Abstract
Background
The measurement of selenium in human plasma is useful to assess deficiency or toxicity. The presence of gadolinium in clinical samples following administration of certain contrast agents used for magnetic resonance imaging can cause a significant positive bias in selenium results when measured using quadrupole inductively coupled plasma mass spectrometry (Q-ICP-MS).
Methods
A mathematical equation to correct for gadolinium interference was assessed using both patient samples and commercial quality control/external quality assurance (QC/EQA) materials spiked with gadolinium. Samples were analysed using an Agilent 7900 ICP-MS operated in ‘narrow peak’ (half-mass) mode. Accuracy was evaluated by comparing corrected selenium results with target concentrations.
Results
Corrected results were found to be accurate at all gadolinium concentrations tested (2, 4, 10 and 20 mg/L). Average recoveries ranged from 97.4 to 106.5%. Results for QC/EQA materials were within specified target ranges. Within-run imprecision was <3%, and between-run imprecision was <4.3%, demonstrating robustness.
Conclusions
The correction equation described here is a simple method to correct for gadolinium interference on plasma selenium measurement using ICP-MS. This approach eliminates the need for specimen recollections, and improves patient care by reducing laboratory turnaround times and preventing delays in diagnosis/treatment.
Introduction
Selenium is an essential trace element with a significant role in human health, including production of active thyroid hormone (T3), fertility and redox regulation of the cell. 1 Selenium in the form of selenocysteine (a structural analogue of cysteine where the sulphur atom is replaced by selenium) is an integral component of selenoproteins. At least 25 selenoproteins including biologically important enzymes such as iodothyronine deiodinase, thioredoxin reductase and glutathione peroxidase have been identified in humans. 1 Selenium deficiency may manifest a wide range of symptoms, including those of hypothyroidism. Cases of selenium toxicity have also been described;2,3 however, deficiency is more common.
The measurement of selenium in biological fluids is used to assess nutritional status, and is now routinely performed using inductively coupled plasma mass spectrometry (ICP-MS). The concentration range between deficiency and toxicity is narrow, underscoring the need for accurate selenium determination. The presence of gadolinium in a clinical sample is well known to interfere with selenium analysis using quadrupole ICP-MS, causing a significant positive bias. This interference arises because gadolinium has a relatively low second ionization potential (12.09 eV), and therefore forms double charged ions in the argon plasma which have the same mass-to-charge ratio (m/z) 4 as selenium isotopes (Table 1). Interference has been observed at gadolinium concentrations as low as 0.2 mg/L in serum. 6
Gadolinium chelates are often used as contrast agents for magnetic resonance imaging (MRI) procedures. Gadolinium-based contrast agents (GBCA) were first introduced in 1983, 7 and there are now 30 million patient administrations per year. 8 Hence, gadolinium interference must be considered when measuring selenium in clinical samples. A number of strategies have been described to obviate this interference. Bishop et al. 9 have used a triple quadrupole ICP-MS (ICP-QQQ-MS) with oxygen mass-shift to enable detection of all major selenium isotopes in the presence of gadolinium. Triple quadrupole instruments are more expensive than single quadrupole instruments however, and this may be a limiting factor for some laboratories. Sector field (SF) ICP-MS instruments have sufficient resolution to resolve gadolinium and selenium isobars 5 ; however, these instruments are very expensive for routine use and are usually restricted to research applications. Ryan et al. 10 have described an approach using the minor selenium isotope 82Se; however, it was noted that 82Se should not be used as the sole isotope for monitoring due to potential interference from 81Br1H. 10 82Se also has a lower natural abundance than 78Se (Table 1), and therefore lower analytical sensitivity. A method using hydrogen collision gas has also been described. 11 Here, we describe a mathematical equation to correct for gadolinium interference, yielding a ‘corrected selenium’ result.
Materials and methods
Reagents and materials
All calibration standards, blanks and plasma samples were diluted for analysis using 1% ammonium hydroxide (Ajax Finechem, Auburn, Australia) containing 0.05% Triton-X100 (BDH, Poole, UK), 0.05% EDTA (Sigma Aldrich, St. Louis, USA), 1% isopropanol (Merck, Darmstadt, Germany) and 1000
Instrumental conditions
An Agilent 7900 ICP-MS (Agilent Technologies, Santa Clara, USA) was used for the analysis, with the following relevant m/z monitored: 78 (78Se), 78.5 (157Gd2+), 125 (125Te internal standard). Helium was used as a collision gas. Instrument conditions are outlined in Table 2.
