Abstract
Despite recent advances in care, women with diabetes in pregnancy are still at increased risk of multiple pregnancy complications. Offspring exposed to hyperglycaemia in utero also experience long-term health sequelae, affecting neurocognitive and cardiometabolic status. Many of these adverse consequences can be prevented or ameliorated with good medical care, specifically to optimize glycaemic control. The accurate assessment of glycaemia in pregnancy is therefore vital to safeguard the health of mother and child. However, there is no consensus about the best method of monitoring glycaemic control in pregnancy. Short-term changes in insulin dosage and lifestyle, with altered appetite, insulin sensitivity and red cell turnover create difficulties in interpretation of standard laboratory measures such as HbA1c. The ideal marker would provide short-term feedback on daily or weekly glycaemic control, with additional capability to predict pregnancies at high risk of suboptimal outcomes. Several novel biochemical markers are available which allow assessment of dynamic changes in glycaemia over weeks rather than months. Continuous glucose monitoring devices have advanced in accuracy and provide new opportunities for robust assessment of glycaemia in pregnancy. Recent work from the continuous glucose monitoring in pregnant women with type 1 diabetes trial (CONCEPTT) has provided information about the ability of different markers of glycaemia to predict pregnancy outcomes.
The aim of this review is to summarize the care for women with pre-existing diabetes in pregnancy and to highlight the important role of glycaemic monitoring in pregnancy.
Introduction
The birth of a healthy baby is a cause for celebration. Advances in maternity care over the last century have made pregnancy safer for both mother and child than ever before.1,2 The majority of women can now expect to have a healthy and uncomplicated pregnancy with delivery of a healthy baby at the end of nine months. However, for women with pre-existing type 1 or type 2 diabetes, pregnancy still represents great challenges and positive outcomes are not guaranteed.3,4 Maintaining strict glycaemic control both before conception and during pregnancy is fundamental to reducing risks for mother and child.4,5 The aim of this review is to summarize the clinical care of women with pre-existing type 1 and type 2 diabetes in pregnancy, and to discuss the challenges of monitoring glycaemic control in this population.
Prevalence, diagnosis and classification of pre-existing diabetes in pregnancy
Around 700,000 babies are born every year in England and Wales, and at least 3300 pregnancies occur in women with pre-existing diabetes (Figure 1).3,6 Recent data from the National Pregnancy in Diabetes Audit suggest that broadly half of these pregnancies occur in women with type 1 diabetes, and half in women with type 2 diabetes, but there is considerable regional variation due to local socioeconomic factors, obesity rates and ethnicity.3,6 Data from Scotland demonstrate a gradual increase in prevalence over 15 years of both type 1 diabetes and type 2 diabetes in pregnancy, culminating in rates of 1:210 and 1:504, respectively, in 2013. 7
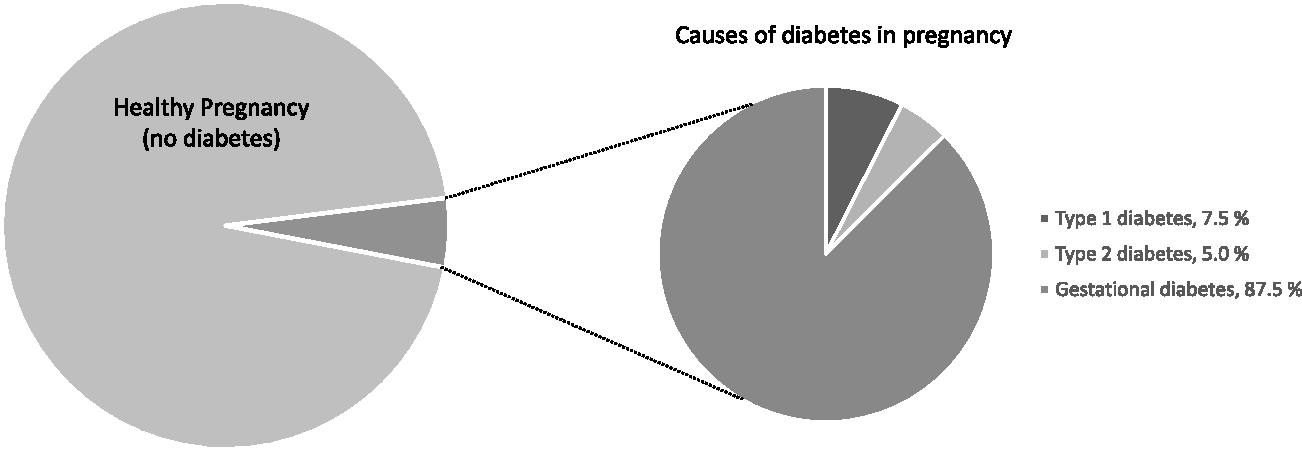
Prevalence and causes of diabetes in pregnancy in England and Wales (data from 2015). 3 Type 2 diabetes in pregnancy rates are steadily increasing.
