomarkers potentially explains their limited clinic application. 29
Investigators of Translational Research Investigating Biomarker Endpoints in Acute Kidney Injury (TRIBE-AKI) trial showed urine IL-18 along with urine and plasma NGAL concentrations within a clinical prediction model for AKI were able to enhance the precision of clinical and laboratory data for prediction of AKI. 31 , 32
The 10th acute disease quality initiative (ADQI) consensus designated a more pathophysiological approach to AKI, leading to a classification of biomarkers into stress, injury and functional biomarkers on the basis of their capacity to detect stress and damage or to reflect functional changes. The 10th ADQI marked the first proposal to use biomarkers as AKI diagnosis and staging criteria. 29 Moreover, combining stress, injury and functional biomarkers may provide a tool to comprehend the mechanisms and pathophysiology of AKI better, and to determine the prognosis and time of intervention more precisely. 29 , 33 , 34
When biomarkers are used, many aspects should be considered, including identification of the best biomarkers for each purpose, i.e. risk assessment, diagnosis and prognosis, recognition of different thresholds in each setting and AKI on chronic kidney disease or other common diseases. 29 Nonetheless, the 22nd ADQI consensus conference suggested the use of biomarkers to depend on local resources and be for primary and secondary preventions of AKI. 34
Discovery and validation of [TIMP-2]×[IGFBP7]
In September 2014, the marketing of Nephrocheck® was allowed by the FDA for risk assessment of moderate to severe AKI in critically ill patients. 23
The approved test is an in vitro diagnostic device that quantitatively measures IGFBP7 and TIMP-2 in human urine by fluorescence immunoassay on the ASTUTE140® Meter. The test is a single-use cartridge comprised of assays for the two biomarkers on a membrane test strip enclosed in a plastic housing that employs a sandwich immunoassay technique. A clinical urine sample (mixed with labelled fluorescent conjugate) is applied to the test cartridge; then the cartridge is inserted into the bench-top/table-top meter for incubation, reading and result calculation.
One year later, an application of the same test to a laboratory immunochemistry analyser was marketed. 35 The VITROS® NephroCheck® Test is comprised of two separate immunometric immunoassays for TIMP-2 and IGFBP-7, based on mouse monoclonal antibodies (anti-TIMP-2 or anti-IGFBP-7) and a chemiluminescent detection. The AKIRisk™ Score is calculated and reported by the analyser as the product of the measured concentrations of the two biomarkers, TIMP-2 and IGFBP-7 (ng/mL), divided by 1000. AKIRisk™ Score measured by VITROS Systems has been recently reported to have an acceptable precision (coefficient of variation of about 10% around the first cut-off level, 0.3, and less than 5% at the second one, 2.0), good limits of detection (0.003) and wide range of linearity (0.002–13.8). 36 In comparison with the Astute 140® Meter NephroCheck®, a point of care test system at the moment widely used in many clinical settings, 37 VITROS test has reported having a good correlation (r: 0.96; slope 1.07), and a very high negative predictive value (95.24%).
There are many advantages in using an immunochemistry analyser, instead of a meter based on a single cartridge: higher confidence from biomedical scientists and more standardized management of samples, analysis, quality controls, recording, verifying and reporting results. It is evident how it could be helpful in the management of critical patients, where nephrologists, intensivists and laboratory medicine diagnostic service are required to work in a multidisciplinary team.
While the meter is preferable in a clinical context, in a laboratory setting probably the main constraint using an immunoanalyser is determined by the number of tests performed to sustain the costs of the instrument. Moreover, the assay is only available on one immunoassay platform, and it makes unlikely that a laboratory would purchase the platform solely for the Nephrocheck test.
