Abstract
Background
In the evaluation for hypercortisolism (Cushing’s syndrome), the 1 mg overnight dexamethasone suppression test has an important role, but false-positive results can occur due to low serum dexamethasone. Given the high intraindividual reproducibility of post-dexamethasone suppression test serum cortisol concentrations, we investigated the chance of success of repeating a non-suppressed dexamethasone suppression test if serum dexamethasone is low.
Methods
We retrospectively analysed the results of 1901 consecutive dexamethasone suppression tests performed in our laboratory from February 2011 to November 2018. Serum dexamethasone and cortisol were measured by LC-MS/MS, and both were reported. The 2.5 and 5th percentiles of serum dexamethasone in suppressed dexamethasone suppression tests were investigated as cut-off value. Then, we retrospectively determined the success rate of repeating an initial, non-suppressed dexamethasone suppression test in 131 patients, stratified by initial serum dexamethasone.
Results
At serum dexamethasone concentrations between the 2.5 and 5th percentiles (3.2–3.9 nmol/L), significantly more non-suppressed dexamethasone suppression tests were observed (27/67) than in the control group of 1357 tests having serum dexamethasone ⩾6 nmol/L (40% vs. 30%, P = 0.047), indicating that 3.9 nmol/L is the better cut-off. Overall, 40% of non-suppressed dexamethasone suppression tests were repeated, but repeat testing was performed more often when serum dexamethasone was low. In patients who had initial serum dexamethasone below the cut-off of 3.9 nmol/L, a significantly higher chance of having a suppressed repeat dexamethasone suppression test was observed compared to the control group: 57% (31/54) vs. 26% (15/57), P = 0.001.
Conclusions
Measuring and reporting serum dexamethasone in dexamethasone suppression tests have added value for the selection of patients who might benefit from a repeat dexamethasone suppression test. We suggest a cut-off for serum dexamethasone of ⩾3.9 nmol/L.
Background
In the initial evaluation for hypercortisolism (Cushing’s syndrome), the 1 mg overnight dexamethasone suppression test (DST) has an important role. In this test, the patient is instructed to take 1 mg of dexamethasone in the evening, and serum cortisol (s-CORT) is measured the next morning between 08:00 and 09:00. In healthy subjects, s-CORT is suppressed below 50 nmol/L. However, at a sensitivity of 95%, the DST has a specificity of only 80%. 1 One well-known cause for falsely positive (i.e. non-suppressed) DSTs is the use of exogenous oestrogens, which increase cortisol-binding globulin and therefore total s-CORT (but not the free fraction). 1
It has long been known that false-positives can also occur due to insufficient serum concentrations of dexamethasone (s-DXM), e.g. caused by poor compliance, fast metabolism, smoking or obesity. A cut-off value of 5.6 nmol/L s-DXM (by radioimmunoassay) has been proposed. 2 Recently, the 2.5 percentile of s-DXM in 208 negative DSTs was reported as 3.3 nmol/L, and it was shown that s-DXM below this threshold occurred in one-quarter of non-suppressed DSTs.3,4
If one initial screening test (i.e. 24 h urine free cortisol, DST or late night salivary cortisol) is abnormal, repeating the test should be considered, according to the Endocrine Society. 1 However, no proof has been given that repeating DSTs where the s-DXM is insufficient is beneficial. On the contrary, post-DST s-CORT concentrations were shown to have high intraindividual reproducibility. 3 Our hypothesis was that the chance of a suppressed repeat DST would be higher if s-DXM was low in the first test than if s-DXM was already sufficient.
Methods
s-DXM and s-CORT can be conveniently measured simultaneously by LC-MS/MS. 5 This assay has been operational in our laboratory since February 2011. As of November 2018, 1901 DSTs have been carried out, in which s-CORT and s-DXM were measured and reported simultaneously. Using these data, we first investigated the optimal cut-off value for s-DXM. Based on literature, 2 our laboratory currently uses a reference value of ⩾6 nmol/L. Others proposed the 2.5 percentile, 3 but since the reference range for s-DXM is one sided, it is more customary to use the 5th percentile. We compared the proportion of non-suppressed DSTs between the 2.5 and 5th percentiles to the group ⩾6 nmol/L. Then, we retrospectively analysed the data of all tests, in which s-CORT was incompletely suppressed (⩾50 nmol/L) and which had been repeated in the same patient. Comparisons were made between groups having different concentrations of s-DXM. Details are given in the Supporting Information.
Results
From 1244 DSTs achieving adequate suppression of s-CORT, the 2.5 percentile of s-DXM was calculated as 3.2 nmol/L, close to the value of 3.3 nmol/L found in a smaller series. 3 The 5th percentile was 3.9 nmol/L. Of all DSTs with s-DXM between 3.2 and 3.9 nmol/L, 40% (27/67) were non-suppressed. This proportion was significantly higher (P = 0.047, one-sided binomial test) than the 30% non-suppressed DSTs found in the control group of 1357 tests with s-DXM ⩾6 nmol/L.
