Abstract
Background
Anti-Müllerian hormone is marker of ovarian and testicular reserve. The clinical use of this hormone requires proper standardization of reference intervals. The aims of this study were to validate the Anti-Müllerian hormone Gen II immunoassay, to establish Anti-Müllerian hormone reference intervals in healthy subjects, and to evaluate the influence of hormonal contraceptives, smoking, and body mass index on Anti-Müllerian hormone.
Methods
The validation of the Anti-Müllerian hormone Gen II assay (Beckman Coulter Company, TX, USA) was performed using a simplified protocol recommended by Clinical Laboratory Standard Institute. One-hundred and thirty-three healthy females and 120 males were prospectively selected for this study.
Results
The analytical and functional sensitivities of the Anti-Müllerian hormone Gen II immunoassay were 0.02 and 0.2 ng/mL, respectively. Intra-assay coefficients ranged from 5.2 to 9.0%, whereas inter-assay precision ranged from 4.6 to 7.8% at different concentrations. In females, Anti-Müllerian hormone showed progressive decline with increasing age (r = −0.4, p < 0.001), whereas in males, age showed no influence on Anti-Müllerian hormone concentrations. In females, Anti-Müllerian hormone concentrations did not differ between users and non-users of hormonal contraceptives, smokers, and non-smokers and obese and lean individuals. However, there was a negative and significant correlation between Anti-Müllerian hormone and body mass index in males (r = −0.3, p = 0.008).
Conclusions
Anti-Müllerian hormone Gen II assay was reliable for determining serum Anti-Müllerian hormone concentrations. Anti-Müllerian hormone concentrations declined with aging and presented a wide inter-individual variability. The lack of influence of hormonal contraceptives, smoking, and obesity on Anti-Müllerian hormone in both sexes allowed us to refine the normative concentrations for the Brazilian population.
Introduction
Anti-Müllerian hormone (AMH) is a 140-kDa dimeric glycoprotein hormone belonging to the transforming growth factor-β superfamily and it is therefore involved in cell growth and differentiation processes. 1
The role of AMH in human physiology was first described in the regression of the Müllerian ducts during sexual differentiation of the male fetus. In males, AMH is produced by Sertoli cells of the testes in high concentrations, from fetal life to the onset of puberty; at this stage the elevation in testosterone concentrations inhibits AMH secretion, which slowly regresses to residual concentrations in adulthood. 2
Historically, the determination of serum AMH in males has been used to confirm or exclude the presence of testicular tissue and evaluate Sertoli cells function mainly, in prepubertal boys, at which age testosterone concentrations are still very low. 3 Clinical application of AMH in males has been reported in sexual development disorders 4 and in the differential diagnosis between constitutional growth and pubertal delay and isolated hypogonadotrophic hypogonadism. 5 In adults, AMH concentrations can be applied to male fertility assessment, but so far the data are inconsistent. 6
In females, AMH is expressed by ovarian follicles granulosa cells from the 23rd week of gestation. 7 AMH synthesis involves the initial recruitment of primary follicles from the pool of primordial follicles and persists in preantral and antral small follicles. Once these follicles become sensitive to follicle stimulating hormone (FSH) expression, AMH concentrations decrease until they reach undetectable concentrations in antral follicles greater than 8 mm. 8 This pattern of expression suggests that AMH concentrations may reflect the number of remaining primordial follicles, in fact, Hansen et al. 9 reported a strong positive correlation between AMH concentrations and the number of ovarian primordial follicles. AMH is secreted from the ovary into circulation and can be measured in serum. It is well known that AMH concentrations in females are detectable at birth and gradually increase until the end of puberty, reaching peak concentrations around 16 years of age. Thereafter AMH concentrations remain relatively stable until age 25, when they begin to show a strong negative correlation with age, demonstrating longitudinal decline until undetectable concentrations are reached about 5 years before the menopause.10–12
Recent studies indicate that AMH is the best marker of ovarian reserve compared to chronological age (CA), 13 and to other markers such as serum FSH, oestradiol (E2), inhibin B (INHB), and antral follicle count on day 3 of the follicular phase of the menstrual cycle. 14 Clinical applicability of AMH in females has been described not only in ovarian reserve assessment, but also as predictor of menopausal transition, 15 extent of ovarian damage after chemotherapy, 16 gynecological surgery, 17 ovarian failure assessment, as in Turner syndrome, 10 in the diagnosis of polycystic ovary syndrome (PCOS), 18 as well as predictor of ovarian response in assisted human reproduction. 19
Reference intervals for AMH in both sexes are crucial for further clinical interpretation. Until 2010, two tests for AMH measurement were commercially available, one developed by Diagnostic Systems Lab (DSL) and the other by Immunotech Beckman Coulter, which shows discrepant results. 20 The consolidation of these two companies provided the scientific world with a more sensitive and reproducible test, the AMH Gen II immunoassay, which uses DSL antibodies and Immunotech (IT) standard curve.20,21
In this study, we validated the AMH Gen II immunoassay and established reference intervals for AMH in healthy Brazilian adult subjects. Furthermore, we evaluated the influence of hormonal contraceptive use, smoking, and body mass index (BMI) on AMH concentrations.
