Abstract
Background
Specimen labelling and patient identification are significant contributors to the rate of error in the preanalytical phase of laboratory medicine. This study aimed to investigate the prevalence and nature of preanalytical quality monitoring practices for patient identification and specimen labelling errors in Irish clinical laboratories.
Methods
A survey was developed by the Clinical Biochemistry Unit, Trinity College Dublin and the Irish External Quality Assessment Scheme (IEQAS), with the intention of gathering key information from each laboratory. Thirty-nine questions were organized into seven subsections covering general information, labelling requirements, information availability, rejection criteria, error monitoring, error reporting and interest in participation in an external quality assessment scheme. The survey was sent electronically to 63 laboratory quality managers at 55 laboratories in Ireland.
Results
A total of 39 responses (61% response rate) provided information on 94 separate laboratory departments or disciplines. Laboratories reported varying practices and requirements for labelling specimens and all accepted handwritten preprinted request forms. All (100%) respondents had defined rejection criteria both for specimen labelling and request form completion. Unsurprisingly, the rejection criteria differed between the various laboratory disciplines. Almost all respondents provided information to clinical staff on labelling requirements, but just over half provided training on the same. A large percentage of laboratories (74%) monitored the rate of specimen-labelling errors; however, only 46% had defined target limits for acceptable rates of error.
Conclusion
The survey observed a wide variation in collection, recording and monitoring of errors but also confirmed significant interest in improving preanalytical monitoring and data collection.
Introduction
It is widely accepted that the preanalytical phase is the most perilous stage of the laboratory testing process. Incorrect patient identification and subsequent specimen-labelling inaccuracies are hazardous for patients. Attempts to reduce these errors represent a significant challenge for clinical and laboratory staff alike. In a study looking at the risk occurrence associated with various aspects of the phlebotomy process, specimen labelling and patient identification were identified as having the highest risk. These two steps are critical to ensure that the correct results are associated with the correct patients. 1 Failure to correctly identify patients can have wide ranging implications. At the most serious end of the spectrum, this can lead to diagnostic error, misdiagnosis, unnecessary treatment, which may be potentially harmful or cause a delay in treatment. At the less harmful but no less serious end, it may just result in a repeat test. Both scenarios will have an impact on the patient’s mental health as a result of the unnecessary stress and anxiety caused.2,3 There is also an economic impact of these errors ranging from the cost of a repeat test to litigation. In addition, these errors can be difficult or impossible to identify in the laboratory, especially if samples are barcoded at source. 2 Although laboratories employ processes to identify and mitigate errors in real time, the recording of these errors may sometimes be haphazard, and there is a lack of standardization in how and what is recorded.3–5
The requirement to comply with the BS EN ISO 15189:2012 standard has undoubtedly contributed to significant improvements in the analytical phase in the last 10 years. However, the same rigorous methodologies have not yet been applied to the pre or post analytical phases. Lack of agreement or consistency on monitoring and standardization at national and international levels is evident. This led our research team to develop an electronic questionnaire-based survey to investigate current mechanisms in use for identification, recording, monitoring and mitigation of errors in specimen labelling and request form completion, and also to assess willingness to participate in an External Quality Assessment (EQA) scheme.
Materials and methods
An electronic survey consisting of 39 structured questions was developed by the Clinical Biochemistry Unit at TCD in conjunction with the Irish External Quality Assessment Scheme (IEQAS). Questions concerning specimen labelling and patient identification practices were organized into seven subsections covering general information, labelling requirements, information availability, rejection criteria, error monitoring, error reporting and willingness to participate in an EQA scheme. The survey was emailed to 63 laboratory quality managers based at 55 laboratories in Ireland in June 2018. The survey file is available online as supplementary material.