Most women with type 1 diabetes will have had many years of diabetes duration before pregnancy, but this is not the case with many women with type 2 diabetes. Most centres will identify women every year with severe hyperglycaemia in pregnancy, usually undiagnosed type 2 diabetes, using standard diagnostic pathways to identify gestational diabetes. The classification of patients with newly identified hyperglycaemia in pregnancy which appears more severe than gestational diabetes is challenging. Although the diagnosis of type 2 diabetes should be confirmed after pregnancy, recent guidance about management of diabetes in pregnancy during the pandemic suggests that women with HbA1c ≥48 mmol/mol in pregnancy should now be treated as having likely type 2 diabetes in pregnancy. 8 The guidance of the National Institute of Health and Care Excellence (NICE) in 2015 did not stipulate exact criteria for identifying undiagnosed pre-existing type 2 diabetes in pregnancy, but recommended that clinical teams ‘measure HbA1c levels when women are diagnosed with gestational diabetes, to identify women who may have pre-existing type 2 diabetes’. 3 Guidance from the World Health Organization in 2013 classified ‘diabetes in pregnancy’ as a distinct diagnosis to gestational diabetes. 9 ‘Diabetes in pregnancy’ could be diagnosed based on a fasting glucose ≥7.0 mmol/L or a glucose of ≥11.1 mmol/L on a random sample or after a 2-h oral glucose tolerance test. The recommendations of the International Association of the Diabetes in Pregnancy Study Groups (IADPSG) from 2010 defined ‘overt diabetes in pregnancy’ as requiring a fasting glucose ≥7.0 mmol/L or a random glucose of ≥11.1 mmol/L or an HbA1c ≥48 mmol/L. Other forms of pre-existing diabetes, including monogenic forms, pancreatic diabetes and cystic fibrosis also occur in pregnant women but as highly specialist topics, are beyond the scope of this review.3,10,11
Pre-existing diabetes in pregnancy: A final frontier
Despite recent advances in the care of adults with diabetes, the clinical management of women with pre-existing diabetes in pregnancy remains an area of particular challenge. The St Vincent declaration in 1989 set a target that women with pre-existing diabetes in pregnancy should have comparable pregnancy outcomes to the general antenatal population within five years. 12 Even now, 32 years later, that target has not been achieved and unfortunately, is unlikely to be achieved in the near future. 4
Women with pre-existing diabetes in pregnancy are more likely to suffer multiple adverse pregnancy outcomes including miscarriage, stillbirth, congenital anomaly and neonatal death.6,7 They are also at risk of other pregnancy-related conditions, such as pre-eclampsia, preterm delivery and polyhydramnios. Their infants are more likely to be large-for-gestational age, which causes problems at delivery, resulting in shoulder dystocia, birth injury and high numbers of caesarean deliveries, often at 36–38 weeks’ gestation. 7 After birth, infants born to mothers with diabetes are at higher risk of neonatal jaundice, neonatal hypoglycaemia and are more likely to need treatment in the neonatal intensive care unit.4,13 There is also increasing evidence that exposure to hyperglycaemia in utero leads to longer term neurocognitive and cardiometabolic effects upon offspring.14–16
Many of these risks can be reduced by exemplary glycaemic control, both before conception and during pregnancy. 17 NICE guidelines recommend that women with diabetes aim to have an HbA1c <48 mmol/mol (<6.5%) prior to stopping contraception. 3 Women with HbA1c >86 mmol/mol (10%) should be advised to avoid pregnancy, due to the high risk of congenital malformations. 3 Further measures to reduce risk include starting 5 mg folic acid daily prior to conception and stopping potentially teratogenic medications, such as ACE inhibitors. 3 Unfortunately, despite these recommendations, many women start pregnancy with poor glycaemic control: only 15% of women with type 1 diabetes and ∼40% with type 2 diabetes have a first trimester HbA1c within the recommended range and <50% are on folic acid at any dose prior to conception. 6 Pre-pregnancy clinics are available for women who are planning pregnancy, but awareness and engagement can be low and many pregnancies are unplanned.5,18 Without pre-pregnancy planning, many women will present for their first antenatal appointment from 8 to 12 weeks, when the period of fetal organogenesis is nearly complete.
