Abstract
We hereby describe the rare case of a 59-year-old patient presenting with marked hyperamylasaemia mimicking acute pancreatitis upon admission. Investigation of co-existent hypokalemia revealed the presence of ectopic adrenocorticotropic hormone secretion, leading to the final diagnosis of small cell lung cancer, exhibiting dual paraneoplastic syndromes including Cushing Syndrome and hyperamylasaemia. Although, paraneoplastic syndromes occur commonly, paraneoplastic hyperamylasaemia especially in the context of dual paraneoplastic syndromes occurring simultaneously, is extremely rare. Such misleading manifestations require a high index of suspicion on behalf of the physician, so that an underlying malignancy is not missed, and a final diagnosis combining all clinical and laboratory findings is reached. In turn, in rare cases common biochemical markers such as amylase can be used as a useful follow up index driving further management.
Keywords
Introduction
Patients can present with multiple paraneoplastic syndromes at the same time, especially when the tumour arises from neuroendocrine cells. Although rare, there have been reports in the literature of patients with two or more paraneoplastic syndromes complicating small cell lung cancer (SCLC).1–6 We also found a single report of a patient with two sequential paraneoplastic syndromes concurrently with SCLC. 7 Recently, there has been a case report of SCLC secreting both adrenocorticotropic hormone (ACTH) and calcitonin at the same time. 8 To the knowledge of these authors, this is the fourth – the first in the last 28 years – reported case of amphicrine intrathoracic tumour resulting in paraneoplastic hyperamylasaemia and Cushing Syndrome (CS).9,10
Case presentation