The survey was piloted in one large (University Hospital Limerick) and one smaller (Portiuncula University Hospital) laboratory, and the questions were adjusted based on feedback before the main circulation. A printable copy and electronic version (Microsoft Excel 2013®) of the questionnaire was provided in the email. The purpose of the study was also noted in a statement, along with information about and contact details for the research team. Recipients were also advised that any data provided would be anonymized, so no individual person or laboratory could be identified in reports. Each questionnaire contained a simple instruction for participants requesting completion of all questions. Trial runs indicated that this should take no more than 5–15 min to complete. Recipients were advised that the survey would close after four weeks, and a reminder email was issued 10 days after the initial one with a final reminder sent before the closing date to those institutions that had not yet replied. All data were entered and collated in Microsoft Excel 2013®.
Results
A total of 39 (61%) responses were received from a range of departments and disciplines (Figure 1(a)). Respondents reported varying practices and requirements for labelling specimens (Figure 1(b)). All accepted (100%) handwritten preprinted request forms. Departments reported different requirements for the number of patient identifiers on specimens and forms (Figure 1(c)). Interestingly, there was a preponderance for a greater number of patient identifiers on the manual preprinted request form, with 58% of respondents requiring three patient identifiers. Fifty-one per cent of departments advised that the labelling requirements differed across their laboratories (data not shown). Of these, 11 respondents provided further comments, all related to more stringent requirements for blood transfusion. Almost all respondents provided information to clinical staff on labelling requirements (100% provided sample labelling information, 95% provided information on completing request forms). This was via a laboratory handbook in almost all laboratories (97%). In addition, other resources were used which included sample label/request form (49%), departmental websites (25%), leaflets/posters (15%) and direct queries (62%). However, only 57% provided training on labelling requirements (Figure 1(d)).