Standard pregnancy physiology and care: Further challenges to glycaemic control
Pregnancy offers unique challenges to maintaining strict glycaemic control, as insulin sensitivity varies considerably over the three trimesters. Typically, a period of enhanced insulin sensitivity begins around weeks 12–20 and can be associated, especially in type 1 diabetes, with hypoglycaemia and reduced insulin treatment requirements. 3 Careful titration of doses is needed to prevent harm to both mother and child. Conversely, after around 20 weeks, insulin resistance develops and can worsen as pregnancy progresses. This results in large increases in insulin treatment requirements and if unmanaged, can result in uncontrolled hyperglycaemia and ketoacidosis. 3 Ketoacidosis in pregnancy can be disastrous – around 20–40% of offspring will not survive. 3
In addition to the effects of gestation upon insulin resistance, pregnancy can also influence maternal appetite, behaviour and nutrition. Pregnancy-related nausea and vomiting and hyperemesis gravidarum can make prandial insulin dosing challenging, especially in the first trimester. Towards the end of pregnancy, steroid injections for threatened preterm labour, or the normal processes of labour and delivery are unpredictable and can all cause additional variability in glycaemic control.19,20
Optimizing glycaemic control to minimize complication rates
Measures to improve glycaemic control are the cornerstone of management of women with pre-existing diabetes in pregnancy. 3 All women with diabetes in pregnancy are advised to keep fasting blood glucose concentrations below 5.3 mmol/L and postprandial concentrations below 7.8 mmol/L or 6.4 mmol/L at 1 and 2 h after a meal. 3 These targets are strict and challenging to achieve. In addition to pharmacological treatments, particularly insulin, careful modification of diet, selection of low glycaemic index foods, portion control and regular exercise are all important aspects of disease management. 3
Women with established type 1 diabetes should all be on insulin prior to pregnancy. Many women will be treated with a basal bolus regimen, with prandial doses adjusted for carbohydrate intake (for example, using principles of dose adjustment for normal eating, DAFNE).3,21 Increasing numbers of women now have access to technologies such as continuous subcutaneous insulin infusion (the insulin pump) especially if they attended prepregnancy planning sessions.
Women with type 2 diabetes may be on no treatment, metformin or insulin prior to pregnancy.
Women who are not already meeting glycaemic targets often need to commence medication as soon as possible after pregnancy is identified. Even if they have been stable on metformin alone prepregnancy, women with type 2 diabetes often need large doses of insulin during pregnancy, as placental hormones contribute to worsening insulin resistance.22,23 Some women with type 2 diabetes are diagnosed for the first time during pregnancy. This poses particular challenges for affected patients: the urgent need for education to understand their condition, to start insulin therapy, learn carbohydrate counting and adhere to strict glycaemic targets can feel overwhelming.