A 59-year-old Caucasian male was referred to our hospital by a primary care unit initially presenting for an episode of mild epigastrial pain and a feeling of abdominal fullness. At the time the patient complained for gradually increasing lower limb oedema, as well as, loss of appetite during the last 20 days. He complained of no other symptoms whatsoever, while his medical history was unremarkable, except for regular agricultural activities, pets and consumption of unpasteurized dairy products. He admitted consuming around 500 mL of wine/day for the last 20 years, being a 40 pack/year smoker and not doing regular health check-ups. Physical examination revealed increased body mass index with truncal obesity in excellent condition, vitals of BP 170/90, 72 bpm, T: 37℃, moon facies, mild epigastrial tenderness and lower limb oedemas bilaterally; otherwise unremarkable (Figure 1(A) and (B)). Regular laboratory studies revealed marked hyperamylasaemia and hypokalemia, together with mildly elevated aminotransferases and increased glucose value (fasting 14.8 mmol/L) (Figure 2). In the context of obesity and alcohol consumption, the diagnosis of acute pancreatitis was made. Interestingly, his CXR also exhibited a nodule on the right upper lobe. Following unfinding cardiological assessment, the patient was then referred to our tertiary hospital to manage further his pancreatitis, oedemas and investigation of his nodule.
(A) Cushingoid face and (B) marked ankle oedema was observed upon presentation. (C) Full body CT scan revealed presence of abnormal tissue at the level of upper right pulmonary lobe as well as co-lateral hilar lymphadenopathy. (D) Secondary lesions were observed in the liver; however, no signs of inflammation were noted in the pancreas or peripancreatic area. (E) Tissue biopsies following bronchoscopy revealed small cell carcinoma of the lung exhibiting densely packed small tumour cells with darkly stained oval to spindle nuclei without nucleoli and scant cytoplasm (Hematoxylin and Eosin, ×40). (F) CD56 immunoreactivity with a membranous staining pattern (×40). Schematic representation of amylase, potassium and Ht level through the course of the disease. Vertical axis represents laboratory values, horizontal axis represents time of presentation in the primary and tertiary unit, as well as, days of hospitalization in the latter.