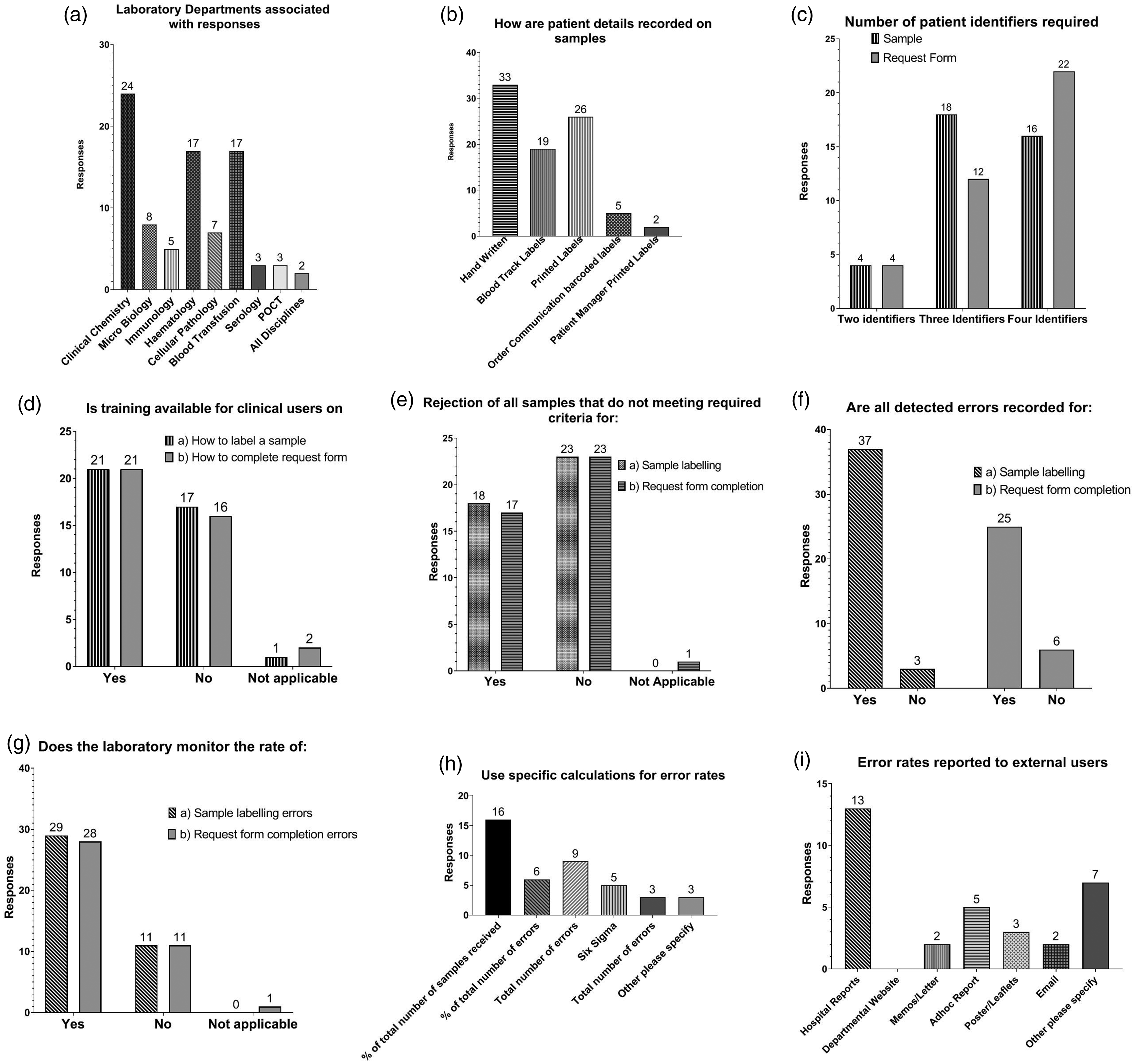
Respondents’ answers to questionnaire. Individual graphs labelled (a) to (i) represent answers to nine questions. The number of responses are on the y-axis, answer options on the x-axis. (a) Laboratory departments associated with responses, (b) how are patient details recorded on samples, (c) number of identifiers required, (d) is training available for clinical users on, (e) rejection of samples that do not meet required criteria for, (f) are all detected errors recorded for, (g) does the laboratory monitor the rate of, (h) use specific calculations for error rates, (i) error rates reported to external users.
All (100%) respondents had defined rejection criteria both for specimen labelling and preprinted request form completion. Unsurprisingly, the rejection criteria differed across disciplines in laboratories, and not all samples without the required identifiers were being rejected (Figure 1(e)). Respondents from blood transfusion laboratories identified a strict sample labelling requirement and subsequent rejection policy. Twenty-seven respondents provided additional commentary on their rejection criteria. Thirteen laboratories used a ‘disclaimer form’ completed by clinicians, where changes were made to sample labelling in the case of irreplaceable samples (e.g. cerebrospinal fluid, biopsies or bronchial washings).
A large percentage (74%) of laboratories monitored the rate of specimen-labelling errors availing of various methodologies (Figure 1(h)). However, only 46% had defined target limits for acceptable rates of error (Figure 1(g)). An important aspect of all error monitoring is feedback to the clinical users. In 77% of respondent laboratories, clinical users were made aware of their mistakes, and in 67%, there was a reporting mechanism to communicate error rates to areas outside of the laboratory (Figure 1(i)). In addition, 69% of respondents reported that clinically significant sample labelling errors were also reported to hospital risk management.