Of all DSTs, 657 were non-suppressed. In 260 cases, a follow-up DST was available (40%). Of each patient, only the first pair of tests was taken and the maximum time interval was set at one year, leaving a study population of 131 patients. Repeat testing was performed more often when initial s-DXM was low, and s-DXM concentrations in follow-up DSTs were higher than in initial DSTs (Supporting Information).
In the control group who had an initial s-DXM ⩾6 nmol/L, 26% (15/57) showed a suppressed repeat test. The probability of having a suppressed repeat DST was significantly higher at 57% (31/54, P = 0.001) in patients having an initial s-DXM below the determined cut-off of 3.9 nmol/L. Figure 1 illustrates the percentage of suppressed repeat DSTs as a function of initial s-DXM.
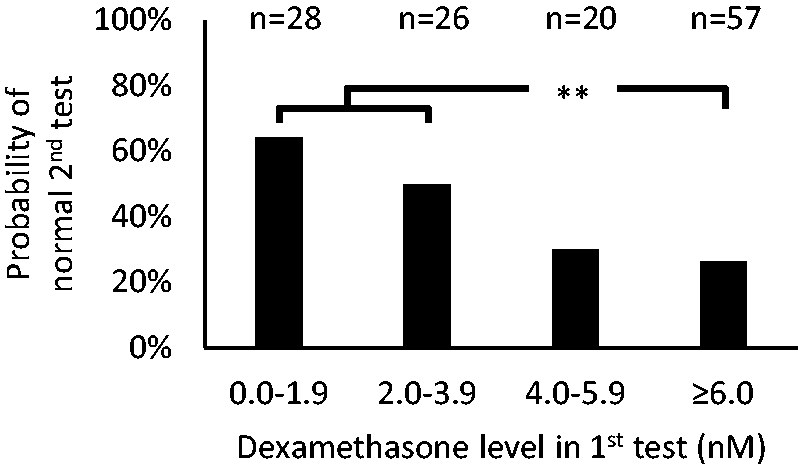
Percentage of patients who show a suppressed repeat dexamethasone suppression test as a function of the dexamethasone concentration attained in the initial test. **P = 0.001 by one-sided Fisher’s exact test.
Discussion
Although it is known that a significant part of false-positive DSTs can be explained by low s-DXM,2,3 the present study is the first to investigate whether a DST should then be repeated. Overall, 40% of non-suppressed DSTs were repeated. Repeat testing was performed more often, and the chance of having a suppressed repeat DST was significantly higher in patients having low s-DXM initially. Concomitantly, the median s-DXM in follow-up tests was higher than in initial tests. We hypothesize that for repeat DSTs, the patients received better instruction and/or a higher dose of dexamethasone. For all DSTs, our laboratory provides an interpretative comment, and all s-DXM concentrations are reported. In most interpretative comments of DSTs with s-DXM <3 nmol/L, the clinical biochemist mentioned the possibility of no or wrongly timed administration of dexamethasone. In many comments of included DSTs with s-DXM between 2 and 3 nmol/L, the possibility of the patient being a fast metabolizer was also mentioned. It seems plausible that these comments have led to repeat testing and to actions that increased s-DXM.
The optimal cut-off for s-DXM cannot be sharply defined. However, our finding of a significantly increased proportion of positive DSTs if s-DXM is 3.2–3.9 nmol/L demonstrates that the 5th percentile of suppressed DSTs (3.9 nmol/L) is a more appropriate cut-off than the 2.5 percentile.
A peculiar finding is that even in the control group, 26% of repeat DSTs were suppressed. Possible explanations are the known limited specificity of the DST even if dexamethasone is sufficient,2,3 false-negative repeat tests and patient selection bias. Intercurrent diseases or (pharmacological) interventions may have played a role especially in the control group due to their long median time interval (Supporting Information). The most obvious intervention (stopping of oestrogen-containing contraceptives) seems of little importance to our results, as the proportion of women of reproductive age was low in all groups (Supporting Information).
Conclusion
Based on our data, we conclude that measuring and reporting s-DXM in DSTs has added value for the selection of patients who might benefit from a repeat DST. We suggest a cut-off for s-DXM of ⩾3.9 nmol/L. Furthermore, it is plausible that interpretative comments by a clinical biochemist help in ensuring adequate repeat testing.
Supplemental Material
Supplemental material for Retrospective analysis of repeated dexamethasone suppression tests – the added value of measuring dexamethasone
Supplemental Material for Retrospective analysis of repeated dexamethasone suppression tests – the added value of measuring dexamethasone by Albert J de Graaf, AH Leontine Mulder and Johannes G Krabbe in Annals of Clinical Biochemistry
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
JK.
Contributorship
JK and LM conceived the study, AG performed data analysis and wrote the first draft of the article. All authors reviewed and edited the article and approved the final version of the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