Inductively coupled plasma mass spectrometry parameters for determining ‘Corrected Se’.
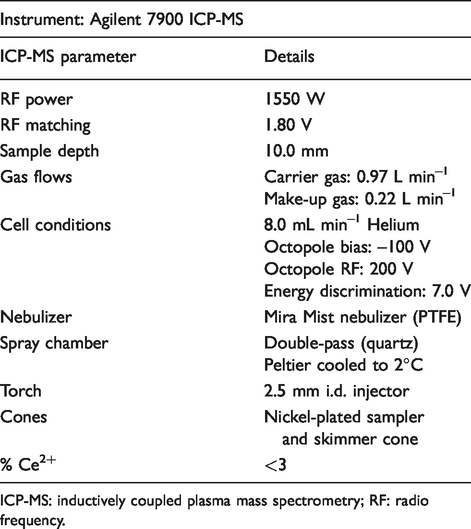
ICP-MS: inductively coupled plasma mass spectrometry; RF: radio frequency.
Using a higher resolution setting on the instrument (‘narrow peak mode’), the signal for 157Gd2+ can be measured at m/z 78.5. The contribution of 156Gd2+ on 78Se+ can therefore be calculated using the known distribution of isotopic abundance for gadolinium (156Gd: 20.47%, 157Gd: 15.65%). The following equation was used to correct for interference
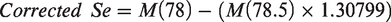
In this equation,
Methods
Sample preparation
Aliquots of plasma specimens and QC/EQA materials were diluted with an alkaline diluent (as outlined above), giving a final dilution factor of 20. Diluted samples were vortex-mixed for 5 s before analysis on the ICP-MS.
Thirteen de-identified human plasma specimens that would otherwise be discarded were used to evaluate this method. The concentration of selenium (78Se) in each specimen was determined using an existing in-house ICP-MS method. Subsequently, each specimen was subdivided into four aliquots and spiked with 2–20 mg/L of gadolinium. QC/EQA materials (
The repeatability of corrected results was assessed by replicate (
Results
Corrected and uncorrected results are shown in Table 3 (spiked plasma samples) and Table 4 (spiked QC/EQA materials). All specimens spiked with gadolinium demonstrated a significant positive bias in uncorrected selenium results. The magnitude of the positive bias increased proportionally with gadolinium concentration, with selenium recoveries up to 1375% greater than known concentrations (Table 3). Figure 1 shows the average recovery of patient specimens plotted against gadolinium concentration.
Recovery of selenium (corrected and uncorrected) in patient samples spiked with gadolinium.
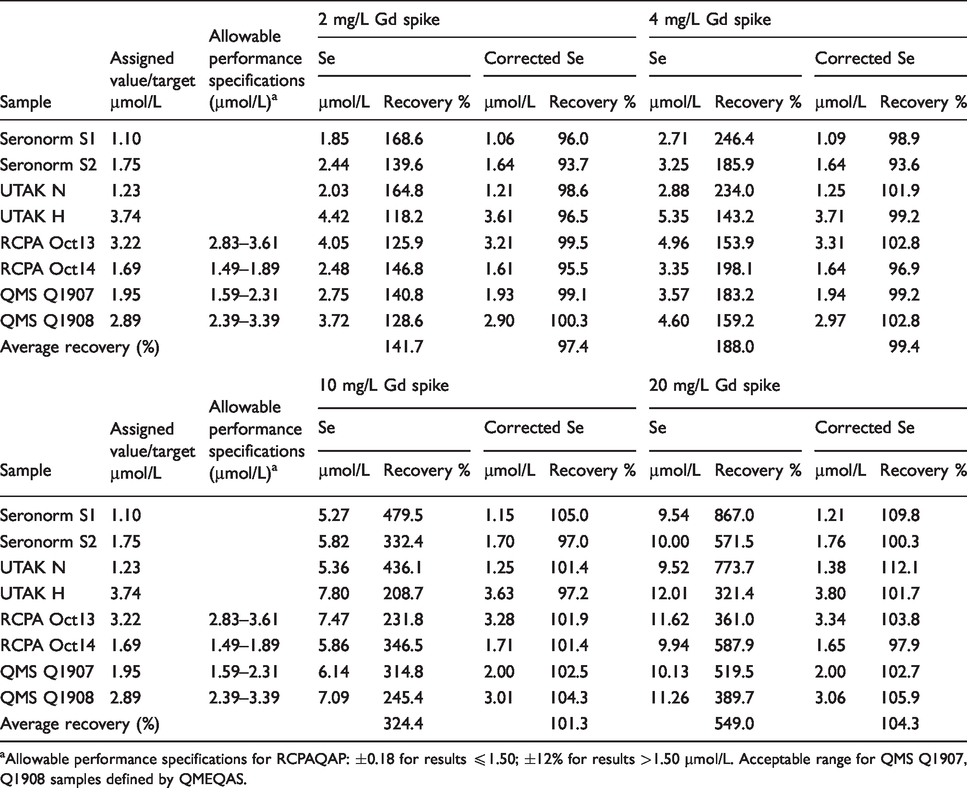
aAllowable performance specifications as defined by RCPAQAP: ±0.18 for results ≤1.50; ± 12% for results >1.50
Recovery of selenium (corrected and uncorrected) in QC/EQA materials spiked with gadolinium.
