Abstract
Background
Measurement of serum neuron-specific enolase (NSE) for neuro-prognostication post out-of-hospital cardiac arrest (OHCA) is recommended by international guidelines. There is, however, a lack of consensus regarding the cut-offs and time points to use. In addition, NSE is particularly susceptible to haemolysis interference. This study aimed to define the optimum NSE testing strategy to support the intensive care unit (ICU).
Methods
Patients admitted to ICU post-OHCA over 16 months had NSE measured. The outcome was survival to ICU discharge. NSE at 0 h, 24 h, 48 h, 72 h and change in NSE (ΔNSE) were assessed for prognostic accuracy using receiver operator characteristic curve analysis. The magnitude of haemolysis interference was quantified by spiking haemolysate into paired serum.
Results
There is a consistent linear increase in NSE with increasing haemolysis, independent of baseline NSE concentration. A haemolysis index acceptance threshold was defined as 20. There were 142 patients, and 82 survived to ICU discharge. The NSE parameter with best predictive performance was ΔNSE at 48 h, which had an area under the receiver operator characteristic curve of 0.91. A cut-off of >0
Conclusions
Application of a stringent haemolysis interference threshold and measurement of NSE at two time points enabled us to achieve excellent discrimination. Increasing NSE over the first 48 h, suggestive of an ongoing reperfusion injury to the brain, is a strong predictor of poor outcome.
Introduction
Out-of-hospital cardiac arrest (OHCA) is an acute medical emergency with an incidence of approximately 28,000 per year 1 and a survival to hospital discharge of around 9% 2 in the UK. Sudden cessation of blood flow causes a global hypoxic injury, and this depletion of oxygen and energy leads to cellular and tissue damage. Following return of spontaneous circulation (ROSC), reperfusion injury occurs. The mechanism of reperfusion injury is poorly understood but is postulated to be mediated by increased concentrations of radical species, causing inflammatory and oxidative damage, and ultimately leading to oedema and further cell death. 3 Brain tissue is highly sensitive to reperfusion injury because of its high need for oxygen and its high content of polyunsaturated fatty acids; therefore, neurological damage is a major cause of death in those who survive to ROSC. 4
Provision of evidence-based post-OHCA care, principally encompassing prompt coronary assessment and a period of sedation with targeted temperature management (TTM), has led to improvements in survival for patients admitted to an intensive care unit (ICU), with survival rates now around 50%. 5 For those patients admitted to ICU post-OHCA, there is a vital need to undertake accurate and unambiguous neuro-prognostication, to guide both clinical decision-making and also best interest discussions with families. Current guidelines advocate a multimodal approach including clinical assessment (e.g. review of pupillary and corneal reflexes, neurological examination), electrophysiological investigations (e.g. electroencephalogram [EEG] and somatosensory evoked potential [SSEP]), imaging and measurement of biomarkers. 6 Measurement of serum neuron-specific enolase (NSE) was included in the most recent edition of the European Resuscitation Council’s (ERC) guidelines for post resuscitation care. 5
Neurons contain a high concentration of NSE; therefore, release of NSE into the circulation from dying neurons may indicate the extent of neuronal damage. It has been known for several years that elevated NSE in serum correlates with poor neurological outcome post-OHCA.
7
There is also a high concentration of NSE within erythrocytes,
8
and measurement of serum NSE is highly susceptible to haemolysis, both intravascular and
Our laboratory introduced NSE testing in 2017 to support the ICU in following the ERC guidelines.
The present study has three goals, which will allow us to define the optimum testing strategy for NSE in routine clinical use:
To determine the magnitude of haemolysis interference in order to define a threshold for specimen acceptance. To investigate whether absolute NSE concentration or the change from baseline (ΔNSE) is most useful for neuro-prognostication. To investigate the longitudinal monitoring of NSE in order to define the optimum time point for NSE sampling.
Methods
Patients admitted to the ICU at the University Hospitals Bristol post-OHCA and who had serum NSE measured on admission were eligible for the study. Exclusion criteria were (a) death within the first 72 h and (b) no subsequent NSE concentration available. In the first phase of the study, from October 2017 to May 2018, a single subsequent sample for NSE was taken at 48 h. In the second phase of the study, from May 2018 until February 2019, subsequent samples were taken at 24 h, 48 h and 72 h. The time windows were defined as ±12 h.