The test was soon validated by a number of relevant studies. The Sapphire study was a two-stage (discovery and validation) multicentre observational trial in critically ill patients at risk of AKI. 19 The primary endpoint of this study was to validate a biomarker to predict moderate to severe AKI (KDIGO stage 2 or 3) within 12 h of sample collection among critically ill patients. In the discovery stage, Kashani et al. enrolled 522 patients among three distinct cohorts and examined 340 proteins, with the aim of identifying the best biomarkers for AKI. Urine TIMP-2 and IGFBP7 were recognized as the best performing candidates, with an area under the receiver operating curve (AUC) of 0.80 for the arithmetic product of TIMP-2 and IGFBP7. They were found to be superior to many other existing biomarkers, including NGAL, plasma cystatin C, KIM-1, IL-18 and L-FABP. The subsequent prospective multicentre study validated these two biomarkers in a population of 728 patients with critical illnesses (e.g. cardiovascular and respiratory dysfunction) and without evidence of moderate to severe AKI at the time of enrolment. [TIMP-2]×[IGFBP7] demonstrated robust diagnostic performance, which significantly improved clinical decision-making in AKI risk assessment and stratification.
Using data from the Sapphire study, two cut-offs for [TIMP-2]×[IGFBP7] (0.3 (ng/mL)2/1000 and 2.0 (ng/mL)2/1000) were prospectively tested and verified in the Opal multicentre study by including additional 154 critically ill patients. Opal study confirmed the high sensitivity and negative predictive value for the cut-off of 0.3 (89% and 97%, respectively), and the high specificity and moderate positive predictive values for the cut-off of 2.0, i.e. 95% and 49%, respectively. 38
The Topaz study was a multicentre prospective study of 420 critically ill patients (cardiovascular and respiratory impairments) to validate the use of urinary [TIMP-2]×[IGFBP7] combined with a clinical model for risk assessment. This study showed the absolute risk for AKI was seven times higher in patients with [TIMP-2]×[IGFBP7] >0.3 compared with those with values <0.3. 39
Characteristics of cell cycle arrest biomarkers
Cell cycle arrest is a protective mechanism meant to prevent the division of cells with damaged DNA, thus permitting adequate repair. Cell cycle division is required for cell proliferation. For cells to divide, they must complete several tasks in organized, predictable steps to complete the cell cycle. These steps include G1 (gap1), S (synthesis), G2 (gap2), M (mitosis). 40 , 41 The entire process is controlled by cyclins, cyclin-dependent kinases (CDK) and cyclin-dependent kinases inhibitors. 19 During the cell cycle, DNA content is proofread, and if the process detects any defects, the normal division and repair process can become maladaptive. Therefore, cells can remain in the G1 or G2 phases instead of re-initiating the cell cycle or can leave the cell cycle in the late G1 phase, situations leading to senescence or apoptosis, respectively. 42
TIMP-2 and IGFBP7 are known as cell cycle arrest biomarkers. TIMP-2, a 24-kDa protein, inhibits matrix metalloproteinase activity and stimulates p27 expression, playing a role in cell cycle regulation, while IGFBP7 is a 29-kDa protein, member of the IGFBP superfamily; it directly increases p21 and p25 expression, potently inducing G1 cell cycle arrest. 43 In response to stress or insult, renal tubular cells may express or secrete TIMP-2 and IGFBP7 in an autocrine or paracrine fashion to induce G1 cell cycle arrest in tubular cells in order to prevent the proliferation of injured cells. 44 Additionally, cell cycle arrest seems to play a role in senescence and maladaptive repair. 45
Risk and development of AKI
Timely identification of the patients at risk of developing AKI is the cornerstone of early interventions that might ensure the preservation or recovery of an adequate renal function. For this particular purpose, different care bundles have been designed. While some care bundles simply aim at monitoring serum creatinine and the use of nephrotoxins,46–48 others integrate demographics, vitals, laboratory data, clinical interventions and other diagnostics into machine learning prediction models.49–51 Health information technologies and biomarkers are tools that could potentially provide the chance to identify patients who are at risk of AKI earlier and provide a more efficient path to preventive measures.
The utility of cell cycle arrest biomarkers to predict AKI in different fields other than the intensive care unit (ICU) population has been extensively explored.