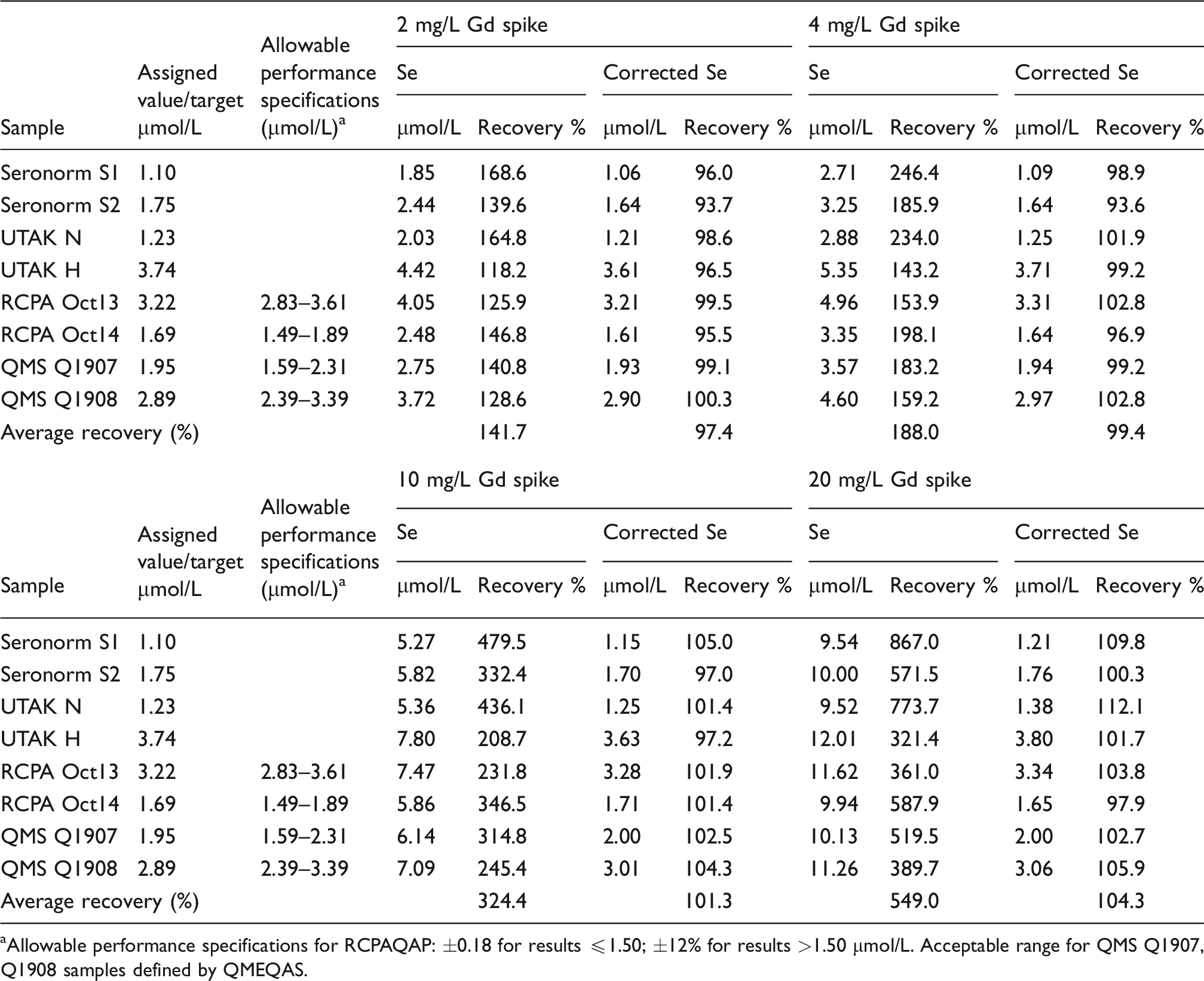
aAllowable performance specifications for RCPAQAP: ±0.18 for results ≤1.50; ±12% for