Materials and methods
This study was approved by the Ethics Committee of the Hospital das Clinicas da Faculdade de Medicina da Universidade de São Paulo, Brazil. The written informed consent was obtained from all volunteers.
Health professionals from Hospital das Clinicas da Faculdade de Medicina da Universidade de São Paulo were invited to participate in the study. Between February 2012 and August 2013, 253 healthy volunteers age 18–50 years were selected, of which 133 were females and 120 males. All participants answered a questionnaire containing personal data, height, weight, medical history, tobacco smoking (yes/no), and reproductive history, as well as hormonal contraceptive use in females.
In females, blood collection was performed between the 2nd and 7th day of the follicular phase of the menstrual cycle. Females who declared hormonal contraceptive use were instructed to collect the blood at the time of the menstrual flow. In addition to AMH measurement, a simultaneous collection for luteinizing hormone (LH), FSH, E2, and INHB measurement was performed. Blood sampling for AMH measurement, as well as for LH, FSH, total testosterone, and INHB measurement was carried out in males.
The exclusion criteria for females included chronic diseases, menstrual irregularity, PCOS, infertility, hysterectomy, oophorectomy, and serum LH and FSH concentrations out of the reference ranges. For males, the exclusion criteria included history of chronic diseases, infertility, orchiectomy, and gonadotrophins and total testosterone concentrations out of reference ranges.
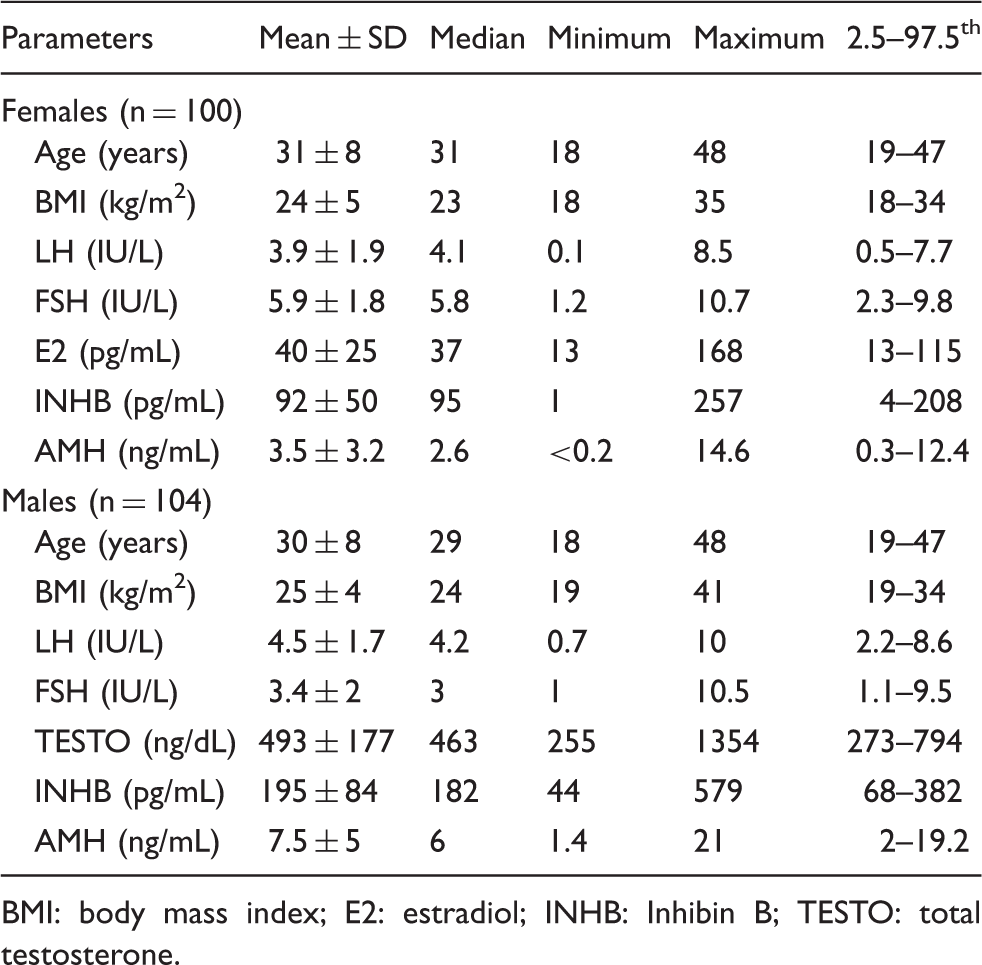
In females, the main causes of exclusion were: menstrual irregularity (n = 1), infertility (n = 2), polycystic ovaries (n = 11), and abnormal serum LH and FSH concentrations (n = 19); therefore, 100 healthy females were studied, with CA ranging from 18 to 48 years (mean = 31 ± 8 years). In males, the main cause of exclusion was abnormal LH, FSH, and total testosterone concentrations (n = 16); therefore, 104 healthy males were studied with CA ranging from 18 to 48 years (mean = 30 ± 8 years).
Hormonal evaluation
All hormonal measurements were performed in the Hormone Laboratory of Hospital das Clinicas da Faculdade de Medicina da Universidade de São Paulo. Blood samples (20 mL) were collected by venipuncture in a dry tube. After centrifugation of blood and serum separation, samples were stored at −20℃ for a period of up to 3 months before being analyzed.