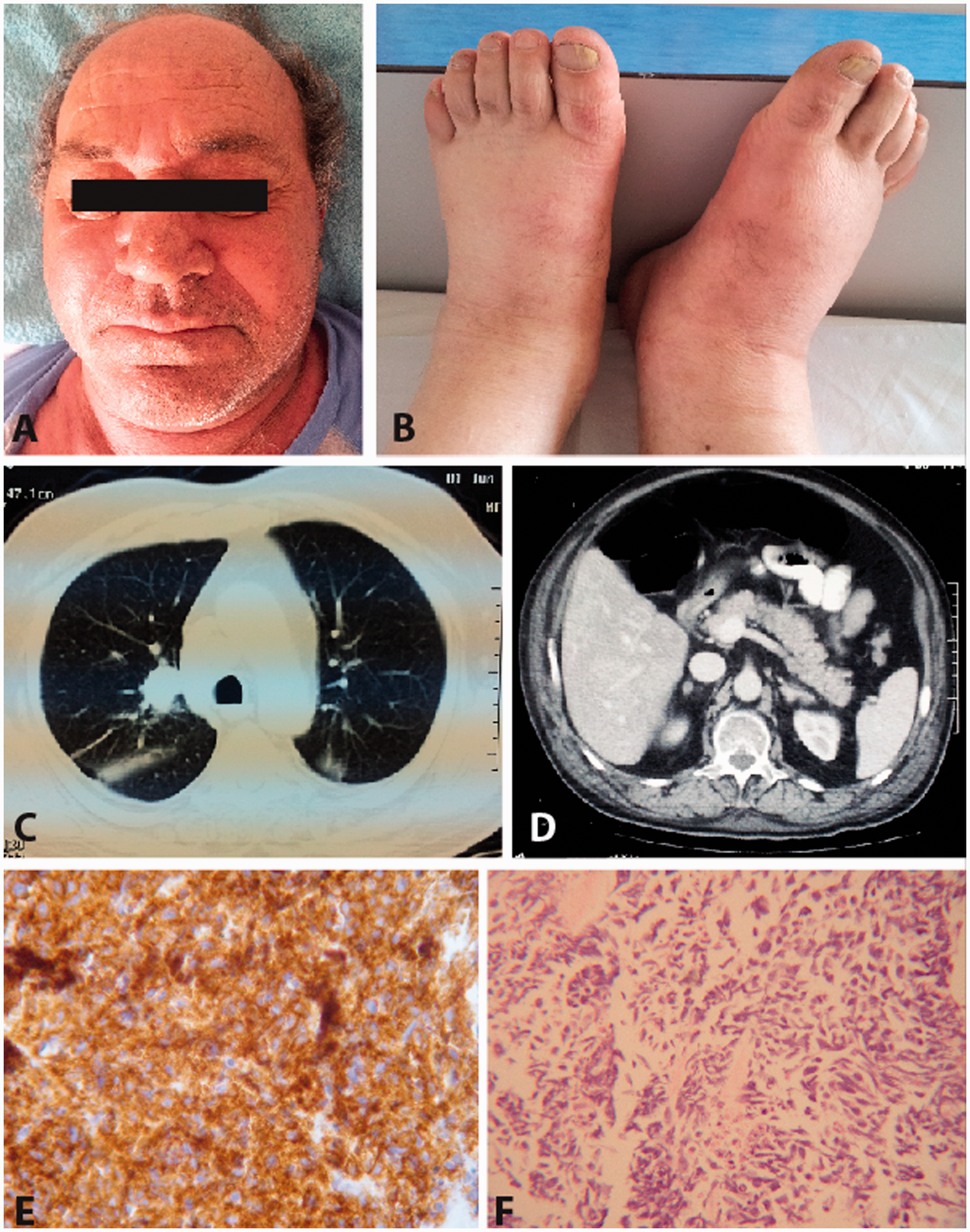
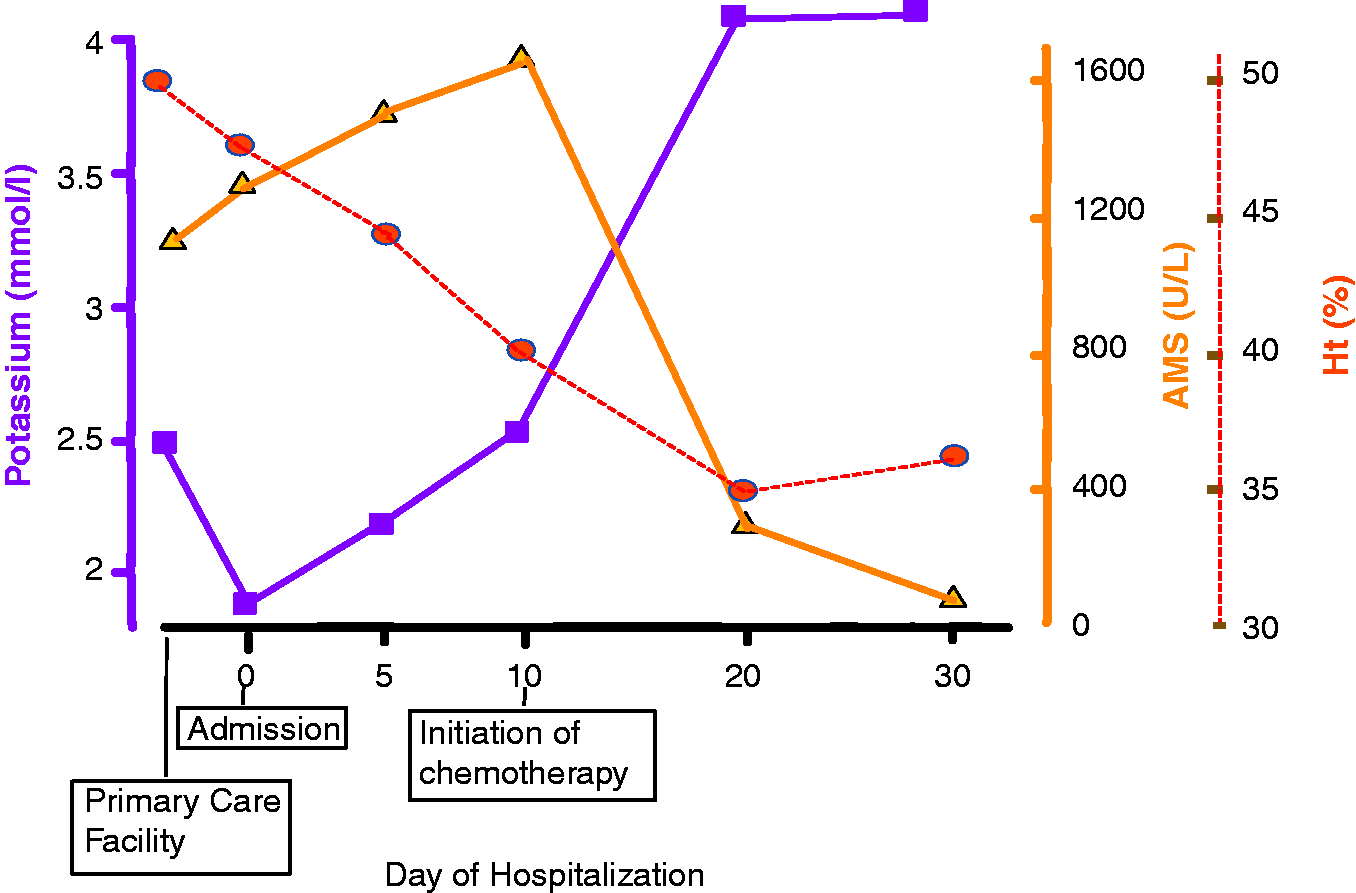
Upon admission, a full body CT scan was scheduled to assess his pancreatitis and pulmonary nodule. In parallel, an endocrinology panel of tests was ordered to look into excessive hypokalemia. Notably, serum cortisol and ACTH levels were found to be extremely elevated, i.e. >1750 nmol/L and >67.7 pmol/L, respectively. Hormone levels remained equally elevated even following dexamethasone administration (8 mg), in the absence of adrenal or sellar focal lesions, being suggestive of ectopic ACTH disease. In the meantime, imaging revealed no signs of inflammation or other disturbances in the pancreas or peripancreatic tissue, in contrast to the right upper pulmonary lobe, where the presence of abnormal tissue was observed (Figure 1(C) and (D)). At this time amylase levels remained elevated, the majority of salivary type isoenzyme, urine amylase were markedly elevated accordingly, while the calculated ratio of amylase to creatinine clearance was within normal limits (<5%). Based on the isoamylase patterns and on the radiologic findings, an acute pancreatitis was excluded; the neoplasm appearing to be the major source of the hyperamylasaemia and CS. Bronchoscopy specimens came back to set the diagnosis of SCLC (Figure 1(E) and (F)). Upon electrolyte improvement following kenoconazole and potassium administration, the patient was immediately put on carboplatin and etoposide, and responded well as shown by electrolyte and enzymatic rapid improvement (Figure 2). Interestingly, amylase levels subsided to be normalized by the end of the second cycle of chemotherapy.