results >1.50
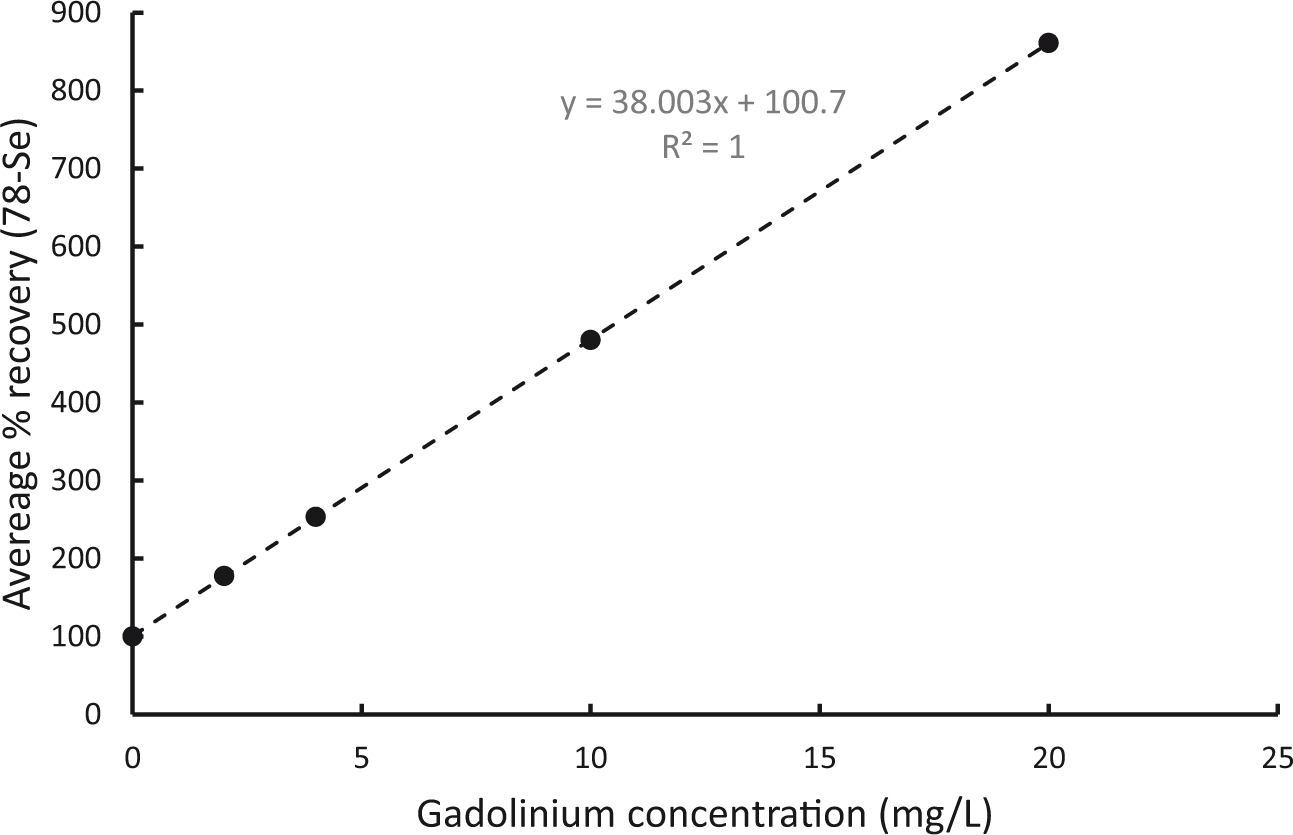
Effect of gadolinium on (uncorrected) selenium results.