Two recent meta-analyses separately showed that urinary [TIMP-2]×[IGFBP7] is a valid predictor for cardiac surgery-associated AKI (CSA-AKI) within 24 h. 52 , 53 Su et al. analysed 10 articles with a sample size of 747 patients undergoing elective cardiovascular surgery. They found the sensitivity and specificity of [TIMP-2]×[IGFBP7] to be 0.77 and 0.76, respectively. This is while the positive and negative likelihood ratios were 3.26 and 0.32, with a diagnostic odds ratio of 10.1 and AUC of 0.83. Meanwhile, Tai et al. examined eight studies comprising 552 patients undergoing cardiac surgery. The authors reported the sensitivity and specificity of [TIMP-2]×[IGFBP7] to be 0.79 and 0.76, respectively. This is while the positive and negative likelihood ratios were 3.49 and 0.31, with a diagnostic odds ratio of 14.9 and AUC of 0.87. The different cut-off thresholds, heterogeneity in the AKI diagnosis threshold, and time of measurement might have limited these results. In a recent study, Levante et al. showed an increased accuracy when [TIMP-2]×[IGFBP7] was included in a clinical model for AKI diagnosis. 54 Moreover, two independent studies on cardiosurgical population demonstrated that the routine measurement of cell cycle arrest and subsequent activation of an acute kidney response team have been associated with a significant decrease in postoperative severe AKI after cardiac surgery without increases in cost or length of stay. 55 , 56 For all these reasons, the Enhanced Recovery After Surgery Society recently published the guidelines for perioperative care of cardiac surgery, recommending the use of [TIMP-2]×[IGFBP7] for early identification of patients at risk and to guide an intervention strategy to reduce AKI. 57
Cell cycle arrest biomarkers are also studied in major non-cardiac surgical operations with promising results. Gunnerson et al. showed that a single urinary [TIMP-2]×[IGFBP7] test accurately identified patients at risk of developing AKI stage 2 or 3 within 12 h of ICU post-surgery admission accurately. 58 Gocze et al. conducted a clinical trial assessing 107 surgical patients at high risk of AKI and found that [TIMP-2]×[IGFBP7] had an AUC for risk of any AKI to be 0.85. 59 The early biomarker-prediction of imminent AKI followed by implementation of KDIGO care bundle replicated the results in cardiac surgery, reducing AKI severity, length of ICU and hospital stay in patients after major non-cardiac surgery.
[TIMP-2]×[IGFBP7] was also shown to be a predictor of AKI in many other settings like decompensated heart failure, 60 cardiac arrest,61–63 emergency department 64 and sepsis.65–67 There are also some other investigations in post-chemotherapy toxicity, 68 , 69 and obstetric-related critical illness, 70 , 71 which shows cell cycle arrest biomarkers’ performance to be suboptimal.
Cell-cycle arrest biomarkers and prognosis
Urinary TIMP-2 and IGFBP7 can be found soon after an insult occurs, and rapidly decline if the damage is not perpetuated. 72 Based on the recent information, it appears that these biomarkers are not only associated with an increased risk of AKI but may also be inversely related to the functional recovery of AKI or be a predictor of adverse outcomes.