Patients received care in line with ERC guidelines. 5 Patients were admitted to the emergency department where they were assessed by the ICU and cardiology teams. If deemed appropriate by the cardiology team, the patient was transferred to the cardiac catheter laboratory and underwent coronary angiography. On admission to the ICU, the patients were sedated with propofol and alfentanil for 72 h. All patients during the study period received TTM. From October 2017 until October 2018, patients were cooled to 33°C. Since October 2018, the ICU has taken part in the TTM-2 trial, 11 which randomizes patients to either normothermia (a core body temperature of up to, but no higher than, 37.7°C) or hypothermia (33°C). After a 24 h period of TTM, patients were slowly re-warmed to normothermia. Once the patient’s temperature was >36°C (and after 40 h of sedation if enrolled in TTM-2), sedation was stopped, and neurological function was assessed after 72 h. Measurement of serum NSE formed a part of this standard care since 2017; however, in light of the uncertainty around how best to interpret results, no cut-offs were specified for NSE concentrations for the duration of this study.
The primary outcome of this study was survival to ICU discharge. Non-survivors were further categorized into either primary neurological death due to hypoxic brain injury or non-neurological death. Neurological death was diagnosed according to the ERC guidelines. 5 This was a service evaluation study following implementation of international guidelines, and as such no ethics committee approval was required.
Serum NSE was measured using a commercial immunoassay (Roche Diagnostics GmbH, Mannheim, Germany). Specimens were centrifuged and separated within 1 h of being drawn, and the serum was stored frozen at −20°C until analysis. The linear range of the assay is 0.5–370
Paired serum and EDTA whole blood specimens from patients undergoing routine NSE monitoring were used to investigate the interference from haemolysis. A haemolysate was produced using the osmotic shock method: The whole blood sample was centrifuged and the plasma was removed. The red cells were washed with isotonic saline before an equal volume of deionized water was added; the sample was mixed and then subjected to a freeze–thaw cycle. After sedimentation of cell debris, the resulting haemolysate was diluted and then spiked into the paired serum. Four spikes were made, with HI increments of 5, 10, 15 and 20.