AMH concentrations were measured using AMH Gen II assay (Beckman Coulter Company, TX, USA), a non-competitive enzyme-linked immunosorbent assay (ELISA) that uses two antibodies directed against different regions of the AMH molecule. The preparation and storage of reagents, calibrators, and controls, as well as the test procedure, were performed according to the manufacturer’s recommendations. No samples were submitted to preanalytical dilution. Twenty microliters of the calibrators, controls, and samples were incubated with 100 μL of the assay buffer in microtiter plate wells coated with anti-AMH antibody. After incubation and washing, the wells were treated with 100 μL of the anti-AMH detection antibody labeled with biotin. After a second incubation and washing step, the wells were incubated with 100 μL of streptavidin–horseradish peroxidase. After a third incubation and washing step, the wells were incubated with 100 μL of the tetramethylbenzidine substrate. Lastly, 100 μL of an acidic stopping solution was added. The degree of enzymatic turnover of the substrate was determined by dual wavelength absorbance measurement at 450 nm as primary test filter and 630 nm as primary reference filter. The absorbance measured was directly proportional to the concentrations of AMH in the samples. A set of AMH Gen II calibrators was used to plot a calibration curve of absorbance versus AMH concentration. The AMH concentrations in samples were calculated using this calibration curve which ranged from 0.2 to 21 ng/mL. To obtain serum AMH in pmol/L, multiply the results for ng/mL by 7.14.
LH and FSH were assessed by an immunofluorometric assay AutoDELFIA (PerkinElmer, Wallac, Finland) and by electro-chemiluminescence immunoassay (ECLIA) Cobas Analyses Elecsys (Roche Diagnostics, Indianapolis, USA). Estradiol and total testosterone were assessed by fluoroimmunoassay AutoDELFIA (PerkinElmer, Wallac, Finland) and by ECLIA Cobas Analyses Elecsys (Roche Diagnostics, Indianapolis, USA). INHB was assessed by an ELISA, Inhibin B Gen II (Beckman Coulter Company, TX, USA). The intra- and inter-assay coefficients of variation were less than 5% for all automated assay and less than 15% for manual assays in all concentrations.