Meersch et al. showed that among post-cardiac surgery patients, the decline in [TIMP-2]×[IGFBP7] values were a strong predictor of renal recovery, with an AUC of 0.79. 73 In the post kidney transplant patients, it seemed that only TIMP-2 is a promising biomarker in the prediction of the development and duration of delayed graft function (DGF), as shown in the studies by Pianta et al. 74 and Bank et al. 75 In contrast, Yang et al. found the product of TIMP-2 and IGFBP7 to be a good predictor of DGF following deceased-donor kidney transplant. 76
Daubin et al. studied a population of critically ill patients and found that the values of cell cycle arrest biomarkers were higher in transient versus persistent AKI at early hours after ICU admission. 77 Aregger et al. showed that IGFBP7 predicted mortality, renal recovery, and severity and duration of AKI in a small cohort of critically ill patients. 78
Koyner et al. performed a post hoc analysis of the Sapphire study, 79 confirming that [TIMP-2]×[IGFBP7] measured early in the setting of critical illness was associated with a nine-month composite outcome of all-cause mortality or prescription of renal replacement therapy (RRT). Recently, Xie et al. found a strong association between positivity of cell cycle arrest biomarkers and the composite endpoint of the need for RRT and all-cause mortality. 80 Jia et al. conducted a systematic review and meta-analysis on the prognostic value of cell cycle arrest biomarkers patients at high risk of AKI. 81 Examining four studies with 277 patients undergone major or cardiac surgeries found [TIMP-2]×[IGFBP7] had an AUC of 0.92 (sensitivity analysis confirmed the homogeneity of pooled study).
In predicting mortality, cell cycle arrest biomarkers showed a fair performance in recent studies. Gocze et al. found an AUC of 0.77 in determining 28-day mortality. 59 Dewitte et al. found an AUC of 0.81 for predicting mortality in a prospective observational study of 57 ICU patients with AKI. 82 In a pediatric population study by Westhoff et al., 83 the authors found AUC values of 0.79 and 0.88 for [TIMP-2]×[IGFBP7] in predicting 30-day and three-month mortality, respectively.
[TIMP-2]×[IGFBP7] was also evaluated for the risk of mortality or poor neurological outcome in a cohort of 195 cardiac arrest patients by Beitland et al. 61 The authors noted there was no association between these biomarkers and neurological outcomes.
Guiding management
Recently, there is more academic interest in the incorporation of cell cycle arrest biomarkers within the prediction models aiming the guidance of the application of the KDIGO bundle for AKI risk.
Recently Zaouter et al. evaluated the prediction of AKI by using a combined approach with renal resistive index (RRI) and cell cycle arrest biomarkers measurements at different time points during the first day of ICU admission in an elective cardiac surgery population. They note neither RRI nor cell cycle arrest biomarkers detected CSA-AKI occurrence in the first postoperative week. 84
Gocze et al. conducted a randomized trial for implementation of the KDIGO care bundles to prevent AKI among patients admitted to the ICU after major surgical procedures who had elevated cell cycle arrest biomarkers. 85 They showed a reduction in AKI occurrence, length of ICU, and hospital stay, while no difference was found in terms of RRT, in-hospital mortality, and major adverse kidney events at hospital discharge.
A prospective randomized controlled intervention trial 64 was performed in a high-risk cohort for the AKI in the emergency department. After screening 257 patients with urinary [TIMP-2]×[IGFBP7] > 0.3, the enrollees received immediate one-time nephrological consultation. The endpoints were one-day and three-day AKI, need for RRT, length of stay, and death. Although the results showed no difference between the intervention and non-intervention groups, the authors outlined a trend of study endpoints in favour of intervention, suggesting a better selection of population might be warranted.
Given the complexity of critically ill patients and the heterogeneity of kidney injury in this particular context, the appropriate timing of biomarker measurement continues to be a significant issue. 86 Whether a single or repeated measurement of biomarkers should be considered to assess dynamic changes among critically ill patients adequately remains to be observed in future studies. In addition, the integration of cell cycle arrest biomarkers with the clinical context or other diagnostic techniques needs to be evaluated. 23 , 87
Clinical implementation
Nephrocheck®, as a biomarker of AKI prediction, has excellent potential for clinical implementation. It is a simple test with a rapid turnaround time of approximately 20 min. Only 0.1 mL of urine is required to complete the quantitative analysis. When it is used in conjunction with clinical prediction models or AKI e-alerts, it has the potential to improve the quality of care and patient outcomes.