Data analysis was performed using Excel (Microsoft Corp., Redmond, USA) and SPSS Statistics (IBM Corp., Armonk, USA). Non-normally distributed data are presented as the median and interquartile range and are compared by the Mann-Whitney U test. A
Results
Investigation of haemolysis interference
There were 13 serum specimens with paired EDTA whole blood. Their baseline NSE concentrations and HI were 9–193
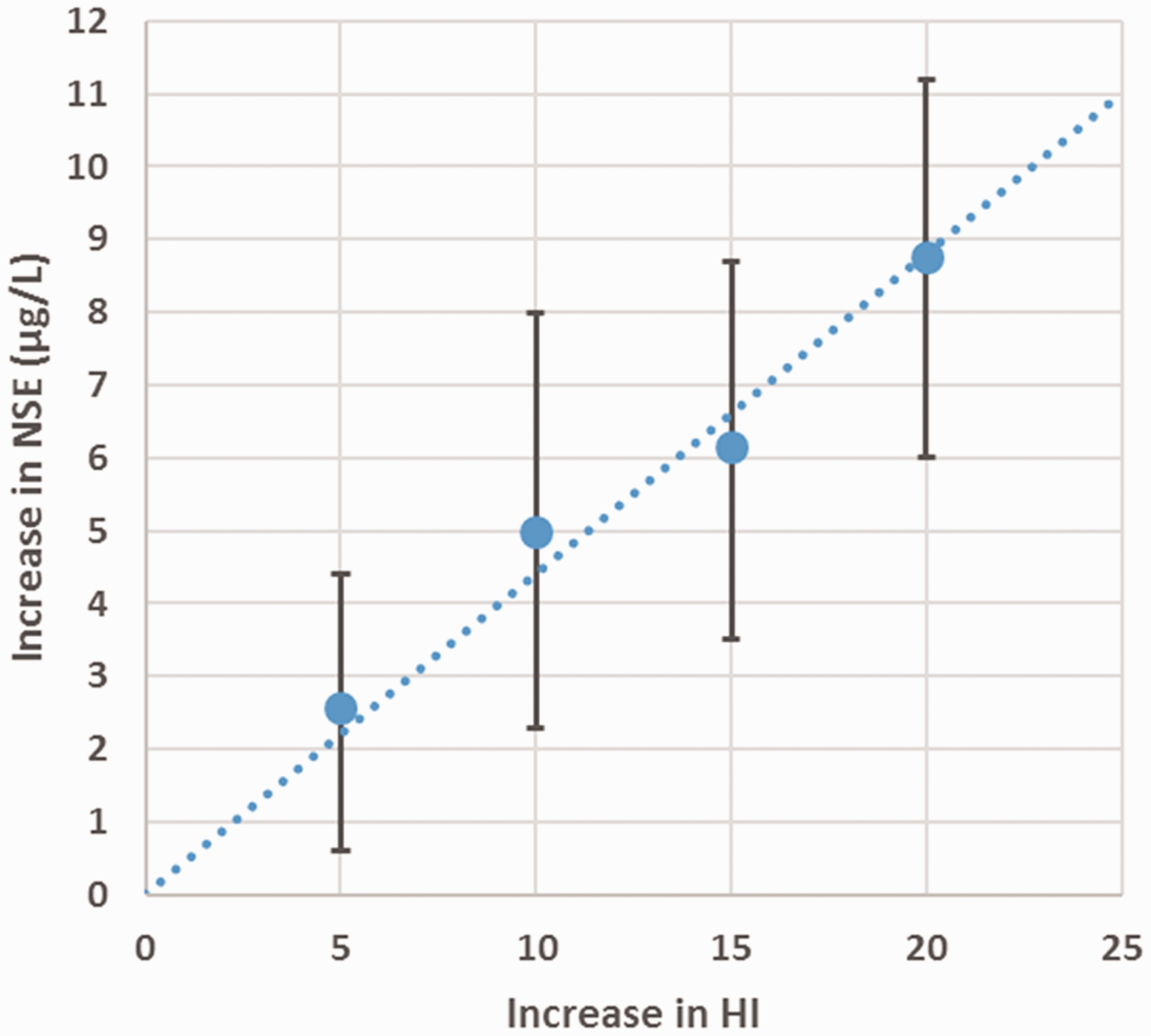
Effect of increasing haemolysis on measured serum NSE, average of 13 samples. Circles represent mean and error bars the range. The dotted line is the linear regression line.
Having established that haemolysis interference is linear and is independent of starting NSE concentration, it was possible to define the haemolysis interference threshold in terms of the change in NSE that would be expected. We defined this threshold as HI = 20, below which the error due to haemolysis should be less than 9
Consequently, all specimens received in the laboratory had HI evaluated first: if HI ≤ 20, then NSE was measured, if HI > 20, then ICU was contacted to request a repeat specimen. Based on this criterion, the sample rejection rate was 10%. The median HI of accepted samples in the remainder of the study was 7, with an interquartile range of 4–11.
Patient results
A participant flow diagram is shown in Figure 2. The numbers of patients at 24, 48 and 72 h differ because of incomplete collection of samples during the second phase of the study. Sixty of these patients died on ICU; 36 (60%) were male with a median age of 63 (IQR: 56–73). Eighty-two survived to ICU discharge; 61 (74%) were male, with a median age of 59 (IQR: 52–73). Of the patients who died, 47 (78%) had hypoxic brain injury as the primary cause of death. There were 13 non-neurological deaths; the primary cause of death in these patients was cardiogenic shock (6 patients), multiorgan failure secondary to sepsis (4), aspiration pneumonia (1), stroke (1) and severe chronic obstructive pulmonary disease (1).
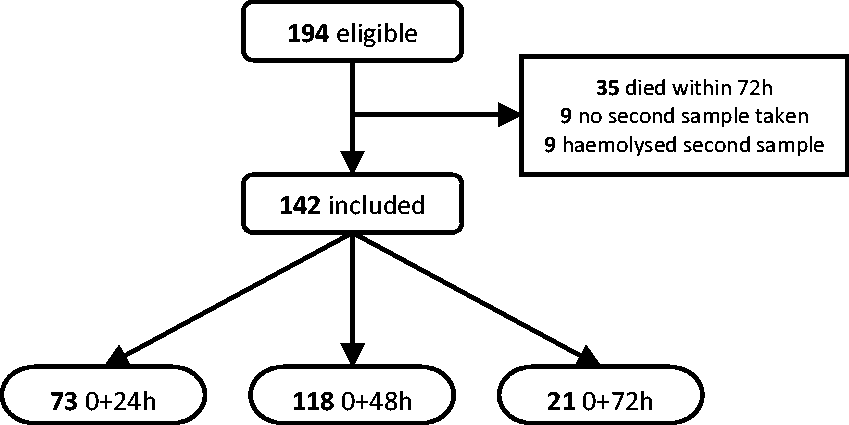
Participant flow diagram indicating the number of patients with pairs of samples at each of the time points.