In part because of limited opportunities for new pharmacological treatments in pregnancy, there has been a focus on increasing use of novel technologies, which can support women to attain the strict glucose targets required. These include continuous glucose monitoring, insulin pumps and closed loop systems.
Novel technologies: Continuous glucose monitoring systems
Continuous glucose monitoring systems have much improved in recent years and involve a sensor placed in the subcutaneous tissue connected via Bluetooth to a receiver device, which could form part of a computer or mobile phone. Most devices measure glucose using electrochemical enzymatic reactions involving glucose oxidase. A typical monitoring device can give over 300 glucose measurements per day, and the patterns of glycaemic change are often more useful than the absolute values (see Figure 2 for a typical report). Capillary blood glucose readings are used to calibrate the device, but many devices are now calibration free, as analytical performance has improved substantially. Factory calibration of electrochemical readings is made with venous glucose, allowing some comparison despite the difference in matrix.
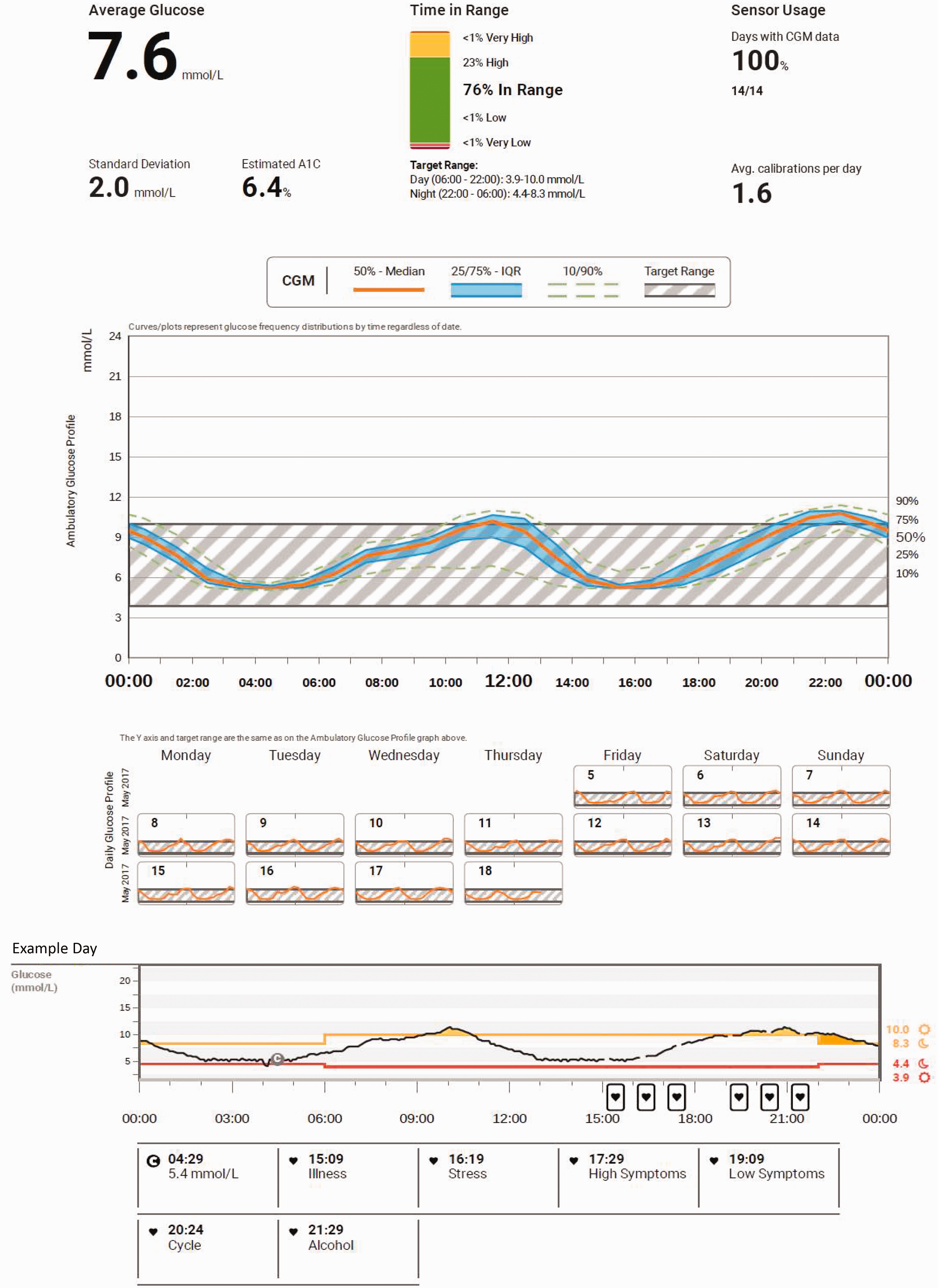
A report for a continuous glucose monitoring system in a patient with type 1 diabetes planning pregnancy. The report shows widespread daily variation even in a patient with good glycaemic control. Note that accepted targets for pregnancy are 3.5–7.8 mmol/L.