Discussion
Misidentification of a patient and subsequent specimen-labelling errors should be viewed as sentinel events and a significant public health-care issue. 2 The European Federation of Clinical Chemistry and Laboratory Medicine (EFLM) has called for harmonization of patient identification and subsequent tube labelling to ensure that specimen tubes are clearly and unambiguously linked to the patient, thereby reducing a significant patient risk. 6 International and European recommendations support the active positive identification of patients with the use of two and preferably three identifiers. Although the stage in the phlebotomy process that tube labelling should occur differs between the EFLM and the Clinical and Laboratory Standards Institute (CLSI) documents, both advise that labelling must occur in the presence of the patient.7,8 Further to this, there is an acknowledgement of the importance of monitoring errors. 6
The results of the survey indicate a wide variation in collection, recording and monitoring of sample labelling and request form completion errors. A similar observation was made by Cornes et al. for the recording of preanalytical errors. 9
Overall, positive practices were found in respect of patient identifier requirements, and all respondents had defined rejection criteria. However, there was considerable variation in the rejection criteria across different departments and even within the same disciplines, highlighting a need for improved evidence-based standardization. When asked, respondents recognized that there may be issues (such as pressure from clinicians) that prevent stricter enforcement of rejection criteria, and this is a crucial hindrance for laboratories. There are many reasons why a laboratory may not wish to reject samples; however, a restrictive specimen rejection policy has demonstrated a significant reduction in specimen-labelling errors in a 10-year study in Taiwan. That study also further highlighted the importance of reporting errors to originating departments. 10 Respondents identified a number of mechanisms for reporting errors; however, none availed of the departmental website as a communication option. As laboratory professionals, it is critical that samples that do not meet the sample acceptance criteria are rejected given the high risk to the patient.6–8 Many arguments against this relate to cost and inconvenience. However, this is greatly outweighed by the implications of an identification error which may result in harm and litigation. Only five respondents indicated the use of computerized order entry labels on specimens, 33 reported acceptance of handwritten patient identifiers, 19 used blood track labels and 26 used other printed labels. It would not be inconceivable to expect that increased use of automated barcoded specimen labels would in fact reduce the error rates. However, this may not always be the case. In 2015, Ford found in a Q-Probes study that despite an increase in the use of technology (where use of barcode armband readers increased from 8.1% in 2007 to 37.9% in 2015), there was no noticeable reduction in the rate of ‘wrong blood in tube’ errors during the same period. 11 This suggests that although technology has a role, it is not the only requirement for improvement.
The survey exhibited a significant opportunity for improvement in the area of training. Education of our clinical colleagues will undoubtedly have a role to play in reducing errors as demonstrated by Soderberg et al. 12 The importance of education and re-education was highlighted as a positive process practice improvement in test tube labelling for clinical staff attending re-education as opposed to those who had not. This area may be important not just for clinical staff but also education for scientific staff, students and our phlebotomy colleagues. 13 It is essential that the education is a continuous process with feedback and discussion, as this has been shown to have the best outcome in terms of adherence to changes. 7 This education should be part of a competency-based system for the whole phlebotomy process and should follow a programme of delivery and revalidation as for other hospital and laboratory processes and as outlined in the EFLM phlebotomy guideline. 14
Finally, the survey responses also confirm that there is substantial interest in the area of preanalytical error monitoring in Irish laboratories, which would include participation in initiatives to advance preanalytical quality monitoring and data collection. A significant improvement in this area would be the introduction of a national EQA scheme. The vast majority of respondents were highly supportive of and would participate in such a scheme, and a clear willingness was expressed to develop improved preanalytical error reporting and monitoring.
Supplemental Material
ACB903076 Supplemental Material1 - Supplemental material for Monitoring and capturing patient identification errors in laboratory medicine
Supplemental material, ACB903076 Supplemental Material1 for Monitoring and capturing patient identification errors in laboratory medicine by A Leonard, G Boran, A Kane and M Cornes in Annals of Clinical Biochemistry
Supplemental Material
ACB903076 Supplemental Material2 - Supplemental material for Monitoring and capturing patient identification errors in laboratory medicine
Supplemental material, ACB903076 Supplemental Material2 for Monitoring and capturing patient identification errors in laboratory medicine by A Leonard, G Boran, A Kane and M Cornes in Annals of Clinical Biochemistry
Footnotes
Acknowledgements
The team would like to acknowledge the help of the staff at the Irish External Quality Assessment Scheme for their assistance in distributing the survey.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
GB.
Contributorship
GB and AL researched literature and conceived the study. AK, AL and GB were involved in questionnaire development and data analysis. AL, GB and MC wrote the first draft of the article. All authors reviewed and edited the article and approved the final version of the article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