Predictive power of NSE
Absolute NSE and ΔNSE were higher in those who died at every time point (Figure 3). The difference between groups was statistically significant at every time point except 0 h (
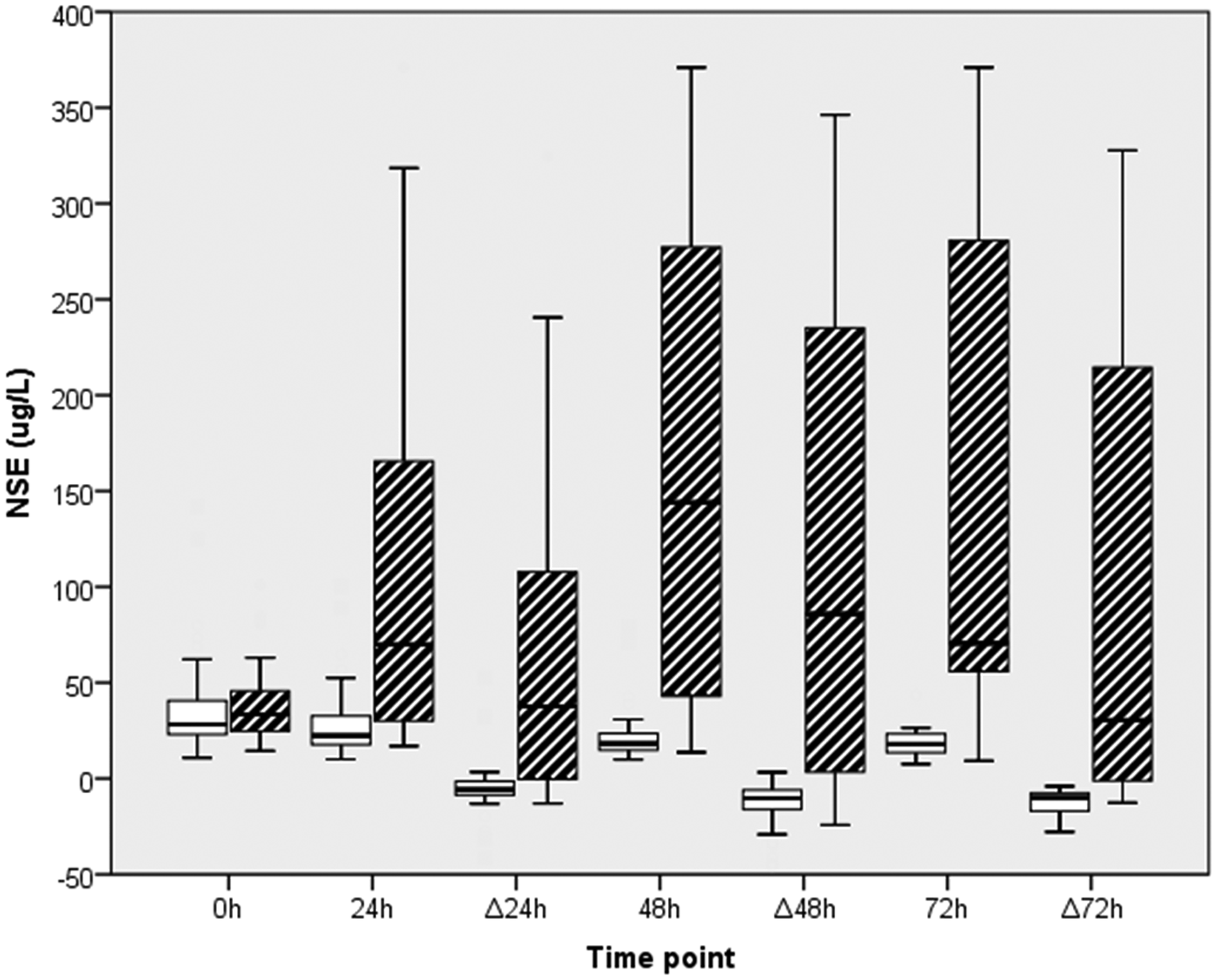
Box and whisker diagram of NSE results.