Most continuous glucose monitoring systems now provide substantially better accuracy than standard glucometers. Formal assessment of continuous glucose monitor accuracy involves a comparison with the reference standard (Yellow Springs International, YSI instrument). The metric used in regulatory assessment is the mean absolute relative difference (MARD) between the novel method and the reference method. The current generation of continuous glucose monitoring devices have a MARD of 9–11% which compares favourably to many glucometers (MARD 15–20%).24–26 In pregnancy, similar accuracy levels have been described, despite the changes in haemodynamic status. 27 While laboratory measurement of glucose offers highly accurate blood glucose quantitation, the preanalytical problems of glucose transport provides much lower accuracy overall, with a typical MARD <10%. For example, Dunseath et al. assessed the accuracy of a laboratory analyser (Randox Daytona Plus) in comparison to the YSI analyser using a 1-h delay in specimen processing to mimic standard hospital procedures. 28 The laboratory analyser measured glucose with a MARD of 6.3% (data from Dunseath et al., personal communication). As continuous glucose monitoring devices improve in future, it is likely they will have comparable accuracy to standard laboratory glucose quantitation.
In a recent work from the continuous glucose monitoring in women with type 1 diabetes in pregnancy trial (CONCEPTT), an international multicentre randomized controlled trial has provided further evidence for continuous glucose monitoring technology in type 1 diabetes. 13 The use of continuous glucose monitoring was associated with improved glycaemia with a 100-min increase in the time-in-target (i.e. the time spent in the target range of 3.5–7.8 mmol/L measured using continuous glucose monitoring). There was also a small improvement in HbA1c (2 mmol/mol; 0.2%, P = 0.02). This improved glycaemic control resulted in a profound improvement in neonatal outcomes, with reduced rates of large-for-gestational-age and admission to the neonatal intensive care unit. As the number needed to treat was eight to prevent one admission to the neonatal intensive care unit, the intervention was considered cost effective for widespread use.13,29 These findings resulted in adoption of continuous glucose monitoring devices in the National Health Service and in many other countries internationally for women with pre-existing diabetes in pregnancy. 3
In some ways, continuous glucose monitoring is a complex intervention, and its effects upon glycaemic control are likely to be multifactorial. The daily use of continuous glucose monitoring provides an opportunity to learn with real-time feedback about the impact of dietary choices, carbohydrate counting and matched insulin dosing upon glycaemia. Women can learn the best time and duration of exercise to minimize hyperglycaemia and prevent hypoglycaemia. A further benefit of continuous glucose monitoring is that hypoglycaemia can be easily identified, and even prevented, with alarm settings which identify a falling glucose trajectory. Figure 2 shows a continuous glucose monitoring report for a patient with reasonably good glycaemic control.