Calculated results for corrected selenium were all in good agreement with target values. Average recoveries ranged from 97.4 to 103.9% for specimens spiked with 2–10 mg/L gadolinium. At 20 mg/L gadolinium, average recoveries for patient specimens and QC/EQA materials were 106.5 and 104.3%, respectively. Further, all corrected results were within the allowable performance specifications as defined by RCPAQAP. Results for QMEQAS samples (
Within-run imprecision was 1.3% CV (
Histograms of uncorrected selenium results in QC/EQA materials (Figure 2) and corrected results (Figure 3) demonstrate the efficiency of the correction equation.
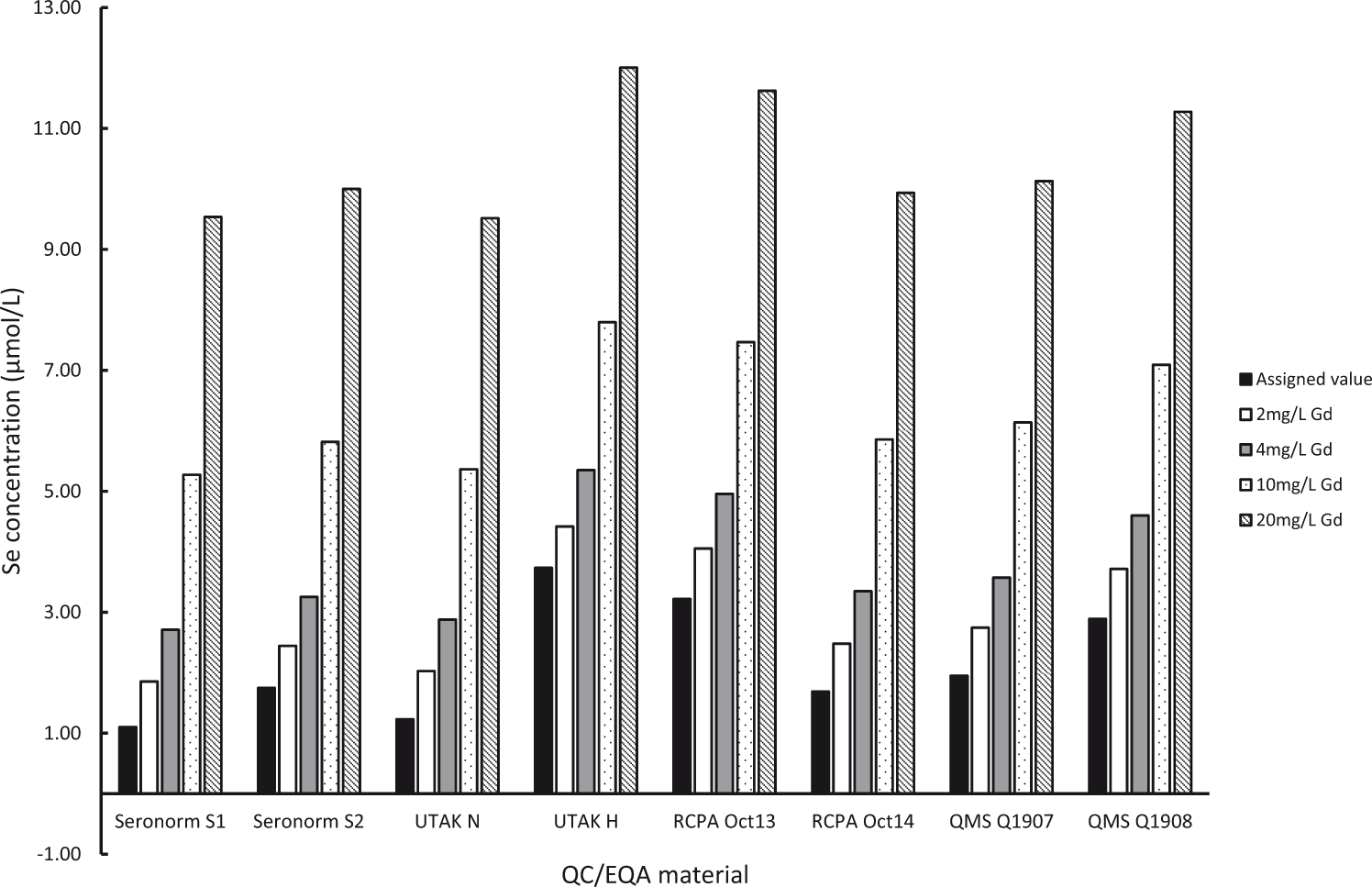
Measured Se concentration (uncorrected) in QC/EQA materials spiked with Gd.