Validation of ELISA AMH Gen II
The validation of AMH Gen II assay was performed according to the simplified protocol established by the Hormone Laboratory of Hospital das Clinicas da Faculdade de Medicina da Universidade de São Paulo according to recommendations of the Clinical Laboratory Standards Institute.22–24 Validation was performed according to the following protocol: (1) Analytical sensitivity: the zero standard of the calibration curve was measured 10 times in a single assay. The mean and standard deviation (SD) of absorbance were calculated. Analytical sensitivity was defined as the concentration of AMH corresponding to the mean absorbance plus two SD. (2) Functional sensitivity: two serum samples with AMH concentrations of 0.23 and 0.38 ng/mL were measured in triplicate for five consecutive days. The mean, SD, and coefficient of variation (CV) were calculated. Functional sensitivity was defined as the lowest AMH concentration with CV <20%. (3) Precision: it was expressed as CV for within run (intra-assay) and between run (inter-assay) variability. To estimate intra-assay CV, four samples with AMH concentrations of 0.4, 1.0, 3.6, and 11.3 ng/mL were measured 10 times in a single assay. To evaluate inter-assay CV, four samples with AMH concentrations of 0.4, 0.9, 3.2, and 18.8 ng/mL were measured in triplicate for 5 consecutive days. The mean, SD, and CV were calculated. (4) Linearity: two samples with AMH concentrations of 0.3 and 18 ng/mL were mixed and diluted in six different concentrations. These six diluted samples were assayed in duplicate and the observed results were plotted against the expected concentrations. Expected concentrations were calculated by dividing the concentration from the undiluted sample by the dilution factor used. Then, the % recovery was calculated by (observed value/expected value) × 100. (5) Parallelism: one sample with AMH concentration of 20.9 ng/mL was diluted with a diluent provided by the manufacturer in three different concentrations. These three diluted samples were assayed in duplicate and the observed results were plotted against the expected concentrations. Expected concentrations were calculated by dividing the concentration from the undiluted sample by the dilution factor used. Then, the % recovery was calculated as described earlier. (6) Stability of samples: Nine fresh serum samples were promptly assayed in duplicate on the day of collection. Samples were stored in a freezer (−20℃) for 90 days and reassayed in duplicate. Then, the % recovery was calculated as described earlier. (7) Method comparison: The AMH Gen II Beckman Coulter assay was evaluated against the AMH IT assay using 26 serum samples measured in duplicate and the correlation coefficient was calculated.
Statistics
Statistical analysis was performed using Sigmastat statistical program for Windows, version 3.5. Descriptive statistics (mean, median, SD, CV, minimum, maximum, and percentiles) was used to present clinical and laboratory data. The AMH reference intervals were established as results within the 2.5 and 97.5 percentiles.
The association between the use of hormonal contraceptives, smoking, and BMI on serum AMH concentrations was evaluated using Student’s t-test or analysis of variance (ANOVA) for data with normal distribution and the Mann–Whitney U test or Kruskal–Wallis test for data with non-normal distribution. Pearson’s correlation coefficient, age-adjusted when pertinent, was calculated between AMH concentrations and other numerical variables. Statistical significance was set at p < 0.05.
Results
Validation of the AMH Gen II assay
The analytical sensitivity of the method corresponded to 0.02 ng/mL and the functional sensitivity was 0.2 ng/mL. The intra-assay precision CV was 7.3, 5.2, 5.5, and 9.0% at AMH concentrations of 0.4, 1.0, 3.6, and 11.3 ng/mL, respectively. The inter-assay precision CV was 7.8, 4.6, 6.4, and 5.9% at AMH concentrations of 0.4, 0.9, 3.2, and 18.8 ng/mL, respectively. The method linearity ranged from 0.2 to 17.3 ng/mL. The parallelism study showed that the samples may be diluted to 1:10. In the stability study samples, the % recovery in the concentration of AMH in the sample stored at −20℃ for 90 days when compared to baseline varied between 93 and 120%, with a mean variation of 6.3%. The concentrations obtained with the AMH Gen II assay showed poor concordance with concentrations obtained in the IT assay (median 7.9 [1.8–19.2] versus 6.0 [0.8–26.3]). The percentage of discordance between both assays ranged from −22.7 to 85%. Linear regression analysis results were as follows: r = 0.862, p < 0.001; AMH Gen II = −1.464 + [0.85 × AMH IT] (data not shown).