Novel technologies: Insulin pumps
Continuous subcutaneous insulin infusion (CSII or insulin pumps) consistent of an insulin-containing pump connected via a narrow tube to a small needle placed in the subcutaneous tissue. Pumps deliver a continuous infusion of short-acting insulin throughout the day and night, considered a more physiological approach to insulin delivery, and can be programmed to give additional boluses of insulin before meals. Many pumps can give the patient a recommendation about the bolus dose based on the carbohydrate content of a meal. The major advantage of an insulin pump in pregnancy is that it can be adjusted to give a different basal rate which can help address variations in insulin sensitivity and prevent hypoglycaemia.
Despite the widespread use of pump therapy, there is a lack of good quality evidence for its use in pregnancy. There are no large prospective randomized controlled trials using pump therapy in pregnancy. Several retrospective studies have compared pregnancy outcomes with pump therapy to women on multiple daily insulin doses (MDI) regimens and had conflicting results.30–32 This is almost certainly due to baseline differences between women on pumps and women on MDI.31,33 In spite of these limitations, pump therapy is widely used in type 1 diabetes, especially for women with suboptimal control on MDI regimens, or who have frequent hypoglycaemia. 3
Novel technologies: Closed loop (‘artificial pancreas’)
A relatively new development is the use of an ‘artificial pancreas’ which combines a ‘closed-loop’ of a glucose sensor (continuous glucose monitoring) with a pump (continuous insulin infusion) and a computer algorithm. The sensor provides a glucose measurement which is fed into the algorithm resulting in a change to the insulin infusion rate. This allows variations in the insulin infusion rate to match minute-by-minute changes in glucose concentrations. Closed-loop insulin delivery has been studied in 16 women with type 1 diabetes in pregnancy as a cross-over study. The use of the closed loop system was associated with improved glycaemic control with no increase in rates of hypoglycaemia 34 and was well tolerated. 35 A larger randomized controlled trial is underway to assess maternal and neonatal outcomes to assess if improvements in glycaemia are sustained and offer improved pregnancy outcomes.
A further development may include the use of hybrid closed loop systems, which use the closed loop for basal insulin administration with additional prandial bolus doses administered by patients. 36 This appears to give further reduction in hyperglycaemia, with no increase in hypoglycaemia. 37 However, hybrid closed loop systems have not generally been used in pregnancy, although three cases were recently published showing some possible benefits with no obvious adverse effects. 38 Further investigation is needed before these enter widespread use.
Monitoring of glycaemic control in pregnancy
Strict control of glycaemia before and during pregnancy is crucial to achieve optimal pregnancy outcomes for both mother and child. However, from a biochemical perspective, pregnancy offers unique challenges to the accurate assessment of glycaemic status. Two approaches are needed: firstly to assess the short-term dynamic changes in glucose control which arise from differences in insulin sensitivity, insulin dosing and nutritional intake; and secondly, to measure and monitor longer term glycaemic markers which provide prognostic or predictive information. A perfect biochemical marker of glycaemic control has yet to be identified for use in pregnancy, but there are several novel or established glycaemic markers available which may prove useful in future, including fructosamine, glycated albumin, 1,5 Anhydroglucitol (1,5 AG) and glycated CD59 (gCD59). Furthermore, continuous glucose monitoring has also been used for monitoring of control and prediction of suboptimal outcomes.
Monitoring of short-term glycaemia: Capillary monitoring using a glucometer
Home blood glucose monitoring has an important role but reflects relatively short-term changes in glycaemic control. Furthermore, it is dependent upon the patient doing the test at the correct time and in the correct manner to achieve an accurate result. Although self-monitoring of blood glucose remains an important part of pregnancy care, its use is likely to be superseded by continuous glucose monitoring systems in future.
Monitoring of short-term glycaemia: Continuous glucose monitoring (interstitial glucose)
The use of continuous glucose monitoring as an intervention to improve glycaemia has been described above. However, the primary aim of these devices is to monitor glycaemic control in the short term. Recent work suggests that continuous glucose monitoring metrics also provide predictive information.