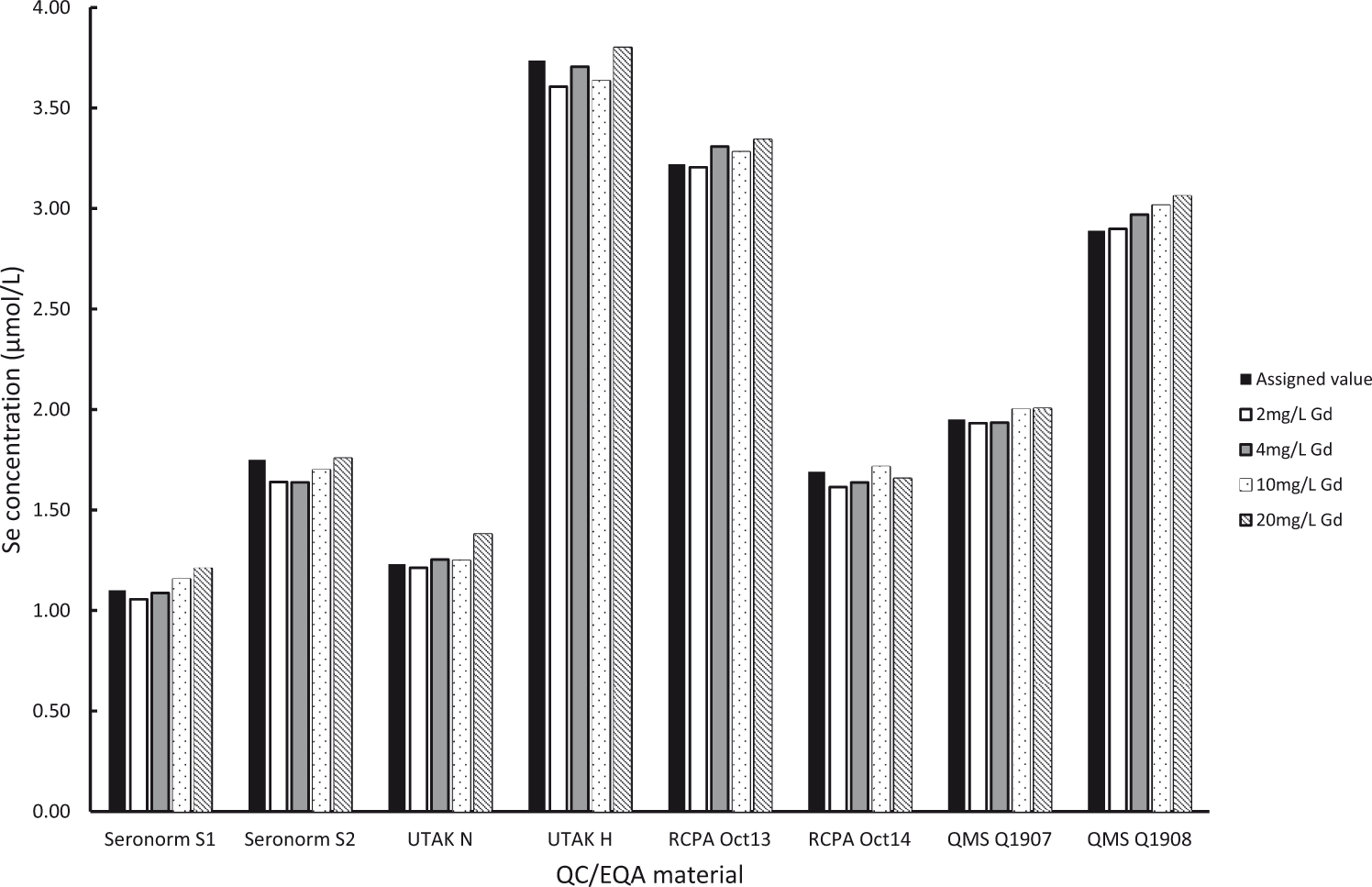
Measured Se concentration (corrected) in QC/EQA materials spiked with Gd.