Discussion
Marked hyperamylasaemia associated with ectopic production by nonpancreatic tumours appears to be a rare phenomenon. Occasional reports have documented an association of hyperamylasaemia with lung cancer, ovarian cancer and recently pheochromocytoma.11,12 Amylase-producing tumours of the lung include primarily adenocarcinoma and secondly undifferentiated small-cell carcinoma, comprising in total 1 to 3% of all lung cancers.13–16 Small-cell lung cancer originates from the Kulchitsky cell, a cell of neural crest, which is present beneath the epithelial lining cells of the lungs. These tumours are able to secrete polypeptide hormones and enzymes and bear antigenic markers of both the neural crest and epithelium. 17 It has been suggested that except for rare cases of tumours producing the enzyme, increased a-amylase activity in serum and pleural fluid associated with carcinoma of the lung may be due to an increase in the amylase contained in normal lung tissues; but the cellular site of production is uncertain.18,19 The presence of tumour cells may cause an inflammatory response resulting in activation and release into the blood stream of the enzyme. In every reported case of hyperamylasaemia in lung cancer patients, the a-amylase present in the serum or in the tumour tissue has been described as of the salivary amylase type.14,16,20
In our case report, strong evidence indicates hyperamylasaemia as a paraneoplastic phenomenon. Firstly, the marked salivary type hyperamylasaemia in the absence of inflammatory conditions of both the pancreas and salivary glands and secondly, the significant decrease of amylase values soon after the starting chemotherapy. Unfortunately, unavailability of salivary gland type amylase antibody did not allow further histopathological detection of enzymatic activity in the neoplastic tissue that would provide definite evidence of paraneoplastic secretion. Interestingly, in our case, the paraneoplastic hyperamylasaemia behaved like a specific tumour marker, since its fall reflected tumour response to chemotherapy. Similarly, in previous cases when signs of a relapse appeared, the serum amylase level again rose subtly in comparison to other tumour markers.21–23 Minami et al. 13 and Kitazawa et al. 24 reported the serum amylase levels to be a potentially useful tumour marker for both the diagnosis of lung cancer and the follow-up. On the other hand, Uno et al. 25 reported that the serum amylase levels were not associated with the severity of the disease. However, the clinical efficacy of this tumour marker remains controversial because some previous studies have shown that the level of amylase synthesized in the tumour tissue does not correlate with hyperamylasaemia.21,22
CS occurs in approximately 1 to 5% of SCLCs. In a large retrospective study, 14 of 840 (1.6%) patients with SCLC had clear-cut findings of ectopic ACTH production. 26 It is mainly attributed to ectopic expression of ACTH, or rarely corticotrophin releasing hormone, and presents with the rapid onset of hypertension, edema, hypokalemia and glucose intolerance as typically shown in our patient. 27 SCLC accounts for ∼75% of all cases of ectopic ACTH syndrome. Cancer patients generally present with electrolyte disturbances rather than typical Cushingoid features (buffalo hump, striae) because the hypercortisolism is acute and the patients generally do not survive long enough for morphologic changes to occur. Patients with SCLC and CS tend to have more extensive disease and exhibit less response to chemotherapy, and they are more likely to die prematurely. 28 Retrospective studies have shown that median survival in SCLC patients with CS ranges from 3.5 to 5.5 months, suggesting that the presence of ectopic ACTH syndrome is a poor prognostic factor.29,30 Another study found that although ketoconazole administered with chemotherapy did not appreciably prolong survival (median survival, 19 weeks), it did provide a palliative hormonal response in two-thirds of the patients (median duration, 25 days). 31 Most patients died as a result of progressive malignant disease and loss of hormonal control by ketoconazole; bacterial and opportunistic infections were a common cause of morbidity.
In conclusion, our patient represents a rare case of dual paraneoplastic syndromes, including paraneoplastic hyperamylasaemia and CS, occurring simultaneously. Although rare, the presence of more than one paraneoplastic syndrome should be considered when unexpected biochemical abnormalities are observed in the clinical setting and should prompt further investigation. In our case, the presence of uncommonly encountered paraneoplastic hyperamylasaemia initially misled diagnosis. Interestingly though, during the course of disease amylase behaved as a tumour marker reflecting response to chemotherapy.
Footnotes
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Written consent has been obtained prior to use of data from patient’s file and publication, while care has been given so that no information indicates subject’s identity.
Guarantor
CG.
Contributorship
KA, DS and DV were the attending physicians, OK and VK performed the endocrinology consults, EG prepared, examined biopsy specimen and provided Figure 1(e) and (![]() ), KA researched literature and wrote the manuscript, DS, VK and CG corrected the manuscript, CG supervised the case study. All authors reviewed and approved the final version of the manuscript.
), KA researched literature and wrote the manuscript, DS, VK and CG corrected the manuscript, CG supervised the case study. All authors reviewed and approved the final version of the manuscript.