Normative concentrations of AMH and influence of hormonal contraceptive use, smoking, and BMI
Females
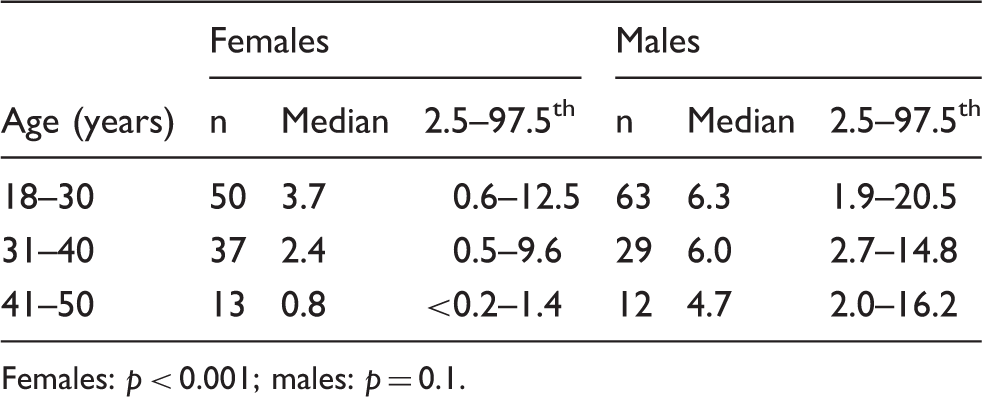
In the group of 100 healthy females, 72 were not using hormonal contraceptives and 63 were non-smokers. Regarding body weight, 66 had BMI up to 25 kg/m2, 24 were overweight (BMI between 26 and 30 kg/m2), and 10 were obese (BMI > 30 kg/m2). There was a significant negative correlation between CA and serum AMH (r = −0.4, p < 0.001) (Figure 1(a)). The median and the 2.5–97.5 percentile for AMH concentrations in the total group were 2.6 and 0.3–12.4 ng/mL, respectively. The clinical and laboratory characteristics of the 100 females included in the control group are summarized in Table 1. AMH concentrations distributed by CA are presented in Table 2. High variability and overlap of AMH concentrations were observed among all CA groups. All females older than 40 years had AMH concentrations ≤1.4 ng/mL and six aged 18–30 and 11 aged 31–40 years (19%) also had AMH concentrations ≤1.4 ng/mL.