The AUC the ROC curves was calculated for absolute NSE and ΔNSE at each time point, along with the cut-off and sensitivity (Table 1 and Supplementary Table). It was not possible to define a cut-off with 100% specificity at 0 h because the highest NSE concentration at 0 h was in a patient who survived. The 13 patients who died of non-neurological causes were excluded, and the sensitivity was re-calculated (Table 1). The highest AUC was achieved for ΔNSE at 48 h. The optimum cut-off with the best combination of sensitivity and specificity is defined as the point closest to the top left corner of the ROC curve. This was 0
Predictive performance of absolute NSE and ΔNSE at each time point.
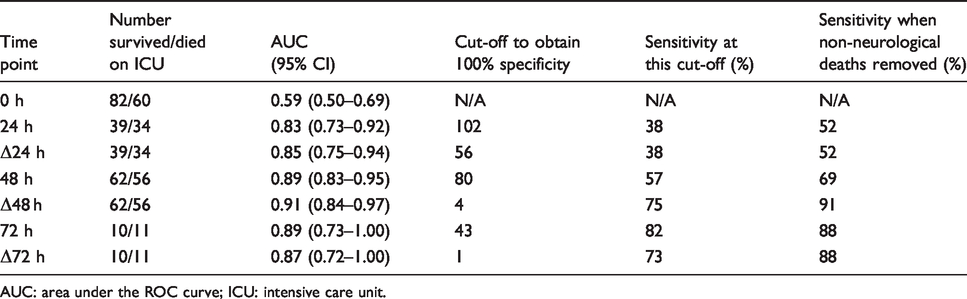
AUC: area under the ROC curve; ICU: intensive care unit.
By comparing ΔNSE as a percentage with published NSE reference change values (RCV, +30% or −23%), 12 it is possible to determine whether the ΔNSE is significant. In the survivors group, 50 (81%) patients had a significant decrease at 48 h, 12 (19%) had a non-significant change (the 2 positive ΔNSE were +3% and +18%). In the all-cause death group, 43 (73%) patients had a significant increase at 48 h, 7 (12%) had a non-significant change and 9 (15%) had a significant decrease. In the neurological death group, 41 (87%) patients had a significant increase, 3 (6%) had a non-significant change and 3 (6%) had a significant decrease.
Longitudinal NSE data
NSE was measured at 0 and 24 h on 73 patients, 0, 24 and 48 h on 51 patients and 0, 24, 48 and 72 h on 11 patients. Median NSE increased from 0 to 24 h and 24 to 48 h, before falling back from 48 to 72 h in patients who died; this pattern was more exaggerated for neurological death. In contrast, median NSE fell between each of the four time points in patients who survived (Figure 4). There were two patients who survived with a positive ΔNSE at 24 h but a negative ΔNSE at 48 h. In the subgroup of patients with samples at both 48 h and 72 h, the AUC was still higher at 48 h for both absolute NSE (0.91 vs. 0.88) and ΔNSE (1.00 vs. 0.87).