At San Bortolo Hospital in Vicenza, a protocol titled nephrology rapid response team (NRRT), including a multidisciplinary team of nephrologists and intensivists 88 has been implemented that incorporates the power gained by automatic electronic alerts and cell cycle arrest biomarkers to improve the care of patients with AKI. 87 During the initial triage, critically ill patients were divided into low-, moderate- and high-risk categories within the screening process using the AKI risk assessment fantastic four (ARA F4) model. 89 , 90 The electronic algorithm, using patients’ clinical status, past medical and surgical history, physical examination and laboratory results, provided the ARA F4 score for each individual patient. As [TIMP-2]×[IGFBP7] is likely to be more precise in predicting AKI among moderate or high-risk individuals when compared with the low-risk population, 30 we collected urine samples from moderate to high-risk patients to perform Nephrocheck®. Tests are performed by using the VITROS® NephroCheck® Test on VITROS® 3600 Immunodiagnostic System (Ortho-Clinical Diagnostics, Raritan, NJ, USA). We demonstrated that [TIMP-2]×[IGFBP7] on ICU admission had a fair performance in predicting AKI, especially within the first four days of admission. 76 A value of ≤ 0.3 (ng/mL)2/1000 was used to monitor and provide standard AKI prophylaxis, while measures > 0.3 (ng/mL)2/1000 indicated the need for nephrology consultation for the patient management. 19 Moreover, a cut-off value of > 2 (ng/mL)2/1000 suggested the highest risk of AKI. Finally, the AKI Management protocol at San Bortolo Hospital in Vicenza, included a checklist of preventive and protective measures, as suggested by the KDIGO AKI guideline. 7 This checklist is designed to be implemented within 12 h after the release of the Nephrocheck® result. 87 The ultimate goals of the NRRT were fewer episodes of AKI, a decrease in the severity of AKI episodes, and mitigated need for dialysis. As reported in a recent publication, 91 the practical results of the NRRT implementation, triggered by a positive Nephrocheck results were remarkable. NRRT protocol was compared to a standard practice in two subsequent years (2017 versus 2016). In the same hospital the number of AKI patients decreased from 314/year to 281/year and number of patients requiring renal replacement therapy decreased from 121/year to 99/year.
Nephrocheck test is currently FDA approved for use 23 in the United States and commercially available in several European countries including France, Germany, Italy, Austria, Switzerland, United Kingdom and Spain. Recently, National Institute of Health Excellence (NICE) did not recommend its routine use for patients admitted in ICU. 92 The questions not solved according to the NICE committee were which patients could benefit from the test and by how much. Instead, several institutions, early adopters of AKI biomarker technology, had very similar experiences with the clinical use of the test. Clinical experts from Europe and North America published a guidance for Nephrocheck test use, which tried to help determine how the test is being and to identify which patients would be appropriate for testing, how the results are interpreted, and what actions would be taken based on the results. 93
Conclusion
In recent years, the use of traditional and newer functional and damage biomarkers of AKI has allowed better identification of patients at risk for AKI. The impact of biomarkers on individual patient management is increasing and gaining positive evidence. Close monitoring of available biomarkers such as serum creatinine and urine output is strongly associated with improved patient outcomes. Further to that, an integrated approach using new biomarkers may trigger implementation of care bundles to optimize renal protection and AKI prevention. This will likely be the new frontier for the next years leading to an expanding collaboration between renal medicine, intensive care and clinical and laboratory medicine, implementing an effective multidisciplinary approach to the high risk, critically ill patient.
Footnotes
Acknowledgements
This article was prepared at the invitation of the Clinical Sciences Reviews Committee of the Association for Clinical Biochemistry and Laboratory Medicine
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding Research funding to Dr. Kashani’s institution from Astute Medical Inc. 2012. Prof. Ronco declares speaker honoraria from Astute Medical, ASAHI, Toray, FMC, B. Braun, GE, Bellco. The funding organizations played no role in the writing of the report; or in the decision to submit the report for publication.
Ethical approval
Not applicable.
Guarantor
Ronco Claudio.
Contributorship
All the authors have accepted responsibility for the entire content of this submitted manuscript and approved the submission.