Correlation between serum AMH concentrations and chronological age in females (panel (a)) and in males (panel (b)). Clinical and laboratory characteristics of the subjects included in the control groups. BMI: body mass index; E2: estradiol; INHB: Inhibin B; TESTO: total testosterone. Serum AMH concentrations (ng/mL) in healthy subjects according to chronological age. Females: p < 0.001; males: p = 0.1.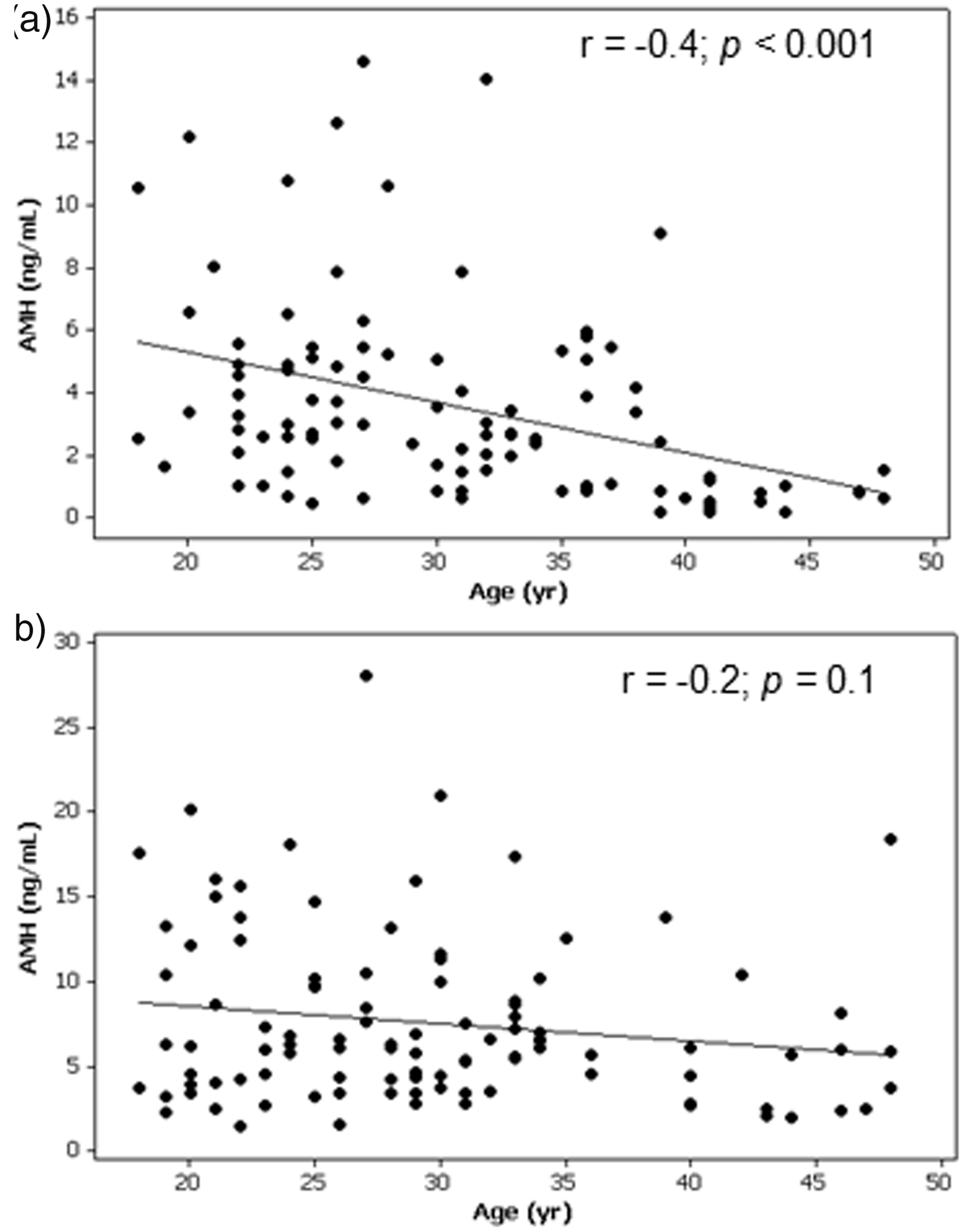
Serum AMH concentrations (ng/mL) in females user and non-user of oral contraceptives.
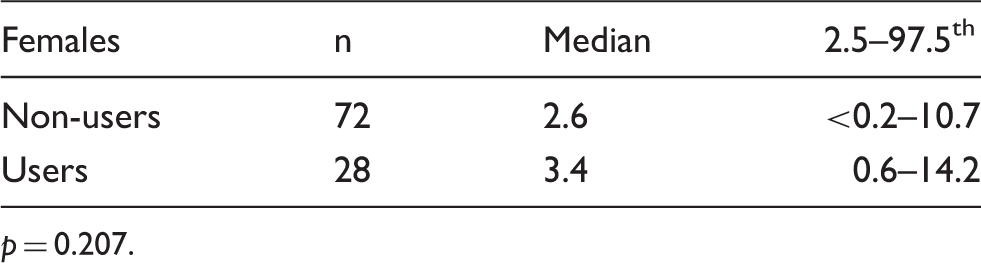
p = 0.207.
Serum AMH concentrations (ng/mL) in smokers and non-smokers subjects.

Females: p = 0.149; males: p = 0.637.
Serum AMH concentrations (ng/mL) in healthy subjects according to BMI.
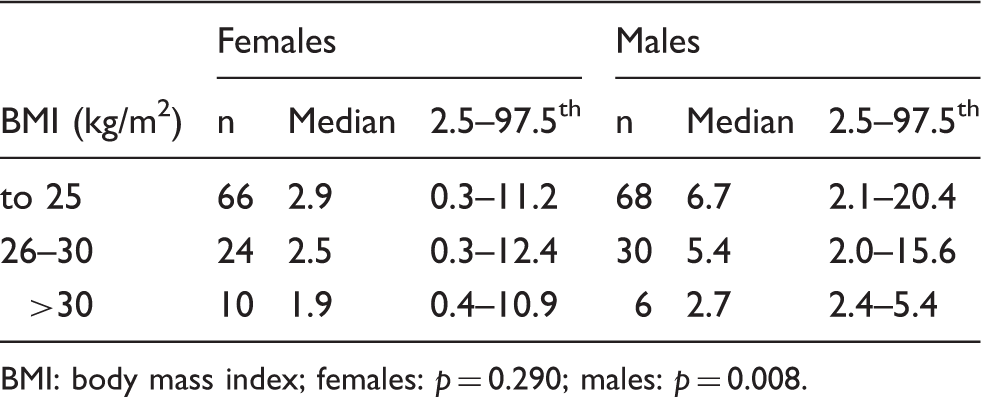
BMI: body mass index; females: p = 0.290; males: p = 0.008.