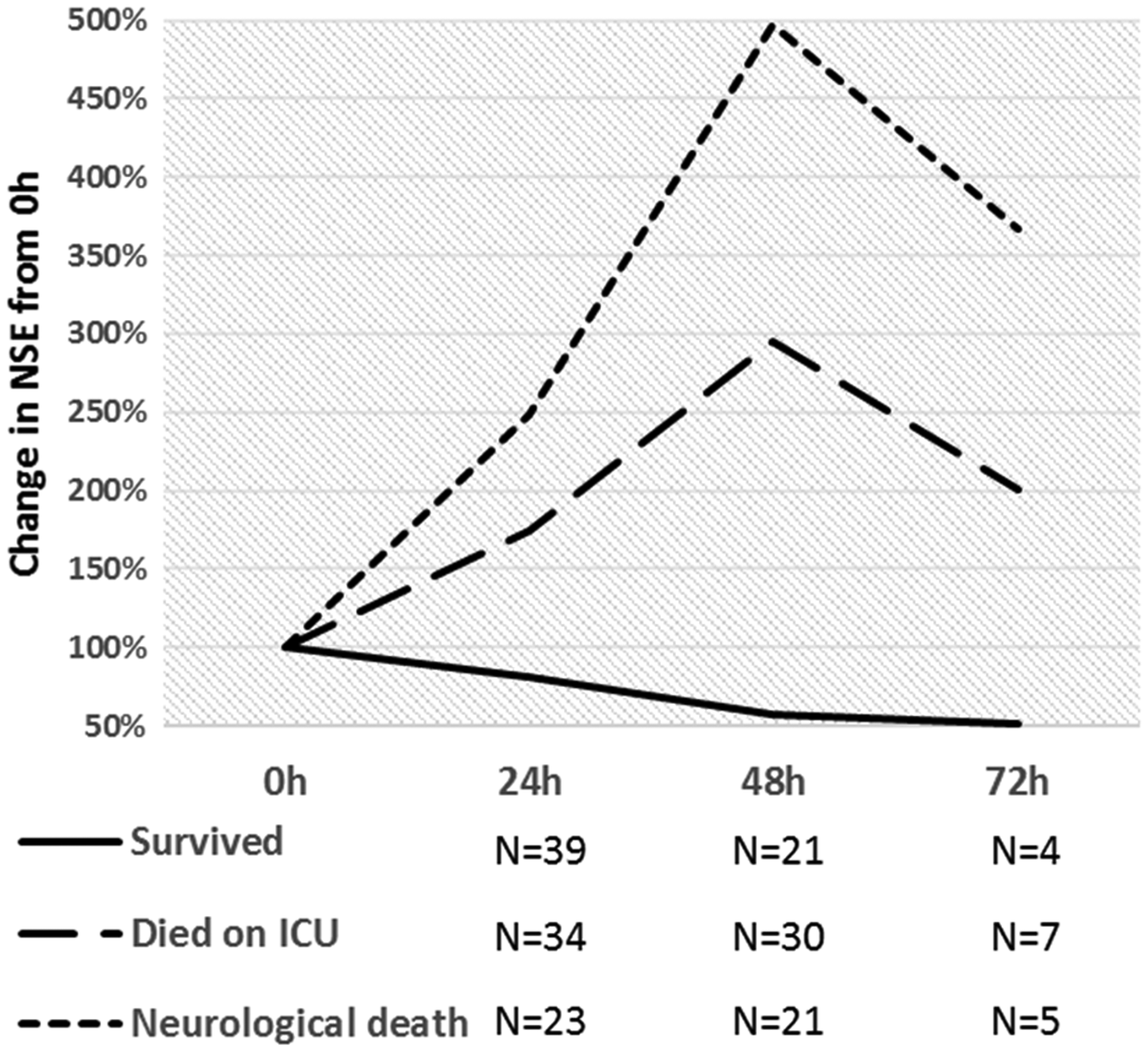
Longitudinal NSE data.
Discussion
Measurement of serum NSE as a part of multimodal neuro-prognostication post-OHCA has been recommended best practice since 2015. The guidelines state that ‘high NSE’ makes a poor outcome very likely, and that ‘sampling at multiple time points is recommended’. 5 The purpose of this study was to provide clarity on both these points. We attempted to determine the optimum time for the NSE sampling by daily measurement up to 72 h. Unfortunately, there was incomplete adherence to this sampling protocol, with low numbers of samples at 72 h in particular. The significant overlap in NSE concentrations at 0 h between the surviving and died on ICU groups makes this time point in isolation of no use for neuro-prognostication (Figure 3). In contrast, both absolute NSE and ΔNSE at 24 h, 48 h and 72 h provided a good separation between the groups, as shown by the high AUC (Table 1). The data support the use of a 48 h interval over a 24 h interval: both AUC and sensitivity are much higher at 48 h. The low patient numbers mean that the AUC estimates for 72 h are less reliable. However, the longitudinal data provide some evidence that NSE concentrations start to fall again by 72 h in those who died (Figure 4), which suggests that it becomes a less useful discriminant. The best separation between the surviving and died on ICU groups was achieved with ΔNSE at 48 h (Table 1). Taken together, these findings point to 48 h being the best time point for NSE sampling. A strong case for 48 h sampling can also be made for logistical reasons. Excluding patients in whom there is very rapid deterioration, decisions about prognosis are not made until 72 h later, at which point the EEG and SSEP can be performed. NSE data are therefore not required at 24 h, but waiting until 72 h adds a further delay for the collection and processing of the sample; 48 h provides the best compromise. The fact that sampling at 72 h is not as useful logistically may explain why we had so few samples available at this time point.