Discussion
The measurement of selenium in biological samples is a technical challenge. Selenium has a high ionization potential (9.75 eV), and isotopic abundance is distributed among its six stable isotopes (Table 1); therefore, analytical sensitivity is relatively low compared with other elements. Each isotope of selenium also suffers from significant spectral interference from isobaric polyatomic ions (e.g. 40Ar40Ar on 80Se, and 40Ar38Ar on 78Se), or isobaric interference from isotopes of other elements (e.g. 82Kr on 82Se). While polyatomic interferences can largely be obviated using a collision cell with helium kinetic energy discrimination, this approach is ineffective for double-charged interference.4,15 The correction equation evaluated here was found to produce accurate results for selenium at the tested gadolinium concentrations up to 20 mg/L. Repeatability of corrected results within-run and between-run was good (CV <5%), demonstrating robustness.
The observed positive bias of 4.3–6.5% at gadolinium concentrations of 20 mg/L may be attributable to signal overlap (‘tailing interference’) of 155Gd2+ (m/z 77.5) on 78Se, which is not corrected for in this approach. Correction equations are also inherently less accurate when the magnitude of the correction is significantly higher than the concentration of the measurand (we observed an order of magnitude difference between corrected and uncorrected results in samples spiked with 20 mg/L gadolinium). A further source of error may arise due to the so-called space-charge effect. This type of matrix effect is caused by the mutual electrostatic repulsion of positively charged ions in the mass spectrometer. In the presence of high concentrations of a heavy matrix element such as gadolinium, ions from lighter elements such as selenium will be preferentially lost from the ion beam causing a reduction in signal.4,16 Tellurium was used as an internal standard to mitigate this effect. Space-charge effects due to gadolinium can theoretically cause inaccuracy in the quantitation of any element (not only selenium). Day et al. 17 reported an approximately 15% reduction in signal for gallium, germanium, yttrium and rhodium in the presence of 1000 mg/L gadolinium. This gadolinium level is significantly higher than that typically observed in samples collected post MRI, however. For example, in a pharmacokinetic analysis of four different gadolinium chelates (gadodiamide, gadopentetate, gadoversetamide and gadoteridol) in 12 healthy individuals, Brown et al. 12 showed that average serum gadolinium concentrations were all less than 20 mg/L within approximately 3 h of administration. The accuracy of the method described here has not been established at gadolinium concentrations higher than 20 mg/L; therefore, caution is recommended in samples collected less than 3 h after administration of the contrast agent.
Gadolinium interference can cause artefactually elevated selenium results,6,10 which may lead to misclassification of selenium status (either masking a deficiency, or misclassifying selenium toxicity). The degree of interference is also affected by instrument tuning conditions which affect double-charged ion formation, such as radio frequency power, nebulizer gas flow and sample depth. These parameters can be optimized to attenuate the interference to some extent. Optimal instrument tuning is therefore expected to minimize potential sources of error or bias in the mathematical correction (outlined above). The correction equation described here significantly improves the reliability of selenium results in patient samples. While perhaps slightly less accurate than interference mitigation strategies such as ICP-QQQ-MS which physically removes the interferent, this correction equation can be used with a single-quadrupole ICP-MS which is much more common in routine biochemistry laboratories (and also markedly less expensive). If gadolinium is detected at significant levels in a clinical sample, a laboratory may be forced to cancel the test and request a specimen recollection (usually after appropriate time is allowed for gadolinium to clear from the circulation). For example, Day et al. 17 reported 42 cancelled serum selenium requests within one year at the Department of Laboratory Medicine and Pathology, Mayo Clinic, Rochester, MN, USA. Implementation of a correction equation approach to gadolinium interference will significantly reduce the time and cost associated with such recollections and lead to improved patient management.
Conclusion
The correction equation described here is a simple method to correct for gadolinium interference on plasma selenium measurement using an Agilent 7900 ICP-MS. This approach eliminates the need for specimen recollections and improves patient care by reducing laboratory turnaround times, and preventing delays in diagnosis/treatment.
Footnotes
Acknowledgements
We are very grateful to Mr. Greg Ward, Mr. Robert Flatman, Dr. David Kanowski, and Dr. Lee Price of Sullivan Nicolaides Pathology for their help in reviewing the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
SW.
Contributorship
All authors were involved in designing the study. SW wrote the first draft of the article. All authors reviewed and edited the article and approved the final version of the article.