Males
In the group of 104 healthy males, 62 were non-smokers, 68 had normal BMI, 30 were overweight, and six were obese. There was no significant correlation between CA and serum AMH concentrations (r = −0.2, p = 0.1) (Figure 1(b)). The median and the 2.5–97.5 percentile for AMH concentrations in the total group were 6.0 and 2.0–19.2 ng/mL, respectively. The clinical and laboratory characteristics of the 104 males included in the control group are summarized in Table 1. AMH concentrations distributed by CA are presented in Table 2. Similarly to females, high variability and overlap of AMH concentrations were observed at all ages.
There was no statistically significant difference in serum AMH between smoker and non-smoker males (p = 0.637) (Table 4). However, there was a significant and negative correlation among AMH concentrations and the different BMI categories (r = −0.3, p = 0.008); however, AMH concentrations of overweight and obese males were within the same range of those with normal BMI (Table 5). We observed a significant correlation between serum AMH and LH, FSH, and INHB concentrations, respectively (r = −0.3, p = 0.002; r = −0.4, p < 0.001; r = 0.4, p < 0.001). Nevertheless, we found no significant correlation between AMH and total testosterone.
Discussion
To our knowledge, this is the first study to report reference intervals for serum AMH in Brazilian subjects. In this study, we validated the AMH Gen II assay, established normal AMH concentrations in healthy Brazilian females and males, and evaluated the influence of hormonal contraceptive use, smoking, and BMI on serum AMH concentrations.
The AMH Gen II assay met all acceptance criteria for method implementation in the laboratory routine. Analytical and functional sensitivity, as well as intra- and inter-assay precision, linearity, and parallelism were compatible with the previously published validation data of this same assay.21,25 AMH concentrations were compared between fresh samples and those stored at −20℃ for 90 days, with a mean of 6.3%. Two recent studies have evaluated the stability of samples stored at −20℃ using AMH Gen II assay, but the results were discordant.21,26 While Kumar et al. 21 reported variation of AMH concentrations <1% in samples stored for 7 days, Rustamov et al. 26 found a variation of 23% in samples stored for 5 days. Although the stability study design is different, our results are in agreement with the first one. We also compared AMH concentrations measured by the Gen II and IT assays and observed that AMH had a significant correlation between the two tests, as described by other authors. However, a high rate of discordance between these two assays was found.21,27 In 2012, Li et al. 27 compared that AMH concentrations obtained by these same assays and observed AMH concentrations obtained by the Gen II were 35% higher than with IT. In our study, we identified widely variable AMH concentrations when measured by Gen II compared to IT (ranging from −22.7 to 85%).
In the present study, there was a significant decline in AMH concentrations with aging and wide inter-individual variability, as reported by other researchers.10–12 Variations in AMH concentrations can be influenced not only by age, but also by the number of follicles producing AMH, 13 presence of PCOS, 18 menopause, 15 and genetic predisposition. 12 All females included in this study reported regular menstrual cycles, no known history of infertility or polycystic ovaries, and normal gonadotropin concentrations. One limitation of the study was a potential inclusion of females with PCOS as sonographic evaluation and investigation of hirsutism and/or hyperandrogenemia were not performed, which could raise upper limit of normal AMH.
In our female group, AMH concentrations <1.4 ng/mL were observed in all females older than 40 years and in 19% of females aged 18–40 years, suggesting that AMH below this concentration could represent impaired ovarian reserve, even with normal gonadotropin concentrations. Recently, Hagen et al. 28 assessed AMH capacity to predict fecundability in healthy females aged 20–35 years and showed that low AMH (0–1.8 ng/mL) in females in their mid-20 s did not predict reduced fecundability, suggesting that low AMH concentrations would not be necessarily related to impaired ovarian reserve. 28 In contrast, females with high AMH concentrations had a high incidence of subfecundity, explained by a possible association with the presence of PCOS. 28 The usefulness of AMH concentrations in predicting menopausal onset was evaluated by Freeman et al. 15 and they demonstrated that for AMH concentrations >1.5 ng/mL, the mean time to menopause was 6 years for females aged 45–48 years, and >13 years for those aged 35–39 years. 15 In the present study, it would be interesting to carry out follow-up of all females with AMH <1.4 ng/mL to determine the rate of fertility, even with low serum AMH concentrations.