The trajectory of serum NSE between the arrest and arrival in ICU is unknown, but we can surmise that there must be an initial rise due to the hypoxic insult. A continuing rise in NSE post admission to ICU represents ongoing neuronal damage post resuscitation, presumably due to reperfusion injury. This theory provides biological plausibility favouring use of ΔNSE over absolute NSE. At our preferred time point of 48 h, ΔNSE had a higher AUC than absolute NSE, although the CI overlapped considerably and a much larger sample size would be required to prove that they differ with statistical significance. The optimum cut-off for ΔNSE at 48 h was 0
We have also shown that by removing patients who died of non-neurological causes from the data, we remove apparent ‘false negatives’, i.e. patients with a falling NSE but poor outcome. For our preferred cut-off of any positive ΔNSE at 48 h, the sensitivity rose from 80% to 91% by doing this. The rationale for this approach is that a poor prognosis due to non-neurological causes can be determined from other clinical parameters: i.e. the ICU physician is not reliant on the serum NSE concentration in these cases. Equally, our data provide a rationale to continue support for the patient with a falling NSE, because if he/she recovers, then the neurological prognosis is good. The proportion of patients who died of neurological causes is slightly higher in this study than in another published series (78% vs. 68%). 4 This may be because we excluded patients who died before 72 h, or that NSE was preferentially ordered for patients in whom neuro-prognostication was the primary clinical concern. A limitation of this study is the lack of clinician blinding to the NSE, which may have introduced bias and perpetuated the ‘self-fulfilling prophecy’ of neuro-prognostication. This was unavoidable due to the nature of the service evaluation study; we were not trying to prove that NSE is useful but rather trying to refine how it can best be used in clinical practice.
Published NSE cut-offs are summarized in Table 2. This selection is limited to more recent studies in which the supportive care given to patients included TTM; there is some evidence that mild hypothermia leads to lower NSE values and alters the AUC. 13 In the present study, all patients received TTM. As part of the TTM-2 trial, 11 (9%) patients were randomized to ‘normothermia’. Removing these patients from the data-set did not alter the AUC. Of note, two of these patients died of hypoxic brain injury and had a negative ΔNSE at 48 h, i.e. they were false negatives. Removing the 11 normothermic patients therefore slightly improved in the sensitivity achieved at 100% specificity: 78% vs. 75% for the preferred measure of ΔNSE at 48 h. It is, however, hard to draw firm conclusions from such a small number of patients.
Published NSE cut-offs to achieve 100% specificity for poor outcome.
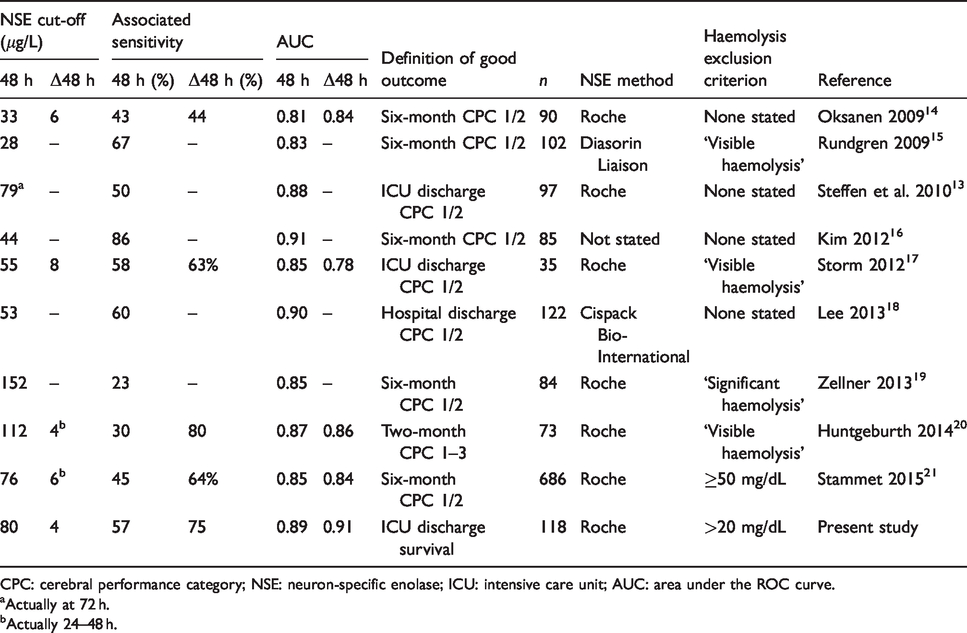
CPC: cerebral performance category; NSE: neuron-specific enolase; ICU: intensive care unit; AUC: area under the ROC curve.