Monitoring of long-term glycaemia for outcome prediction: Continuous glucose monitoring
A secondary analysis of our recent CONCEPTT study assessed if continuous glucose monitoring metrics could provide predictive information. 39 Data were gathered from continuous glucose monitoring (minimum 72 h) in each trimester and compared with biochemical markers of glycaemia taken at the same timepoints. We identified that time in range (TIR; 3.5–7.8 mmol/L, expressed as a percentage of daily values), time above range (TAR) and mean glucose all provided predictive information from the first trimester. In the first and second trimesters, TIR was able to predict preterm birth, large-for-gestational age, neonatal hypoglycaemia and offspring admission to NICU. In the third trimester, TIR was able to predict large-for-gestational age and neonatal hypoglycaemia.
Monitoring of long-term glycaemia for outcome prediction: HbA1c
HbA1c is considered the standard of care for assessment of glycaemia in pregnancy. The formation of HbA1c is generally believed to reflect glycaemia over two to three months in adult life.40,41 HbA1c is widely used in the non-pregnant population and is associated with the long-term risks of retinopathy, nephropathy and mortality in people with diabetes.42,43 However, although it is also used in women with diabetes in pregnancy, HbA1c has some particular limitations in these circumstances. Firstly, as red cell turnover is different during pregnancy,44,45 HbA1c concentrations will also vary as pregnancy progresses and changes in HbA1c may not necessarily reflect changes in glycaemia alone. 46 In addition, iron deficiency is common in pregnancy and may be undiagnosed, resulting in an elevated HbA1c regardless of glycaemia. 47 HbA1c is generally believed to reflect glycaemic control over the preceding two to three months, but this may not be reliable even in a non-pregnant population. 41 It is likely to be even more unpredictable in pregnancy, when red cell lifespan may be much more variable. Due to the short duration of pregnancy, and the need for rapid improvements in glycaemic control over a short time, HbA1c often improves too slowly to assess the benefits of changing treatment strategies.
Nonetheless, HbA1c is still used in pregnancy to assess baseline glycaemic control in women with pre-existing diabetes and to exclude undiagnosed type 2 diabetes in women with gestational diabetes. 3 I recently carried out a comprehensive assessment of the predictive capabilities of HbA1c in women with type 1 diabetes in pregnancy. HbA1c, especially in trimesters 2 and 3, was directly associated with multiple pregnancy outcomes, including preterm birth, large-for-gestational age, neonatal hypoglycaemia and admission to NICU. 39 HbA1c has also been associated with important adverse events including stillbirth and congenital anomalies. 6 Despite the many factors which influence HbA1c concentration in pregnancy, and the resulting limitations, it remains a useful predictive tool. No other single marker was able to improve upon the global predictive capability of HbA1c in pregnancy. 39
Alternative or novel markers of glycaemic control: Glycated albumin and fructosamine
Fructosamine (total glycated serum proteins) and glycated albumin (expressed as a percentage of total albumin) have been considered to offer some benefits above HbA1c in pregnancy. These markers provide a measure of glycaemic control over the last two to four weeks and have been associated with the development of diabetes and microvascular complications in non-pregnant adults.48,49 Although fructosamine and glycated albumin are unaffected by altered haemoglobin concentration, iron deficiency, red cell turnover or ethnicity,50,51 they have other potential disadvantages in pregnancy and are still subject to variability due to the dilutional effects of pregnancy. 52 In pregnancy, fructosamine has been tested as a screening method for gestational diabetes with varying results53,54 but showed no consistent associations with pregnancy outcomes. 55 With an international group of collaborators, I recently identified that fructosamine and glycated albumin are poor predictors of outcomes in women with type 1 diabetes in pregnancy, although fructosamine were associated with the development of pre-eclampsia in the third trimester. Given the associations between pre-eclampsia and protein catabolism and proteinuria, it is likely that concentrations of fructosamine and glycated albumin are altered due to the disease process itself, rather than in advance, negating a predictive role. 56
Alternative or novel markers of glycaemic control: 1,5-anhydroglucitol