To date, there is no literature consensus regarding the influence of hormonal contraceptives use, smoking habit, and obesity on serum AMH concentrations. The use of hormonal contraceptives is a reversible and widespread contraceptive method also widely used in the treatment of reproductive endocrine disorders. 29 The contraceptive effect is primarily established by the suppression of pituitary gonadotropins secretion, which results in decreased ovarian activity, namely the inhibition of follicular growth and ovulation. 29 In this study, we observed no significant statistical difference in AMH concentrations between users and non-users of hormonal contraceptives and our findings are in agreement with the results of Steiner et al. 30 and Li et al. 31 Alternatively, two recent reports showed reduced AMH concentrations in hormonal contraceptive users.29,32
It is also known that smoking exerts adverse effects on reproductive function. 33 Some studies in animal models and humans have shown that toxic substances in tobacco accelerate ovarian follicular depletion and reduce the mean age to menopause onset, but there is little evidence on the exact mechanisms involved in the premature decline in fertility. 33 We did not identify any difference in the mean AMH concentrations between females who smoke and non-smokers. The studies by Waylen et al. 34 and La Marca et al. 11 support our results. However, other authors described reduced AMH concentrations in smoker females.33,35 One weakness of this study is that we considered controls only as ‘smokers’ or ‘non-smokers,’ regardless of the number of cigarettes/day, time of smoking, and the effect of passive smoking.
Obesity overweight has an impact not only on general health, but also on reproductive health. Several studies showed the adverse effects of obesity on fertility in females trying to conceive spontaneously. 36 It is known that increased BMI contributes to reduced rates of spontaneous conception, increased risk of miscarriage, pregnancy complications, reduced response to fertility treatment, menstrual irregularity, and anovulation. 36 In 2008, Su et al. 37 reported a reduction of up to 77% of AMH concentrations in obese females, data also supported by Freeman et al. 38 In contrast, in our group there was no significant difference in mean AMH concentrations according to BMI groups, data also verified by other studies.36,39 The discrepancy between the various studies may be related to the association between BMI and age, i.e. most studies included premenopause females and since BMI increases and AMH concentration decrease with age, the association between BMI and AMH could be influenced by age, although in our analysis the age-adjusted correlation between BMI and AMH did not reach statistical significance. It is evident that the questions related to the influences of hormonal contraceptive use, smoking, and BMI on AMH concentrations in females remain and further studies are necessary to clarify these questions.
In males, in agreement with previously published data, we observed significantly higher serum AMH concentrations than in females, in addition to great variability among individuals.40–42 In contrast to the females, no influence of CA on AMH concentrations was observed in males. We identified no differences in mean AMH concentrations between smoker and non-smoker males. However, we observed significant reduction in AMH concentrations with increasing BMI. Although the group of obese males consisted of only six subjects, their lower mean AMH concentrations fell within the reference range. In 2012, Pietiläinen et al. 42 demonstrated a significant negative correlation between AMH concentrations and BMI and suggested that obesity may reduce Sertoli cell function through an interaction between adipose tissue and Sertoli cells; however, this mechanism has not been elucidated yet.
In conclusion, AMH Gen II assay was reliable for determining serum AMH concentrations. Furthermore, the lack of influence of hormonal contraceptive use, smoking habit, and obesity on serum AMH in both sexes allowed us to refine normative serum AMH concentrations for the Brazilian population, indicating that normative AMH concentrations can be improved through adequate control selection. Finally, the clinical applicability and diagnostic value of AMH serum concentrations in several clinical conditions in both sexes, in which AMH measurements could represent a potential tool, must be demonstrated in further studies.
Footnotes
Acknowledgements
We would like to thank all the staff and volunteers of the Hospital das Clínicas da Faculdade de Medicina da Universidade de São Paulo and ETEC Euro Albino de Souza cooperating with the development of this study. We thank Ms Sonia Strong for the English review.
Declaration of conflicting interests
All authors have nothing to declare.
Funding
This research was funded by Fundação de Amparo a Pesquisa do Estado de São Paulo (FAPESP) processes n° 2011/2129070 and 2012/205258 to VNB and RRW.
Ethical approval
The ethics committee of Hospital das Clínicas da Faculdade de Medicina da Universidade de São Paulo approved this study (REC number: 0666/11).
Guarantor
VNB.
Contributorship
RRW validated the assay; recruited patients, collected samples and analyzed the database, and wrote the article. LPB, MCB, HPLV performed the study design and assay validation interpretation. BBM participated in data interpretation and article revision and VNB supervised the analysis and interpretation of data and revised the article.