aActually at 72 h.
bActually 24–48 h.
The present study differs from previously published work in that the outcome is survival to discharge from ICU, rather than cerebral performance category score, which quantifies the presence and degree of disability. The effect that the different outcome might be expected to have on our data would be to introduce false positives, i.e. patients with a positive ΔNSE at 48 h who survived but with reduced neurological function. However, at our preferred cut-off of 0, specificity is still high at 97%. Only two patients had a positive ΔNSE at 48 h and survived to discharge. Robust estimates of the biological variation of NSE have been recently published, 12 which allow us to determine whether the measured ΔNSE is statistically significant. In these two ‘false positive’ cases, the rise in NSE was below the RCV, and therefore may not reflect a true change. Both patients were observed to be interacting and talking to staff on the ward, one of them (a foreign national) in two languages. However, without further follow-up, it cannot be known for certain whether they have achieved a return to baseline function.
Most of the published studies to date looked only at absolute concentrations of NSE, and found a wide range of cut-offs from 28 to 152
A strength of this study is that we know the NSE data are not subject to significant haemolysis interference. In contrast, many of the published studies in Table 2 do not mention haemolysis or excluded only visible haemolysis, which is both insensitive and subject to variability. Pollution of NSE data with haemolysis interference would be expected to cause increased scatter; this might explain why our AUC is better, but our cut-offs are in keeping with published values.
The linear relationship between the degree of haemolysis and NSE is well known,9,10 and this susceptibility is recognized as a limitation of this biomarker.21,22 Some authors have proposed correction formulae to adjust the measured NSE value according to the HI.23,24 This approach was not considered in the present study because it was felt to introduce another source of error into the NSE concentration. Given that the patients on ICU have easy venous access, typically with central venous and/or peripheral arterial catheters
An alternative biomarker for neuro-prognostication post-OHCA, neurofilament light chain (NFL), has been proposed, with the stated advantage that is insensitive to haemolysis. 22 In our study, we have shown that by validating and then implementing a stringent haemolysis interference threshold, we can achieve similar AUC with NSE as for NFL. 22 NSE has an advantage over NFL in that a readily available commercial immunoassay exists on a major automated platform, allowing rapid and real-time analysis in routine clinical biochemistry laboratories.
The range of NSE concentrations at 48 h in the neurological death group is very broad (14−>370
This study confirms that measurement of serum NSE is a valuable tool for neuro-prognostication in comatose patients post-OHCA, as recommended by the ERC guidelines, and we have shown how this test can be incorporated into the routine clinical laboratory. We have provided a robust estimate of the magnitude of haemolysis interference on NSE measurement using the Roche assay and suggested a practical HI threshold for acceptance of samples. Finally, we have shown that ΔNSE at 48 h is superior to absolute NSE and suggest a cut-off of any positive ΔNSE at 48 h to predict poor neurological outcome.
Supplemental Material
ACB886326 Supplemental Material - Supplemental material for Serum neuron-specific enolase measurement for neuro-prognostication post out-of-hospital cardiac arrest: Determination of the optimum testing strategy in routine clinical use
Supplemental material, ACB886326 Supplemental Material for Serum neuron-specific enolase measurement for neuro-prognostication post out-of-hospital cardiac arrest: Determination of the optimum testing strategy in routine clinical use by Oliver Clifford-Mobley, Frances Palmer, Kieron Rooney, Agnieszka Skorko and Graham Bayly in Annals of Clinical Biochemistry
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
GB.
Contributorship
OCM performed the data analysis and wrote the article. FP performed the haemolysis experiments and reviewed the article. KR and AS provided the clinical outcome data and reviewed the article. GB conceived the study and reviewed the article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