A glycoprotein called 1,5-anhydroglucitol (1,5 AG) has recently been proposed as a marker of glycaemic control (tradename Glycomark). 1,5AG is apparently not metabolized in humans but enters the body through the diet and is excreted in the urine. Glucose competes with 1,5AG re-uptake in the renal tubule, and thus periods of hyperglycaemia promote excessive loss of 1,5AG which takes around two weeks to stabilize. 57 1,5AG has been assessed in women with diabetes in pregnancy and has been found to be inversely associated with birthweight, and in mothers with gestational diabetes, with neonatal hypoglycaemia.58–60 Our own work in women with type 1 diabetes confirmed the association between 1,5AG and large-for-gestational age in all three trimesters, but overall, 1,5AG was not a strong predictor of most other relevant outcomes. 39 1,5AG was also affected by pre-eclampsia in the third trimester, perhaps due to altered renal function. 39
Alternative or novel markers of glycaemic control: Glycated CD59 (gCD59)
Several reports in the literature have identified a novel glycaemic marker called gCD59.61,62 CD59 is a complement protein which prevents cell lysis following complement activation. Glycation of CD59 (‘gCD59’) appears to alter its function and has been implicated in macrovascular complications in diabetes outside of pregnancy.63,64 gCD59 falls within two weeks of a treatment intensification. 61 However, gCD59 has not been thoroughly tested in pregnancy although has shown some promise in early gestational diabetes. 62 We assessed gCD59 performance at 24 weeks in women with type 1 diabetes in pregnancy and identified that it was strongly associated with neonatal complications, including neonatal hypoglycaemia and admission to the neonatal intensive care unit. 39 Although the overall performance of HbA1c was superior, there is opportunity for gCD59 to emerge as a predictor of neonatal complications. 39
Additional aspects of clinical care for women with pre-existing diabetes in pregnancy
In addition to assessing and optimizing glycaemia, clinical care also requires attention to other aspects to reduce complications. All women should be assessed for deep vein thrombosis risk and prophylaxis should be considered. Aspirin is given after 12 weeks to reduce the risk of pre-eclampsia. Improvements in glycaemic control can worsen retinopathy, and regular eye surveillance is important. Many people with diabetes will have other comorbidities, such as thyroid disease, which may also need specific management during pregnancy. 3
Labour and delivery
Women with type 1 or type 2 diabetes in pregnancy are at high risk of perinatal complications. Operative and instrumental deliveries are common, often precipitated by a large-for-gestational-age baby. Therefore, guidelines recommend that women should be admitted for elective delivery between 37 and 38 + 6 weeks of pregnancy. 3 This usually involves an induction of labour or a caesarean section. However, if there are metabolic maternal or fetal complications, delivery before 37 weeks is recommended. Data from the NPID audit suggest that around 65% of pregnant women with type 1 diabetes and 57% of women with type 2 diabetes have a caesarean section. 6
Conclusions
While advances in maternity care have made pregnancy safer for both mother and child in the general population, women with pre-existing diabetes in pregnancy still experience high rates of pregnancy complications which cause short-term and long-term consequences upon the health of their child. Despite the early enthusiasm of the St Vincent declaration, and the advent of many useful technologies since then, the resolution of all morbidity associated with pre-existing diabetes in pregnancy may seem to be an unattainable goal. However, steady progress has been made in the measurement and optimization of glycaemic control in pregnancy, providing some hope for the future.
Footnotes
Acknowledgements
The author would like to thank the diabetes in pregnancy multidisciplinary team at Cambridge Universities NHS Foundation Trust, and all colleagues and collaborators involved in the CONCEPTT clinical trial. This article was prepared at the invitation of the Clinical Sciences Reviews Committee of the Association for Clinical Biochemistry and Laboratory Medicine.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: CLM is supported by the Diabetes UK Harry Keen intermediate clinical fellowship (17/0005712) and the European Foundation for the Study of Diabetes – Novo Nordisk Foundation Future Leader’s Award (NNF19SA058974).
Ethical approval
Not applicable.
Guarantor
CLM.
Contributorship
CLM planned the article and wrote the article.
